Connecting Science to Practice
The authors aimed to provide information on the regulatory
requirements imposed by all 50 US state boards of pharmacy
(BOPs) on out-of-state investigational drug service pharmacies
shipping investigational new drugs (INDs) into their jurisdiction.
Each state BOP was provided with a hypothetical
case, and BOP responses were collected to understand
whether nonresident pharmacy registration or permits are
required and if dispensing pharmacist or pharmacist-in-charge
licensure in the receiving state is needed for interstate shipment
of INDs.
It has been estimated that as many as 20% of clinical trials are either terminated early or fail to meet original recruitment targets.1 This does not come as a surprise, as participant recruitment and retention continue to be leading barriers to clinical trial success. A 2019 survey assessing engagement in clinical trials collected 12,451 responses inquiring about the most burdensome aspect of clinical trial participation.2 Survey results showed that respondents ranked traveling to the clinical trial site as the top burden.2 In the United States, most clinical trial sites are attached to large academic medical centers located in urban areas.3 This centralized approach to conducting clinical trials results in accessibility barriers for residents of rural areas who must travel far distances to participate.4 It is estimated that 70% of potential participants in the United States live more than 2 hours away from their nearest clinical trial site.4 Given the existing access gap, one emerging solution is the adoption of decentralized clinical trial (DCT) elements, including the shipment of direct-to-participant investigational new drugs (INDs), as part of the clinical trial design.4
The Evolution of DCTs and Delivery of Direct-to-Participant Shipments
The FDA defines a DCT as a clinical trial where some or all of the trial-related activities occur at locations other than traditional clinical trial sites.5 The use of DCT elements in clinical trial design is not a novel concept and can be traced back more than a decade. In 2014, Pfizer published results of the REMOTE trial, which was one of the earliest fully DCTs with no in-person interaction to incorporate direct-to-participant IND shipments.6 In December 2019, an industry standard research survey showed that DCT elements had been widely piloted by study sponsors. Notably, 70% of sponsors reported the use of direct-to-participant supply from sites or depots in at least 1 phase 3 trial in the past.7 More recently, the Coronavirus Disease 2019 (COVID-19) pandemic served as a major driver for the continued adoption of DCT elements in clinical trial conduct.8
During the height of the COVID-19 pandemic, direct-to-participant delivery became an essential DCT element to maintain participant retention in clinical trials.8 While COVID-19 was depleting health-system resources due to rising hospitalizations, restricting travel due to lockdown measures, and reducing access to clinical trial sites, the pandemic accelerated a considerable increase in DCTs. During the first year of the pandemic, 76% of pharmaceutical companies, device manufacturers, and contract research organizations reported use of DCT elements in their trial design.9 With this increased adoption, the regulatory framework surrounding interstate shipment of INDs in the United States became more relevant. As a result, a landslide of guidance documents were published by several US authorities supporting DCT adoptions in response to COVID-19.10-14
In early March 2020, the World Health Organization declared COVID-19 a pandemic, triggering a global disruption to standard clinical trial conduct.15 Soon after this declaration, the FDA published interim guidance documents that shaped immediate responses and discussed considerations for continuing active clinical trials.13,14 The guidance includes recommendations on the delivery of low-risk investigational products to participants’ homes and the FDA reporting requirements for ongoing trials that allow shipping INDs as an alternative delivery method.10 The core principles of the FDA draft guidance were focused on ensuring the safety of trial participants, protecting the trial integrity, and maintaining compliance with good clinical practice in conducting clinical trials. The FDA draft guidance highlighted safety considerations for direct-to-participant shipment of investigational products administered at home, such as implementing systems to maintain secure delivery, appropriate storage conditions, and product accountability measures.10
Simultaneously, yet independently, the Division of AIDS Clinical Research and Department of Health and Human Services (DHHS) published several memorandums updating shipping guidance for Cancer Therapy Evaluation Program (CTEP) INDs due to the COVID-19 pandemic.11,16 Before these memorandums, pharmaceutical management branch-supplied agents required the dispensing pharmacy to request authorization to allow shipment of the oral INDs directly from the site to the patients. In June 2020, the DHHS published an updated memorandum that extended the alteration to standard operating procedures through September 13, 2020, allowing sites to ship CTEP IND agents without a request for authorization from the Pharmaceutical Management Branch.11 In 2022, DHHS built on the 2020 memorandums and permanently adopted a policy allowing sites to ship CTEP INDs directly to patients as long as the elements of the policy were satisfied for each shipment.12 CTEP policy elements allowing direct-to-participant shipment include the following requirements: ensure that appropriately qualified shipping container systems are used, document the need for the shipment in the study participant’s medical record at the time the order is written, ensure proper labeling of the drug for patient use, ensure documentation of any patient counseling for CTEP INDs, and comply with any applicable state and federal regulations with respect to shipment of study agents.12
Investigational Drug Services (IDS) pharmacies are responsible for state and federal regulatory compliance when dispensing and shipping investigational agents.12 From a federal perspective, the FDA Code of Federal Regulations (CFR) Title 21, Section 312.1 jurisdiction states that “An investigational new drug for which an IND is in effect in accordance with this part is exempt from the premarketing approval requirements that are otherwise applicable and may be shipped lawfully for the purpose of conducting clinical investigations of that drug.”17 Nevertheless, IDS pharmacies must comply with state and federal law when shipping INDs across state lines. Previously, in 2014, Chelsey McIntyre, PharmD, published nationwide findings regarding state boards of pharmacy (BOPs) interpretations of interstate shipping laws for non-INDs that do not fall under the FDA CFR Title 21, Section 312.1 jurisdiction.18 To date, there has not been a nationwide assessment of state BOPs interpretations of interstate shipping laws for INDs. This study aims to fill that gap in the literature by providing information on the 50 US state-level BOPs regulatory requirements for interstate shipment of INDs dispensed by pharmacies.
