With commentaries by

Seth Wander, MD, PhD
Director, Precision Medicine,
Termeer Center for Targeted Therapies
Director, Translational Research,
Breast Oncology Program
Assistant Professor of Medicine, Harvard Medical School
Massachusetts General Hospital
Boston, MA
Introduction
Hormone receptor (HR)-positive/HER2-negative breast cancer is the most common subtype of breast cancer.1 The cornerstone of adjuvant treatment for any hormone-sensitive breast cancer is endocrine therapy (ET). However, patients with HR-positive/HER2-negative breast cancer, particularly those with early-stage, node-positive disease with certain pathological and genomic features, remain at substantial risk of recurrence and death when treated in the adjuvant setting with ET alone.2 In combination with adjuvant ET, cyclin-dependent kinase (CDK)4/6 inhibitors represent an important class of targeted therapies for both node-negative and node-positive, HR-positive/HER2-negative early breast cancer at high risk of recurrence. Among FDA-approved CDK4/6 inhibitors, high-risk criteria for eligibility are based on varying tumor characteristics such as size, grade, molecular genomic risk profile, and nodal status. One such agent, abemaciclib, was approved in 2021 for high-risk, node-positive, HR-positive/HER2-negative early breast cancer based on the results of the phase 3 monarchE trial.3 Pivotal data from monarchE demonstrated statistically significant improvement in invasive disease-free survival (IDFS) at 4 years for patients receiving 2 years of abemaciclib alongside ET when compared with ET alone.4 With the recent release of landmark 7-year analyses from monarchE, this article provides a timely data review, along with expert commentary provided by Seth Wander, MD, PhD. Dr Wander summarizes long-term outcomes of patients treated with ET alone and those treated with abemaciclib in combination with ET in monarchE. Additional real-world evidence (RWE) reported within this article extends tolerability and dosing patterns for abemaciclib from clinical trial experience to clinical practice.
ET: The Backbone of CDK4/6 Inhibitor Therapy
CDK4/6 inhibitors are used in combination with ET, as ET is supported by decades of clinical trials demonstrating efficacy in patients with HR-positive breast cancers. ET is generally recommended for patients for a minimum of 5 years to reduce recurrence.5 Options for ET selection include selective estrogen receptor modulators (SERMs), such as tamoxifen, or aromatase inhibitors (AIs), such as anastrozole, letrozole, and exemestane, and may be combined with therapies, such as goserelin or leuprolide, to produce ovarian function suppression (OFS) or ablation in premenopausal women.6
The impact of adjuvant ET on reducing the risk of death varies from 10% to 30% according to multiple meta-analyses and clinical trials. The first meta-analysis found that for women with estrogen receptor (ER)-positive disease, 5 years of tamoxifen resulted in a 30% reduction in the risk of death due to breast cancer and a 9.2% absolute reduction in breast cancer mortality at 15 years compared with no tamoxifen treatment (P<.00001) using data from 20 trials and 10,645 patients.7 A second meta-analysis examined 2 trials and 9885 postmenopausal women with ER-positive early breast cancer and found that 5 years of AI therapy offered improvement over tamoxifen, reducing the risk of death due to breast cancer by 15% and an absolute reduction in breast cancer mortality by 2.1% at 10 years (P=.009).8 Several studies have investigated AI therapy for a duration longer than 5 years. Among postmenopausal patients who had already completed 5 years of AI therapy, an analysis of more than 22,000 individuals from 12 trials revealed that extending AI treatment for an additional 5 years resulted in a 10% reduction in risk of death due to breast cancer, but only a nonsignificant 0.6% absolute reduction in breast cancer mortality at 15 years, and a significantly increased risk of bone fracture.9 In the higher-risk setting of premenopausal female patients with HR-positive breast cancer, the SOFT trial examined the addition of OFS to a SERM or AI for 5 years.10 This trial compared tamoxifen alone, tamoxifen with OFS, and exemestane with OFS. The combination of exemestane and OFS emerged as the most effective, reducing the overall risk of death by 26% and absolute reduction in breast cancer mortality by 5.6% compared with tamoxifen alone at a median follow-up of 12 years.10
These same studies demonstrated improvement in disease recurrence, yet the long-term risk of recurrence remained high. Following 5 years of tamoxifen treatment, the 15-year recurrence rates were 33.0% compared with 46.2% for the arm receiving no tamoxifen treatment (rate ratio [RR], 0.61; 95% CI, 0.57-0.65).7 When comparing 5 years of an AI or tamoxifen, the 10-year recurrence rates were 19.1% for the AI group and 22.7% for the tamoxifen group (RR, 0.80; 95% CI, 0.73-0.88).8 After 5 years of extended AI treatment, the 15-year recurrence rates were 11.6% compared with 15.2% for the arm receiving no further treatment (RR, 0.71; 95% CI, 0.61-0.81).9 For premenopausal women receiving 5 years of ET, the 12-year distant recurrence-free interval was 75.1% with tamoxifen alone, 77.7% with tamoxifen plus OFS, and 79.6% with exemestane plus OFS.10
ET in Node-Positive, HR-Positive/HER2-Negative Breast Cancer
With ET alone, "patients with higher-risk HR-positive disease experience recurrence rates on par with triple-negative disease."
