Connecting Science to Practice
Due to the known pharmacokinetic impact of food on
abiraterone exposure, treatment guidelines state that abiraterone
can be dosed at 250 mg daily with a low-fat breakfast
as an alternative to the standard 1000-mg daily dose. The
lower dose strategy has potential benefits for decreasing financial
burden of treatment and improving patient adherence,
although clinical evidence supporting this dosing
strategy remains minimal. This retrospective review sought
to evaluate the effectiveness of these 2 dosing strategies on
prostate-specific antigen and radiographic progression-free
survival outcomes in men receiving abiraterone for the
treatment of metastatic prostate cancer. The results support
low-dose abiraterone as a reasonable alternative if cost or
compliance are limiting factors.
Prostate cancer is the most common cancer type in men, with an estimated 268,490 new diagnoses of prostate cancer in the United States in 2022, accounting for approximately 27% of new cancer diagnoses in male patients.1 Abiraterone, a common treatment for prostate cancer, is a potent antiandrogen that works as a selective irreversible inhibitor of cytochrome P-450c17, an enzyme required for androgen biosynthesis.2 The inhibition of dehydroepiandrosterone leads to the desired effect of decreased testosterone synthesis3; however, abiraterone also inhibits 17-hydroxyprogesterone, a required precursor for the synthesis of cortisol, necessitating that abiraterone be in combination with low-dose prednisone to provide sufficient cortisol for physiologic functioning.3 Abiraterone was initially used in the metastatic, castration-resistant cancer setting, but has recently gained a place in the metastatic, castration-sensitive cancer setting as well.2,4 The National Comprehensive Cancer Network (NCCN) guidelines list abiraterone as a preferred category 1 recommendation for the treatment of castration-sensitive prostate cancer (CSPC) and castration-resistant prostate cancer (CRPC).4
Abiraterone is FDA approved for the treatment of metastatic CRPC and metastatic CSPC as a 1000-mg oral dose taken once daily on an empty stomach with 5 mg of prednisone.3 Early clinical data in healthy individuals demonstrated a 5-fold higher area under the curve (AUC) when abiraterone was administered with a low-fat meal of 300 calories and a 10-fold higher AUC when abiraterone was administered with a high-fat meal of 825 calories compared with overnight fasting.3,5 The initial studies resulting in the FDA’s approval of abiraterone were conducted in the fasting state, leading to the current approved dosage of 1000 mg daily taken on an empty stomach at least 1 hour before or at least 2 hours after a meal.3
Because of the known effect of food on abiraterone exposure and the potential pharmacoeconomic implications of receiving low-dose abiraterone, Szmulewitz and colleagues conducted a randomized phase 2 study to compare the efficacy of 250-mg abiraterone taken with a conventional low-fat breakfast with the standard approved dosing of 1000 mg taken after an overnight fast of at least 8 hours and at least 2 hours before ingesting any food.6 Their study’s results showed that the lower dose of abiraterone taken with food was noninferior to standard-dose abiraterone in the primary outcome of prostate-specific antigen (PSA) reduction at 12 weeks.6
Because of the high cost of anticancer medications, the potential pharmacoeconomic implications of this lower-dose regimen for abiraterone are compelling. Szmulewitz and colleagues estimated a per-patient cost-savings of >$100,000 with the use of low-dose abiraterone in the metastatic CRPC setting.6 An analysis of Medicare Part D data showed that standardizing the prescribing of abiraterone to the low dose could result in an estimated annual savings of $700 million.7 In addition to a lower financial burden, this low-dose abiraterone regimen may also lead to enhanced compliance in patients who only need to receive 1 tablet instead of 4 tablets. Because of the potential reduced financial adverse events (AEs) and improved compliance, the NCCN guidelines have added a statement that abiraterone can be dosed at 250 mg daily after a low-fat breakfast as an alternative to 1000 mg daily, based on the available data.4
Despite a small (N=72) trial population and the use of a biochemical end point rather than a clinically validated surrogate end point, the results of the study by Szmulewitz and colleagues provided data that a lower dose of abiraterone taken with food may be as efficacious as standard dosing and has changed some prescribing practices.6 The results of a survey in India showed that 61.9% of 118 oncologists had already changed to prescribing abiraterone 250 mg in all or in limited-resource settings, whereas 28.8% of the oncologists indicated that they planned to change their practice to prescribe abiraterone 250 mg.8
Based on the available data, in combination with the need to lower financial AEs among their patients, some oncologists within our health system have transitioned to prescribing abiraterone 250 mg, whereas others continue to prescribe the approved dosage of 1000 mg daily. This study seeks to provide real-world data on the efficacy of low-dose abiraterone compared with standard dosing in patients with metastatic prostate cancer.
