Connecting Science to Practice
This study describes observed patterns of concomitant medications
identified during a pharmacist-led, standardized
medication review process for patients undergoing screening
for oncology clinical trials. After evaluating >1000 medication
checks across 240 trials, the authors identified near-universal
use of concomitant medications and found that almost
one-third of reviews included a protocol-prohibited
medication. Despite this, most patients remained eligible
and were ultimately enrolled in the trial, suggesting that
structured medication review by investigational drug service
pharmacists can help overcome this common barrier to trial
participation. This research reinforces the value of pharmacist
involvement in clinical trial screening and demonstrates
how implementation of medication reviews can improve
trial access and operational consistency.
Oncology clinical trials are essential for advancing new therapies, improving patient outcomes, and providing novel treatment options for those who have exhausted standard treatments.1,2 Although the investigational drug is the focal point of clinical trials, managing concomitant medications, defined as all therapies a patient is using during trial screening or participation, is critical to ensuring patient safety, minimizing drug-drug interactions, and maintaining the integrity of clinical outcomes in oncology research. Concomitant medication use is nearly universal among patients undergoing treatment for cancer in phase 1 trials, reflecting the multiple comorbidities and complex needs of patients with cancer.3-5 Although concomitant medication restrictions are essential to protect patient safety and preserve scientific validity, they may also limit trial eligibility for patients who otherwise have few therapeutic alternatives. As a result, patients may be excluded or experience enrollment delays due to medication-related criteria, even when such restrictions are applied for appropriate scientific or safety reasons.3-5 The challenges of managing pharmacokinetic and pharmacodynamic drug-drug interactions have increased, as targeted therapies, immunotherapies, and oral anticancer agents continue to transform oncology care, necessitating oversight of concomitant medication use.6-10
Beyond treatment efficacy and safety concerns, concomitant medication restrictions are key in determining patient trial eligibility.9-11 The use of medications that are explicitly restricted by the study protocol (prohibited medications) can render a patient ineligible for trial participation, limiting trial enrollment and access to potentially beneficial investigational treatments.4,9,11 Moreover, many protocols require washout periods for prohibited medications before a patient can proceed with screening, which may delay enrollment or cause a patient to miss the eligibility window altogether.9,11 Given these challenges, conducting proactive, timely, and thorough concomitant medication reviews is essential to safeguarding trial integrity and preventing avoidable enrollment delays.
Historically, clinical pharmacy specialists embedded within disease-specific inpatient and outpatient oncology clinics conducted concomitant medication reviews at our institution. However, these reviews were time-consuming and can prohibit clinical pharmacists from engaging in other direct patient care activities. In addition, this decentralized approach resulted in variable documentation and inconsistent timing across clinics.
To address these challenges, we transitioned the responsibility of conducting initial concomitant medication reviews for oncology trials to the investigational drug service (IDS) pharmacy team. At the time of implementation, the IDS pharmacy supported >575 active clinical trials, of which approximately 70% were oncology trials. IDS pharmacists specialize in investigational drug protocols, allowing for a streamlined and standardized approach to initial medication reviews. This workflow ensured every patient underwent an initial medication review with a uniform process across all trials and clinics to allow early detection of eligibility issues and reduce downstream screening delays.
Despite these potential advantages, real-world data on the effectiveness of IDS pharmacist-led interventions in optimizing trial enrollment remain limited. This 2-year retrospective descriptive study aimed to characterize observed patterns of concomitant and protocol-prohibited medication use during oncology clinical trial screening and to evaluate associated enrollment outcomes under a standardized pharmacist-led medication review model at a single National Cancer Institute–designated academic cancer center.
Methods
We conducted a 2-year retrospective descriptive analysis of initial concomitant medication reviews for all patients screened for enrollment in oncology clinical trials at a single National Cancer Institute–designated academic cancer center from August 1, 2022, to July 31, 2024. The local institutional review board approved this study and the data collection process.
During patient eligibility screening, an IDS pharmacist reviewed reported home medications to identify any protocol-prohibited drugs. Before this evaluation, the clinical research coordinator and study team reconciled the patient’s medication list, ensuring its accuracy and completeness in the electronic medical record (EMR). The IDS pharmacist then retrieved the verified list from the EMR for final review. Reviews were conducted using a standardized documentation template within the EMR, capturing reviewed and prohibited medications. Pharmacists referenced research protocols, sponsor-provided guidance, and validated tertiary drug-drug interaction reference resources and applied these consistently across reviews. In this analysis, concomitant medications refers to all medications reported during trial screening, whereas protocol-prohibited medications represent a subset of concomitant medications that are explicitly restricted by the study protocol.