Whereas ET demonstrates a reduction in recurrence and mortality rates in the broader HR-positive population, real-world data highlight significant differences in outcomes between high-risk and non−high-risk patients with HR-positive/HER2-negative disease. A retrospective study utilized the Flatiron Health database of electronic health records from approximately 16,000 adult patients with early breast cancer, treated with adjuvant ET. This study showed that node-positive, high-risk disease (as defined by criteria in the monarchE trial) was associated with a greater 5-year recurrence (29.1% vs 8.8%) and 5-year mortality (18.4% vs 6.1%) compared with non−high-risk disease. In contrast, 5-year recurrence (29.1% vs 25.7%) and 5-year mortality (18.4% vs 20.8%) in these patients were similar to those observed in patients with early triple-negative breast cancer (TNBC).11 TNBC is considered particularly aggressive due to its high recurrence rates, limited treatment options, and poor prognosis compared with other breast cancer subtypes.12 Although N1 disease, defined as the presence of 1 to 3 positive lymph nodes, is not typically categorized as high risk, this study revealed that in the presence of additional high-risk features, patients with N1 disease that is grade 3 or a tumor size ≥5 cm face significantly worse outcomes, including a 6.3% higher 5-year mortality risk and a 14.3% higher recurrence risk when compared with N1 non−high-risk disease.11 These findings underscore the importance of recognizing that even limited nodal involvement can represent a doubled risk of recurrence and death when additional clinical or pathological factors are present.
Commentary by Seth Wander, MD, PhD: We know that as risk factors increase (including tumor size, grade, molecular genomic risk profile, and nodal status), patients have higher risks of both local and distant recurrence. I think this real-world data underscores the fact that the subgroup of patients with higher-risk HR-positive disease experience recurrence rates on par with triple-negative disease. Placing these patients into the context of their risk profile is an important reminder for clinicians to view them as a distinct patient population and helps guide appropriate titration of their treatment regimen toward their risk profile.
Historically, treatment options for HR-positive/HER2-negative breast cancer have remained largely the same, with adjustments primarily limited to chemotherapy, choice of anti-estrogen therapy (tamoxifen vs an aromatase inhibitor), the use of ovarian suppression in premenopausal women, or the duration of ET therapy (5, 7, or 10 years). We now have the opportunity to add CDK4/6 blockade, which has conveyed an important and meaningful difference for recurrence risk. We also have the first overall survival signal for adjuvant CDK4/6 inhibitors, and in my opinion, the importance of that cannot be understated.
The IDFS benefit of the CDK4/6 inhibitor abemaciclib for treatment of HER2-negative advanced or metastatic breast cancer has long been established.4 In the early breast cancer setting, abemaciclib is indicated in combination with ET (tamoxifen or an AI) for the adjuvant treatment of adult patients with HR-positive/HER2-negative, node-positive early breast cancer at high risk of recurrence based on an IDFS benefit at 4 years, including in patients with high-risk N1 disease.4 The recent publication of the landmark 7-year analysis of abemaciclib’s pivotal trial, monarchE, marks the first report of a statistically significant increase in overall survival (OS), along with a continued IDFS benefit in patients with high-risk, node-positive, HR-positive/HER2-negative early breast cancer receiving a CDK4/6 inhibitor and ET compared with ET alone.13
Long-Term Outcomes of Abemaciclib: 7-Year Updated Analysis of monarchE
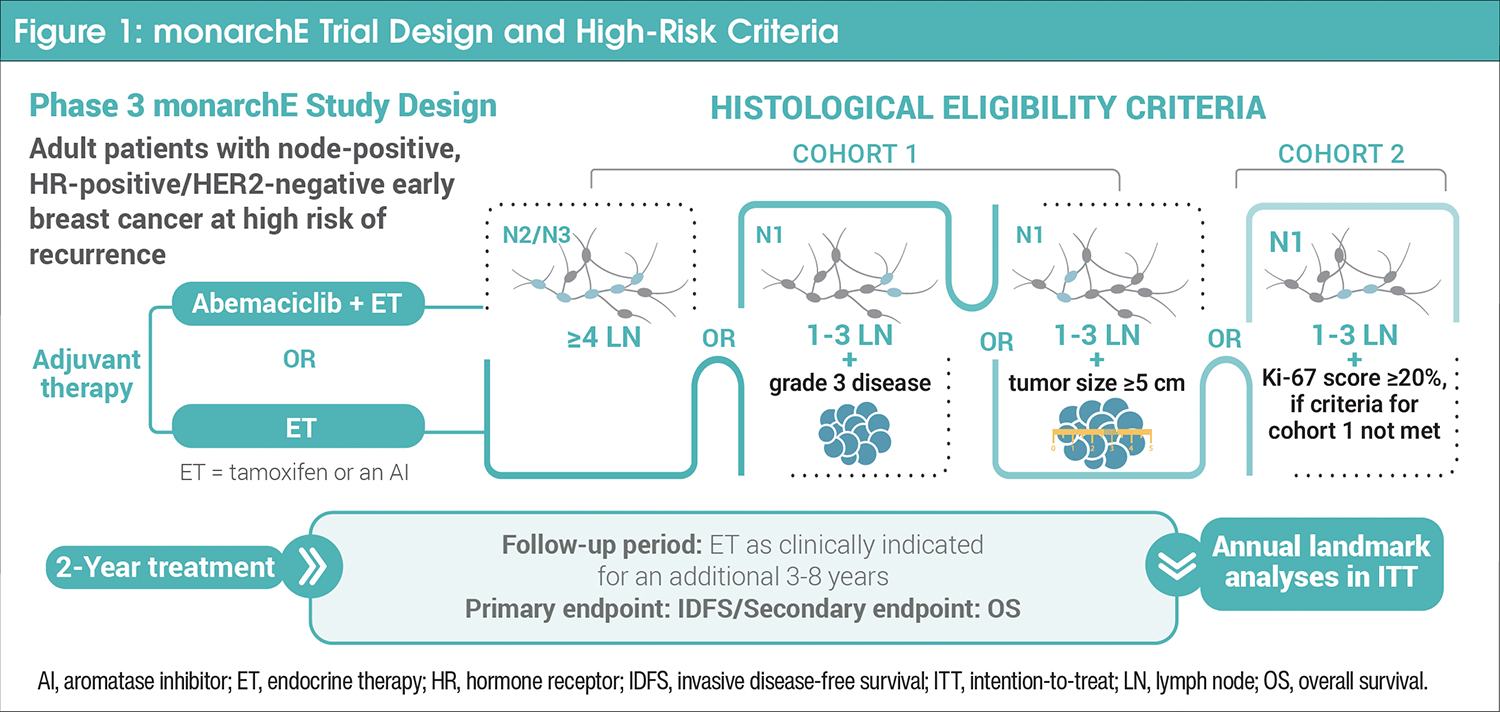
monarchE (NCT03155997) was a global, randomized, open-label, phase 3 study that included patients aged ≥18 years with node-positive, HR-positive/HER2-negative early breast cancer with a high risk of recurrence.13 A total of 5637 patients were randomized in a 1:1 ratio to receive at least 5 years of adjuvant ET with (n=2808) or without (n=2829) 2 years of adjuvant abemaciclib.13 Patients in either arm were assigned to 1 of 2 subgroup cohorts based on tumor pathology. Cohort criteria are outlined in Figure 1.
