Connecting Science to Practice
Pharmacist-led chemotherapy education is considered valuable,
but there are minimal objective data showing its impact.
In this pilot study, 20 patients starting chemotherapy
received education from a pharmacist accompanied by supplemental
materials from a printed handout or from a webpage.
Knowledge retention scores were similar between the
2 groups, with initial and follow-up survey scores remaining
similar across the groups. These observations suggest that
pharmacist-led chemotherapy education, with either of the
supplemental aids, may help patients retain important information
about adverse event management during the course
of their treatment.
Oncology pharmacists are well positioned to serve a unique role on the multidisciplinary care team. Their scope of practice has evolved from traditional dispensing duties to include clinical responsibilities, such as direct patient care, interdisciplinary support, and patient education.1 Chemotherapy education is a critical component of patient care. Pharmacist-led chemotherapy education (PLCE) improves medication adherence, alerts patients to identify potential drug interactions, and engages patients in anticipating and self-managing adverse events (AEs).1 This is particularly important in oncology, where treatments can be highly toxic and maintaining a balance between efficacy and quality of life is essential.
Pharmacists are also integral to the development and implementation of patient education tools.2,3 Satisfaction rates of patients and providers exceed 90% when pharmacists are actively involved in their care or practice.1 The value of pharmacists in multidisciplinary teams has been shown in a variety of practice settings, with studies highlighting their impact on medication adherence, supportive care management, and laboratory monitoring.1,4-8 Although some of these studies include pharmacist-led education as a component, the primary focus is often on clinical interventions and patient satisfaction rather than objective measures of patient knowledge or comprehension.9,10
Although a few studies have reported high patient satisfaction and self-reported understanding after pharmacist education,9-11 there remains a notable gap in the literature regarding the impact of PLCE. A systematic review of pharmacist interventions in the outpatient oncology setting identified counseling as a key component, with positive effects on nausea and vomiting control, patient satisfaction, and medication adherence.12 However, none of these studies objectively quantified how pharmacists’ interventions impacted patients’ knowledge regarding chemotherapy. More recent literature has explored pharmacists’ involvement in oral chemotherapy, although these studies similarly lack a focus on knowledge retention.13
Patient education modalities vary between institutions, ranging from audio or video recordings to one-on-one discussions with a healthcare provider, and each provides unique challenges and benefits.14 In one analysis, audio and video recordings provided benefit to patients with literacy issues, whereas education via a telephone call was not favored.14 Another study showed that patients found written supplemental information beneficial to their understanding, although they still expressed the need for improvement.15 A survey of more than 1000 patients with cancer revealed that they sought online educational materials for reasons including to obtain additional information (92%), because they had a lack of knowledge about what questions to ask (12%), insufficient information was provided by their clinic (11%), and they had limited time during visits to ask questions (5%).16 Despite the existence of multiple modalities to provide education, we continue to have room for improvement in our education tools. At The University of Kansas Cancer Center (KUCC), pharmacists aim to provide PLCE to all patients before the initiation of chemotherapy. To support this, institution-specific education materials have been created and curated by KUCC’s pharmacists.
During the COVID-19 pandemic, telehealth became a more prominent method of delivering education, highlighting the need for accessible and shareable resources. ChemoExperts is a web-based platform designed to meet this need by concisely presenting chemotherapy-related information in 3 easy-to-understand formats: text, audio, and video. This platform also allows patients to easily share educational materials with family, friends, and caregivers. As the pharmacist’s role continues to evolve, demonstrating the value of pharmacist-led education through measurable outcomes is critical. The ability to quantify knowledge gained after PLCE may further solidify the pharmacist’s role in oncology care and support the continued development of educational tools.
The initial period after a cancer diagnosis is often emotionally overwhelming, because patients and their support systems are inundated with complex information about treatments and disease management, which can lead to information overload and reduced recall of key concepts.17 The study team hypothesized that providing information in an accessible, written format alongside pharmacists’ education allows patients to revisit details discussed during education sessions throughout their course of treatment and aids in long-term knowledge retention related to chemotherapy.