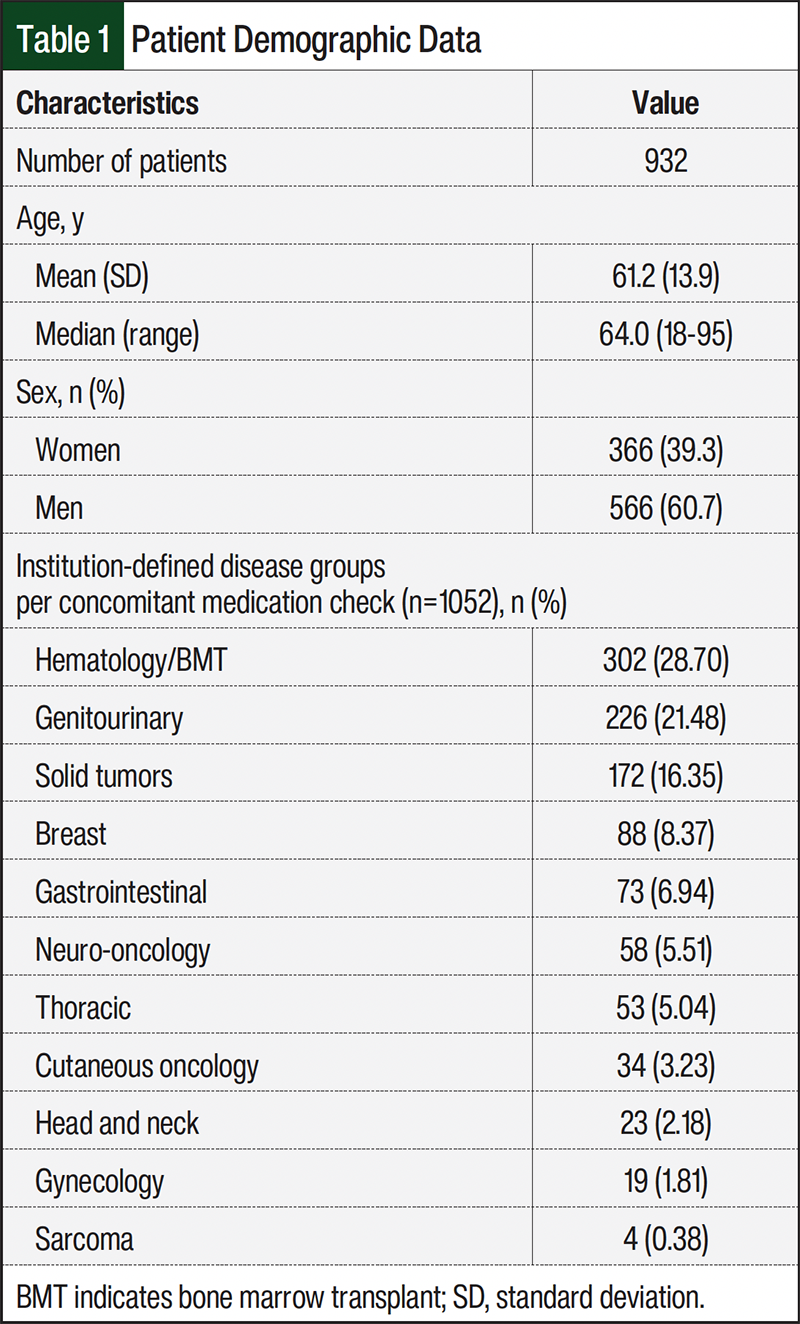
We conducted an EMR chart review of the detailed concomitant-medication report notes. The information collected included the number and identities of concomitant and prohibited medications. Medications were grouped by therapeutic class according to the American Hospital Formulary Service Pharmacologic-Therapeutic Classification system.12 Herbal supplements were defined according to National Cancer Institute guidelines as any biologically based complementary or alternative medicine.13 Information on patient demographics and trial characteristics was also collected, including age, sex, the protocol’s institution-defined disease categories used for internal trial classification, and patient enrollment status.
Data extraction was automated using an R software (version 4.2.3, CRAN.R-project.org) script, followed by periodic audits of key data points and validation by trained and independent IDS pharmacist reviewers to ensure data accuracy and consistency. Medication review notes that were mislabeled or had missing values were excluded, ensuring that only complete reviews were analyzed to maintain consistency across statistical comparisons.