In addition to the criteria in Figure 1, cohort 1 also allowed micrometastatic, multifocal, or multicentric disease, and constituted 91% of the study’s total patients.
The primary endpoint was IDFS.13 IDFS was defined as the time from randomization until the first occurrence of 1 of the following events: ipsilateral invasive disease, ipsilateral locoregional invasive disease, contralateral invasive breast cancer, distant recurrence, second primary noninvasive cancer, or death from any cause.13 Key secondary endpoints included OS and distant recurrence-free survival (DRFS).13 OS was defined as the time from randomization until the date of death from any cause, and DRFS was defined as the time from randomization until the first occurrence of metastatic disease or death from breast cancer. Both primary and secondary endpoints were analyzed in the intention-to-treat (ITT) population, which included both cohort 1 and cohort 2.13 Landmark analyses were performed annually for up to 7 years using the Kaplan-Meier method. Safety was assessed, with serious adverse events (AEs) monitored for up to 5 years from randomization.13
At the 4-year primary efficacy analysis, a significant improvement in IDFS was observed in the ITT population, driven predominantly by patients in cohort 1. IDFS rates for cohort 1 were 85.5% for treatment with abemaciclib plus ET versus 78.6% for treatment with ET alone (hazard ratio, 0.65; 95% CI, 0.57-0.75).4
