Connecting Science to Practice
Clinical oncology pharmacists at WVU Cancer Institute
developed a pathway to identify patients to use home infusion–supplied blinatumomab during planned admissions
lasting <7 days to save inpatient drug acquisition costs. Patients
were initiated on blinatumomab in the outpatient
infusion center utilizing a home infusion–supplied 7-day
duration blinatumomab bag and were subsequently admitted
to the inpatient service with an order to continue therapy
as patient’s own medication. During the study period,
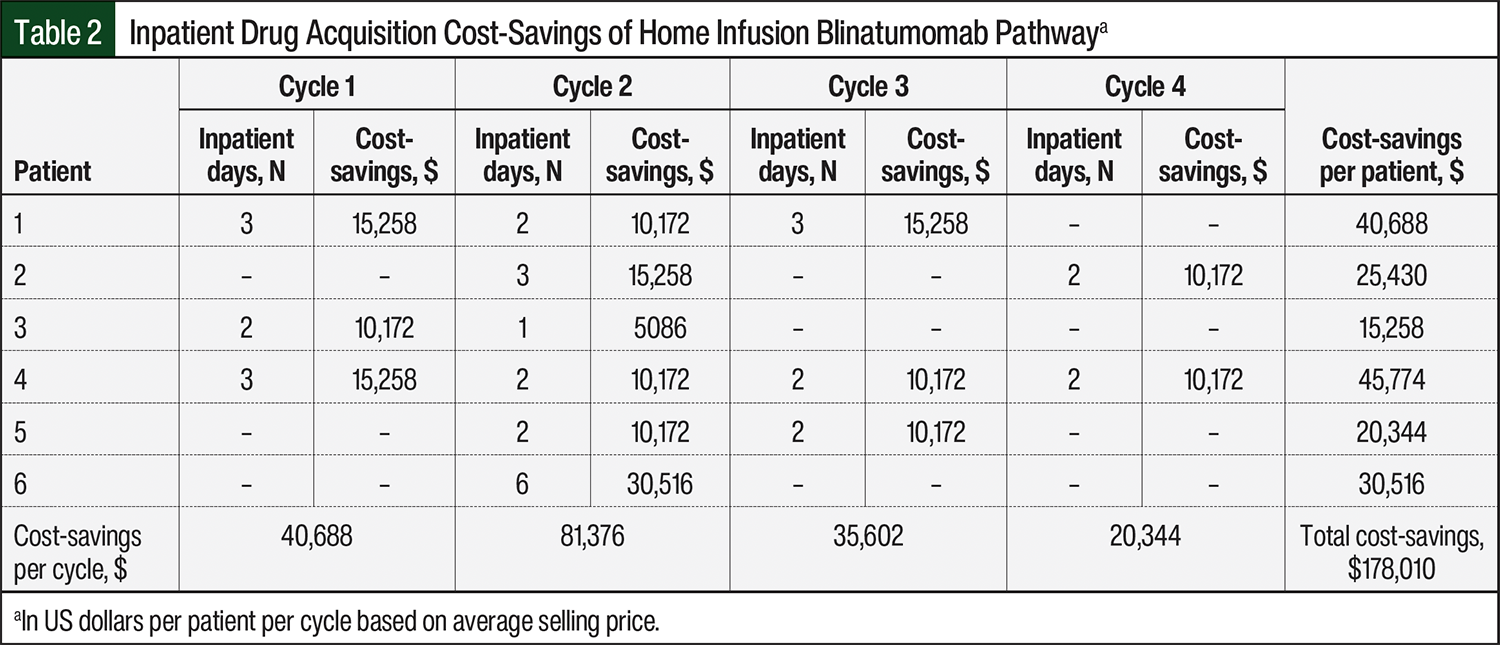
use of this pathway for 6 patients resulted in an actual inpatient
drug acquisition cost-savings of $178,010. The average
cost-savings per cycle was $12,715, and the average cost-savings
per patient was $29,668. This study demonstrates significant
inpatient drug acquisition cost-savings for patients
who require short-term inpatient monitoring after the outpatient
initiation of blinatumomab.
