Connecting Science to Practice
Multiple myeloma (MM) is the most common hematologic
malignancy among Black patients in the United States and
Europe. Studies indicate significant disparities in outcomes
for Black patients, including higher mortality rates. Lenalidomide
is an important immunomodulatory agent frequently
used for newly diagnosed patients with MM but is often associated
with adverse events (AEs) that may lead to dose
modifications. Although subgroup analysis from the GRIFFIN
trial suggests higher rates of AEs in Black patients, data
comparing AEs by race are limited. Our study suggests that,
in real-world practice, Black race is not an independent risk
factor for lenalidomide-related AEs requiring dose modifications.
These results show that lenalidomide-based induction
regimens can be safely tolerated across races with close monitoring
and patient education supporting treatment adherence.
In addition, it has the potential to improve outcomes.
Multiple myeloma (MM) is the second most prevalent hematologic malignancy in the United States with 36,110 new cases estimated in 2025.1,2 The incidence of MM is ≥2 times higher in Black patients, making it the most common hematologic malignancy in this race.3 This is likely a result of a higher prevalence of subclinical precursor states, such as monoclonal gammopathy of undetermined significance and smoldering MM, in Black individuals.4 Black patients are also twice as likely to die from the disease, and racial disparities have been reported across various stages of the disease.1,5 In addition, Black patients more frequently present with underlying anemia and worse renal dysfunction, which can impact treatment.6 These factors, combined with potential disparities in access to healthcare, contribute to the observed higher mortality rates in Black patients.6 However, recent evidence gathered in patients with MM suggests that Black and White patients have comparable outcomes when provided equal access to care.6,7 Our findings further support that race itself is not an independent risk factor for lenalidomide dose modifications, highlighting the importance of patient education and timely clinical intervention.
Lenalidomide is an oral second-generation immunomodulatory agent with potent anti-MM and anti-inflammatory effects. The anti-MM effect is multifactorial, including induction of apoptosis, reduction of cytokines, inhibition of angiogenesis, blocked binding of MM cells to the bone marrow stromal cells, and stimulation of host natural killer cell anti-MM immunity.8 It is a first-line, preferred agent for induction regimens for transplant-eligible patients and transplant-ineligible patients with newly diagnosed MM per the NCCN guidelines.9 The FDA-approved starting dose for lenalidomide in patients with MM is 25 mg on days 1 to 21 of a 28-day cycle.10 The medication is also used off-label at 25 mg on days 1 to 14 of a 21-day cycle.11-14 However, the use of lenalidomide is associated with a number of adverse effects (AEs) and must be reduced in some patients because of comorbidities or AEs.10 Some of the most common AEs requiring dose modifications include neutropenia, thrombocytopenia, and infection.10
Currently, published data describing AEs with lenalidomide by race are lacking; however, a post-hoc subgroup analysis from the phase 2 GRIFFIN study suggests that Black patients may have higher rates of neutropenia and possibly other AEs with lenalidomide, thus prompting more frequent dose reductions.13 The GRIFFIN study evaluated the addition of the anti-CD38 monoclonal antibody daratumumab (D) to lenalidomide, bortezomib, and dexamethasone (RVd) followed by autologous stem cell transplantation in patients with newly diagnosed MM.14 The most common grade 3/4 treatment-emergent AEs (TEAEs) in Black patients were neutropenia followed by lymphopenia.14
Patients who have lenalidomide-associated AEs may require additional pharmacologic management and treatment interruptions. To further evaluate the impact of race on lenalidomide dose modifications, we investigated the incidence of AEs in a real-world data set of Black patients compared with non-Black patients.
Patients and Methods
This single-center, retrospective study, approved by the institutional review board at Emory University and the Winship Cancer Institute, enrolled patients aged ≥18 years with newly diagnosed MM who were treated with lenalidomide as part of induction therapy during the period from January 2010 to June 2022. The inclusion criteria for the study also included patients with newly diagnosed MM, completion of ≥1 cycle of a lenalidomide-based induction regimen at Emory Healthcare, and lenalidomide dosage initiated at 25 mg daily. For this study, “1 cycle” is defined by the standard for the regimen, with duration of either 28 days or 21 days. Patients who received lenalidomide through clinical trials or outside of Emory Healthcare were excluded. Demographic information, including sex, self-reported race, MM type, and disease stage, was extracted from electronic medical records. Race was categorized as Black or non-Black. Additional demographic and clinical characteristics, including weight, lenalidomide dose and frequency, baseline and subsequent serum creatinine levels, platelet counts, absolute neutrophil counts, and dose modifications and interruptions, were also collected. Incidence of dose modifications was categorized as a binary variable and defined as an event occurring if ≥1 lenalidomide dose reduction or treatment interruption occurred during treatment due to AEs.
