Connecting Science to Practice
Hypercalcemia of malignancy is an oncologic emergency
that is often treated with hydration, bisphosphonates, and
calcitonin. Data guiding clinicians on which weight—ideal
body weight (IBW) or actual body weight—to use for optimizing
efficacy while balancing high acquisition costs are
lacking, however. Our study sought to assess corrected calcium
levels 48 hours after the administration of calcitonin
using an IBW-based dosing protocol for calcitonin. This
dosing protocol resulted in similar hypercalcemia of malignancy
resolution at 48 hours and substantial cost-savings
over the 2-year postimplementation period studied. Our
findings represent potential improvements in effectiveness
and reduced costs of care.
Hypercalcemia of malignancy (HCM) is an oncologic emergency that occurs in 20% to 30% of patients with cancer and is a poor prognostic factor.1 The signs and symptoms of HCM are nonspecific, including dehydration, nausea and vomiting, constipation, acute kidney injury, electrocardiogram changes, weakness, and anorexia.1-4 In patients with severe HCM, which is defined as corrected calcium >14 mg/dL, neurologic symptoms may manifest as confusion, altered mental status, and seizures.1-3,5
The current treatment guidelines for adults with severe HCM strongly recommend the use of intravenous (IV) bisphosphonates or denosumab in combination with calcitonin as initial treatment.3 Other pharmacologic therapies, including aggressive hydration, diuretics, calcimimetics, and glucocorticoids, are included in the management of HCM.1-3 Although very effective for HCM, the effect of bisphosphonates is delayed, with results seen in approximately 48 to 72 hours.1 Because of the delayed effect of the bisphosphonates, calcitonin may be administered at the same time as the bisphosphonates to allow for more rapid resolution of the hypercalcemia.
For the treatment of HCM, the dose of calcitonin is 4 to 8 units/kg, which is administered via subcutaneous (SC) or intramuscular (IM) dosing every 12 hours, and must be given with other therapies, including bisphosphonates and fluids.2,6,7 Because of the low volume of distribution of calcitonin (0.15-0.3 L/kg), lower doses of calcitonin may be effective for the treatment of HCM. After approximately 48 hours of treatment, calcitonin exerts a tachyphylaxis effect where the outcomes are diminished, rendering it ineffective despite repeated administration.2 Tachyphylaxis possibly occurs as a result of the downregulation of calcitonin receptors through repeated administration in target organs, such as the bones. Another theory for this tachyphylaxis is antibody production against the drug, because it is derived from salmon, which is a foreign substance.2 Apart from the unwanted tachyphylactic effect of calcitonin, the price of this drug has drastically increased over the past decade. In 2013, the cost of calcitonin increased by 3263%, changing the cost from approximately $68 per unit to approximately $2286 per unit; calcitonin currently costs approximately $1878 per unit.8
Because of this price escalation and the tachyphylactic effect that occurs with calcitonin treatment, pharmacy-implemented dosing protocols have restricted treatment with calcitonin at different institutions.9,10 The purpose of this study is to assess the corrected calcium level at 48 hours after the implementation of a calcitonin dosing protocol using ideal body weight (IBW).
Methods
The institutional review board of the hospital previously approved the study. This is a multicenter, retrospective, noninferiority, pre- and post-quasiexperimental study conducted at St Joseph’s Hospital and Medical Center (SJHMC) in Phoenix, AZ, and Chandler Regional Medical Center (CRMC) in Chandler, AZ. A discern analytics report was used to identify patients who had calcitonin ordered at the study sites during the preprotocol (2014-2016) and postprotocol (2018-2020) time frames. The calcitonin dosing protocol using IBW was implemented at the study’s sites in 2017. Adults admitted to SJHMC or CRMC who received an injectable form of calcitonin for the treatment of HCM were included in the analysis. Patients were excluded from the study if they were pregnant, they were undergoing dialysis, IBW dosing was used in the preprotocol cohort, or if actual body weight dosing was used in the postprotocol cohort.
The calcitonin criteria for treatment and dosing guidelines that were implemented in 2017 at SJHMC and CRMC included a corrected calcium level of ≥12 mg/dL, SC or IM administration, a dose of 4 units/kg IBW every 12 hours, and a maximum number of 4 doses. Protocol compliance was defined as the patient meeting all 4 of the protocol requirements listed above. Additional HCM management beyond the use of calcitonin did not affect protocol compliance, because this was determined by the provider’s individualized discretion and was outside the calcitonin dosing protocol. On calculating the dose of calcitonin using 4 units/kg, protocol compliance allowed a ±15% dose deviation from 4 units/kg IBW to account for rounding to vial size as well as for the volume of drug administered via IM or SC dosing.