Nonparametric tests were used to compare groups after data normality was assessed using the Shapiro-Wilk test (α=.05). Results are presented as medians and interquartile ranges (IQRs), with means and standard deviations included for reference. Differences between key variables and the clinical trial phases were assessed using the Kruskal-Wallis test and Dunn’s post hoc comparison to identify specific phase (phases 1-3) differences with a Benjamini-Hochberg adjustment. We conducted a Mann-Whitney U test to compare outcomes between male and female patients. The chi-square and post hoc pairwise proportions tests were used to evaluate the association between enrollment status by phase. Finally, we used binomial logistic regression modeling to assess the relationship between the total number of concomitant medications and the likelihood of a prohibited medication, with outcomes treated as binary variables. All data analyses and figures were generated using R software (version 4.2.3, CRAN.R-project.org) and the RStudio interface (version 2023.03.0; Posit team, 2023, PBC).
Results
From August 1, 2022, to July 31, 2024, the IDS pharmacist team identified 1103 concomitant medication review notes. After excluding 51 notes that were either created in error or mislabeled as initial reviews for patients already participating in a trial, 1052 initial concomitant medication reviews were included in the final analysis. These reviews corresponded to 932 patients screened for 240 clinical trials. Of the 932 patients, 104 were screened for multiple trials, including 16 who were screened for 3 trials each and 88 who were screened for 2 trials. Clinical trials consisted of phase 1, phase 2, and phase 3 trials across 11 institution-defined disease groups. Patient characteristics are shown in Table 1.
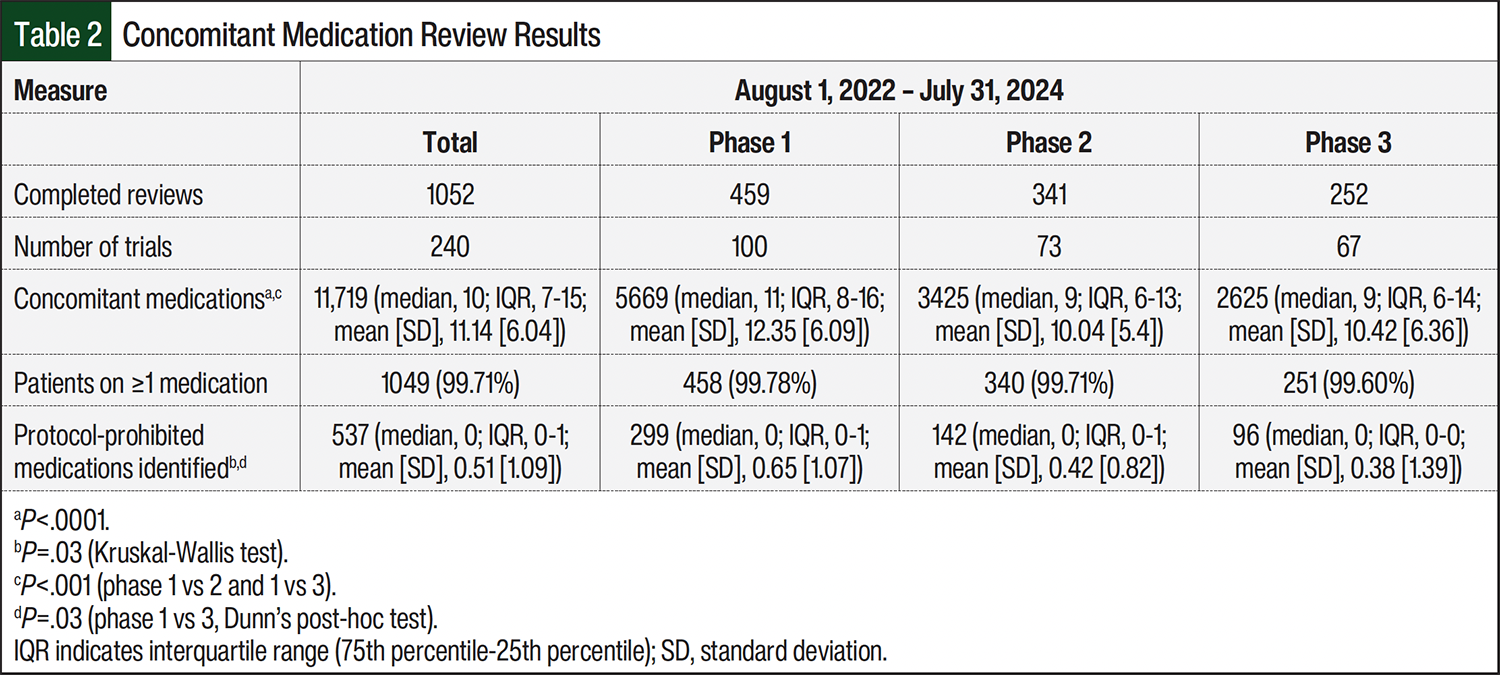
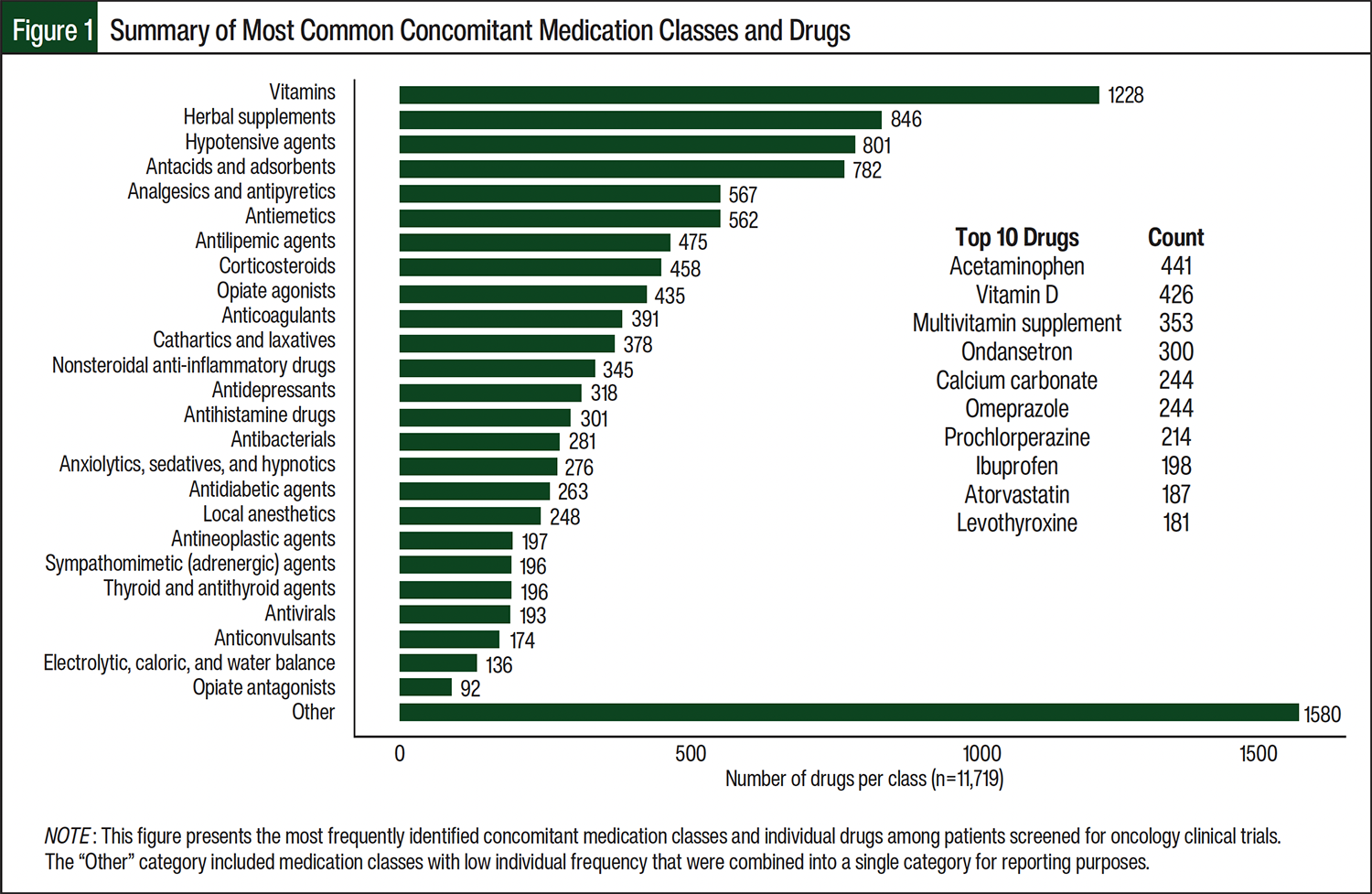
We reviewed 11,719 concomitant medications during trial screening from the 1052 initial reviews. As shown in Table 2, nearly all patients (99.71%) took ≥1 medication, with no significant differences across clinical trial phases. However, the median number of concomitant medications was highest in phase 1 trials (median, 11; IQR, 8-16) compared with phase 2 (median, 9; IQR, 6-13; P<.001) and phase 3 trials (median, 9; IQR, 6-14; P<.001). The most common concomitant medication classes were vitamin and herbal supplements, hypotensive agents, and antacids (Figure 1). The most common concomitant medications included acetaminophen, vitamin D, multivitamins, and ondansetron.