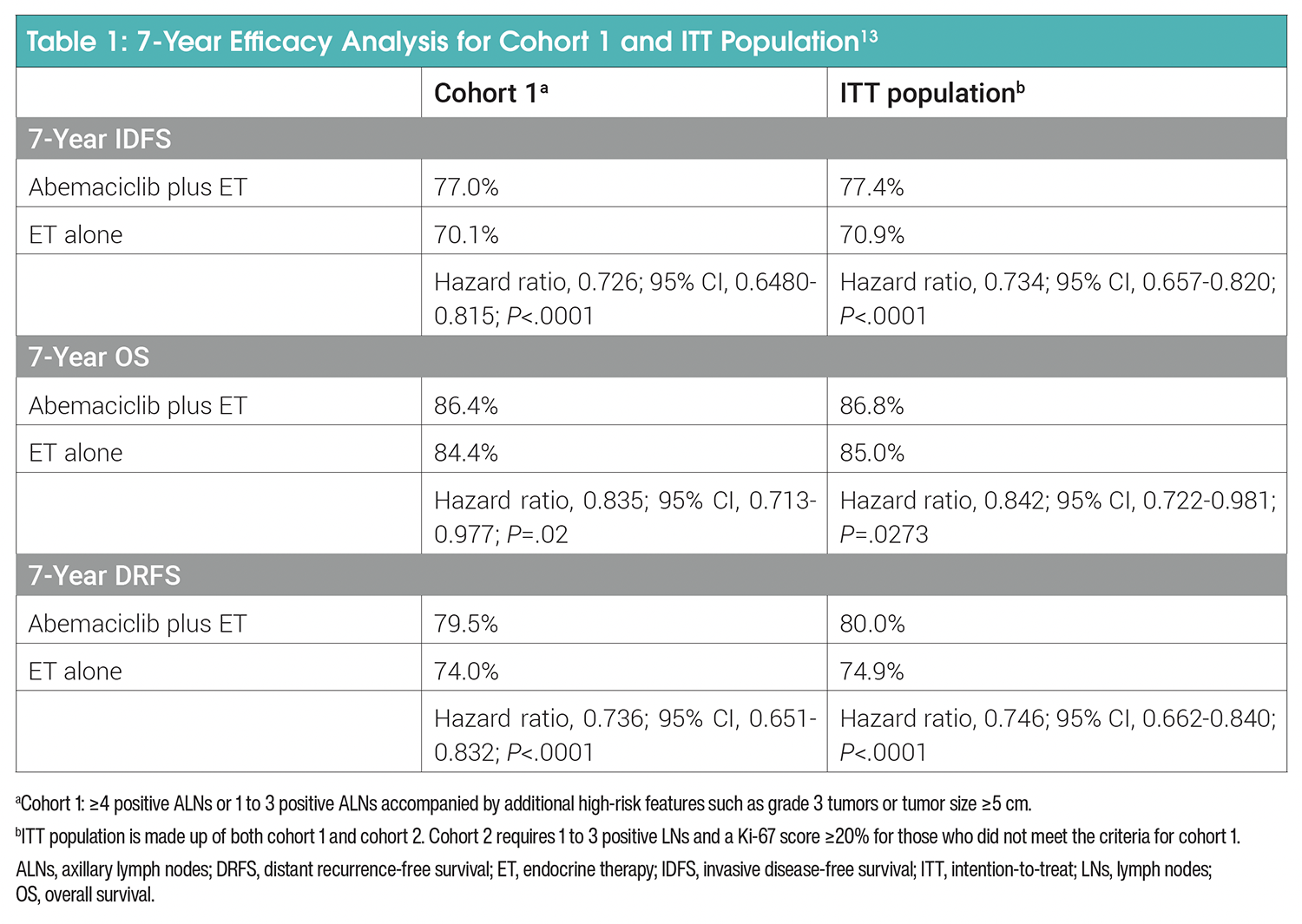
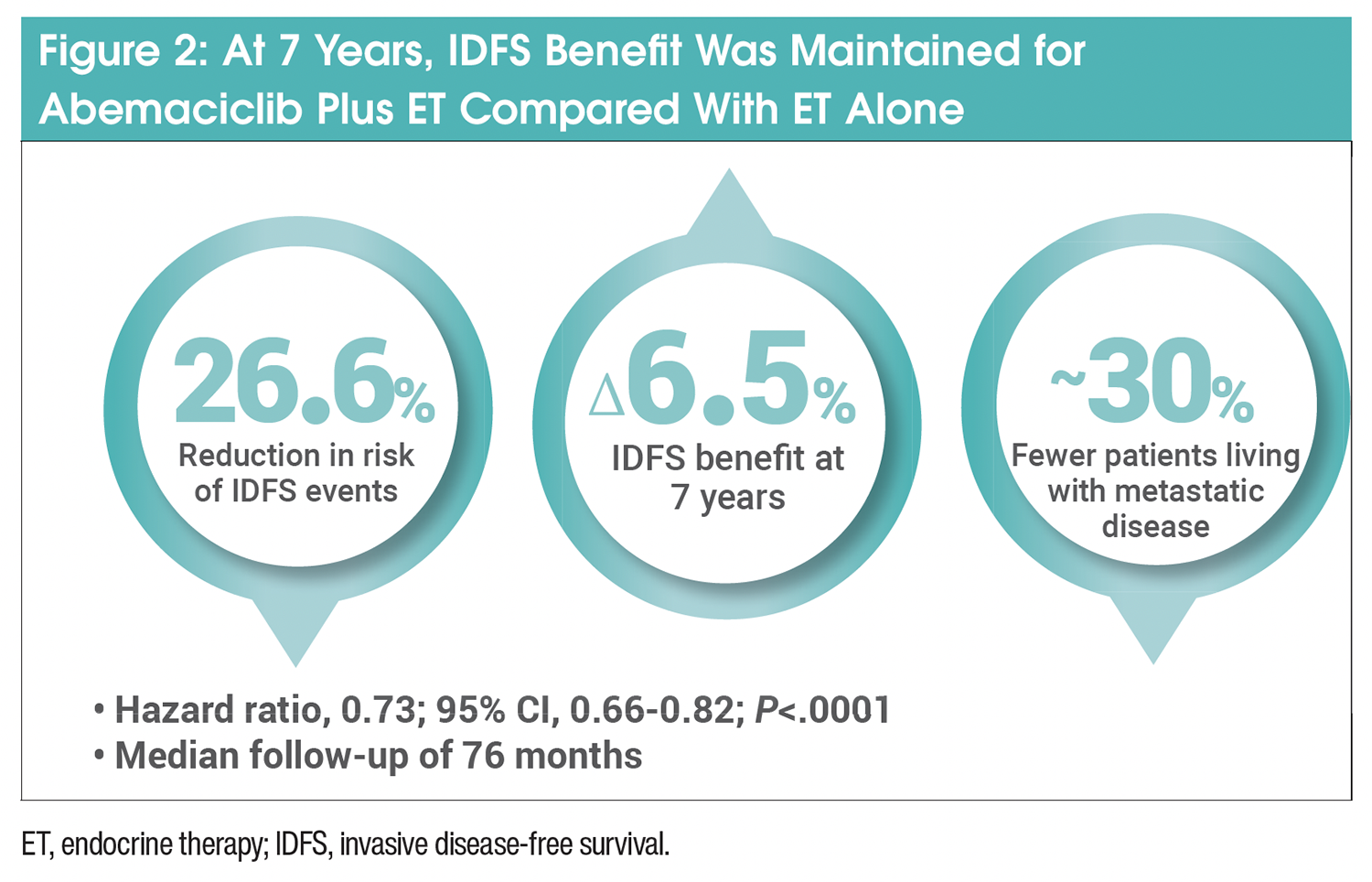
IDFS results were updated at the 7-year landmark analysis (Table 1). In the ITT population (cohorts 1 and 2), an IDFS benefit was maintained at 7 years for patients receiving abemaciclib plus ET, with an absolute improvement of 6.5% (median follow-up time was 6.3 years; hazard ratio, 0.73; 95% CI, 0.66-0.82; P<.0001; Figure 2). The IDFS rate was 77.4% for the abemaciclib plus ET group compared with 70.9% for the ET alone group, and there was a 26.6% reduction in the risk of IDFS events. This benefit was also consistent at 7 years in cohort 1.13
This was consistent across prespecified subgroups regardless of menopausal status, prior treatments, or choice of first ET.13 Most IDFS events involved distant metastatic disease.13 However, there were approximately 30% fewer patients in the abemaciclib arm living with metastatic disease compared with the ET arm (180 patients [6.4%] vs 266 patients [9.4%], respectively).13,14
In the ITT population, treatment with abemaciclib plus ET resulted in a 25.4% reduction in the risk of developing a DRFS event compared with ET alone, with higher rates at 5.1% for abemaciclib plus ET (hazard ratio, 0.75; 95% CI, 0.66-0.84; P<.0001).13 For cohort 1, consistent outcomes for DRFS were also observed (Table 1).