The primary study outcome was corrected calcium levels at 48 hours after the administration of calcitonin using the calcitonin dosing protocol that was implemented using IBW. In patients who were obese, the response was evaluated by using complete response (CR), partial response, and no response. CR was defined as a corrected calcium of <10.8 mg/dL, partial response was defined as a corrected calcium of 10.8 to 11.5 mg/dL, and no response was defined as a corrected calcium of >11.5 mg/dL. The secondary outcomes included protocol compliance, length of stay, change in corrected calcium from baseline (defined as the date of the first dose of calcitonin) up to 72 hours, the average time to calcium resolution (defined as the date of the first dose of calcitonin up to the date of CR), calcitonin-induced hypocalcemia (corrected calcium <8.8 mg/dL), and the potential cost-savings.
Cost Analysis
The average calcitonin wholesale price of the 200-unit/mL vial was $1878.60 at the time of the writing of this article. The mean cost-savings was calculated by determining the difference in the average wholesale price per order of calcitonin between the preprotocol and postprotocol cohorts and multiplying the cost difference by the number of orders in the postprotocol cohort. The median cost-savings would also be calculated if there was great variability between the costs because some patients received higher doses of calcitonin than other patients.
Statistical Methods
Descriptive statistics were reported as means and standard deviations for normally distributed continuous data, medians and interquartile ranges were reported for nonnormal distributed data, and percentages were reported for nominal data. Fisher’s exact test was used for the categorical variables, Student’s t-test was used for the comparison of means, and Wilcoxon rank-sum tests were used for the comparison of medians. An a priori value of P<.05 was used to assess the statistical significance. Power was not predetermined, because we planned to include all eligible patients in the analysis.
Results
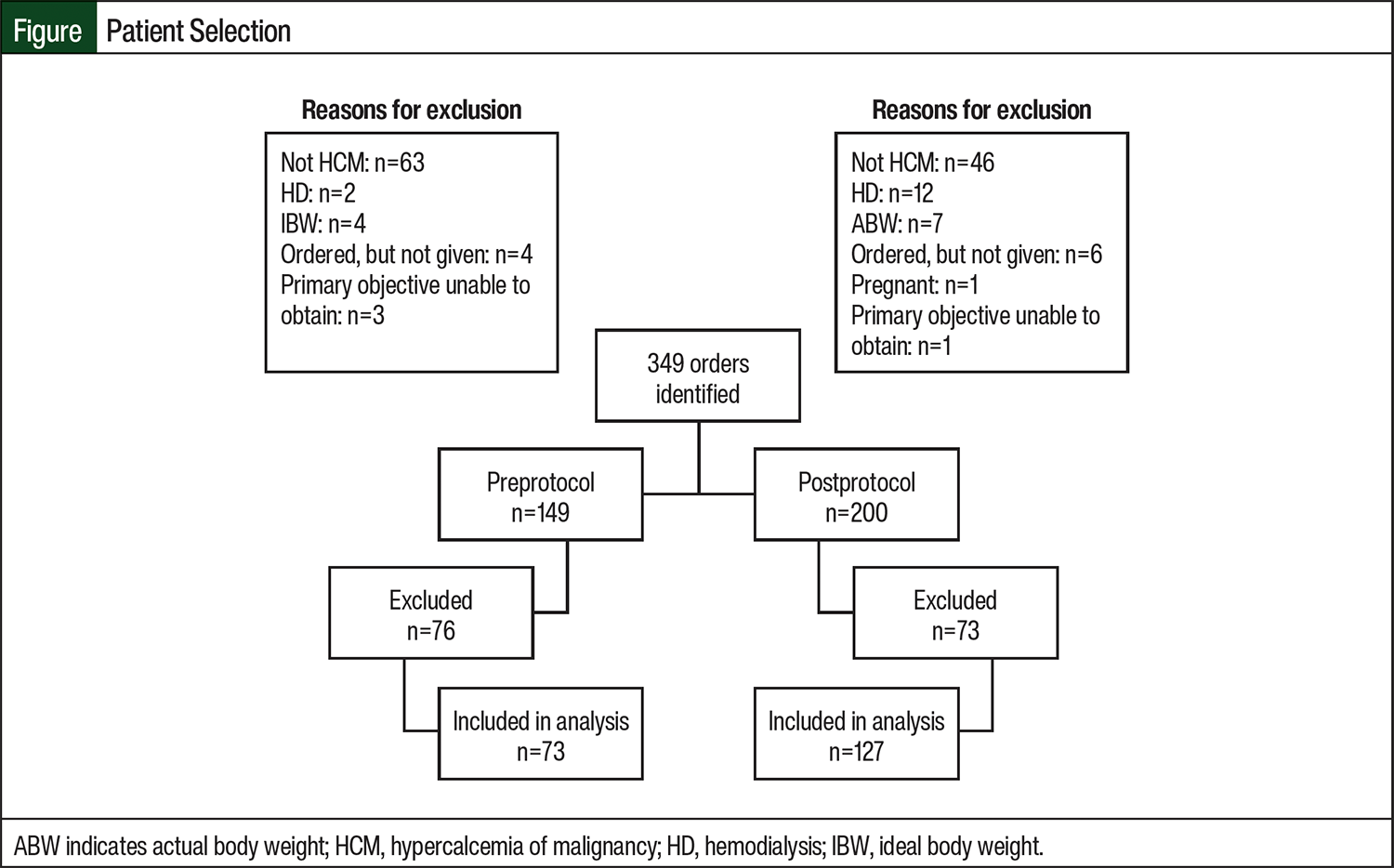
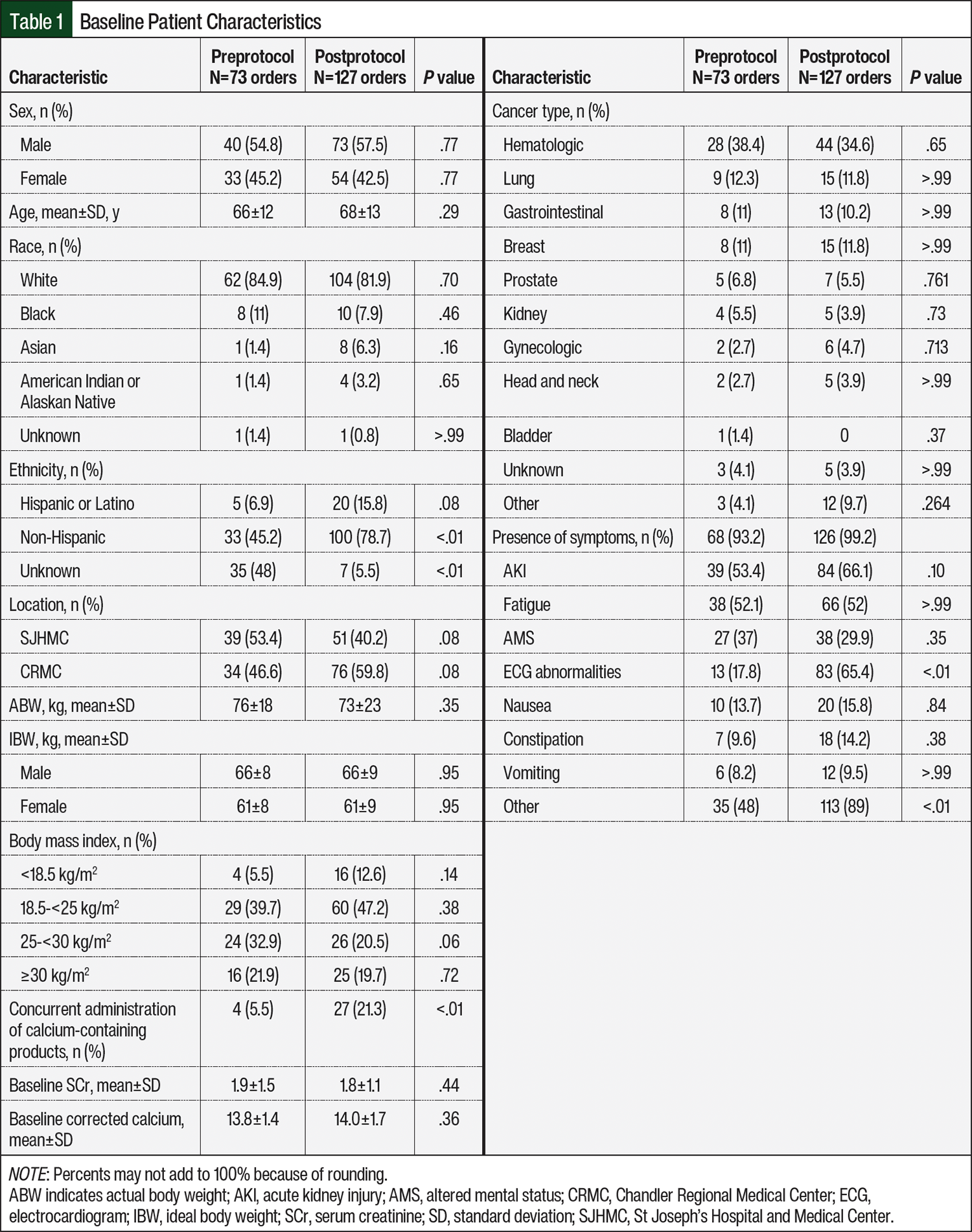
A total of 345 patients and 349 orders were reviewed for study inclusion. The final analysis included 73 orders in the preprotocol group and 127 orders in the postprotocol group (Figure). The main reason for exclusion from the preprotocol and postprotocol groups was the use of calcitonin for indications other than HCM (63 orders in the preprotocol and 46 orders in the postprotocol groups). The complete reasons for exclusion from the study are listed in the Figure. The most common type of cancer in the study patients was hematologic, with 38.4% and 34.6% of patients in the preprotocol and postprotocol groups, respectively (Table 1). More than 90% of patients in both cohorts had symptomatic HCM. The mean baseline corrected calcium was similar between both cohorts, at approximately 14 mg/dL. The baseline characteristics of the study patients are shown in Table 1.