Methods
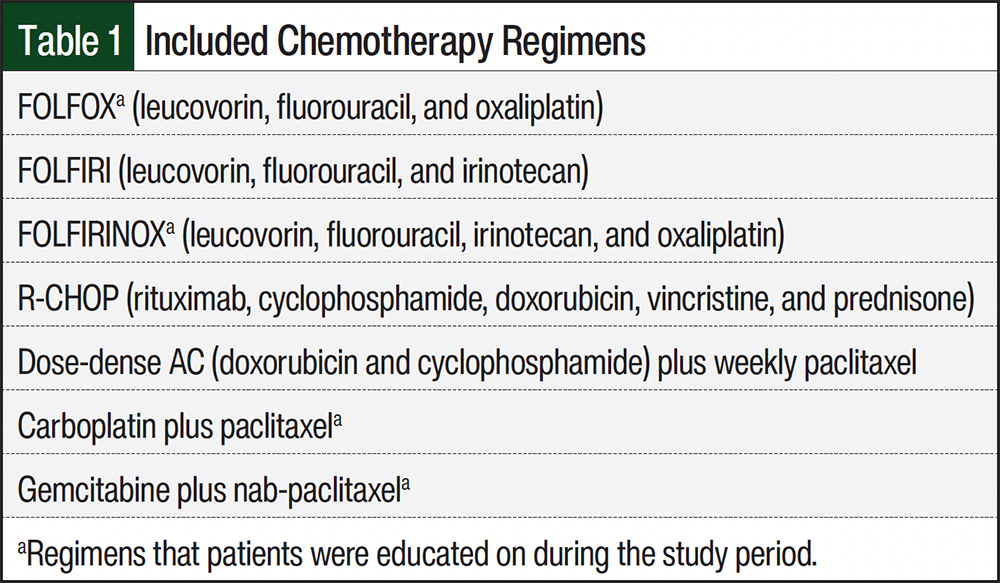
This was a single-center, prospective, survey-based pilot study of adults with cancer who planned to initiate 1 of 7 prespecified chemotherapy regimens in the outpatient setting between November 15, 2022, and April 28, 2023 (Table 1). This study was approved by The University of Kansas Medical Center Institutional Review Board. Written consent was obtained from each study participant before enrollment. The pharmacists conducting the education session screened the patients for eligibility.
The 7 prespecified regimens were selected because they represent first-line regimens in their respective disease states, thus making it likely that patients who initiated these treatments were treatment naïve. Patients were excluded from the study if their planned treatment regimen contained additional therapies, such as concurrent radiation, targeted therapies, oral chemotherapies, or monoclonal antibodies, to avoid confusion with overlapping AEs that would require separate education for management. An exception was made for R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone), because the study team wanted to capture patients from the lymphoma/myeloma clinic, where additive therapies are a standard of care for all lines of treatment. Patients were also excluded if they had previously received the same regimen, were non–English-speaking, or had a documented memory or learning deficit, unless a caregiver was present and was able to be their surrogate during the education session. Pharmacists used their professional judgment to exclude individuals who were emotionally overwhelmed or otherwise unable to retain information at the time of education.
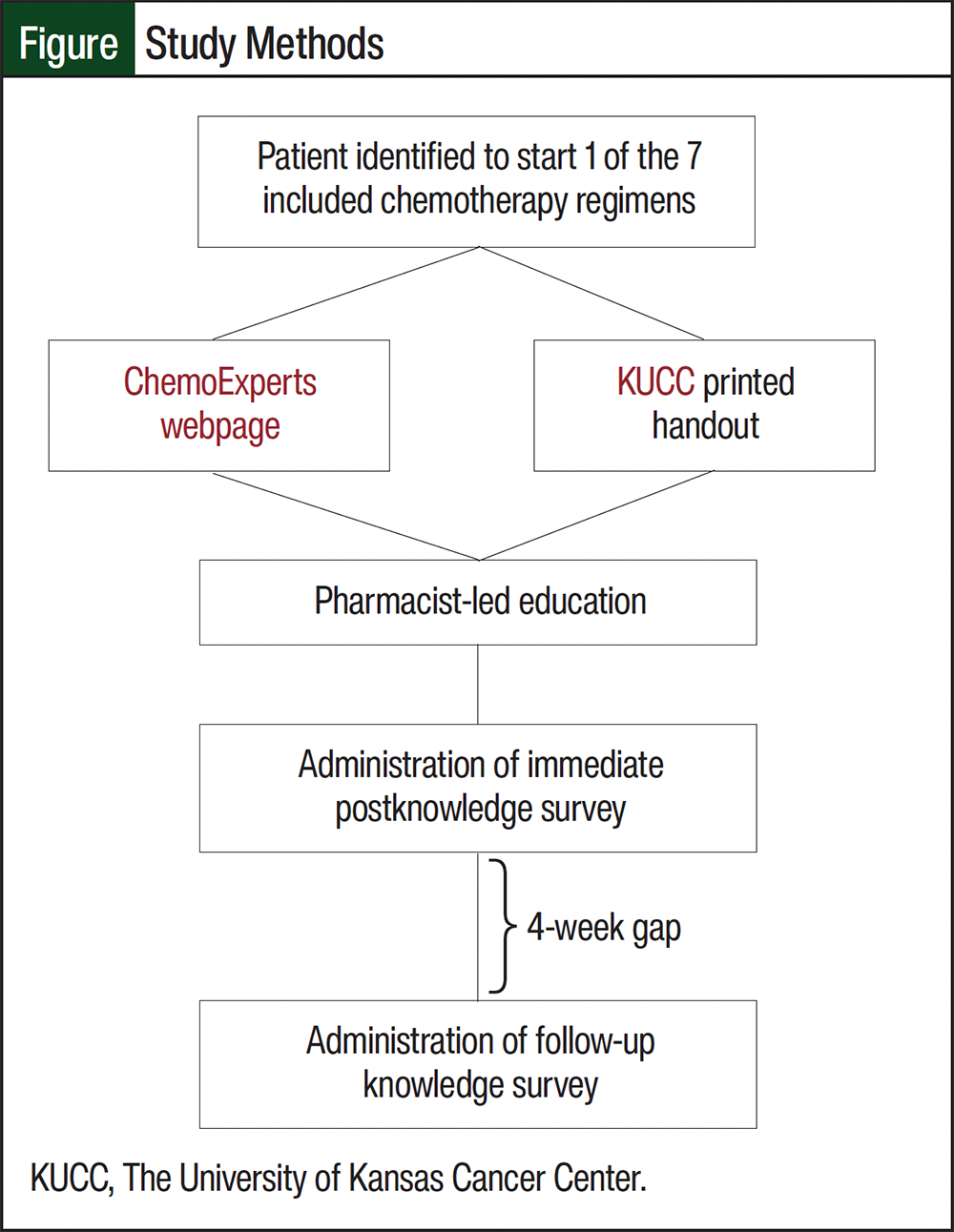
Patients who met the inclusion criteria were then consented and assigned one-to-one to receive pharmacist-led education with supplemental materials of an electronic webpage (ChemoExperts.org) or a KUCC printed handout (Figure). The consenting pharmacist enrolled patients on a spreadsheet, which sequentially assigned the patient a number of 1 through 20. Patients were assigned to the respective supplemental material arms by their enrollment number. Patients receiving an odd enrollment number were assigned to the ChemoExperts arm and patients with an even enrollment number were assigned to the printed handout arm.
Per our standard practice, patients at KUCC receive individual counseling from a pharmacist once their oncologist finalizes their treatment plan. Counseling sessions are comprised of a medication reconciliation, along with an overview of the treatment schedule, agents included in therapy, use of supportive care, anticipated AEs, and instructions of when to contact a physician or clinic. All the information the pharmacist reviews is provided to the patient in a written format. These handouts are created by disease-specific clinical oncology pharmacists by compiling information from tertiary resources and primary literature into patient-friendly language. These handouts are then validated by a second disease-state–specific pharmacist for correctness before use in patient education sessions.