Commentary by Seth Wander, MD, PhD: The 7-year analysis from monarchE is important for a variety of reasons. This represents the longest follow-up data we have for any adjuvant CDK4/6 inhibitor. The results suggest that the benefit is durable and persistent after all participants have completed their adjuvant CDK inhibitor course. You see that the magnitude of benefit in recurrence risk, both overall and distant recurrence risk, is persistent, and actually improved over time compared with the first 2 to 4 years of follow-up. The pattern of the IDFS and DRFS curves suggests that the early intervention with a CDK4/6 inhibitor changes the natural history of the disease, at least in a subset of these patients. The data suggest that there’s a long-lasting effect of using those 2 years of abemaciclib. This may be impacting micrometastatic tumor cells and preventing the later development of overt metastatic disease.
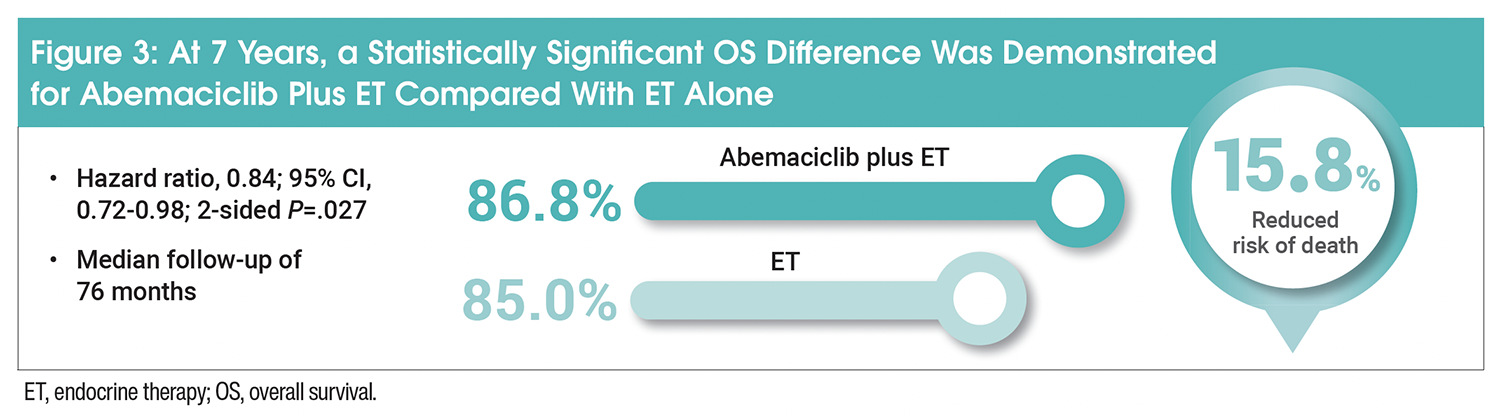
Primary OS analysis was a much anticipated prespecified key secondary endpoint as part of the 7-year analysis. For the ITT population at 7 years, the risk of death was 15.8% lower in the abemaciclib arm compared with ET alone (hazard ratio, 0.84; 95% CI, 0.72-0.98; 2-sided P=.027; Figure 3), meeting the prespecified criteria for statistical significance.14 Increased OS was consistent across prespecified subgroups, including patients aged <65 years, premenopausal patients, and patients utilizing tamoxifen as their first ET.14 In cohort 1, consistent OS outcomes were also observed (Table 1).13
Commentary by Seth Wander, MD, PhD: The 7-year follow-up data from monarchE provide the first statistically significant improvement in overall survival for any CDK4/6 inhibitor in the adjuvant setting. For clinicians who may have been hesitant to use these drugs because of cost or toxicity, we now have a signal for both recurrence risk reduction and overall survival.
At 7 years, the majority of patients without documented distant recurrence continued to receive ET.13 Assessed in the ITT population, 97.0% of patients in the abemaciclib plus ET group remained on ET, with 85.4% receiving therapy for ≥5 years. Similarly, in the ET alone group, 96.7% of patients continued ET, with 82.4% receiving it for ≥5 years. In addition to ET, the most frequently reported first-line treatments following metastatic recurrence were CDK4/6 inhibitors (30.0% vs 47.3%) and chemotherapy (32.7% vs 23.7%) in the abemaciclib plus ET and ET alone groups, respectively.13 Metastatic first-line therapies were also evaluated by time to distant recurrences, with early recurrences defined as those occurring on metastatic first-line treatment or within 12 months of 2-year adjuvant treatment completion/discontinuation, and later recurrences as those occurring >12 months from adjuvant treatment completion/discontinuation.13 In the ITT population, patients with early recurrences in the ET alone arm had higher rates of CDK4/6 inhibitor utilization as their first post-discontinuation treatment compared with the abemaciclib arm (44.7% vs 15.2%), whereas the difference between groups was less pronounced in patients with later recurrences (50.6% vs 43.1%).13
Since the primary safety analysis, no new safety signals were detected, and the AE profiles of both arms were consistent with prior analyses.15 In the safety set, deaths after 30 days of study treatment discontinuation due to AEs in the abemaciclib (n=2791) and ET (n=2800) arms were 1.6% versus 1.1%, respectively.13 Among patients who had post-discontinuation AE data (n=2621 and n=2637, respectively), rates of serious AEs post-discontinuation were similar between the 2 arms (7.5% vs 8.1%).13
Considerations for Clinical Practice
Abemaciclib is associated with a distinct toxicity profile that requires proactive management, and if left unaddressed, can compromise patient adherence, reduce quality of life, and lead to early treatment discontinuation. Common grade 3/4 AEs in monarchE included leukopenia (19%), neutropenia (18.7%), lymphopenia (13.2%), diarrhea (8%), and fatigue (2.9%). Dose interruptions occurred in 62% of patients in monarchE. Grade 3 or 4 AEs included neutropenia, leukopenia, diarrhea, and lymphopenia, which led to permanent discontinuation in 19% of patients.4
Proactive management strategies to mitigate side effects include instructing patients to initiate antidiarrheal therapy, increase oral fluids, and notify their healthcare provider at the first sign of loose stools.4 Additionally, regular monitoring of blood counts is recommended, starting with the initiation of abemaciclib therapy and continuing at least every 2 weeks for the first 2 months, followed by monthly checks for the next 2 months.4
Commentary by Seth Wander, MD, PhD: Side effect management is critical. It is important to provide a thorough and detailed overview of these risks for the patient, making sure they understand what side effects may occur, and reviewing options to help prevent and address these issues as they arise. Patients benefit the most from multidisciplinary team engagement, and should be followed closely during initiation and throughout treatment on these agents.