We identified 474 patients with newly diagnosed MM who were treated with lenalidomide-based induction regimens between the specified date range. After excluding patients who did not complete ≥1 cycle of lenalidomide, who were started on a lower dose, or who received lenalidomide in a clinical trial, 250 patients were included in our analysis.
We analyzed the incidence of dose modifications experienced by patients in the Black and non-Black groups. Descriptive statistics were used to summarize the demographic and clinical data. Association between variables of interest and the race cohort were examined using ANOVA for numerical covariates and chi-square test for categorical covariates. Univariate logistic regression models were used to identify individual prognostic factors for dose modification. A P value of <.05 was considered statistically significant for all statistical analyses. Statistical analysis was conducted using SAS version 9.4 (Cary, NC) and SAS macros developed by Biostatistics and Shared Resource at Winship Cancer Institute.15
Results
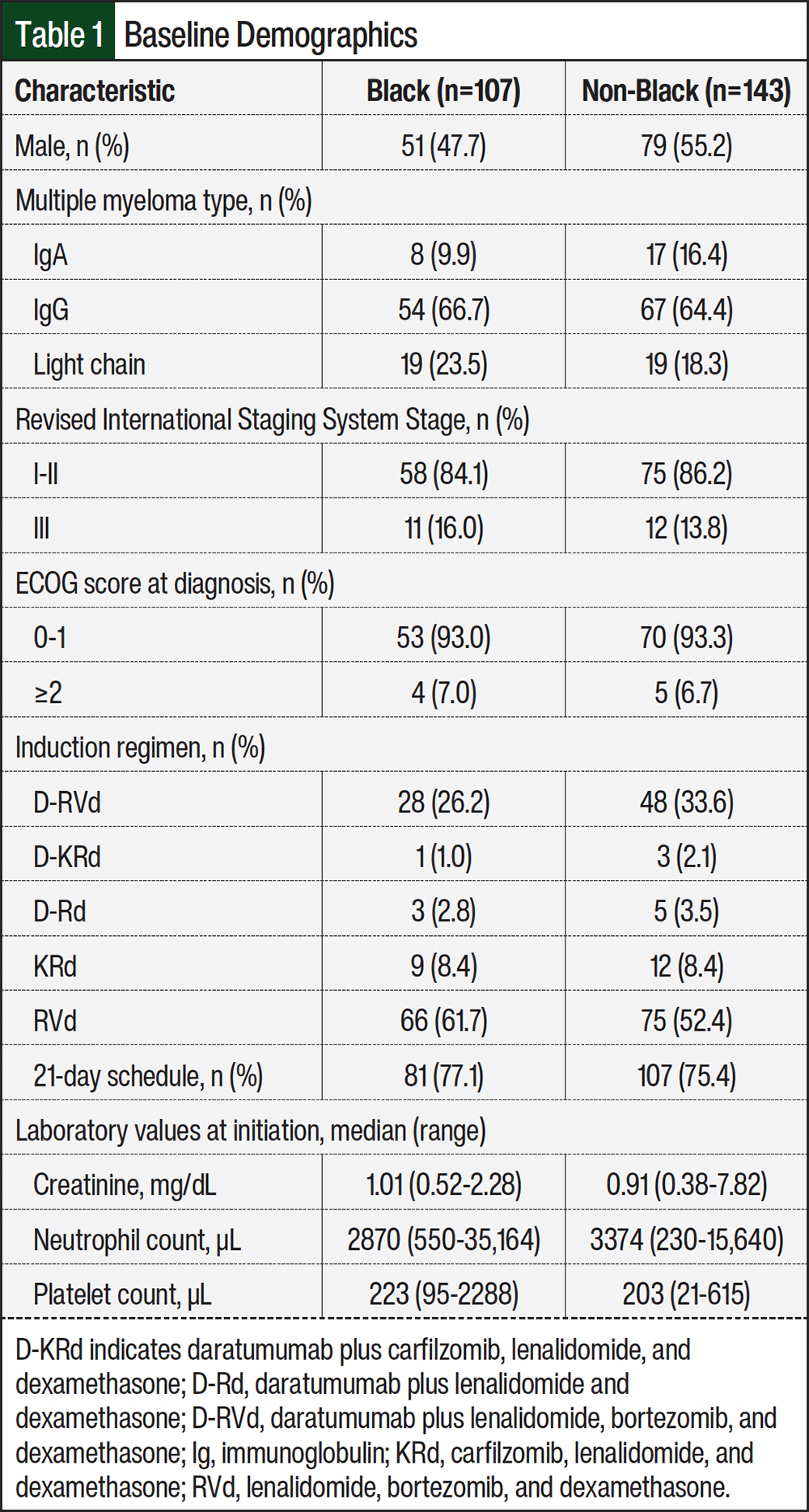
In all, 107 Black and 143 non-Black patients were identified and included in the analysis. Patients’ baseline characteristics are shown in Table 1 and are generally similar, except fewer Black men (47.7%) were included in the analysis compared with non-Black men (55.2%). Similar proportions of Black and