Methods
To determine the regulatory requirements of each state, a case scenario was distributed to all 50 US BOPs in April 2024. The BOPs were contacted through publicly available email addresses of executive directors. Any failure-to-deliver notices or kickback messages required direct telephone communication with the state BOPs to request updated contact information for the state BOPs executive director. All example cases were distributed with the request that the case questions be answered by BOP personnel with insights on state BOP regulations within 3 weeks of initial contact. Email reminders were sent to BOPs personnel who had not responded to the initial case distribution 1 week before the requested deadline. One additional attempt to contact the BOPs was made if no responses were collected in the initial 3-week period. The email inbox was monitored for 6 weeks for response collection. A descriptive analysis including percentages and categorical counts was performed to summarize BOP responses. The intent of the hypothetical case distribution was to ascertain BOPs interpretations of their respective interstate shipping laws regarding INDs. The hypothetical case is shown in Table 1.
Results
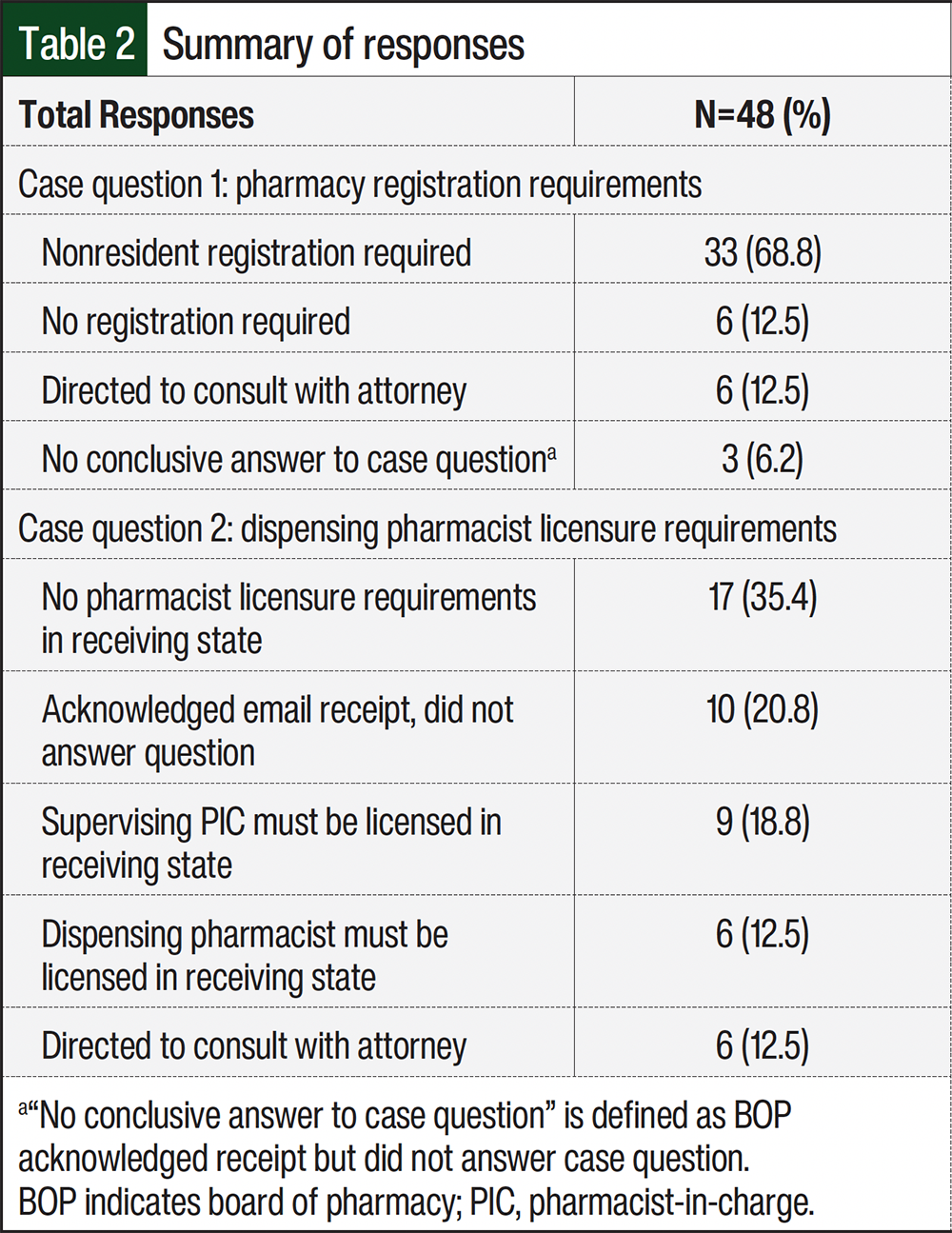
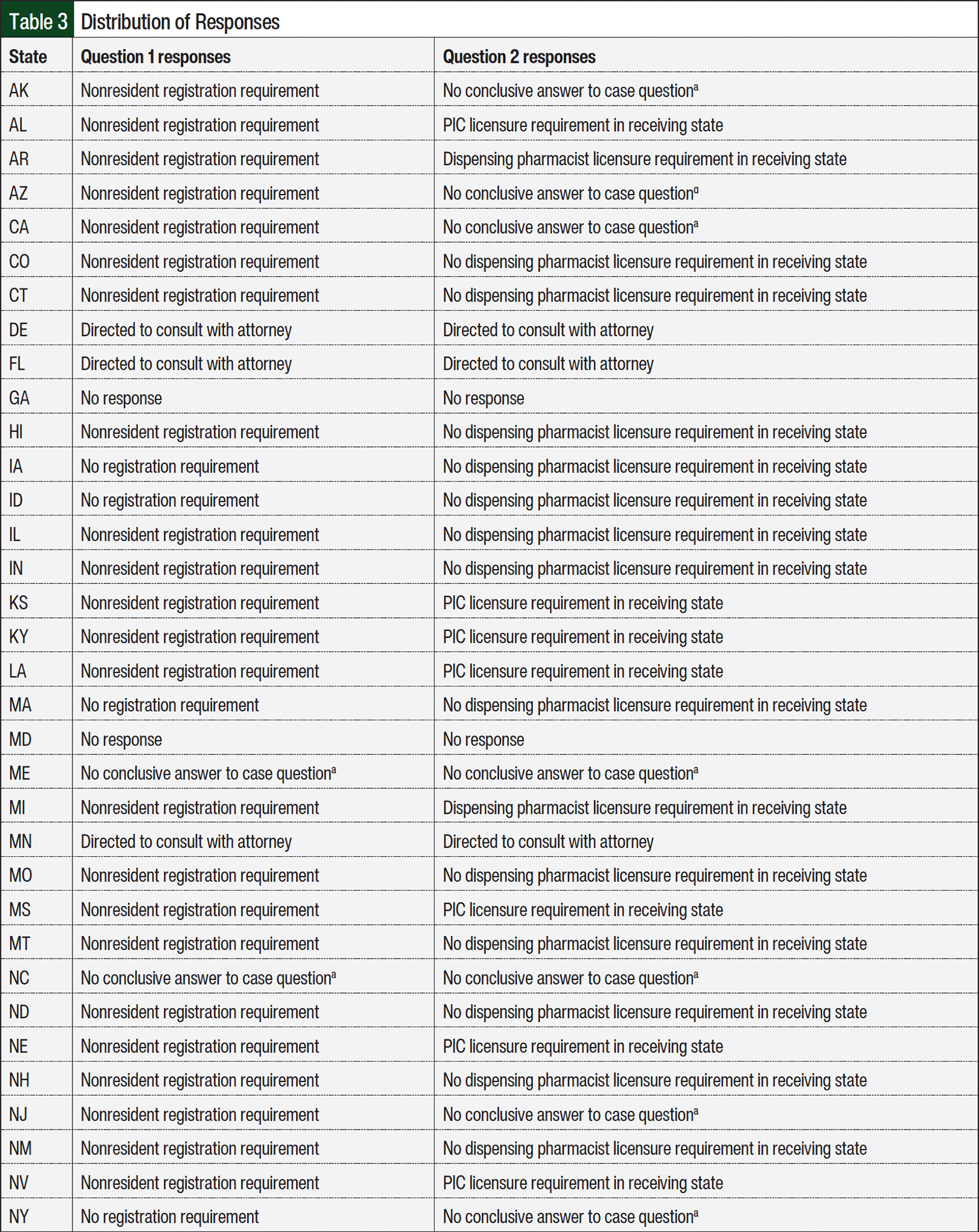
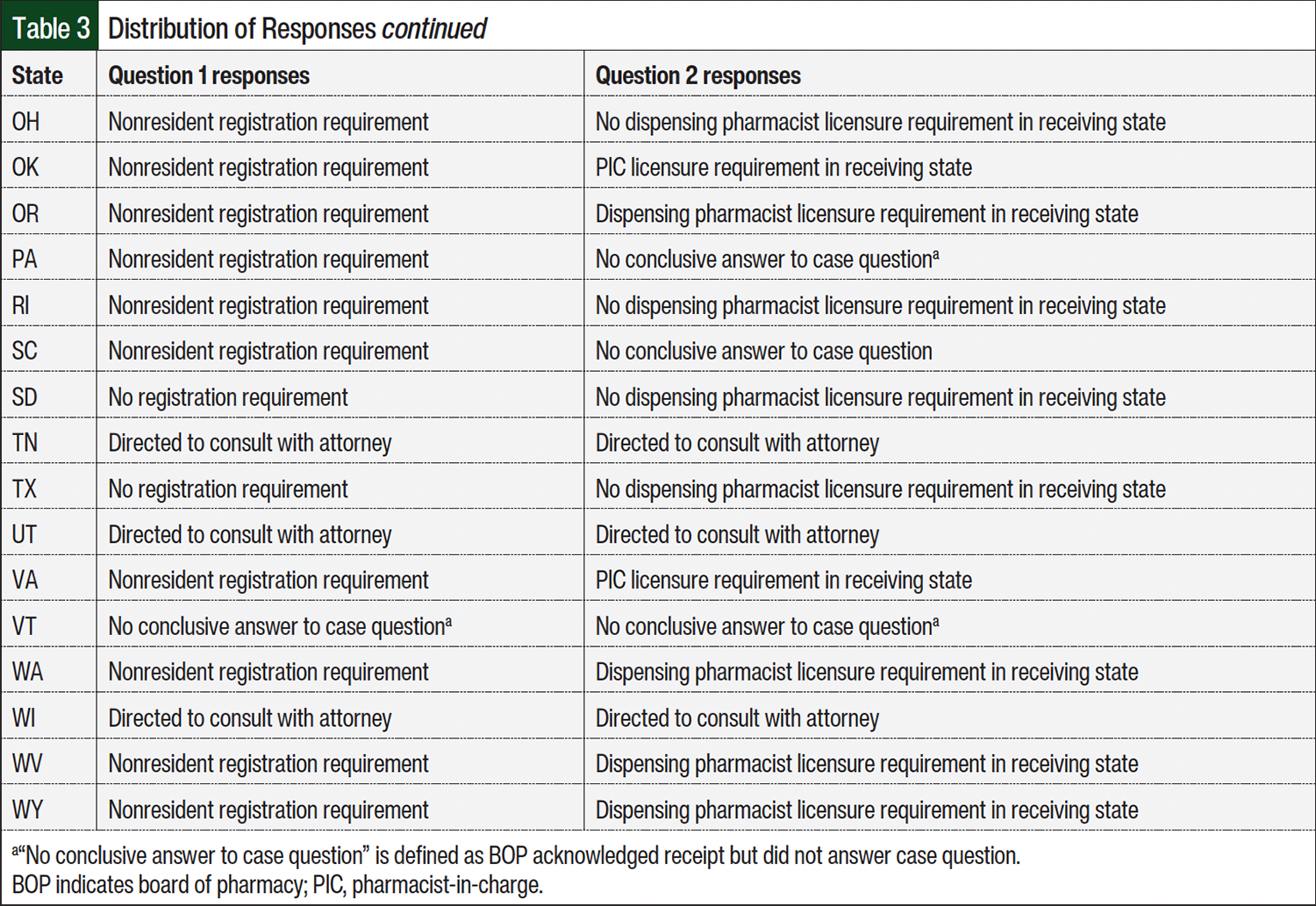
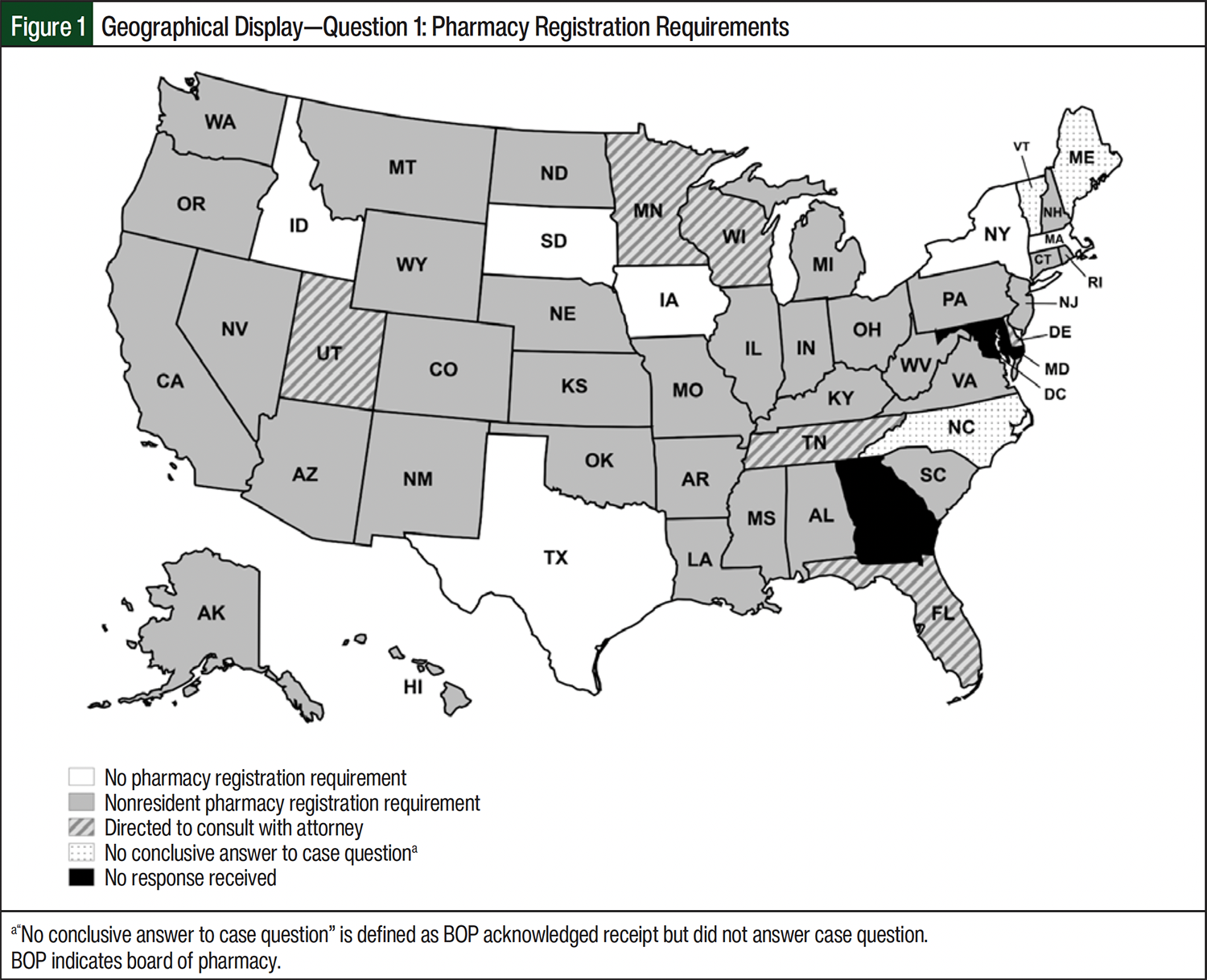
In all, 48 (96%) of the 50 BOPs responded during the 6-week response collection period (April 1, 2024, to May 13, 2024; Table 2). More than half of the responses, 29 (60.4%) of 48, were returned within the first 2 weeks of the response collection period. Table 3 provides a full distribution of responses collected from state BOPs during this study. As seen in Table 1, question 1 in the case asked BOPs about pharmacy registration requirements to ship an IND into their respective states. Pharmacy registration refers to state requirements for out-of-state pharmacies to obtain a permit or license to ship INDs into the state. Permitting requirements may differ from registration depending on state’s language in the law. More than two-thirds of responses from state BOPs, 33 (68.8%) of 48, indicated that pharmacy registration is required for out-of-state IDS pharmacies to register with the state before shipping INDs into the state. Of the responses indicating a pharmacy registration requirement, 17 (51.5%) of 33 state BOPs referred to a nonresident