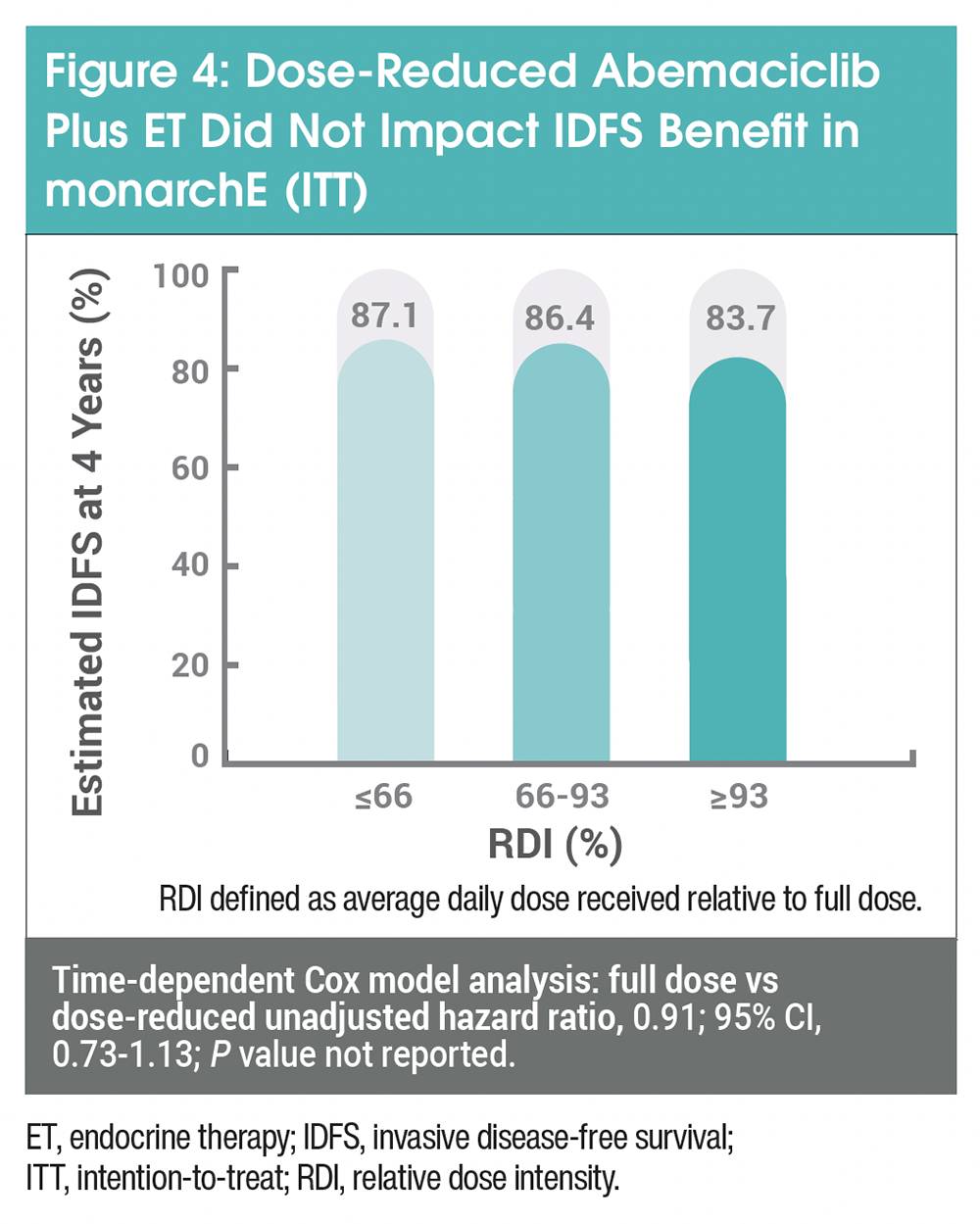
Dose reductions are another proactive management strategy to keep patients on treatment for the recommended duration. This strategy is supported by clinical trial experience demonstrating maintained efficacy at reduced doses and RWE demonstrating the feasibility of achieving treatment persistence in clinical practice. In the monarchE trial, dose reductions due to an AE were employed in 44% of patients, with 17%, 8%, and 5% of AEs attributed to diarrhea, neutropenia, and fatigue, respectively. Abemaciclib was administered orally twice daily (BID) at a full dose of 150 mg, and doses were reduced to 100 mg and then to 50 mg BID if AE issues persisted following the first reduction.16 Data from monarchE’s 4-year long-term analysis demonstrated relatively consistent IDFS across relative dose intensity (RDI) subgroups of abemaciclib plus ET. A time-dependent Cox analysis showed sustained IDFS benefit in full-dose recipients compared with those dose-reduced in both the ITT population (hazard ratio, 0.91; 95% CI, 0.73-1.13; P value not reported; Figure 4) and cohort 1.17 Therefore, certain AEs may be managed by dose reductions while maintaining a primary IDFS benefit.