non-Black patients had an ECOG score >1 and revised International Staging System stage III disease. The most common induction regimen was RVd (Black patients, 61.7%; non-Black patients, 52.4%), followed by daratumumab, lenalidomide, bortezomib, and dexamethasone (D-RVd; Black patients, 26.2%; non-Black patients, 33.6%). Most patients were initiated on 21-day cycles with 14 days of lenalidomide (Black patients, 77.1%; non-Black patients, 75.4%). This is consistent with guideline recommendations, population-based studies, and the demographics of the newly diagnosed MM population at Emory Healthcare.
Overall, 52 (20.8%) patients required dose modification during induction therapy with lenalidomide, including 32 (12.8%) patients requiring ≥1 dose reduction and 34 (13.6%) patients requiring ≥1 treatment interruption. The incidences of dose reductions, therapy interruptions, and dose modifications were similar in the Black and non-Black groups (Table 2). Most patients (77.4%) who required dose reductions completed induction therapy with lenalidomide 15 mg daily.
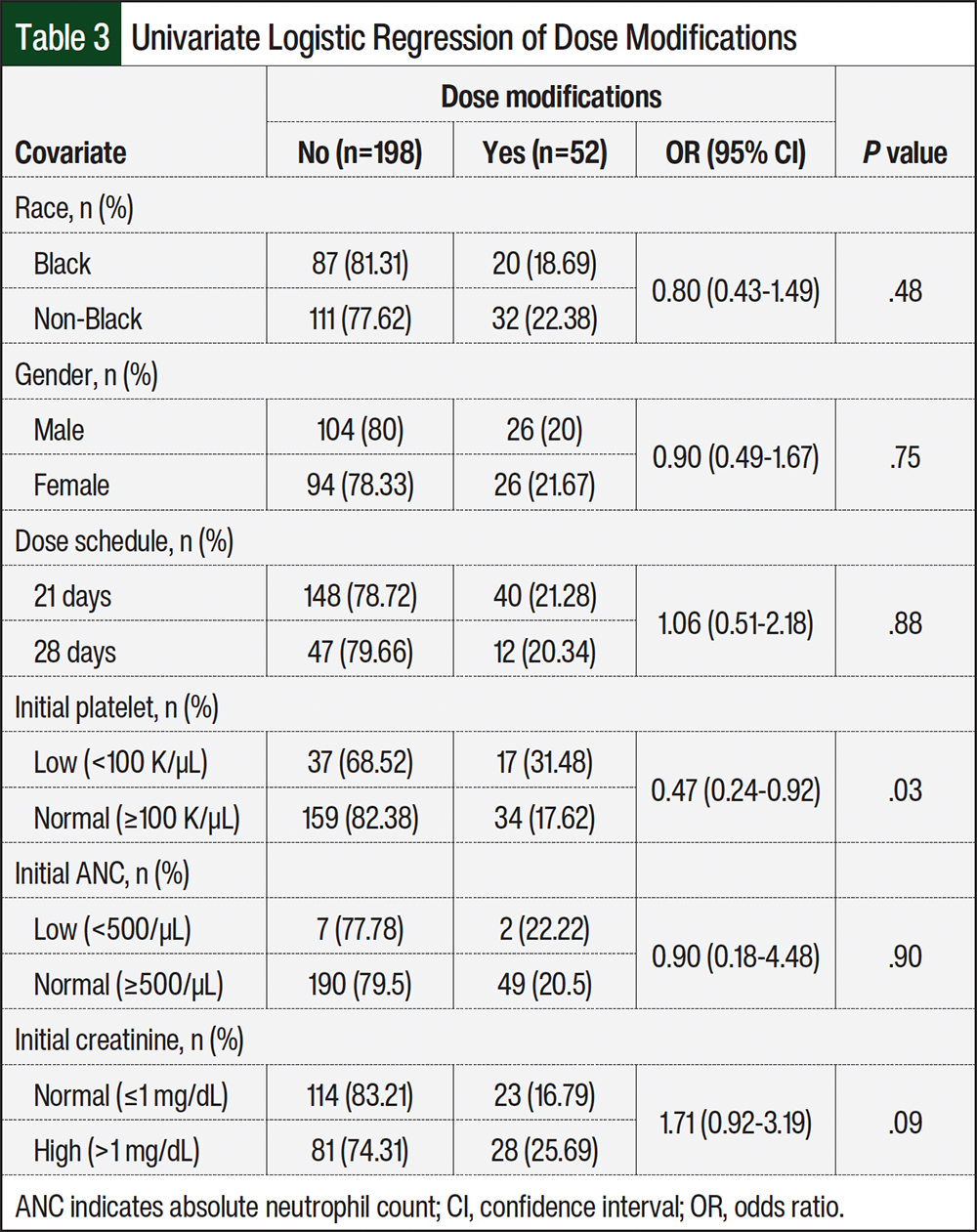
Through univariate logistic regression analysis, we found no significant association between Black race and an increased risk for dose modifications when compared with non-Black patients (odds ratio [OR], 0.80; 95% confidence interval [CI], 0.43-1.49; P=.48; Table 3). Conversely, patients with