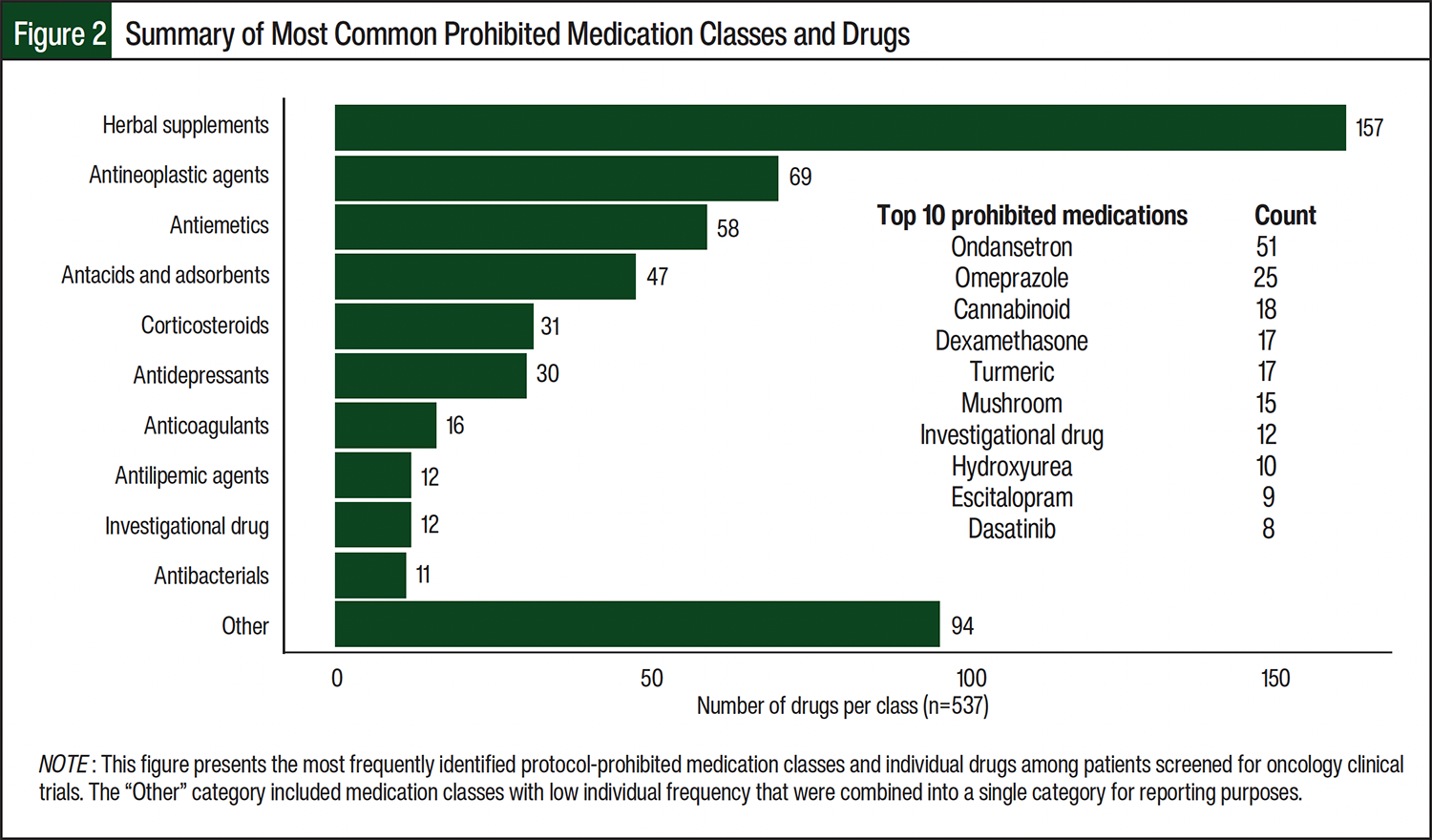
Nearly one-third (321 of 1052 [30.5%]) of reviews were taking ≥1 protocol-prohibited medication, with a total of 537 protocol-prohibited medications identified (Table 2). Patients enrolling in phase 1 trials were significantly more likely to report taking prohibited medications than were those in phase 2 (P<.001) or phase 3 (P<.001) trials. The most common prohibited medication classes across all trials were herbal supplements, antineoplastic agents, and antiemetics (Figure 2). The most frequently identified prohibited drugs included ondansetron, omeprazole, and cannabinoid products.
In addition to describing overall medication use patterns, we conducted a secondary exploratory analysis to evaluate whether medication burden was associated with prohibited medication use. We conducted an exploratory binomial logistic regression analysis to assess whether overall medication burden was associated with the likelihood of prohibited medication use during trial screening. The models predicted that for each additional medication, the odds of taking a prohibited medication increased by 7.5% (odds ratio=1.075; P<.001). However, the model demonstrated limited discrimination (area under the curve, 0.626), and adding covariates did not improve performance. These findings suggest that medication count alone is a weak predictor of prohibited medication use and reinforce the need for individualized, pharmacist-led medication review rather than reliance on medication burden as a screening surrogate.