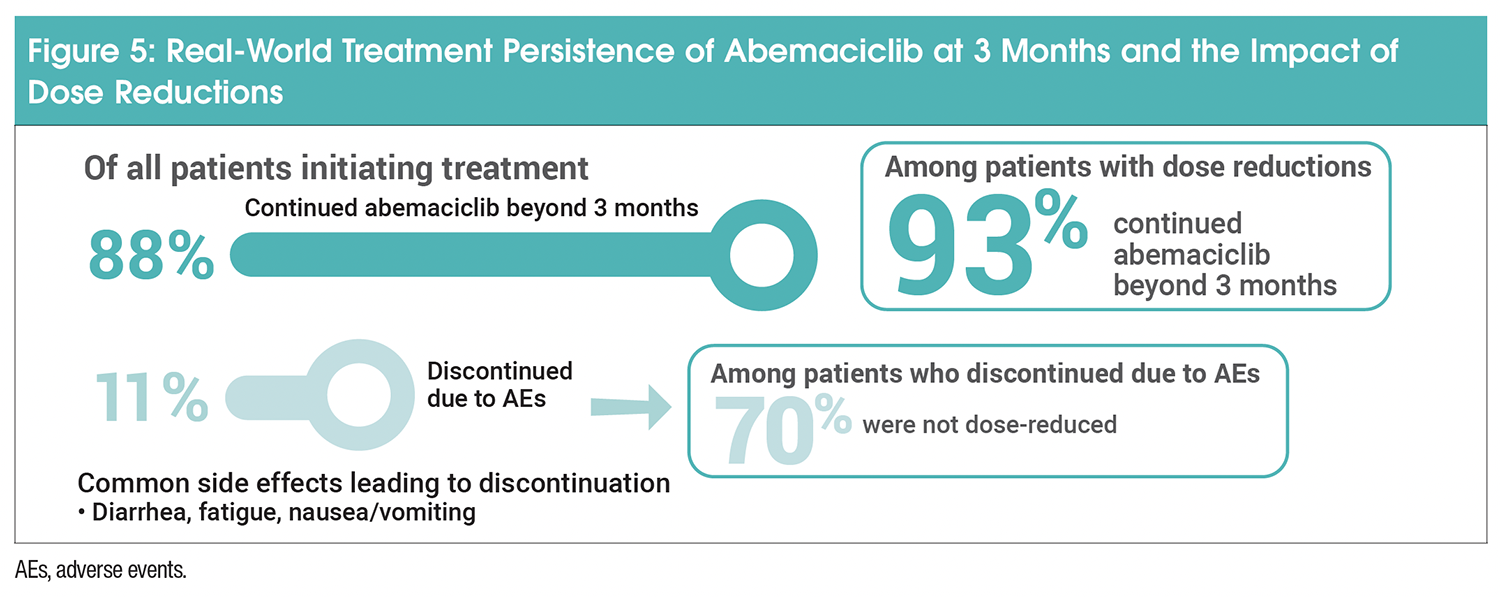
Moving into the real-world setting, a study using 354 patients from the US Flatiron Health database demonstrated that half of patients initiating the full 150-mg dose of abemaciclib had at least 1 dose reduction with a median time to the first dose reduction of 59 days.18 Of note, 88% of patients initiating treatment continued abemaciclib beyond 3 months, whereas for 11% of patients who discontinued treatment due to an AE, 70% were not dose-reduced (Figure 5). Similarly, the majority of patients (75%) continued abemaciclib beyond 6 months.19 More patients (85%) with ≥1 dose reduction compared with patients with no dose reduction (64%) continued abemaciclib beyond 6 months. Among patients who discontinued by 6 months, 70% did not have a dose reduction. Abemaciclib treatment persistence in patients meeting monarchE high-risk criteria (cohort 1) was consistent with that in the overall study population. The proportion of patients who discontinued due to an AE within the first 90 days was 4 times lower for those with ≥1 dose reduction versus those remaining at full dose (5.6% vs 22.4%).19
Commentary by Seth Wander, MD, PhD: In both the metastatic and in the adjuvant setting, data related to the impact of dose reduction is very important for clinicians and patients. We have reliable data from monarchE suggesting that patients who need to pursue dose reduction do not experience significant changes in clinical benefit (measured in IDFS rates). These insights give clinicians and patients more confidence to pursue dose reduction to improve longer-term adherence. In the optimal scenario, we would like to utilize the treatment at the standard dose; for the optimal duration, though, I think we should have a relatively low threshold, when necessary, to reduce the dose to improve persistence and adherence over the full 2-year course.
Given the high rate of dose reductions, the potential impact of dose adjustments and step-up dosing with abemaciclib was investigated. The phase 2 TRADE study assessed the impact of step-wise abemaciclib escalation (50 mg BID for 2 weeks, 100 mg BID for 2 weeks, and 150 mg BID thereafter) on composite AE rates, including discontinuation or inability to maintain the target dose. This dosing strategy resulted in 70.8% of patients reaching the 150-mg dose and maintaining this dose for up to 12 weeks. This exceeds the number of patients in monarchE continuing at the full target dose of abemaciclib at 12 weeks.
Commentary by Seth Wander, MD, PhD: Most patients who will experience significant toxicity on abemaciclib, particularly with diarrhea, will note that these symptoms often peak within the first 4 to 8 weeks on treatment. The TRADE study suggests a significant improvement in the number of patients who can get to the target dose and stay on treatment if you pursue this step-up dosing approach. This will likely provide more confidence for clinicians who would like to consider this approach to minimize toxicity and improve longer-term adherence. This was a very practical and important study to aid clinical management for this large patient population.
Conclusions
These long-term results highlight the importance of staying on abemaciclib therapy. Dose-reducing as indicated by treatment experience or employing a dose-escalation strategy may aid in AE management and treatment persistence. At the 7-year mark, the IDFS benefit was maintained, and DRFS rates and OS were both higher for abemaciclib in combination with ET compared with ET alone. This reinforces a critical role for CDK4/6 inhibitors, such as abemaciclib, combined with ET in the adjuvant treatment of high-risk, node-positive, HR-positive/HER2-negative early breast cancer as demonstrated by the ability to reduce the risk of recurrence and the first statistically significant difference in OS reported for adjuvant CDK4/6 inhibitor treatment.