thrombocytopenia (platelet count <100,000) before lenalidomide initiation were more likely to require dose modifications (OR, 0.47; 95% CI, 0.24-0.92; P=.03). Other covariates, including gender, MM type and revised International Staging System stage of disease, and dose schedule were not found to be independently associated with risk for lenalidomide dose modifications. However, we observed that as baseline performance score worsens, the incidence of dose modifications increases, although this finding did not reach statistical significance for patients with ECOG scores >1 compared with those with ECOG scores of 0-1.
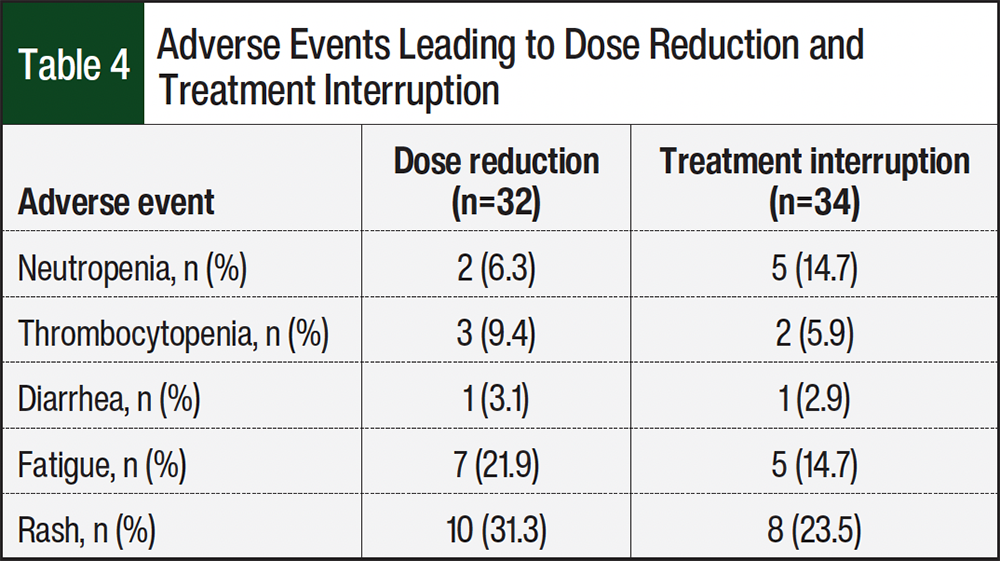
The most common AE leading to dose reductions and treatment interruption was rash (dose reduction, n=10 [31.3%]; treatment interruption, n=8 [23.5%]). Fatigue was the second most common reason for dose reduction (n=7 [21.9%]). Overall, hematologic AEs, including neutropenia or thrombocytopenia, requiring dose modifications in our study were low (n=12 [18.2%]) and frequently managed with treatment interruption (treatment interruption, n=7 [10.6%]; dose reduction, n=5 [7.6%]; Table 4). The observed incidence of neutropenia requiring dose modifications was lower than reported in other randomized trials.11,13,14 Future research should assess whether growth factor support was administered during induction treatment as well as grade of hematologic AE.