pharmacy license requirement in the registration process. In addition, 9 (27.3%) of 33 responses referred to registration via an application requirement. The remaining 7 (21.2%) of 33 responses referred to a nonresident pharmacy permit requirement before shipping INDs into the state. Only 6 (12.5%) of 48 state BOPs indicated that no pharmacy registration is required to ship INDs into the state. Of the remaining 9 (18.7%) of 48 responses, the state BOPs either recommended that the IDS pharmacy consult with an attorney (6 [12.5%] of 48) or did not provide an answer to scenario case question 1 (3 [6.2%] of 48).
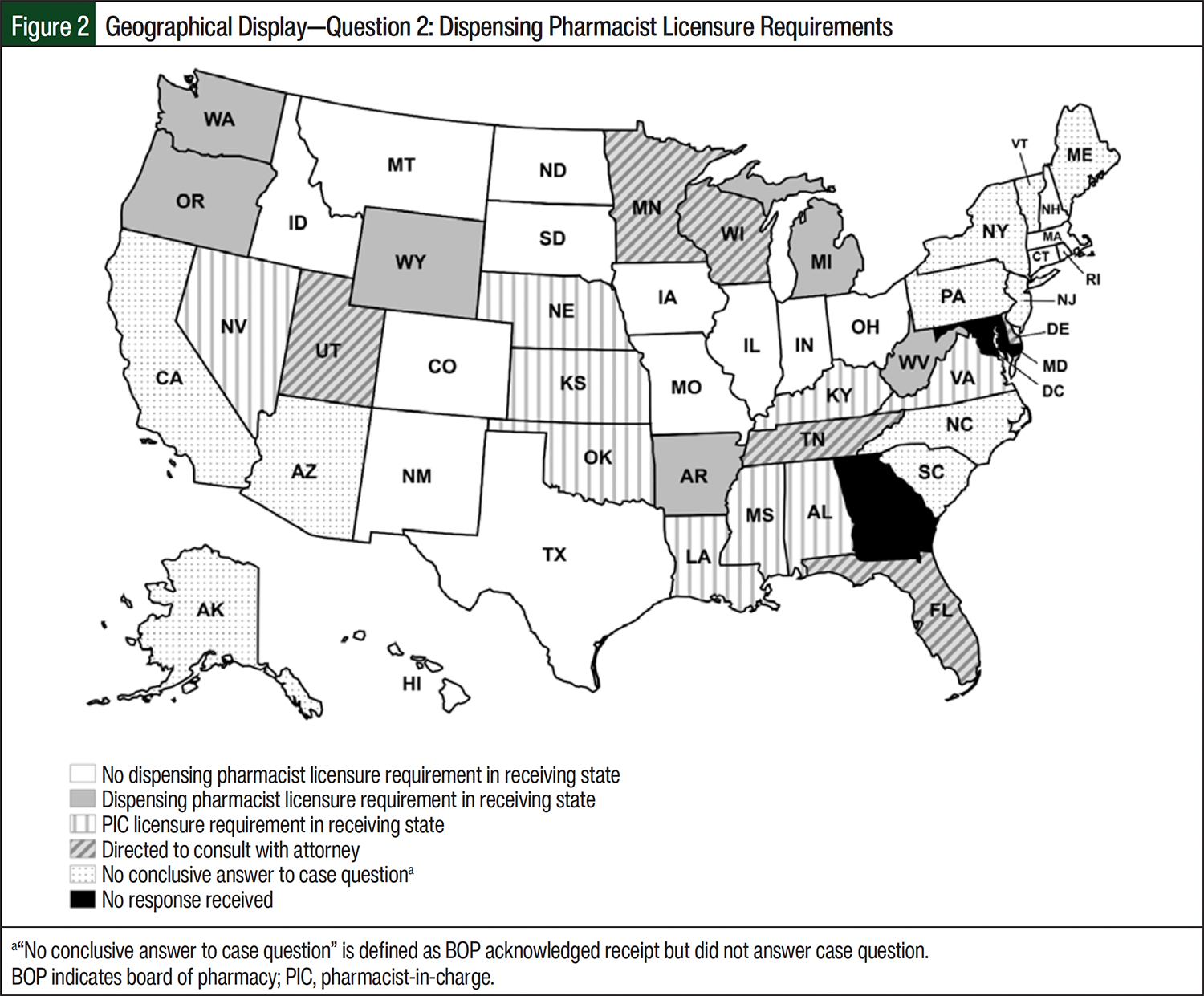
Question 2 inquired about the state BOPs dispensing pharmacist licensure requirements to ship into their respective states. Pharmacist licensure refers to the receiving state BOP requirement for the dispensing pharmacist or pharmacist-in-charge (PIC) to hold an active pharmacist license to practice pharmacy within the receiving state. Nearly one-third of state BOPs responses, 15 (31.3%) of 48, indicated pharmacist licensing requirements for the out-of-state pharmacist before shipping INDs into the state. Of those 15 responses, 9 (60%) of 15 BOPs