To avoid variability between the educational aids for each arm, ChemoExperts customized webpages for each regimen to match the regimen schedule, supportive care, and clinic contact information provided on KUCC-printed handouts. Clinic pharmacists were educated on the study’s enrollment procedures and were oriented to the layout of the ChemoExperts webpages.
During the education, pharmacists used the assigned supplement material to guide chemotherapy counseling. Patients were able to view the supplemental material during the interaction, with the pharmacists referring to the written information as a guide. For the webpage group, patients were shown how to access the page on the clinic’s computer or on their personal device. These patients were also encouraged to explore the audio and video supplements after the completion of the visit. On completion of the education, the pharmacist provided the electronic survey to the patient to complete independently before the end of the visit. The educating pharmacists were blinded to the survey’s knowledge questions to avoid bias and variability from standard education practices.
The survey instrument consisted of 3 sections, including demographic questions, satisfaction questions, and knowledge-based questions (Appendix Table S1). The 4 satisfaction questions used a 3-point Likert scale. The 3-point scale was selected to reduce the burden on the respondents, to expedite survey completion in a clinical setting, and to enhance the clarity of the respondents’ interpretation by reducing response ambiguity.
The survey questions focused on satisfaction with the pharmacist’s education and the provided materials, as well as the perceived value of the pharmacist’s interaction. The knowledge-based questions were all multiple choice, allowing the participant to choose from 4 answers. Five questions were aimed at assessing a patient’s general chemotherapy knowledge, ranging from a chemotherapy’s mechanism of action to AE descriptions. Three additional questions specific to AEs related to the patient’s chemotherapy regimen were included. Regimen-specific questions were created in collaboration with clinical oncology pharmacists who are specialized in the disease state associated with the selected regimens. Because no validated surveys exist to assess chemotherapy knowledge retention in this setting, pharmacists who frequently conduct patient education on the selected regimens recommended the key points. Based on these recommendations, the study investigators developed survey questions to capture the patients’ knowledge of the key points.
The initial survey was administered via REDCap immediately after the education session. Utilizing REDCap’s survey function, a follow-up survey containing the same knowledge and satisfaction questions as the initial survey was scheduled to be sent to the study participants via email communication 4 weeks after education. If the participant did not fill out the follow-up survey at the 4-week mark, REDCap sent reminder emails at week 6 and week 8 after the education session. For patients who did not respond to the emailed survey reminders, clinic pharmacists were able to administer follow-up surveys to patients at subsequent provider or treatment visits within the 8-week follow-up window. Patients who did not complete the follow-up survey within this time frame were considered to be lost to follow-up.