Discussion
Racial disparities in MM treatment and outcomes have been well documented, although with conflicting data.6,7,16 SEER data and other population-level studies indicate that Black patients have significantly lower utilization of autologous transplant and novel agents, which may contribute to worse overall survival.16 Other studies, however, suggest that when access to care is standardized, there is no disparity in outcomes.6,7 The rapidly evolving treatment landscape of frontline and relapsed/refractory MM further complicate interpretation of these disparities, which are likely driven by a combination of biological, clinical, socioeconomic, and other multifactorial influences.
A large single-center analysis within the Veterans Affairs system included 15,717 patients with MM.7 They observed no disparity in the use of novel agents or transplants, and Black patients achieved similar or even superior overall survival compared with White patients.7 Similarly, our single-center study represents a near-equal-access setting, where all patients were treated under standardized clinical protocols with comparable access to treatment, supportive care, and follow-up.
To the best of our knowledge, our study is the first to investigate the impact of race on lenalidomide dose modifications with this large of a sample size. A post-hoc subgroup analysis of the GRIFFIN trial was the only study on lenalidomide AEs in Black patients and reported a high discontinuation rate and lower median dose due to TEAEs.13 Baseline characteristics between the 2 groups included in the GRIFFIN trial were comparable, including similar rates of high-risk cytogenetics and bone marrow involvement, although Black patients overall were younger.13 The authors found that TEAEs leading to treatment discontinuation occurred in approximately 30% of Black patients (D-RVd, 35.7%; RVd, 27.8%) compared with approximately 20% in non-Black patients (D-RVd, 19.3%; RVd, 23.0%). Regarding lenalidomide doses, median lenalidomide dose intensities for Black patients were lower compared with White patients (D-RVd, 81.6% vs 87.7%; RVd, 80.2% vs 96.6%, respectively).13 In addition, the authors found that although overall response rate and complete response rate were similar between the 2 groups, and progression-free survival (PFS) was lower in Black patients.17 This observation added to the concern for disparities in outcomes between Black and non-Black patients with newly diagnosed MM.
In our investigation of 250 patients with newly diagnosed MM who received lenalidomide-based induction regimens, we found that patients who self-report as Black did not have an increased risk for TEAEs that required dose modifications. The incidence of lenalidomide dose modifications was 18.7% in Black patients and 22.4% in non-Black patients. However, we noted that patients with low platelet counts (<100 K/µL) before starting lenalidomide had a higher rate of dose modification. Although treatment-emergent thrombocytopenia leading to dose modification was uncommon, this association suggests that supportive care measures, such as growth factor use or transfusions, may be necessary to support the dose intensity of induction regimens in patients with low platelet counts.