required that the out-of-state PIC be licensed in the state to which the IND would be shipped. The other 6 (40%) of 15 BOPs required that the out-of-state dispensing pharmacist be licensed in the state to which the IND would be shipped. Seventeen (35.4%) of the 48 state BOPs responses indicated no requirements for pharmacist licensure to ship INDs into the state if the dispensing pharmacist held a license in good standing in the state where the IDS pharmacy was located. The remaining 16 (33.3%) of 48 BOPs either recommended that the IDS pharmacy consult with an attorney (6 [12.5%] of 48) or did not provide an answer to scenario case question 2 (10 [20.8%] of 48).
Discussion
This case was developed in response to the growing interest in direct-to-participant IND delivery as a part of DCT adoption. In the United States, pharmacies operate under their state-issued pharmacy licenses, and pharmacists are experts in complying with their own state laws governing the practice of pharmacy. Interstate shipment of INDs broadens the regulatory compliance that pharmacies must satisfy, as the receiving state BOP requirements must also be met when shipping INDs into the receiving state. An interpretation of CFR Title 21, Section 312.1 jurisdiction, which is held by many, is that INDs are exempt from state BOP oversight. Results from our investigation demonstrate that BOPs exercise jurisdiction over INDs pursuant to a prescription. Thus, a clear understanding of IND interstate shipment laws is an essential step in the adoption of this DCT element. As shown in Figure 1, state BOP regulations vary regarding compliance requirements, particularly surrounding nonresident pharmacy registration and nonresident permitting requirements to ship INDs across state lines.
Out-of-state IDS pharmacies must also comply with pharmacist licensure requirements to legally perform interstate shipment of INDs into other states (Figure 2). Individual dispensing pharmacist or PIC licensure requirements held by receiving state BOPs further complicate regulatory compliance. Each state BOP has its own licensure renewal process, costs associated with renewal, and timeline that must be met by pharmacists to maintain an active license to practice. Variations in state BOPs regulatory requirements may discourage IDS pharmacies from offering direct-to-participant services, which can risk regulatory noncompliance in clinical trial conduct.