Given the distribution of participants in this study (566 men and 366 women), we also examined whether sex difference in medication was associated with prohibited medication use. Female patients had a slightly higher median number of concomitant medications than male patients (11 vs 10; P=.005), but no significant difference was seen in prohibited medication use (P=.096).
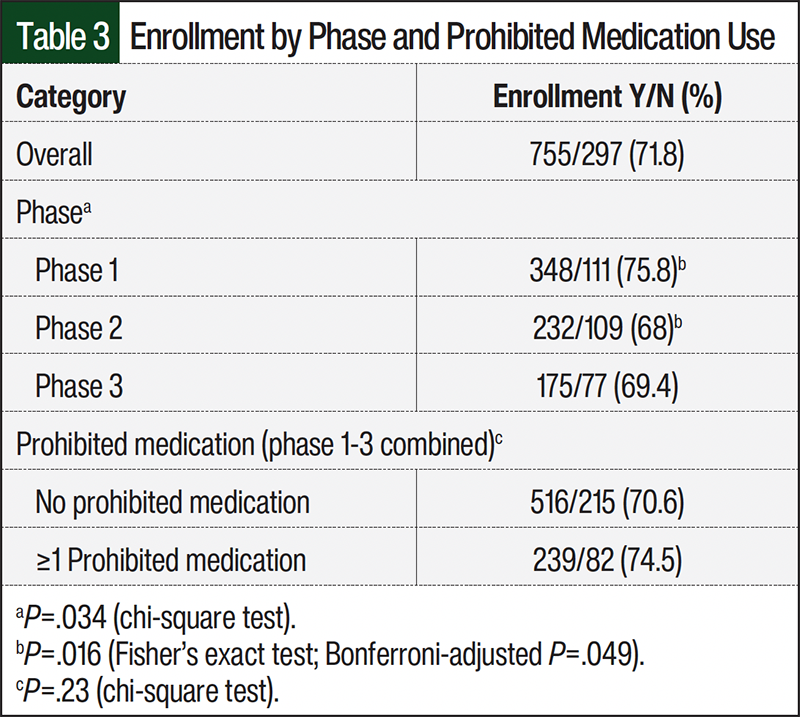
We examined the enrollment status of screened patients. Of the 1052 completed concomitant medication reviews, 755 (71.8%) patients were enrolled in their respective trials (Table 3). Enrollment rates varied by trial phase, with patients screened for phase 1 trials having a higher enrollment rate than those in phase 2 trials (75.8% vs 68%; χ²(2)=6.73; P=.035; post hoc Fisher’s exact test: P=.0163, adjusted P=.049). Of note, the presence of a prohibited medication at screening did not significantly impact enrollment eligibility. Among the 321 reviews identified as taking ≥1 prohibited medication, a total of 239 (74.5%) remained eligible for enrollment by cycle 1 day 1, which is a rate comparable to those who were using no prohibited medications (70.6%; χ²(1)=1.46; P=.227; Table 3).
Discussion
This study evaluated concomitant medication use during oncology clinical trial screening and revealed that nearly all patients (99.7%) took ≥1 medication, regardless of trial phase. Although previous reports identified this trend in phase 1 trials,3-5 we found concomitant medication use was common across all trial phases, with patients in phase 1 trials having the highest median medication use. In addition, almost one-third of patients (30.5%) took ≥1 protocol-prohibited medication before trial enrollment. These findings underscore the importance of accurate eligibility assessments during clinical trial screening that include comprehensive medication histories as key components of this process.14,15
Although protocol-prohibited medications were frequently identified at screening, their presence did not automatically disqualify patients, and most were ultimately able to meet eligibility criteria and enroll. A key factor in mitigating their exclusionary effect was the structured IDS pharmacist–led concomitant medication review process, which was sustained as part of routine clinical trial screening and enabled early identification and resolution of medication-related eligibility issues before enrollment. It is notable that 70% of patients initially flagged as ineligible due to the use of prohibited medication were successfully enrolled, with enrollment rates comparable to those not taking prohibited medications.
This study was conducted at a single institution with an established IDS team, but the findings highlight the broader value of site-level, pharmacist-led medication reviews. Although we could not compare outcomes before and after the implementation of this IDS-led process due to inconsistent and nonstandardized documentation before implementation, the high enrollment rate among patients initially taking protocol-prohibited medications suggests that early identification and management can overcome this common barrier to inclusion.