Although supportive care agents such as thrombopoietin receptor agonists may help maintain dose intensity, further research should explore the potential benefits versus increased thromboembolic risk. In the GRIFFIN study, vascular thromboembolic events (VTEs) occurred in 10.1% of D-RVd and 15.7% of RVd patients, although the utilization of appropriate VTE prophylaxis was limited.18 Given that MM and use of immunomodulatory agents independently increase VTE risk, further research is needed to optimize supportive care measures in this patient subgroup.18-20
Our findings diverge from those reported in the GRIFFIN trial, indicating that race may not have a significant influence on the frequency of lenalidomide dose modifications in real-world patients. In addition, our data suggest a lower occurrence of treatment delays and interruptions in real-world clinical practice compared with what was observed in clinical trials. In the GRIFFIN trial, nearly half of Black and White patients had lenalidomide cycle interruptions or delays (Black patients: D-RVd, 42.9%; RVd, 50.0%; White patients: D-RVd, 43.4%; RVd, 45.9%).13 However, reasons for these treatment delays were not reported. In contrast, our study found that treatment delays or interruptions occurred in only 13.6% of patients, suggesting that lenalidomide-based induction regimens can be safely tolerated without modifications.
The observed differences in treatment delays between the GRIFFIN trial13 and our analysis may be attributed to variations in patient population and limitations in their sample size, with only 32 Black and 161 White patients included in the Griffin trial.13 The higher incidence of dose modifications in the GRIFFIN trial may be due to the trial’s stringent criteria on AE management. In addition, our non-Black cohort included small proportions of Hispanic and Asian patients, which may have contributed to the comparable outcomes observed in our study. Nevertheless, our analysis shows that, in real-world practice, even if a patient develops neutropenia, a dose interruption or reduction may not always be necessary.
Several factors may influence lenalidomide utilization and dosing. An expert consensus hypothesized that chronic kidney disease or a genetic mutation in the ACKR1/DARC gene may lead to more frequent dose modifications in Black patients.21 The ACKR1/DARC variant, which is more prevalent among individuals of African ancestry, is associated with lower baseline absolute neutrophil counts and may be misinterpreted as treatment-related neutropenia, potentially leading to unnecessary dose modifications.21 Other contributing factors include financial toxicity and logistic barriers, such as insurance delays and REMS program complexities.21
An important treatment goal for patients with newly diagnosed MM is to achieve and maintain deep remission to secure long-term disease control.22 Currently, a lenalidomide-based induction regimen is the standard of care for newly diagnosed patients, based on response rate, depth of response, prolongation of remission, and significant overall survival benefit.23 Lenalidomide is believed to exhibit cytotoxic and immunomodulatory effects. Preclinical studies suggest lenalidomide 25 mg (days 1-21 of a 28-day cycle) elicits cytotoxic effects, whereas lower doses of lenalidomide (5-15 mg daily) activate antitumor immunity.24 Although the starting dose of lenalidomide may be reduced in elderly patients, there are limited clinical data to confirm the efficacy and safety of this approach. The impact of lenalidomide dose on clinical outcomes during the induction phase is also not well characterized. However, data in patients with relapsed/refractory MM suggest that full-dose lenalidomide (25 mg) may be directly tumoricidal, whereas reduced-dose lenalidomide provides immunomodulatory effects.24 Subanalysis of data from the MM-009 and MM-010 trials showed a significantly higher median PFS and overall survival if lenalidomide dose reduction occurred after ≥12 months of therapy at 25 mg daily for 21 days of each 28-day cycle compared with those who had earlier dose reductions.24 Although future prospective studies are needed to confirm this theory, these data underscore the importance of maintaining full-dose lenalidomide if tolerated.
Limitations
There are several limitations to our study. The retrospective design may have introduced bias, and information was not always available. Cytogenetic risk profiles, which may influence treatment tolerability, were not assessed. Patients received a variety of induction regimens, and regimen-specific AE profiles could have confounded results. Race was categorized as Black versus non-Black because of the small number of Asian and Hispanic patients, which limited direct comparison with studies that examined Black versus White patients. Although our study did not assess the grade of hematologic AE or use of growth factor support, we evaluated the impact of TEAEs on dose reductions and treatment interruptions. However, these variables should be investigated in future retrospective studies. It is also worth noting that there are currently no established risk factors for lenalidomide-associated AEs. Therefore, it may be worth exploring potential confounding variables including socioeconomic status, pharmacogenomics, and likelihood to report AEs during office visits.