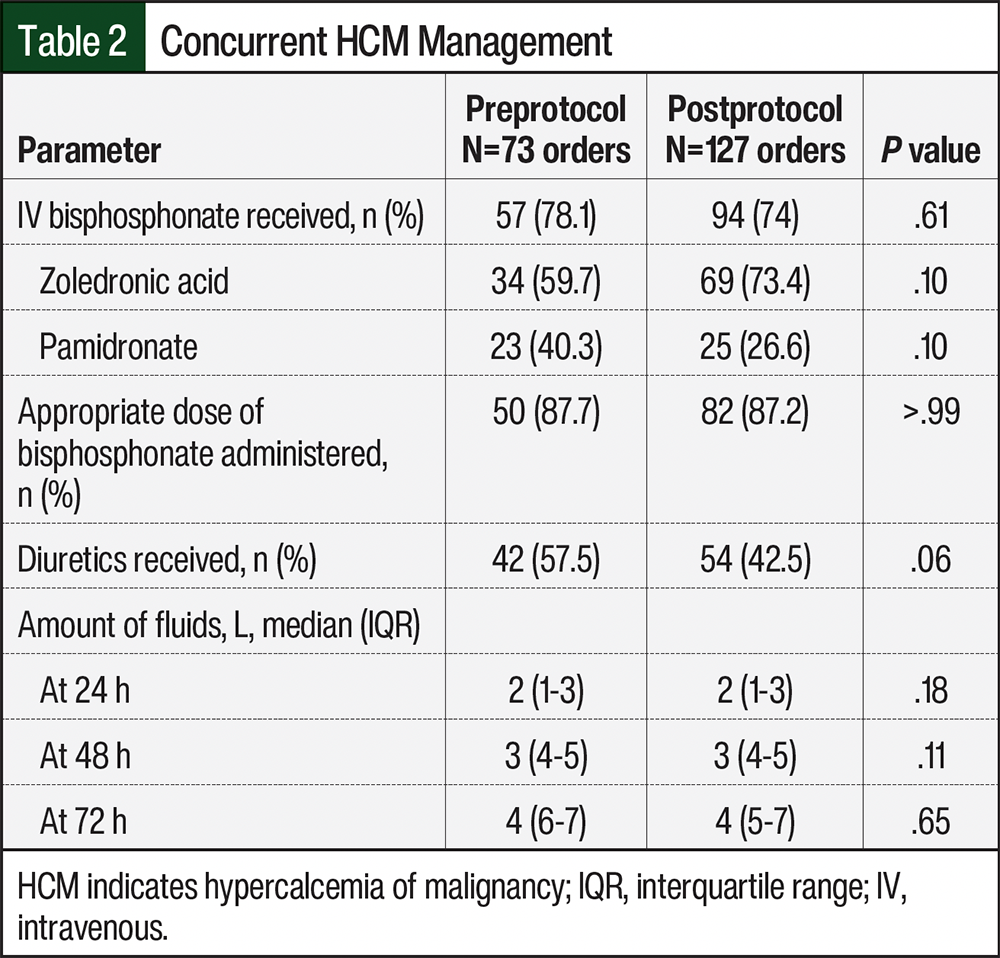
The median numbers of doses per order were 4 in the preprotocol group and 3 in the postprotocol group. The omission of the fourth calcitonin dose was decided by the provider’s discretion outside of the protocol. Treatment with IV bisphosphonates, which included zoledronic acid or pamidronate, was received by 78.1% and 74% of patients in the preprotocol and postprotocol groups, respectively (P=.61; Table 2). Of the patients who received a bisphosphonate, approximately 87% in both cohorts received appropriate doses. The most frequent reason for inappropriate bisphosphonate dosing was underdosing in the setting of mild-to-moderate renal impairment that did not warrant dose reduction. Diuretics were received by approximately 57.5% and 42.5% of patients in the preprotocol and postprotocol groups, respectively. The patients with HCM also received IV fluids, with a median of 4 L at 72 hours. The full details of the concomitant HCM management are summarized in Table 2.