At the end of each patient’s 8-week follow-up survey period, a chart review for the patient’s messages, telephone calls, and clinic encounters was conducted to identify the self-reporting of AEs. The data were collected on whether the patients self-reported any chemotherapy AEs for which they had been instructed to contact their care team.
Statistical Analysis
Data analysis was conducted using descriptive statistics to summarize the demographic information, immediate and follow-up knowledge survey scores, and patient satisfaction responses.
This pilot study was conducted to evaluate the feasibility of incorporating 2 modalities of supplemental education materials alongside pharmacist-led education sessions, followed by patient surveys. The objective data collected included patients’ knowledge retention immediately after pharmacist-led education supplemented with either electronic or printed materials, knowledge retention over time between the groups, overall patient satisfaction, and assessing the incidence of participants reporting self-identified chemotherapy AEs.
Results
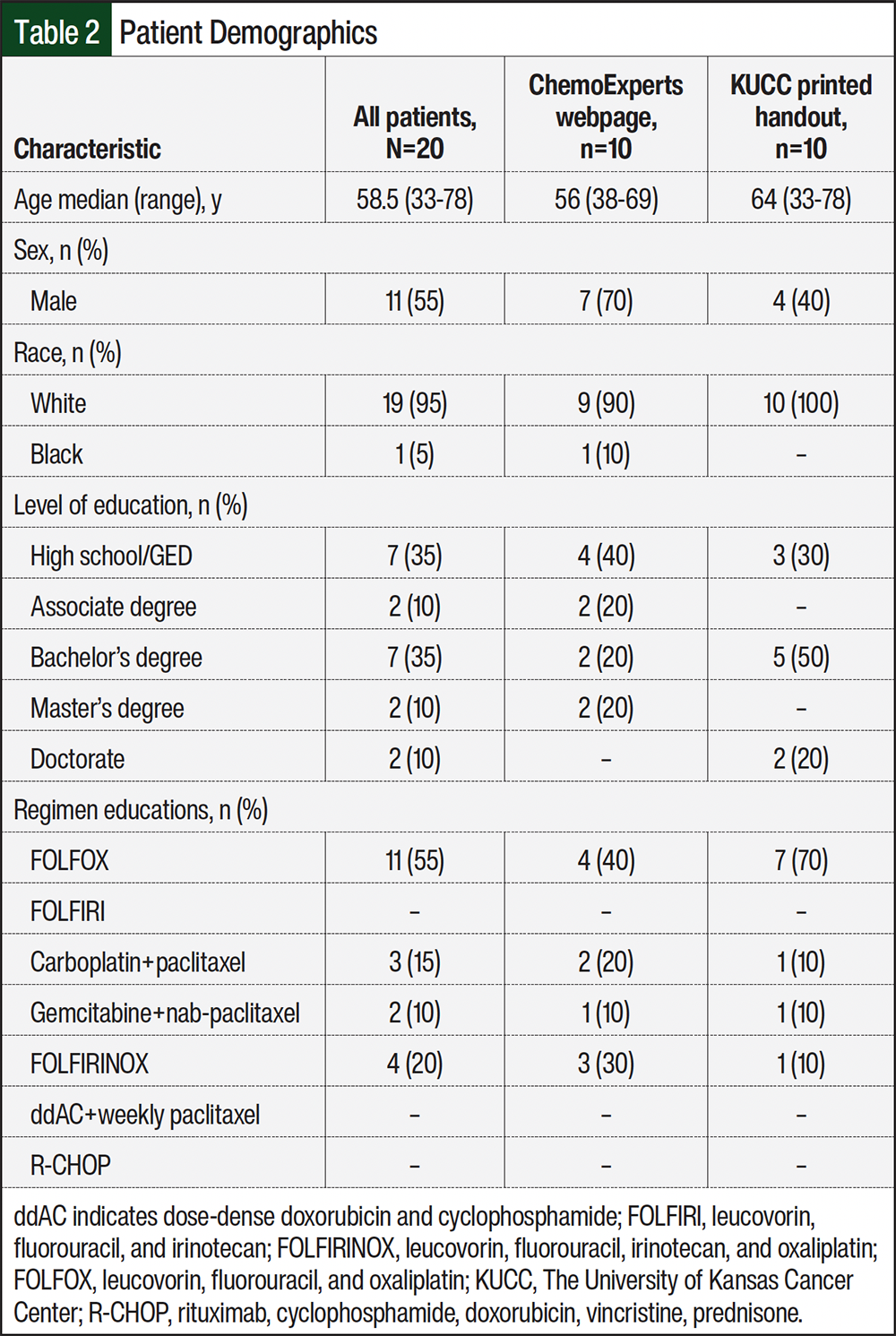
During the study period, 20 patients were enrolled and completed the initial survey. The median patient age was 58.5 years, with most (35%) patients completing a high school or GED-level education. Most (55%) patients received education on FOLFOX (leucovorin, fluorouracil, and oxaliplatin). The demographic and baseline characteristics are presented in Table 2.