Conclusion
This retrospective review demonstrated that Black race is not associated with a higher incidence of lenalidomide dose modifications compared with non-Black patients. However, underlying thrombocytopenia before lenalidomide initiation was associated with higher incidence of dose modifications. Despite frequently reported AEs such as hematologic issues, rash, and fatigue in clinical trials, the experience at our center suggests that lenalidomide-based induction regimens can be safely tolerated in all races with close monitoring and education for patients and healthcare providers. Considering that lenalidomide has significant clinical benefit against MM at various stages, future studies should address the clinical impact of dose modifications in the maintenance and relapsed/refractory settings and aim to identify patient and treatment-related factors that may predict AEs and guide individualized management.
Acknowledgments
The research reported in this article was supported in part by the Biostatistics Shared Resource of Winship Cancer Institute of Emory University and NIH/NCI under award number P30CA138292. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author Disclosure Statement
Dr Hall is a consultant to and on the Advisory Board of Sanofi and is a consultant to CSL-Behring; Dr Murphy is on the Advisory Board of Pfizer and a presentation moderator for Sanofi; Dr Dhodapkar is on the Advisory Board of Janssen, Lava Therapeutics, Sanofi, and BMS; Dr Joseph has received research funding from BMS, AstraZeneca, J&J Oncology, and Pfizer and is a consultant to AstraZeneca and J&J Oncology; Dr Hofmeister received research funding from BMS, Sanofi, and Janssen and is on the Advisory Committee of Janssen and Karyopharm; Dr Kaufman is a consultant to BMS, Ascentage, Genentech, Sanofi, and Sebia; Dr Nooka is on the Advisory Board of and received honorarium from Adaptive Biotechnologies, Amgen, AstraZeneca, BMS, Cellectar BioSciences, GSK, Janssen, k36 Therapeutics, ONK Therapeutics, Pfizer, Sanofi, Sebia, and Takeda and has received grants and research support from Aduro Biotech, Amgen, Arch Oncology, BMS, Cellectis, Genentech, GSK, Janssen, Karyopharm, Kite Pharma, Merck, Pfizer, and Takeda; Dr Lonial is a consultant to Amgen, Novartis, BMS, GSK, Merck, AbbVie, Genentech, Regeneron, Pfizer, Celgene, and Janssen and is on the Board of Directors of and owns stock in TG Therapeutics; Dr Maples is a current employee at Pfizer but was not at the time of this study; Dr Sun, Dr Brumwell, Dr Switchenko, Ms Cao, and Dr Gupta have no conflicts of interest to report.
References
- National Cancer Institute. SEER cancer stat facts: myeloma. Accessed April 1, 2023. https://seer.cancer.gov/statfacts/html/mulmy.html
- Kazandjian D. Multiple myeloma epidemiology and survival: a unique malignancy. Semin Oncol. 2016;43:676-681. doi:10.1053/j.seminoncol.2016.11.004
- National Cancer Institute. SEER Cancer Statistics Review, 1975-2000. Accessed January 15, 2026. https://seer.cancer.gov/archive/csr/1975_2000/
- Landgren O, Gridley G, Turesson I, et al. Risk of monoclonal gammopathy of undetermined significance (MGUS) and subsequent multiple myeloma among African American and white veterans in the United States. Blood. 2006;107:904-906. doi:10.1182/blood-2005-08-3449
- Marinac CR, Ghobrial IM, Birmann BM, et al. Dissecting racial disparities in multiple myeloma. Blood Cancer J. 2020;10:19. doi:10.1038/s41408-020-0284-7
- Ailawadhi S, Jacobus S, Sexton R, et al. Disease and outcome disparities in multiple myeloma: exploring the role of race/ethnicity in the Cooperative Group clinical trials. Blood Cancer J. 2018;8:67. doi:10.1038/s41408-018-0102-7
- Fillmore NR, Yellapragada SV, Ifeorah C, et al. With equal access, African American patients have superior survival compared to white patients with multiple myeloma: a VA study. Blood. 2019;133:2615-2618. doi:10.1182/blood.2019000406