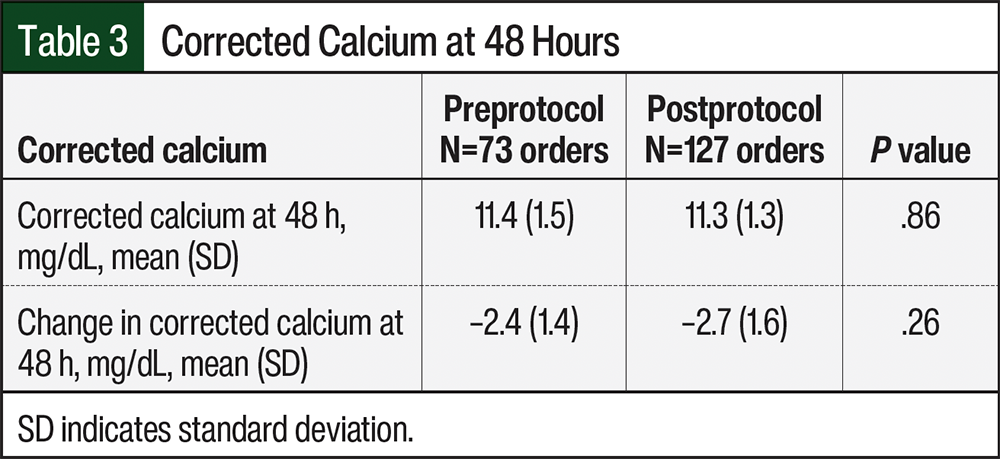
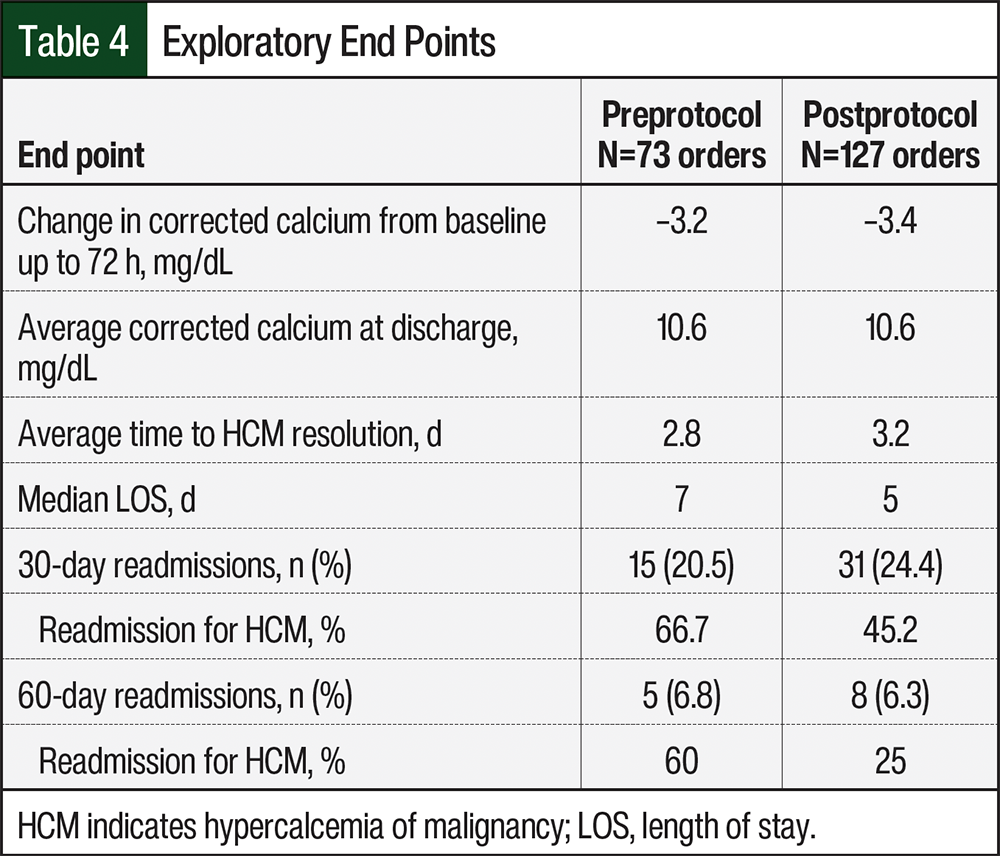
Between the preprotocol and postprotocol implementation cohorts, there was no statistical difference in the corrected calcium levels (11.4 mg/dL vs 11.3 mg/dL, respectively; P=.86) or the decrease in corrected calcium (2.4 mg/dL vs 2.7 mg/dL, respectively; P=.26) after 48 hours of calcitonin administration (Table 3). Also, there was no difference in the exploratory end points (Table 4). The readmission rates for HCM at 30 days and 60 days were lower in the postprotocol cohort, as is shown in Table 4.