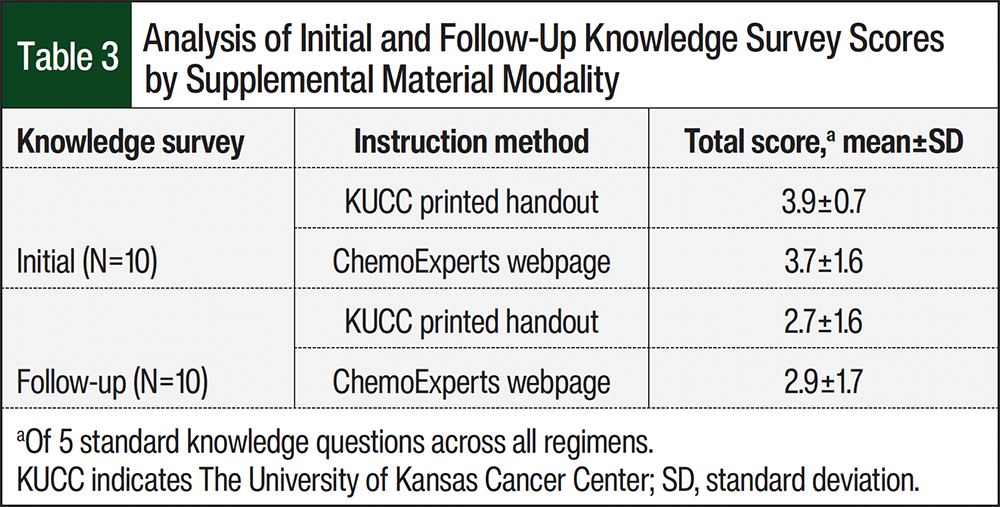
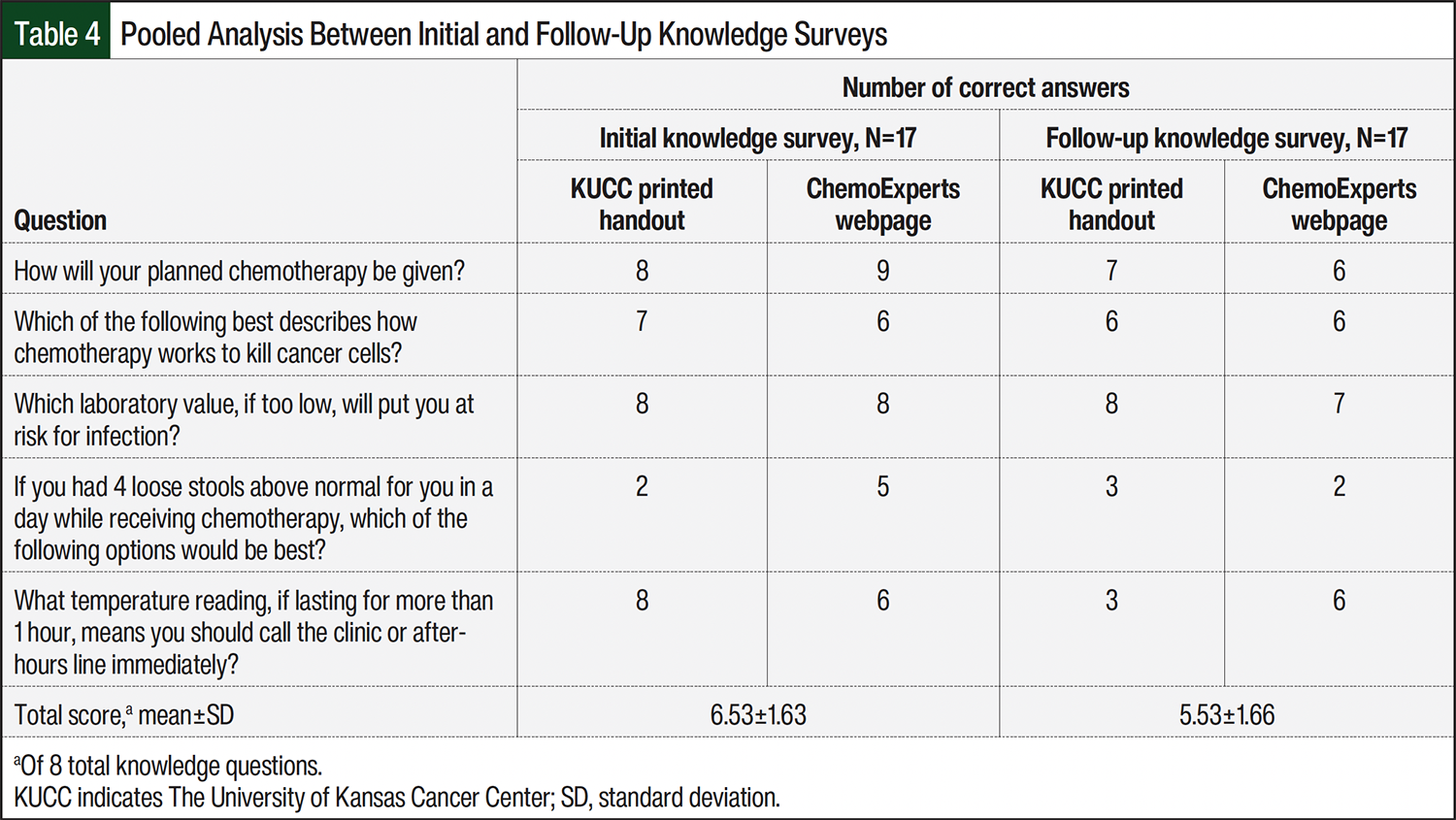
A total of 17 patients completed the initial and follow-up surveys. In all, 3 patients (1 from the webpage group and 2 from the printed handout group) did not complete the follow-up survey, as a result of 1 patient death and 2 patients being lost to follow-up. Based on the 5 knowledge questions common to all regimens, the average initial knowledge scores for the KUCC’s printed material group and the ChemoExperts webpage group were 3.9 and 3.7, respectively (Table 3). For these same groups, the average follow-up knowledge scores were 2.7 and 2.9, respectively (Table 3). The median total knowledge scores were 7 of 8 (87.5%) for both groups on the initial survey and 6 of 8 (75%) for both groups on the follow-up survey. When all of the survey results were pooled, the