The Hematology/Oncology Pharmacy Association (HOPA) has published IDS best practice standards that highlight the importance of establishing standard operating procedures for mailing investigational agents.19 IDS pharmacies without adequate procedures for interstate shipment of INDs are exposed to noncompliance risk if no additional research on specific state BOPs laws is performed. We attribute this potential noncompliance risk to the lack of information regarding best practices for shipping INDs and lack of regulatory uniformity across state BOPs regulatory requirements. To date, BOPs across the United States have failed to standardize requirements to ship INDs across state lines, despite regulatory compliance highlighted in the “Packaging and Shipping of Investigational Products” section of the FDA draft guidance for DCTs.20
The rise in DCT adoptions during the COVID-19 pandemic was a major first step in assessing the preparedness of DCT conduct. According to Clark and colleagues, new approaches for care delivery were established as a result of the pandemic.21 These new approaches redefined expectations for clinical trial conduct and the regulatory framework responsible for its advancement.21 An article by Finnes and colleagues with the HOPA IDS Special Interest Group developed recommendations that reinforced the comments made by Clark and colleagues regarding the advancements in patient care; in particular, the authors described best-practice approaches to the delivery of pharmacy services to patients receiving INDs during the pandemic.22 Similarly, Murphy described the effects of the pandemic on IDS workflows and the significant changes in trial operations resulting from exceptions made to previous prohibitions on shipping INDs, which signaled that direct-to-participant delivery is an acceptable method to manage patients remotely.23 In addition, Murphy strongly encouraged organizations and regional collaboratives to develop best practices for interstate shipping of INDs because currently none exist in the literature. More recently, the Society of Research Clinicians was developed to provide IDS pharmacies a premier community to establish and expand best practice within the IDS setting. Other groups, such as the Southwest Oncology Group Cancer Research Network, offer collaborative opportunities for IDS pharmacists to team up with research leaders interested in DCT adoptions. These professional organizations can serve as a unified voice in lobbying against legislative barriers that may limit clinical trial access.
Interstate shipment of INDs is one example of DCT element adoptions that may improve clinical trial participation. Other examples, such as remote consent, telehealth-supported drug accountability, and alternative delivery models, serve as new approaches that may be explored by IDS pharmacies to further improve access to clinical trials. The downstream benefits of adopting DCT elements may lead to increased accrual rates, decreased time required to complete clinical trial studies, and faster time to market for INDs. Furthermore, adopting DCT elements may also lead to decreased research and development cost and improved disease state representation in study patient populations.
Study Limitations
One limitation of the methods used for this study was the lack of continued dialogue with the BOPs for clarification and inquiry regarding the potential for waivers for related BOP fees or permission to solely follow FDA CFR Title 21, Section 312.1 for interstate shipment of INDs. Other limitations of this study are that the case was entirely hypothetical and did not address INDs dispensed in clinics, agents not dispensed under pharmacy license, or INDs that were not dispensed pursuant to a prescription. Given the nature of recent federal and state support for DCT adoption and the dynamic nature of pharmacy regulations, IDS pharmacies should continuously reference and evaluate state BOPs current guidance and regulations for any updated information on IND interstate shipping requirements. This report is not intended to be legal advice and captured information on state requirements at the time of the survey. To ensure that all current applicable laws have been considered, IDS pharmacies should consult with their own legal counsel before conducting direct-to-participant delivery. The information provided in this study is limited to 50 US states and does not include US districts or territories.
Conclusion
Regulations governing interstate shipment of INDs vary from state to state despite the 21 CFR guidance on interstate shipping for INDs. Although shipment of INDs is covered by federal regulations, there is a lack of standardized requirements at the state level for interstate shipment of INDs. Based on the breadth of insights demonstrated by this investigation, IND-specific regulatory language and statutes should be developed and published by BOPs to prevent discrepancies in interpretation. Furthermore, because IDS pharmacies practice under their respective state BOPs licenses, the authors encourage a relationship to be developed between the FDA and the National Association of Boards of Pharmacy. Given that states have more stringent regulations than those imposed at the federal level, this inconsistency in practice for INDs can lead to barriers for patient access to these novel agents.
Author Disclosure Statement
Dr Murphy is on the Speaker’s Bureau of HOPA, ASHP, and CME Horizon; Dr Dominguez, Dr Serna, and Dr Ataya have no conflicts of interest to report.
References
- Carlisle B, Kimmelman J, Ramsay T, MacKinnon N. Unsuccessful trial accrual and human subjects protections: an empirical analysis of recently closed trials. Clin Trials. 2015;12:77-83.
- Sine S, de Bruin A, Getz K. Patient engagement initiatives in clinical trials: recent trends and implications. Ther Innov Regul Sci. 2021;55:1059-1065.
- Ratcliffe M, Burd C, Holder K, Fields A. Defining rural at the U.S. Census Bureau. United States Census Bureau. December 2016. Accessed September 1, 2024. www.census.gov/content/dam/Census/library/publications/2016/acs/acsgeo-1.pdf
- Betcheva L, Kim JY, Erhun F, et al. Applying systems thinking to inform decentralized clinical trial planning and deployment. Ther Innov Regul Sci. 2023;57:1081-1098.