Blinatumomab is a bispecific T-cell engager that facilitates an interaction between CD19 on B cells and CD3 on T cells.1 T cells then elicit an immune response that has proven its effectiveness against specific indications of acute lymphoblastic leukemia (ALL). Blinatumomab was previously approved for the treatment of CD19-positive B-cell ALL in patients who reached a first or second complete remission with minimal residual disease (MRD), and for the treatment of relapsed or refractory CD19-positive B-ALL.1
Recent data from the ECOG1910 trial demonstrate that consolidative blinatumomab leads to impressive relapse-free survival and improved overall survival in patients with newly diagnosed Philadelphia chromosome–negative B-ALL who reach MRD-negative remission, which will increase the number of patients who are eligible to receive blinatumomab therapy.2 These data, along with similar data from MD Anderson in patients who have received hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone induction,3 led to an additional FDA approval for blinatumomab and incorporation into the NCCN treatment guidelines, which recommend the use of blinatumomab in the consolidative setting for patients with CD19-positive Philadelphia chromosome–negative B-ALL after receiving multiphase chemotherapy.4 Although blinatumomab has successfully addressed important treatment gaps in ALL, its dosing scheme poses operational challenges and is costly when inpatient inventory is used for patients requiring short-term inpatient monitoring based on the prescribing information for blinatumomab. These factors emphasize the need for a cost-effective way to seamlessly deliver this unique therapy in a manner that is safe and convenient for patients and institutions.
Blinatumomab is frequently administered as a continuous intravenous infusion via portable infusion pump for a total of 28 consecutive days.1 The infusion may be compounded as a 270-mL preparation administered over 24 or 48 hours, or as a 110-mL preparation administered over 7 days.1 The choice of infusion duration should be made with consideration of the frequency of infusion bag exchanges and the weight of the patient. On treatment initiation, patients are recommended to be admitted for a duration of 2, 3, or 9 days, depending on their indication and treatment cycle, for the monitoring of cytokine release syndrome, neurologic adverse events, and other adverse reactions for at least the first 2 cycles of therapy.1
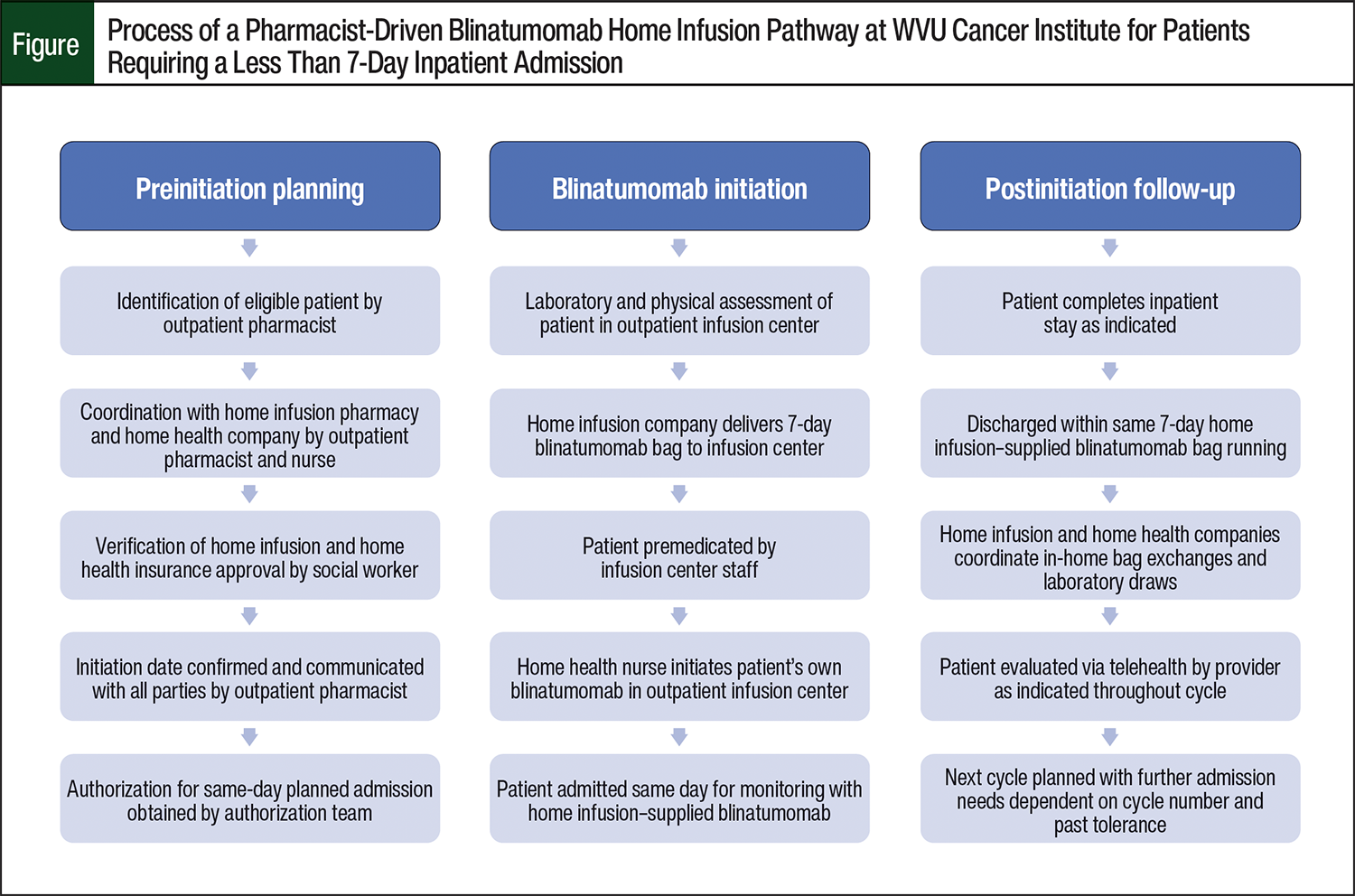