- Musolino C, Allegra A, Innao V, et al. Inflammatory and anti-inflammatory equilibrium, proliferative and antiproliferative balance: the role of cytokines in multiple myeloma. Mediators Inflamm. 2017;2017:1852517. doi:10.1155/2017/1852517
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: multiple myeloma. Version 3.2023. Accessed March 10, 2023. www.nccn.org/professionals/physician_gls/pdf/myeloma.pdf
- Revlimid. Prescribing information. Bristol-Myers Squibb Company; 2023. Accessed February 18, 2026. https://packageinserts.bms.com/pi/pi_revlimid.pdf
- Richardson PG, Jacobus SJ, Weller EA, et al. Triplet therapy, transplantation, and maintenance until progression in myeloma. N Engl J Med. 2022;387:132-147. doi:10.1056/NEJMoa2204925
- Durie BGM, Hoering A, Abidi MH, et al. Bortezomib with lenalidomide and dexamethasone versus lenalidomide and dexamethasone alone in patients with newly diagnosed myeloma without intent for immediate autologous stem-cell transplant (SWOG S0777): a randomised, open-label, phase 3 trial. Lancet. 2017;389:519-527. doi:10.1016/S0140-6736(16)31594-X
- Nooka AK, Kaufman JL, Rodriguez C, et al. Daratumumab plus lenalidomide/bortezomib/dexamethasone in Black patients with transplant-eligible newly diagnosed multiple myeloma in GRIFFIN. Blood Cancer J. 2022;12:63. doi:10.1038/s41408-022-00653-1
- Voorhees PM, Kaufman JL, Laubach J, et al. Daratumumab, lenalidomide, bortezomib, and dexamethasone for transplant-eligible newly diagnosed multiple myeloma: the GRIFFIN trial. Blood. 2020;136:936-945. doi:10.1182/blood.2020005288
- Liu Y, Nickleach DC, Zhang C, et al. Carrying out streamlined routine data analyses with reports for observational studies: introduction to a series of generic SAS® macros. F1000Res. 2018;7:1955. doi:10.12688/f1000research.16866.2
- Ailawadhi S, Frank RD, Advani P, et al. Racial disparity in utilization of therapeutic modalities among multiple myeloma patients: a SEER-Medicare analysis. Cancer Med. 2017;6:2876-2885. doi:10.1002/cam4.1246
- Nooka AK, Kaufman JL, Rodriguez C, et al. An end-of-study subgroup analysis of black patients from the phase 2 Griffin study of daratumumab (DARA) plus lenalidomide, bortezomib, and dexamethasone (RVd) in patients with transplant-eligible newly diagnosed multiple myeloma (NDMM). Blood. 2022;140:10154-10156. doi:org/10.1182/blood-2022-162473
- Sborov DW, Baljevic M, Reeves B, et al. Daratumumab plus lenalidomide, bortezomib and dexamethasone in newly diagnosed multiple myeloma: analysis of vascular thrombotic events in the GRIFFIN study. Br J Haematol. 2022;199:355-365. doi:10.1111/bjh.18432
- Kristinsson SY, Pfeiffer RM, Björkholm M, et al. Arterial and venous thrombosis in monoclonal gammopathy of undetermined significance and multiple myeloma: a population-based study. Blood. 2010;115:4991-4998. Erratum in: Blood. 2011;117:3938. doi:10.1182/blood-2009-11-252072
- Carrier M, Le Gal G, Tay J, et al. Rates of venous thromboembolism in multiple myeloma patients undergoing immunomodulatory therapy with thalidomide or lenalidomide: a systematic review and meta-analysis. J Thromb Haemost. 2011;9:653-663. doi:10.1111/j.1538-7836.2011.04215.x
- Banerjee R, Biru Y, Cole CE, et al. Disparities in relapsed or refractory multiple myeloma: recommendations from an interprofessional consensus panel. Blood Cancer J. 2024;14:149. doi:10.1038/s41408-024-01129-0
- Fenk R, Giagounidis A, Goldschmidt H, et al. Efficacy and tolerability of high- versus low-dose lenalidomide maintenance therapy of multiple myeloma after autologous blood stem cell transplantation. Clin Cancer Res. 2020;26:5879-5886. doi:10.1158/1078-0432.CCR-20-0841
- Holstein SA, McCarthy PL. Immunomodulatory drugs in multiple myeloma: mechanisms of action and clinical experience. Drugs. 2017;77:505-520. doi:10.1007/s40265-017-0689-1
- Dimopoulos MA, Hussein M, Swern AS, Weber D. Impact of lenalidomide dose on progression-free survival in patients with relapsed or refractory multiple myeloma. Leukemia. 2011;25:1620-1626. doi:10.1038/leu.2011.126