total average scores for the initial surveys and their coupled follow-up surveys were 6.53 ± 1.63 and 5.53 ± 1.66, respectively (Table 4).
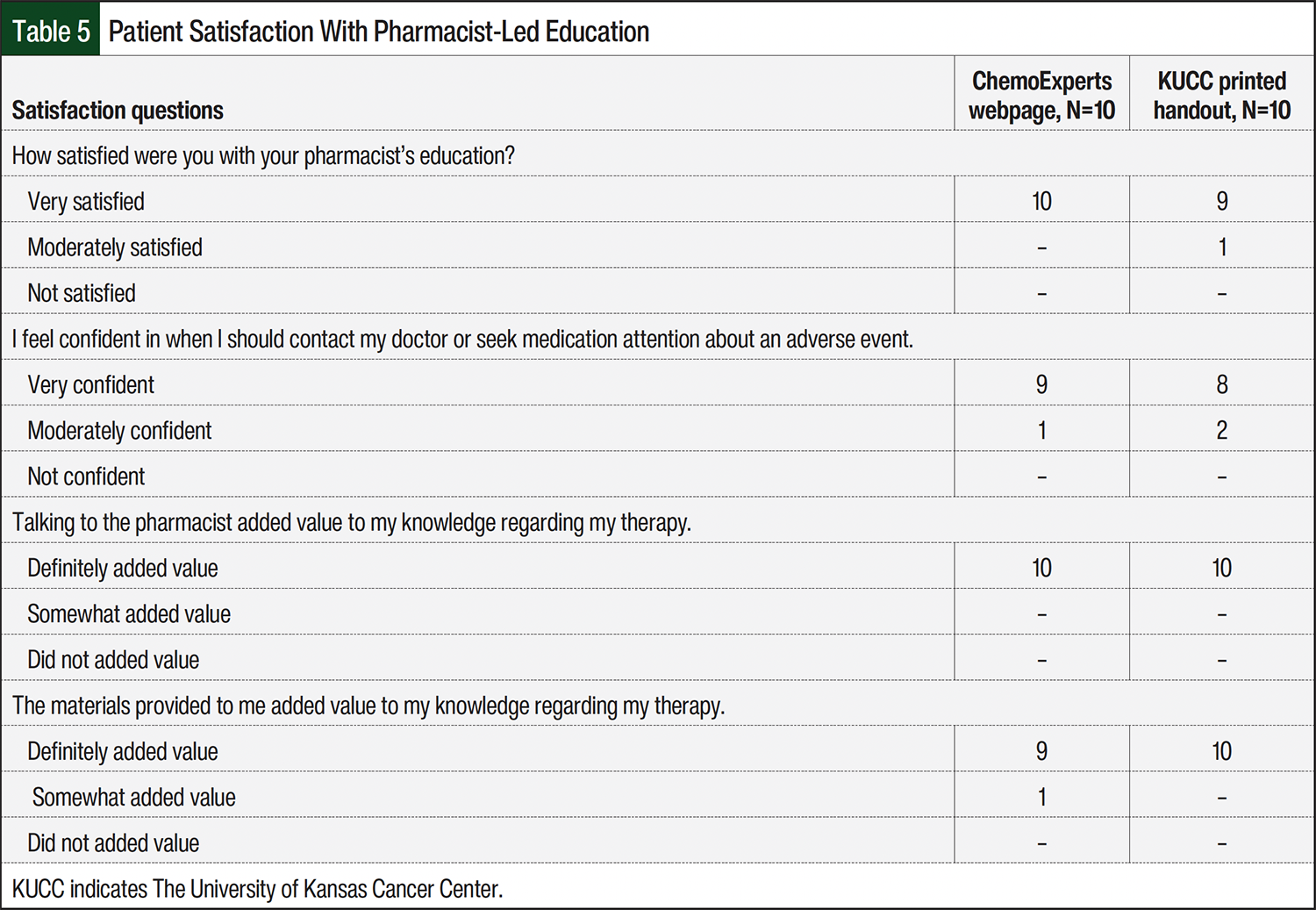
The satisfaction survey showed that 19 of 20 patients (95%) were very satisfied with the pharmacist’s education (Table 5). All patients felt that speaking with the pharmacists definitely added value to their knowledge, whereas 19 of 20 (95%) patients felt that the materials definitely added value. Of the 20 patients, 17 (85%) felt very confident about when they should contact their doctor or seek medical attention about an AE, whereas 3 of 20 patients felt moderately confident. A chart review of all 20 patients revealed that 50% of patients reached out to their respective clinic, via telephone or electronic health record message, to report an AE that was counseled on.