Commentary by Seth Wander, MD, PhD: I think the 7-year follow-up data for monarchE is very important for all breast cancer clinicians. It’s the longest follow-up we have for any early-stage deployment of CDK4/6 inhibitors. This data, along with the dose-reduction and escalation studies that we’ve been discussing, greatly informs our understanding of how to deploy this drug in this high-risk patient population. The subgroup analyses from the adjuvant CDK4/6 trials suggest that there’s benefit across these broad and diverse patient populations. We want to engage all of our partners, including nursing, pharmacy, and nutrition, to help our patients and improve the overall treatment experience. This multidisciplinary team approach has the potential to optimize symptom burden and quality of life, enhancing adherence and, in turn, improving overall recurrence risk and survival.
References
- NIH SEER. Cancer Stat Facts: Female Breast Cancer Subtypes. Accessed December 5, 2025. https://seer.cancer.gov/statfacts/html/breastsubtypes. html
- Salvo EM, Ramirez AO, Cueto J, et al. Risk of recurrence among patients with HR-positive, HER2-negative, early breast cancer receiving adjuvant endocrine therapy: a systematic review and meta-analysis. Breast. 2021;57:5-17.
- VERZENIO [prescribing information]. 2021. Lilly USA, LLC; Indianapolis, IN.
- VERZENIO [prescribing information]. 2025. Lilly USA, LLC; Indianapolis, IN.
- Bekes I, Huober J. Extended adjuvant endocrine therapy in early breast cancer patients-review and perspectives. Cancers (Basel). 2023;15:4190.
- NIH. Hormone Therapy for Breast Cancer. January 26, 2026. Accessed February 25, 2026. www.cancer.gov/types/breast/treatment/hormone-therapy
- Early Breast Cancer Trialists’ Collaborative Group, Davies C, Godwin J, et al. Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet. 2011;378:771-784.
- Aromatase inhibitors versus tamoxifen in early breast cancer: patient-level meta-analysis of the randomised trials. Lancet. 2015;386:1341-1352.
- Braybrooke J, Bradley R, Hills RK, et al. Extending the duration of endocrine treatment for early breast cancer: patient-level meta-analysis of 12 randomised trials of aromatase inhibitors in 22,031 postmenopausal women already treated with at least 5 years of endocrine therapy. Lancet. 2025;406:603-614.
- Francis PA, Fleming GF, Láng I, et al. Adjuvant endocrine therapy in premenopausal breast cancer: 12-year results from SOFT. J Clin Oncol. 2023;41:1370-1375.
- Rugo HS, et al. Presented at: ESMO Breast Cancer Congress. May 14-17, 2025; Munich, Germany. Poster 215P.
- Bianchini G, Balko JM, Mayer IA, et al. Triple-negative breast cancer: challenges and opportunities of a heterogeneous disease. Nat Rev Clin Oncol. 2016;13:674-690.
- Johnston S, Martin M, O’Shaughnessy J, et al. Overall survival with abemaciclib in early breast cancer. Ann Oncol. 2026;37:155-165.
- Johnston SR, Martin M, O’Shaughnessy J, et al. monarchE: primary overall survival (OS) results of adjuvant abemaciclib + endocrine therapy (ET) for HR+, HER2-, high-risk early breast cancer (EBC). Presented at: ESMO Congress 2025. October 17, 2025; Berlin, Germany. Abstract LBA13.
- Johnston SRD, Toi M, O’Shaughnessy J, et al. Abemaciclib plus endocrine therapy for hormone receptor-positive, HER2-negative, node-positive, high-risk early breast cancer (monarchE): results from a preplanned interim analysis of a randomised, open-label, phase 3 trial. Lancet Oncol. 2023;24:77-90.
- Protocol in: Johnson RD, Harbeck N, Hegg N, et al. Abemaciclib combined with endocrine therapy for the adjuvant treatment of HR+, HER−, node-positive, high-risk, early breast cancer (monarchE). J Clin Oncol. 2020;38:3987-3998.
- Goetz MP, Cicin I, Testa L, et al. Impact of dose reductions on adjuvant abemaciclib efficacy for patients with high-risk early breast cancer: analyses from the monarchE study. NPJ Breast Cancer. 2024;10:34.
- Hudson K, Gathirua-Mwangi W, Cui ZL, et al. Clinical characteristics and treatment persistence in US patients with HR+/HER2-, node positive early breast cancer treated with abemaciclib: real-world study from first year after approval. Presented at: San Antonio Breast Cancer Symposium. December 10-14, 2024; San Antonio, TX. Poster P1-11-29.
- Soliman H, Dent S, Liepa AM, et al. Treatment persistence and dose modifications in US patients with HR+, HER2-, node-positive, early breast cancer treated with adjuvant abemaciclib. Presented at: San Antonio Breast Cancer Symposium. December 9-12, 2025; San Antonio, TX. Poster PS5-05-09.
For full prescribing information see:
www.accessdata.fda.gov/drugsatfda_docs/label/2025/208716s019lbl.pdf










Commentary by Seth Wander, MD, PhD: Endocrine treatment is the backbone of therapy for HR-positive/HER2-negative breast cancer. This is the fundamental building block of any treatment regimen—all of the other tools we utilize are critically important and convey significant benefits, but these are organized around a foundation of maintenance therapy with endocrine agents. This type of breast cancer is addicted to estrogen receptor signaling to survive and grow; by integrating optimal anti-estrogen therapy, we’re attempting to block signals that the cancer cells need to survive and recur.