Notably, the inpatient drug acquisition of blinatumomab is costly, with an average selling price of $5085.96 per single-dose 35-µg vial as of October 2023.5 After a patient is discharged from inpatient monitoring, bag exchanges are often performed at an outpatient infusion center or through the coordination of home infusion and home health services, as previously reported.6 To further capitalize on the conveniences of the home infusion pathway, clinical oncology pharmacists at the WVU Cancer Institute Mary Babb Randolph Cancer Center developed a novel workflow to also use these services to eliminate inpatient drug acquisition costs for patients in need of treatment with blinatumomab who require an admission of <7 days. This pharmacist-driven home infusion pathway for blinatumomab uses the home infusion pharmacy drug supply as the patient’s own supply during and after admission in addition to home health bag exchange services, as outlined in the Figure.
The patients who were eligible for the pathway were adults with an expected inpatient duration of <7 days to avoid needing a bag exchange while admitted; therefore, all patients received a dose of 28 µg daily. After identification of an eligible patient, the outpatient pharmacist coordinated with the clinic teams to verify home infusion and home health benefits and accessibility. The selection of specific providers for these services was driven by the respective patient’s insurance coverage and geographic service areas. This pathway was implemented in January 2021.
Although this process requires substantial planning by the outpatient pharmacist and clinical teams, it allows the cost of blinatumomab to be billed to the patient’s insurance and for blinatumomab to be supplied by a home infusion pharmacy before admission rather than the patient incurring significant drug costs in the inpatient setting, which has not previously been reported. This process also substantially decreases the logistical challenges of blinatumomab therapy for patients by decreasing travel and time spent in the infusion center, as previously outlined in publications that focused on home infusion utilization.6-8 Furthermore, this process increases outpatient chair space availability and decreases infusion pharmacy workload. The inpatient drug acquisition cost-savings of this novel workflow are important to quantify so the value of the workflow can be assessed.
The primary objective of this study was to determine the actual inpatient drug cost-savings that were incurred as a direct result of the use of a pharmacist-driven home infusion pathway for blinatumomab since its inception.