It is important to note that medication-related ineligibility is just one of many factors in trial screening, but our data indicate that patients taking prohibited medications were not at an increased risk of ineligibility compared with those who were not taking prohibited medications, after accounting for other eligibility criteria, such as performance status, prior therapies, organ function, and comorbid conditions. Assuming that other exclusion factors were similarly distributed across both groups, this finding suggests that protocol-prohibited medications did not pose an additional barrier to enrollment when proactively managed. Thus, standardized medication assessments may reduce preventable delays and support more efficient trial enrollment.
In addition, we explored whether medication burden alone could predict trial ineligibility risk. Whereas patients with a higher number of concomitant medications were more likely to be taking protocol-prohibited medications, total medication count alone was not a strong predictor of prohibited medication use or enrollment outcomes. This suggests that patients receiving multiple medications are not uniformly high risk, highlighting the importance of individualized medication reviews.
We also examined potential demographic influences, specifically whether patient sex influenced concomitant medication use. Manteuffel and colleagues suggest that women are prescribed more medications than men.16 Consistent with these findings, we observed a significant difference in the median number of reported concomitant medications when comparing female and male patients. This increased medication burden, however, did not result in an increased risk of taking protocol-prohibited medications. These findings reinforce that total medication count alone does not inherently indicate a greater likelihood of ineligibility and underscore the importance of evaluating medication use on a case-by-case basis.
A notable finding was the high prevalence of herbal supplements, which were one of the most common drugs used and the most common prohibited drug classes. Yet, research protocols differ widely regarding herbal supplement use, from outright prohibition to an absence of restrictions. Although some herbal supplements have well-documented interactions, many lack clear pharmacokinetic and pharmacodynamic profiles,17 leading to inconsistent recommendations and uncertainty for patients and investigators. Without explicit regulatory or protocol guidance, oncologists and trial staff are left to determine potential risks, often without standardized resources. Given their widespread use and potential for clinically significant interactions, herbal supplements warrant greater consideration in trial design to ensure reliable study outcomes while respecting patient preferences. Given their frequent designation as protocol-prohibited, routine interrogation for herbal supplements use during screening is essential to ensure that eligibility assessments are accurate and complete.
Limitations
This study has several limitations inherent in its retrospective, single-center design that may limit generalizability. However, the consistency of our results with the literature supports their relevance regarding concomitant medication use in oncology clinical trials. Although medication lists were reconciled before review, some home medications may have been underreported or inaccurately documented, a known limitation of retrospective chart reviews that may have affected data completeness.
Trial eligibility represents another important limitation, as enrollment decisions are influenced by multiple factors beyond concomitant medication use, including performance status, prior therapies, and comorbid conditions. This study was not intended to evaluate all causes of ineligibility but to describe medication-related exclusions and their association with enrollment outcomes. This study reflects current protocol-defined eligibility practices and does not account for emerging efforts to modernize clinical trial eligibility criteria. Direct comparisons between the prior decentralized review processes and the standardized IDS-led approach were not feasible due to inconsistent documentation before implementation, limiting our ability to assess changes in screening efficiency or enrollment outcomes over time.
In addition, although our findings suggest that patients on protocol-prohibited medications adjusted their regimens to meet eligibility criteria, we did not systematically capture how these adjustments were made (eg, discontinuation vs substitution), limiting our insight into how protocol restrictions were operationally resolved. During the study period, IDS pharmacist recommendations were documented in the EMR and communicated to study teams as part of routine screening workflows; however, the timing of these communications relative to protocol-specific screening windows and whether any patients were unable to resolve medication-related restrictions were not captured in this retrospective analysis.
A detailed evaluation of implementation processes, pharmacist training, and long-term sustainability was beyond the scope of this analysis.
Conclusion
This study provides valuable insight into concomitant and protocol-prohibited medication use during oncology clinical trial screening. Nearly all screened patients reported taking concomitant medications, and almost one-third were taking a protocol-prohibited medication, highlighting the complexity of medication management during trial eligibility assessment. It is important to note that most patients identified as taking protocol-prohibited medications ultimately enrolled, underscoring the role of timely, structured, pharmacist-led medication review in identifying and addressing medication-related eligibility barriers early in the screening process.
As clinical trial designs continue to evolve with increasingly complex eligibility criteria, standardized approaches to concomitant medication review may improve trial accessibility, enhance operational efficiency, and promote consistent screening practices across disease groups and trial phases. Future research should focus on larger, multicenter studies to evaluate the implementation and impact of pharmacist-led medication review frameworks, including their effects on enrollment timelines, eligibility resolution strategies, and downstream trial efficiency and their role in supporting safe trial participation through proactive management of potential drug-drug interactions.