Subgroup Analysis
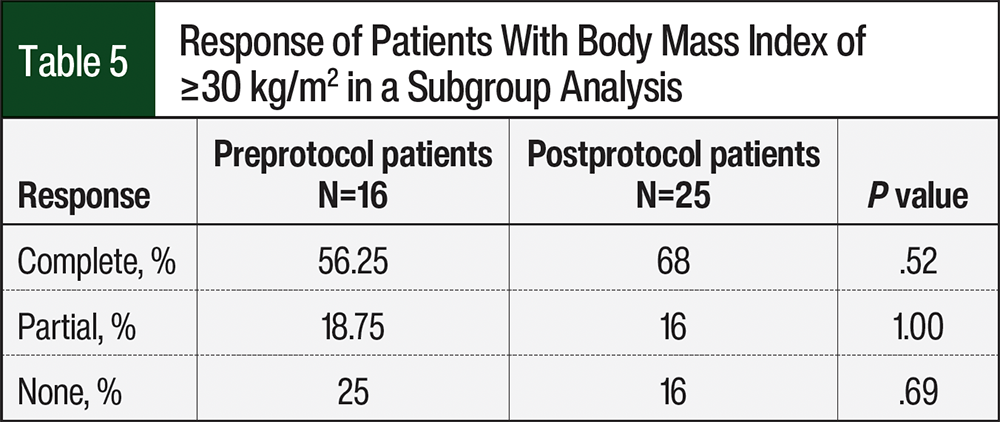
In a subgroup analysis of patients with a body mass index ≥30 kg/m2, there were 16 patients and 25 patients in the preprotocol and postprotocol groups, respectively (Table 5). The CR rate was numerically higher in the postprotocol group than in the preprotocol group (68% vs 56.25%, respectively; P=.52), the partial response was similar between the cohorts (16% vs 18.75%, respectively; P=1.00), and no response was higher in the preprotocol group (25% vs 16%; P=.69).
Cost Analysis
There was a total of 5.1 doses per admission, with an average of 1187 units per admission, in the preprotocol group versus 3.2 doses per admission, with an average of 792 units per admission, in the postprotocol group. The median cost of the treatment course was $7514 versus $7327 in the preprotocol and postprotocol groups (P=.33), with the mean cost of $11,583 versus $7437 in the preprotocol and postprotocol groups (P<.01). The mean difference in the cost per order between the preprotocol and postprotocol groups was $4100, resulting in a total of $520,700 cost-savings from 2018 to 2020 after the implementation of the calcitonin dosing protocol in 2017.
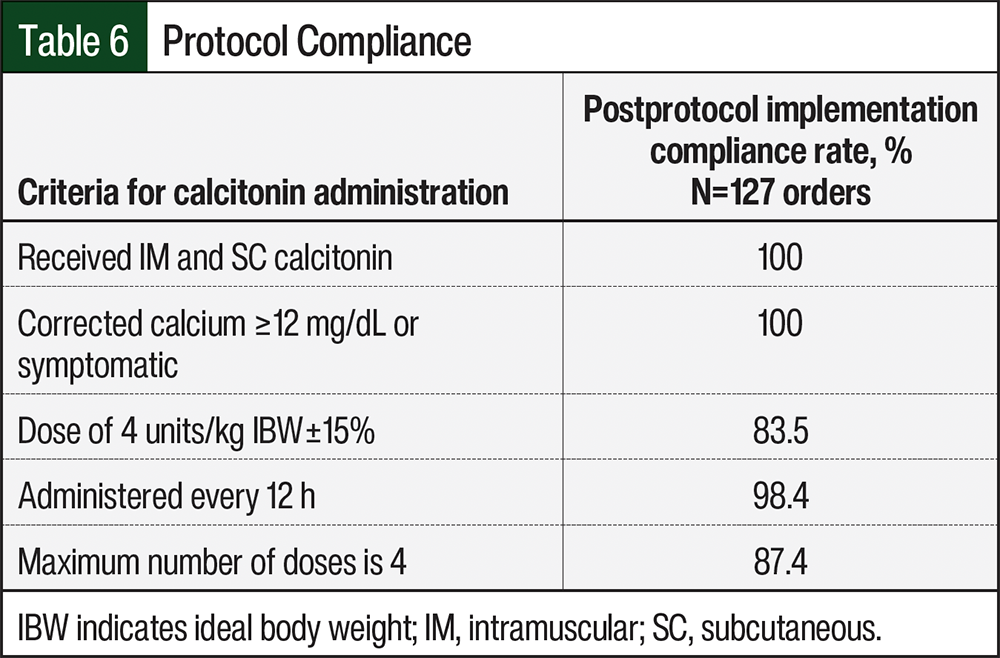
Protocol compliance was assessed with each of the 4 criteria to meet the administration of calcitonin. The restriction to the route of administration (IM and SC), as well as the restriction to give calcitonin only if the corrected calcium was ≥12 mg/dL or if the patient was symptomatic, was met at 100%. With the protocol implementation, 87.4% of orders were received, with a maximum of 4 calcitonin doses (Table 6). The dosing restriction criterion of calcitonin to 4 units/kg (±15%) was met in 83.5% of orders. The frequency of calcitonin to be given every 12 hours was met in 98.4% of orders. Table 6 shows details on the patients’ compliance to the protocol.