Methods
To determine the actual inpatient drug acquisition cost-savings of this process, a chart review of patients who received blinatumomab from January 2021 to December 2023 at the WVU Cancer Institute Mary Babb Randolph Cancer Center was performed. Adults receiving blinatumomab for ALL at a dose of 28 µg/day were included if they received blinatumomab through the pharmacist-driven home infusion pathway for blinatumomab (Figure) and were immediately admitted to WVU Cancer Institute’s transplant and cellular therapy service at Ruby Memorial Hospital for short-term (<7 days) inpatient monitoring per the prescribing information for blinatumomab after outpatient initiation for at least 1 cycle of therapy. Of note, patients who had planned admissions requiring a stay of longer than 7 days were not eligible to utilize the pathway and were therefore excluded from this analysis because of the inability to request a bag exchange through a home infusion pharmacy while the patient is admitted to prevent billing issues.
The inpatient drug acquisition cost-savings were calculated by first determining the number of patients using the pharmacist-driven home infusion pathway for blinatumomab within the study period and then assessing the number of cycles with a planned admission per patient who used the pathway. Each admission was further reviewed to determine the number of inpatient days per patient per cycle in those who received home infusion–supplied blinatumomab, therefore foregoing the need for an inpatient supply of blinatumomab. Each day of therapy received in the inpatient setting was then considered to save the inpatient acquisition cost equivalent to a full single-dose vial (35 µg) of blinatumomab to account for drug waste. The inpatient drug acquisition cost-savings were determined based on the average selling price at the time of the data collection initiation in October 2023. Descriptive statistics were employed for data analysis.
Results
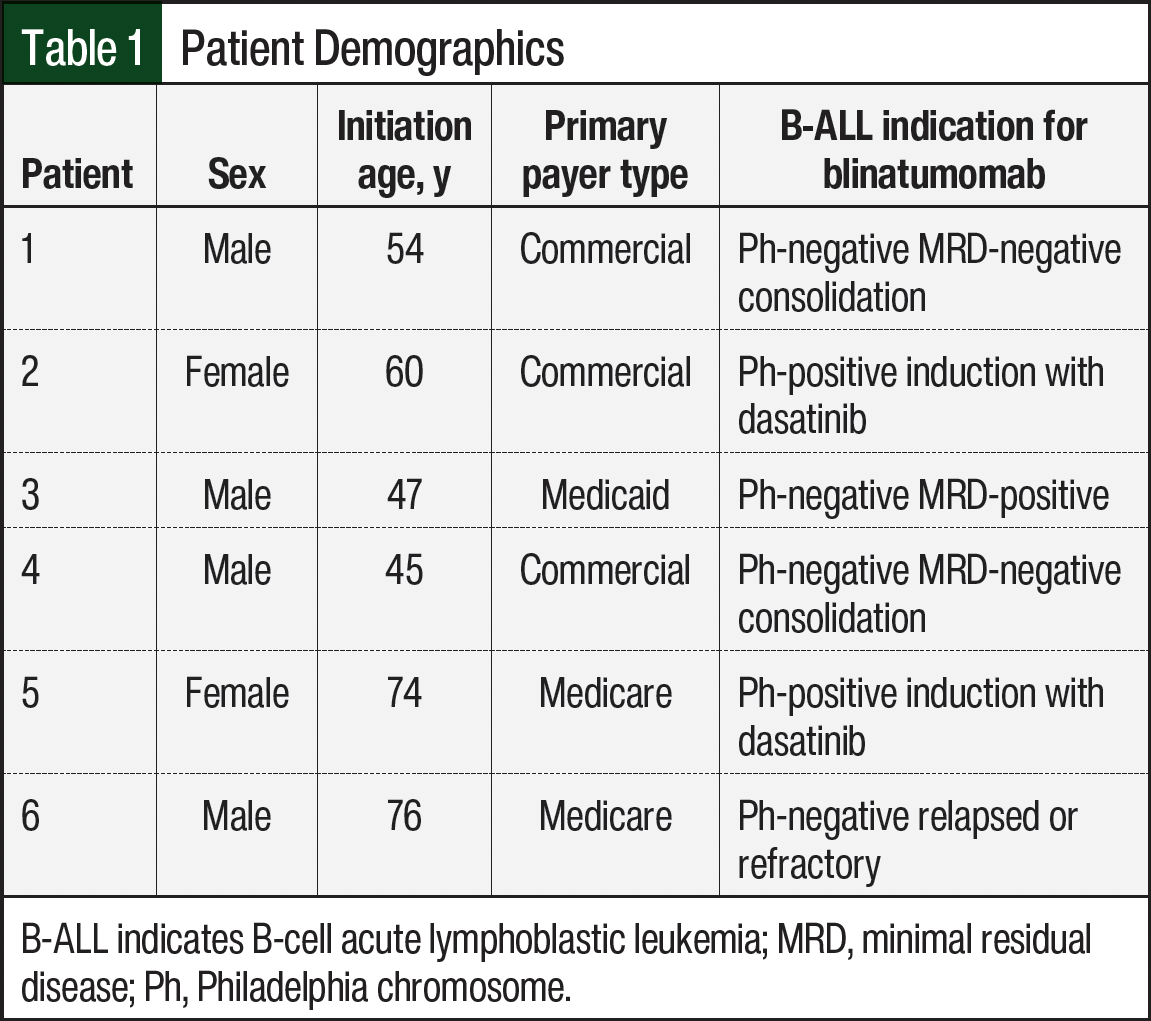
A total of 11 patients received blinatumomab during the study period and were screened for inclusion. In all, 6 patients met the inclusion criteria for the actual drug cost-savings analysis. The demographics of the study patients are shown in Table 1. Of the 5 patients who were excluded from the study, 4 were ineligible for the program as a result of a required admission of >7 days for relapsed disease without a need for subsequent treatment cycles, and 1 patient was unable to utilize home infusion services because of a lack of insurance coverage. Of the 6 patients included in the study, 3 had commercial insurance and 3 had either Medicaid or Medicare coverage (Table 1). These 6 patients accounted for a total of 14 separate admissions requiring a <7-day stay in which the pharmacist-driven home infusion pathway for blinatumomab was used to eliminate the inpatient drug acquisition cost.