Discussion
To our knowledge, this pilot study represents the first prospective, survey-based evaluation exploring 2 different modalities of supplemental educational materials alongside verbal PLCE. Overall, the study procedures demonstrated feasibility, because the pharmacists were able to integrate the web-based supplement into their routine education sessions. The survey administration flowed seamlessly into the established workflow, and the patients were receptive to the method of survey administration. Together, these observations support the practicality of conducting a larger study of this nature.
Among the regimens that were analyzed, FOLFOX was the most common regimen that was educated about. This can be attributed to the large number of patients seen by the 6 providers in the gastrointestinal medical oncology clinic at KUCC. Pharmacists’ integration in patient education is well established in this clinic, with most educational encounters occurring in person.
When comparing the average score for the 5 common knowledge questions for the modalities of supplemental material and both time points, the observed differences between the groups were minimal. The median follow-up survey responses in both groups showed a slight decrease from the initial survey’s scores. There was also an observed difference between the pooled initial and follow-up survey scores. These findings suggest that although patients’ knowledge may decrease as the time from PLCE increases, a supplemental aid, irrespective of modality, may help patients retain knowledge for weeks after the education sessions. This is relevant to oncology patients, whose treatment and AE management can span several months to years.
When analyzing the answers to individual survey questions, the most frequent correctly answered question addressed how chemotherapy would be administered. In contrast, the most frequent incorrectly answered question focused on diarrhea management (Appendix Table S1). This question assessed whether patients knew how to appropriately manage diarrhea that occurred “≥4 times above the normal number of stools in a day.” Although most participants selected “take over-the-counter medications,” this response was not entirely incorrect, because patients should continue the use of antidiarrheal medications. Diarrhea that occurs ≥4 times above normal is classified as a Common Terminology Criteria for Adverse Events grade 2 AE and may warrant a reason for chemotherapy dose modifications. Thus, the most appropriate response in this scenario is to contact their healthcare provider, highlighting an important educational opportunity to reinforce self-management and the escalation of care. These results highlight a gap in patient understanding and suggest an area for improvement in patient counseling and the supplemental materials.
Notably, 50% of patients self-reported various AEs, which may reflect the patient awareness gained through PLCE. These findings underscore the importance of patient education in promoting active engagement in care. As such, it is the responsibility of the healthcare team to ensure that educational information is delivered in a clear, accessible, and patient-centered manner. The results from the satisfaction portion of the survey reinforce pharmacist-led chemotherapy education as a vital component of oncology care, enhancing patient understanding, engagement, and self-management. Integrating pharmacists into educational initiatives can enhance patient empowerment and promote safe, more-informed participation in treatment.
Limitations
We acknowledge several limitations affecting the accrual and external validity of this pilot study. First, the study’s procedures were not easily implemented into telehealth and telephone-based educations, which limited patients’ enrollment.
Second, regimens containing additive therapies, such as monoclonal antibodies, concurrent radiation, targeted therapy, and oral chemotherapies, were excluded to avoid confusion with overlapping AEs. As a result, enrolled patients may not be the most accurate snapshot of all current treatment and education practices within our institution.
Third, several patients declined enrollment because of a preference for printed handouts over webpage-based materials, which was often related to age or unfamiliarity with technology. This should be considered when recommending supplemental materials, because web-based resources may be more suitable for patients who are comfortable with technology and supported by geographically distanced caregivers.
We also acknowledge that the small number of participants in this pilot study limits the ability to draw firm conclusions between the 2 groups. Our enrollment was limited by project time constraints and a high number of excluded patients, as previously mentioned. Finally, given that pharmacists’ education practices may vary between institutions, our findings may not be generalizable to other cancer centers and their patient education workflows.