Discussion
The intention of the calcitonin dosing protocol was to reduce drug cost and use while not having a detrimental effect on the resolution of HCM. The implementation of this calcitonin dosing protocol at 2 tertiary hospitals resulted in similar rates of HCM resolution, including in patients with a body mass index of ≥30 kg/m2, while providing the institutions with substantial cost-savings. Treatment with bisphosphonates in the preprotocol and postprotocol periods was associated with higher CR rates compared with those who did not receive a bisphosphonate, reiterating that treatment with bisphosphonates is the mainstay of therapy for HCM. The main reason for patients not receiving a bisphosphonate was acute kidney injury, followed by patients recently receiving a bisphosphonate for the management of bone disease.
From 2013 to 2015, the total inpatient cost of calcitonin treatment increased from approximately $2.3 million to approximately $55.2 million.8 The unit price of calcitonin increased from approximately $68 per unit in 2013 to approximately $2286 per unit in 2015, which is an approximate 3263% increase in 2 years.8 As a result of this dramatic increase of cost, some institutions have implemented calcitonin-restrictive criteria to reduce unnecessary drug use and minimize the cost of care with calcitonin. The findings from 2 recent studies of the implementation of a calcitonin protocol were consistent with the findings from our study, which are summarized below.9,10
A 2020 study by Shah and colleagues assessed the clinical and economic implications of restricting calcitonin.9 Their institution’s restrictive calcitonin protocol included symptomatic hypercalcemia, ionized calcium >1.5 mmol/L and/or corrected calcium ≥13 mg/dL, a maximum number of 4 doses, and administration of calcitonin every 12 hours.9 This protocol also implemented a stop date for the calcitonin order, which required physicians to assess the appropriateness of continuing treatment with calcitonin after 2 doses. The results of Shah and colleagues’ study showed no difference in calcium resolution before and after the implementation of the protocol, with a cost-savings of approximately $460,251.9
Similarly, a 2022 study by Marte and colleagues evaluated their pharmacy-driven calcitonin protocol for the treatment of HCM, 2 years preprotocol and postimplementation of the protocol in 2016.10 This institution’s restriction criteria included a one-time calcitonin dose for a corrected calcium level of >14 mg/dL, followed by a serum calcium level 6 hours after administration. If the serum calcium level decreased after the administration of calcitonin, the pharmacist scheduled 3 additional doses of calcitonin. If the serum calcium level did not decrease, the pharmacist discontinued the calcitonin order per the protocol. The results of this study were consistent with the findings of our study and those of Marte and colleagues, and showed no difference in HCM resolution before and after the implementation of the protocol, with a significant cost-savings of approximately $294,000 in 1 year.10 Although our study did have a larger cohort in the preprotocol and postprotocol implementation period than in these studies,9,10 our results remained similar in that implementing a restrictive calcitonin protocol results in no difference in HCM resolution while also providing a significant cost-savings.
Although there were consistent findings in the studies by Shah and colleagues and Marte and colleagues in regard to the implementation of their institution’s pharmacy-driven calcitonin protocol, 1 addition that our study added was the use of an IBW for calcitonin dosing.9,10 With the low volume of distribution of calcitonin (0.15-0.3 L/kg), lower doses are expected to remain effective for the treatment of HCM.7 Most of our patients had a body mass index of 18.5 kg/m2 to <25 kg/m2, which accounts for approximately 39.7% and 47.2% of patients in the preprotocol and postprotocol groups, respectively. Approximately 20% of patients in the preprotocol and postprotocol groups were obese, with a body mass index of ≥30 kg/m2. The results of our exploratory subgroup analysis of patients who were obese showed that there was a similar trend in HCM resolution in the preprotocol and postprotocol groups. Although the use of IBW may be favorable to reduce costs on account of the recent price escalation of calcitonin, this dosing regimen remains controversial as a result of the limited data because there are currently no optimal dosing guidelines that are defined for patients who are obese; however, we hope that this will be explored in future studies to assess the differences in outcomes.
Other exploratory end points that were assessed included the readmission rates at 30 days and 60 days. The 30-day readmission rates in the preprotocol and postprotocol groups were 20.5% and 24.4%, respectively, and the 60-day readmission rates were 6.8% and 6.3%, respectively. There were fewer 30- and 60-day readmissions specifically for HCM in the postprotocol group versus the preprotocol group (Table 4), but this is unlikely to be related to the dosing protocol. In addition, future studies should investigate cost-savings and HCM resolution outcomes by further restricting treatment with calcitonin in patients with symptomatic HCM and a corrected calcium level of >14 mg/dL based on the recent guideline recommendations.3
With the implementation of the restriction criterion for a maximum of 4 doses, there was a significantly lower number of doses administered to our patients, which supports the belief that more calcitonin doses received does not equal a greater effect on hypercalcemia, because of the tachyphylactic effect after approximately 48 hours of treatment with calcitonin. In our study, at 48 hours, the corrected calcium level was reduced by 2.4 mg/dL and 2.7 mg/dL from baseline in the preprotocol and postprotocol groups, respectively. Although we expect tachyphylaxis at 48 hours, there was still a further reduction in corrected calcium at 72 hours, with 3.2-mg/dL and 3.4-mg/dL reductions from baseline in the preprotocol and postprotocol groups, respectively. This confirms that calcitonin, when received with other standard-of-care therapies including bisphosphonates and IV fluids, plays a large role in the resolution of HCM, resulting in the continued reduction of calcium after 48 hours of therapy. In our study, there was no difference in HCM resolution with this restriction criteria and a higher incidence of hypocalcemia, with rates of 9.6% and 7.8% in the preprotocol and postprotocol groups, respectively.