A breakdown of the inpatient drug acquisition cost-savings per patient per admission is shown in Table 2. During the allotted study period, the use of the pharmacist-driven home infusion pathway for blinatumomab resulted in an actual inpatient drug acquisition cost-savings of $178,010. The average cost-savings per cycle (total cost-savings divided by 14 admissions) was $12,715, and the average cost-savings per patient (total cost-savings divided by 6 patients) was $29,668. Of note, only 1 of the 6 study patients received blinatumomab through this pathway from January 2021 to December 2022. The remaining 5 patients using this pathway resulted in an annualized cost-savings of $162,751 from January 2023 to December 2023. This represented an 83% capture rate of patients who were eligible for the program in 2023, with the only missed opportunity resulting from a lack of insurance coverage, as previously outlined.
Discussion
Our study demonstrates the feasibility of a pharmacist-driven home infusion pathway for blinatumomab and highlights the substantial impact on inpatient drug cost-savings for planned short-term inpatient monitoring of blinatumomab recipients, despite the low number of patients included. We recognize that recently published data on 9 patients also demonstrate the safety and feasibility of outpatient digital monitoring of patients who initiate blinatumomab for the treatment of MRD-positive B-ALL, although this study had slow enrollment leading to the subsequent termination of the study after a total accrual of 10 patients.7,8 In addition, the study used a digital monitoring system and required manual blood pressure measurements every 3 to 6 hours, which may not be realistic for every health system to implement or every patient or caregiver to perform.6
Although this chart review quantifies the substantial inpatient drug acquisition cost-savings of a pharmacist-driven home infusion pathway for blinatumomab, it should be noted that future predicted annualized cost-savings are likely underestimated considering that the time period of this review preceded the most recent FDA approval of blinatumomab in the consolidative setting for the treatment of patients with Philadelphia chromosome–negative B-ALL. Hence, we expect our actual annualized cost-savings to increase over time as more patients use the pathway and as the data from the program’s impact mature. In addition, the focus of this chart review was strictly inpatient drug acquisition cost-savings, and therefore our estimates do not account for the indirect cost-savings associated with the use of the pathway, such as nursing resources, pharmacy productivity, or infusion chair time saved.