Conclusion
This pilot study demonstrated the feasibility of incorporating web-based versus printed supplemental materials alongside PLCE. Patients’ knowledge retention scores were similar between the 2 educational material groups, suggesting that either modality alongside pharmacist-provided education had a similar impact on patients’ chemotherapy knowledge. The knowledge scores were also similar between the initial and follow-up survey results, which suggests sustained patient understanding. The patients reported that pharmacist-directed education as part of their oncology interdisciplinary team led to high patient satisfaction and added value to their overall knowledge. Given the exploratory nature of this pilot study, larger studies are needed to confirm these findings and to further evaluate the impact on patients’ outcomes.
Acknowledgment
Contributions to this manuscript were provided by the ChemoExperts Foundation.
Author Disclosure Statement
Dr Mahmoudjafari is on the Advisory Boards of BMS, Genentech, Janssen, Pfizer, and Sanofi; Dr Betrabet, Dr Cannon, and Dr Cascone have no conflicts of interest to report.
References
- Segal EM, Bates J, Fleszar SL, et al. Demonstrating the value of the oncology pharmacist within the healthcare team. J Oncol Pharm Pract. 2019;25:1945-1967.
- Mackler E, Segal EM, Muluneh B, et al. 2018 Hematology/Oncology Pharmacist Association best practices for the management of oral oncolytic therapy: pharmacy practice standard. J Oncol Pract. 2019;15:e346-e355.
- Hematology/Oncology Pharmacy Association. Scope of hematology/oncology pharmacy practice. 2013. Accessed September 24, 2023. www.hoparx.org/documents/111/HOPA13_ScopeofPracticeBk1.pdf
- Mekonnen AB, McLachlan AJ, Brien JA. Effectiveness of pharmacist-led medication reconciliation programmes on clinical outcomes at hospital transitions: a systematic review and meta-analysis. BMJ Open. 2016;6:e010003.
- Milosavljevic A, Aspden T, Harrison J. Community pharmacist-led interventions and their impact on patients’ medication adherence and other health outcomes: a systematic review. Int J Pharm Pract. 2018;26:387-397.
- Jia X, Zhou S, Luo D, et al. Effect of pharmacist-led interventions on medication adherence and inhalation technique in adult patients with asthma or COPD: a systematic review and meta-analysis. J Clin Pharm Ther. 2020;45:904-917. doi:10.1111/jcpt.13126
- Presley B, Groot W, Pavlova M. Pharmacy-led interventions to improve medication adherence among adults with diabetes: a systematic review and meta-analysis. Res Social Adm Pharm. 2019;15:1057-1067.
- Thapa P, Lee SWH, Kc B, et al. Pharmacist-led intervention on chronic pain management: a systematic review and meta-analysis. Br J Clin Pharmacol. 2021;87:3028-3042.
- Muluneh B, Schneider M, Faso A, et al. Improved adherence rates and clinical outcomes of an integrated, closed-loop, pharmacist-led oral chemotherapy management program. J Oncol Pract. 2018;14:e324-e334.
- Crespo A, Tyszka M. Evaluating the patient-perceived impact of clinical pharmacy services and proactive follow-up care in an ambulatory chemotherapy unit. J Oncol Pharm Pract. 2017;23:243-248.
- McKee M, Frei BL, Garcia A, et al. Impact of clinical pharmacy services on patients in an outpatient chemotherapy academic clinic. J Oncol Pharm Pract. 2011;17:387-394.
- Colombo LRP, Aguiar PM, Lima TM, Storpirtis S. The effects of pharmacist interventions on adult outpatients with cancer: a systematic review. J Clin Pharm Ther. 2017;42:414-424.
- Dürr P, Schlichtig K, Kelz C, et al. The randomized AMBORA trial: impact of pharmacological/pharmaceutical care on medication safety and patient-reported outcomes during treatment with new oral anticancer agents. J Clin Oncol. 2021;39:1983-1994.
- Valenti RB. Chemotherapy education for patients with cancer: a literature review. Clin J Oncol Nurs. 2014;18:637-640.
- Villarreal-Garza C, Ferrigno AS, De la Garza-Ramos C, et al. Effect of receiving a customizable brochure on breast cancer patients’ knowledge about their diagnosis and treatment: a randomized clinical trial. Cancer Med. 2023;12:15612-15627. doi:10.1002/cam4.6215
- Gilreath, JA, Stuart, JA, Wilds B, et al. Patient and caregiver experience with an online, open-access, standardized, evidence-based, cancer drug and supportive care educational platform. Presented at: ASCO 2021. June 4, 2021; virtual. Abstract e18811.
- Jensen JD, Carcioppolo N, King AJ, et al. The cancer information overload (CIO) scale: establishing predictive and discriminant validity. Patient Educ Couns. 2014;94:90-96. doi: 10.1016/j.pec.2013.09.016