- Center for Drug Evaluation and Research. Conducting clinical trials with decentralized elements. US FDA. May 2, 2023. Accessed September 1, 2024. www.fda.gov/regulatory-information/search-fda-guidance-documents/decentralized-clinical-trials-drugs-biological-products-and-devices
- Orri M, Lipset CH, Jacobs BP, et al. Web-based trial to evaluate the efficacy and safety of tolterodine ER 4mg in participants with overactive bladder: REMOTE trial. Contemp Clin Trials. 2014;38:190-197.
- Agrawal G, Xue J, Moss R, et al. No place like home? Stepping up the decentralization of clinical trials. McKinsey & Company. June 10, 2021. Accessed September 1, 2024. www.mckinsey.com/industries/life-sciences/our-insights/no-place-like-home-stepping-up-the-decentralization-of-clinical-trials
- Castañeda R. Direct to patient: rocky road to remote drug delivery in clinical trials. Clinical Trials Arena. February 10, 2022. Accessed September 1, 2024. www.clinicaltrialsarena.com/features/direct-to-patient-the-rocky-road-to-remote-drug-delivery-in-clinical-trials/
- Oracle. The accelerated evolution of clinical trials in a pandemic environment. Research Report. November 2020. Accessed September 1, 2024. https://go.oracle.com/researchacceleratedtrials?elqCampaignId=257896
- US FDA. Considerations for the conduct of clinical trials of medical products during major disruptions due to disasters and public health emergencies; guidance for industry, investigators, and institutional review boards. September 2023. Accessed September 1, 2024. www.federalregister.gov/documents/2023/09/21/2023-20474/considerations-for-the-conduct-of-clinical-trials-of-medical-products-during-major-disruptions-due
- Department of Health & Human Services. Interim guidance for patients on clinical trials supported by the NCI Cancer Therapy Evaluation Program: shipment of oral IND agents to clinical trial subjects. June 2, 2020. Accessed September 1, 2024. www.swog.org/sites/default/files/docs/2020-06/CTEP%20Oral%20IND%20Agent%20Shipments.2-JUN-2020.pdf
- Department of Health & Human Services. Guidance for shipment of oral IND agents to clinical trial subjects on clinical trials sponsored by the National Cancer Institute Cancer Therapy Evaluation Program. January 1, 2022. Accessed September 1, 2024. https://dctd.cancer.gov/research/ctep-trials/for-sites/agent-management/oral-agent-shipment-policy.pdf
- US FDA. FDA Guidance on conduct of clinical trials of medical products during COVID-19 pandemic guidance for industry, investigators, and institutional review boards. April 2, 2022. Accessed January 7, 2026. www.hhs.gov/ohrp/sites/default/files/fda-covid-guidance-2apr2020.pdf
- US FDA. Conducting clinical trials during the COVID-19 public health emergency. April 30, 2020. Accessed January 7, 2026. www.fda.gov/media/ 137496/download
- Cucinotta D, Vanelli M. WHO declares COVID-19 a pandemic. Acta Biomed. 2020;91:157-160.
- Department of Health & Human Services; National Institutes of Health; National Institute of Allergy and Infectious Diseases, Division of AIDS. Coronavirus Disease 2019 (COVID-19) and DAIDS HIV/AIDS Network Clinical Research Studies. March 13, 2020. Accessed March 20, 2026. www.hptn.org/sites/default/files/inline-files/DAIDS%20COVID%20Guidance%2003132020.pdf
- Code of Federal Regulations Title 21. US Department of Health and Human Services. January 22, 2024. Revised April 1, 2024. Accessed September 1, 2024. www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfCFR/CFRSearch.cfm
- McIntyre C. Regulations guiding the interstate shipment of investigational product. J Pharm Pract. 2014;27:101-105.
- Amin SR, Avila JG, Boron MJ, et al. HOPA investigational drug service best practice standards. Hematology/Oncology Pharmacy Association. Revised July 20, 2018. Accessed September 1, 2024. www.hoparx.org/documents/109/HOPA16_IDS_Guidelines.reviewed_2018.pdf
- Department of Health & Human Services. Decentralized clinical trials for drugs, biological products, and devices: guidance for industry, investigators, and other stakeholders, draft guidance. May 2023. Accessed September 1, 2024. www.fda.gov/media/167696/download
- Clark E, Gordon J, Rao N, et al. Overcoming the cost of healthcare transformation through partnerships. McKinsey & Company. August 11, 2022. Accessed September 1, 2024. www.mckinsey.com/industries/healthcare/our-insights/overcoming-the-cost-of-healthcare-transformation-through-partnerships
- Finnes HD, Child B, DeFrates S, et al. Adapting investigational drug services during a pandemic: recommendations for future preparedness from the Hematology/Oncology Pharmacy Association Investigational Drug Services Special Interest Group. Am J Health Syst Pharm. 2022;80:e67-e73.
- Murphy J. Clinical trial management in the post-pandemic era: reflections of an investigational drug service on the past two years. Hematology/Oncology Pharmacy Association. 2022. Accessed September 1, 2024. http://archive.hoparx.org/images/hopa/advocacy/News/HOPANews_Vol19_Issue3-v7-4web.pdf