Although an assessment of reimbursement was beyond the scope of this study, our center services a high percentage of patients covered by insurance who utilize diagnosis-related group-based payments, which would not be expected to provide separate reimbursement for inpatient drug acquisition costs.
Last, although the development of oncolytic home infusion services offered through our institution are still in infancy, our future aspirations include offering home infusion drug supply for blinatumomab through an internal pharmacy, which will present an opportunity to capitalize further on the inpatient drug acquisition cost-savings by also increasing internal revenue for our home infusion services and offering more comprehensive patient care. This would also capitalize on further drug savings through 340B drug pricing in the outpatient home infusion setting.
Limitations
Our study has limitations. This study reflects a single-center experience with a limited sample size based on chart review. Also, the outcomes are descriptive in nature and do not include a full assessment of potential reimbursement or the indirect cost-savings.
Conclusion
Blinatumomab is a logistically challenging and financially burdensome therapy with an expected increase in utilization based on recent data that demonstrate improved clinical outcomes in patients with newly diagnosed B-ALL. To eliminate inpatient drug acquisition costs, clinical oncology pharmacists at the WVU Cancer Institute Mary Babb Randolph Cancer Center developed a pathway to seamlessly deliver this unique therapy. Although the utilization of the pathway requires extensive and precise multidisciplinary planning, the results of this chart review demonstrate successful implementation and utilization of a pharmacist-driven home infusion pathway for blinatumomab, resulting in significant inpatient drug acquisition cost-savings for patients who require short-term inpatient monitoring after the outpatient initiation of treatment with blinatumomab.
Author Disclosure Statement
Dr Seago, Dr Yingling, Dr Cumpston, and Dr Gallagher have no conflicts of interest to report.
References
- Blincyto (blinatumomab) injection, for intravenous use [prescribing information]. Amgen; December 2024. Accessed May 1, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2024/125557s030lbl.pdf
- Litzow MR, Sun Z, Paietta E, et al. Consolidation therapy with blinatumomab improves overall survival in newly diagnosed adult patients with B-lineage acute lymphoblastic leukemia in measurable residual disease negative remission: results from the ECOG-ACRIN E1910 randomized phase III National Cooperative Clinical Trials Network Trial. Blood. 2022;140(suppl 2):LBA-1.
- Jabbour E, Short NJ, Jain N, et al. Hyper-CVAD and sequential blinatumomab for newly diagnosed Philadelphia chromosome-negative B-cell acute lymphocytic leukaemia: a single-arm, single-centre, phase 2 trial. Lancet Haematol. 2022;9:e878-e885.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): acute lymphoblastic leukemia. Version 3.2024. Accessed December 20, 2024. www.nccn.org/professionals/physician_gls/pdf/all.pdf
- ASP Pricing File. Centers for Medicare & Medicaid Services. Revised October 10, 2024. Accessed November 21, 2024. www.cms.gov/medicare/payment/part-b-drugs/asp-pricing-files
- Bojilova-Dor L, Pinkney K, Cauff B, et al. Successful outpatient administration of blinatumomab infusion in pediatric patients with acute lymphoblastic leukemia. Blood. 2021;138(suppl 1):4028.
- Pardee TS, Varela J, Jeyakumar D, et al. Outpatient blinatumomab with digital monitoring in patients with measurable residual disease positive (MRD+) B-ALL in a phase 4 study. J Clin Oncol. 2024;42(suppl 16):Abstract 6544.
- Feasibility Study to Evaluate Outpatient Blinatumomab in Subjects With Minimal Residual Disease (MRD) of B-precursor Acute Lymphoblastic Leukemia (ALL). NLM identifier: NCT04506086. Updated October 16, 2024. Accessed November 15, 2024. www.cancer.gov/research/participate/clinical-trials-search/v?id=NCI-2020-06780&r=1