Limitations
This study has limitations, including its retrospective design, adherence rates to the protocol that were not as high as we anticipated, a difference between the sample size in the cohort populations, and inconsistent timing of the laboratory draws. After the implementation of the IBW dosing protocol for calcitonin, the hospital implemented an HCM electronic order set with built-in restriction criteria to aid in adherence. The main reason for the difference in sample size is that the oncology services at SJHMC have substantially grown in recent years.
Another limitation is the potential for confounding factors, such as treatment with bisphosphonates for other indications in patients with cancer or a lack of bisphosphonate administration during index hospitalization as a result of recent administration for an indication other than HCM. A total of 2 patients in the postprotocol group received denosumab, which can potentially limit the interpretation of corrected calcium reduction at the time of discharge and can pose a risk for a confounding factor. Our facility did not have a formal fluid protocol to guide providers; thus, fluids were administered at doses that were lower in our study than is recommended in the current guidelines.3 Despite these factors, patients responded to their multifaceted HCM regimen appropriately.
Although the length of stay was shorter in the postprotocol group, we cannot assume that this resulted from the IBW dosing protocol for calcitonin. In these patients, there are many other reasons for hospitalization other than HCM, including comorbid conditions at the time of admission, a new cancer diagnosis requiring extensive workup as an inpatient, as well as a difference in performance status and ECOG scores, for which patients may require a longer time to reach their baseline. These confounding factors contribute to the decision of discharging a patient, which would affect the patient’s length of stay.
When calculating the cost analysis, we were not able to capture drug waste; thus, were not able to accurately estimate the total cost of calcitonin, including waste. Because the calcitonin vials at our facility are multidose, we expect minimal drug waste, thus the minimal effect on the total cost. Because of the different aspects of our restrictive protocol, we were unable to determine if the use of IBW for the dose calculations for calcitonin or the maximum of 4 doses of calcitonin was the primary driver of the estimated cost-savings.
Conclusion
The price escalation of calcitonin over recent years has had a substantial financial effect on healthcare systems. This study’s results showed no difference in HCM resolution using an IBW dosing protocol for calcitonin at the tertiary hospitals in this study. In addition, there were significant annual cost-savings that totaled $260,350. To our knowledge, this is the first study reporting HCM resolution outcomes with calcitonin that is dosed based on IBW. Future research should focus on validating these outcomes in a prospective study.
Author Disclosure Statement
Dr Savaya, Dr Mychajlonka, Dr Radosevich, and Dr Patanwala have no conflicts of interest to report.
References
- Goldner W. Cancer-related hypercalcemia. J Oncol Pract. 2016;12:426-432.
- Rosner MH, Dalkin AC. Onco-nephrology: the pathophysiology and treatment of malignancy-associated hypercalcemia. Clin J Am Soc Nephrol. 2012;7:1722-1729.
- El-Hajj Fuleihan G, Clines AC, Hu MI, et al. Treatment of hypercalcemia of malignancy in adults: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2023;108:507-528.
- Chorin E, Rosso R, Viskin S. Electrocardiographic manifestations of calcium abnormalities. Ann Noninvasive Electrocardiol. 2016 Jan;21(1):7-9.
- Nardone R, Brigo F, Trinka E. Acute symptomatic seizures caused by electrolyte disturbances. J Clin Neurol. 2016 Jan;12(1):21-33.
- Vakiti A, Anastasopoulou C, Mewawalla P. Malignancy-Related Hypercalcemia. StatPearls Publishing; 2024.
- Miacalcin (calcitonin salmon) injection, for intravenous use [prescribing information]. Viatris; August 2021. Accessed May 22, 2022. https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?type=display&setid=89cd4f46-b88a-4c10-b25c-47352280dc4e
- NORC at the University of Chicago. Trends in Hospital Inpatient Drug Costs: Issues and Challenges. October 11, 2016. Accessed May 23, 2022. www.aha.org/system/files/2018-01/aha-fah-rx-report.pdf
- Shah SP, Peters MJ, Usery JB, Twilla JD. Clinical and economic implications of restrictions on calcitonin utilization in a health system. Hosp Pharm. 2020;55:164-167.
- Marte F, Sanchez M, Neel S, et al. Evaluation of a pharmacy-driven calcitonin protocol for treating hypercalcemia in hospitalized patients: a multisite study. J Pharm Pract. 2022;35:874-878.