Author Disclosure Statement
Dr Maughan is a paid consultant/advisor to AbbVie, Astellas, AVEO Oncology, Bayer Oncology, Bristol-Myers Squibb, Clovis, Exelixis, Janssen, Lilly, Merck, Peloton Therapeutics, Pfizer, Sanofi, Telix, Tempus, and Xencor; Huntsman Cancer Institute has received research funding from Bavarian-Nordic (Inst), Bristol-Myers Squibb (Inst), Clovis (Inst), Exelixis (Inst), Genentech (Inst), and Merck on Dr Maughan’s behalf. Dr Maughan is a member of a drug safety monitoring board for AVEO Oncology. Dr Hill and Dr Froerer have no conflicts of interest to report.
References
- Corbaux P, Bayle A, Besle S, et al. Patients’ selection and trial matching in early-phase oncology clinical trials. Crit Rev Oncol Hematol. 2024;196: 104307. doi:10.1016/j.critrevonc.2024.104307
- National Comprehensive Cancer Network. Clinical Trials. NCCN Clinical Practice Guidelines in Oncology. Accessed February 28, 2025. https://www.nccn.org/patientresources/patient-resources/additional-resources/clinical-trials
- Marcath LA, Finley CM, Wong SF, Hertz DL. Drug-drug interactions in subjects enrolled in SWOG trials of oral chemotherapy. BMC Cancer. 2021;21:324. doi:10.1186/s12885-021-08050-w
- Borad MJ, Curtis KK, Babiker HM, et al. The impact of concomitant medication use on patient eligibility for phase I cancer clinical trials. J Cancer. 2012;3:345-353. doi:10.7150/jca.4714
- Wisinski KB, Cantu CA, Eickhoff J, et al. Potential cytochrome P-450 drug–drug interactions in adults with metastatic solid tumors and effect on eligibility for phase I clinical trials. Am J Health Syst Pharm. 2015;72:958-965. doi:10.2146/ajhp140591
- van Leeuwen RWF, Brundel DHS, Neef C, et al. Prevalence of potential drug–drug interactions in cancer patients treated with oral anticancer drugs. Br J Cancer. 2013;108:1071-1078. doi:10.1038/bjc.2013.48
- van Leeuwen RWF, Jansman FGA, van den Bemt PMLA, et al. Drug–drug interactions in patients treated for cancer: a prospective study on clinical interventions. Ann Oncol. 2015;26:992-997. doi:10.1093/annonc/mdv029
- Maggiore RJ, Dale W, Gross CP, et al. Polypharmacy and potentially inappropriate medication use in older adults with cancer undergoing chemotherapy: effect on chemotherapy-related toxicity and hospitalization during treatment. J Am Geriatr Soc. 2014;62:1505-1512. doi:10.1111/jgs.12942
- McGahey KE, Weiss GJ. Reviewing concomitant medications for participants in oncology clinical trials. Am J Health-Syst Pharm. 2017;74:580-586. doi:10.2146/ajhp151052
- Malifarge L, Deppenweiler M, Italiano A, Lortal B. Impact of medication reconciliation in oncology early phase studies: a drug-drug interaction retrospective study. JCO Oncol Pract. 2024;20:386-392. doi:10.1200/op.22.00281
- Rodríguez-Mauriz R, González-Laguna M, Perayre-Badia M, et al. Pharmaceutical care in the screening process of phase I oncohaematological clinical trials. Eur J Hosp Pharm. 2025;32:522-527. doi:10.1136/ejhpharm- 2024-004168
- AHFS Pharmacologic-Therapeutic Classification System. American Society of Health-System Pharmacists. Accessed December 12, 2024. www.ashp.org/products-and-services/ashp-licensing/ahfs-therapeutic-classification?loginreturnUrl=SSOCheckOnly
- Complementary and Alternative Medicine. National Cancer Institute. Updated September 10, 2024. Accessed December 12, 2024. www.cancer.gov/about-cancer/treatment/cam
- Son H, Kim J, Kim C, et al. Pharmacist-led interdisciplinary medication reconciliation using comprehensive medication review in gynaecological oncology patients: a prospective study. Eur J Hosp Pharm. 2018;25:21-25. doi:10.1136/ejhpharm-2016-000937
- Vega TGC, Sierra-Sánchez JF, Martínez-Bautista MJ, et al. Medication reconciliation in oncological patients: a randomized clinical trial. J Manag Care Spec Pharm. 2016;22:734-740. doi:10.18553/jmcp.2016.15248
- Manteuffel M, Williams S, Chen W, et al. Influence of patient sex and gender on medication use, adherence, and prescribing alignment with guidelines. J Womens Health (Larchmt). 2014;23:112-119. doi:10.1089/jwh.2012.3972
- Posadzki P, Watson L, Ernst E. Herb–drug interactions: an overview of systematic reviews. Br J Clin Pharmacol. 2013;75:603-618. doi:10.1111/j.1365-2125.2012.04350.x