Methods
This multicenter, retrospective cohort study included patients with prostate cancer aged ≥18 years who received a prescription for abiraterone between July 23, 2018, and July 22, 2022. The patients who received a prescription for abiraterone 250 mg daily to be taken with food were included in the low-dose cohort. The patients who received a prescription for abiraterone 1000 mg daily were randomly selected for study inclusion and were initially matched in a 2:1 fashion to be included in the standard-dose cohort. The charts were reviewed from the patients’ initiation of abiraterone to December 31, 2022. The start date for all of the time-to-event analyses was the date of the initial abiraterone prescription for 250 mg for patients in the low-dose cohort and for 1000 mg in the standard-dose cohort. The unavailable data points were censored for patients receiving abiraterone past the observation period (beyond December 31, 2022). This investigation was approved by the Sanford Institutional Review Board.
The patients included in this study received abiraterone for the diagnosis of metastatic CSPC or metastatic CRPC. The patients with locally advanced prostate cancer who received abiraterone for a set duration of 2 years in combination with radiation therapy were excluded from this study. Patients who were receiving abiraterone at an outside facility for which the start of therapy date was unknown were not included in this analysis. Patients who were receiving abiraterone 250 mg as a dose modification as a result of AEs or at any dose modification because of hepatic impairment were also excluded.
The primary end point was radiographic progression-free survival (rPFS) in the low-dose cohort versus the standard-dose cohort. rPFS was defined as the time from the initiation of abiraterone to disease progression based on radiographic findings, as determined by the physician, or death from any cause. The secondary outcomes included PSA PFS, combined radiographic and PSA PFS, and the percent change in PSA at week 12. PSA progression was defined using the criteria set by the Prostate Cancer Clinical Trials Working Group as the time from the start of therapy to the first PSA increase that is ≥25% and ≥2 ng/mL above the nadir and is confirmed by a second value more than 3 weeks later. If no decline from baseline was documented, PSA progression was defined as a 25% increase from the baseline value, along with an increase in absolute value of ≥2 ng/mL after 12 weeks of treatment.
Statistical Analysis
The study outcomes were reported as count and percentage for the categorical variables. The continuous variables were summarized as median and interquartile range. Statistical comparisons were conducted using chi-square tests for the categorical variables and student t-tests for the continuous variables. A Kaplan–Meier survival analysis was used for all of the time-to-event analyses. A univariate analysis of variables of interest was also conducted.
Results
Of the 100 patients in this study, 24 received abiraterone 250 mg daily to be taken with food (ie, the low-dose group) and 76 received abiraterone 1000 mg daily to be taken on an empty stomach (ie, the standard-dose group). A total of 14 patients who were initially included in the low-dose cohort previously received abiraterone 1000 mg and were excluded from this analysis. The median patient age was 74 years in the low-dose group and 71.3 years in the standard-dose group (Table 1). The baseline PSA, as documented at the time of the initiation of abiraterone, was lower in the low-dose group than in the standard-dose group. There was a larger percentage of patients with an ECOG performance score of 0 in the low-dose group, whereas the standard-dose group had a larger percentage of patients with a Gleason score of 9 or 10 or that were not reported. The standard-dose group had a higher percentage of patients with metastatic CRPC than the low-dose group. Most of the patients were receiving abiraterone as their first or second line of treatment in the metastatic setting (Table 1).
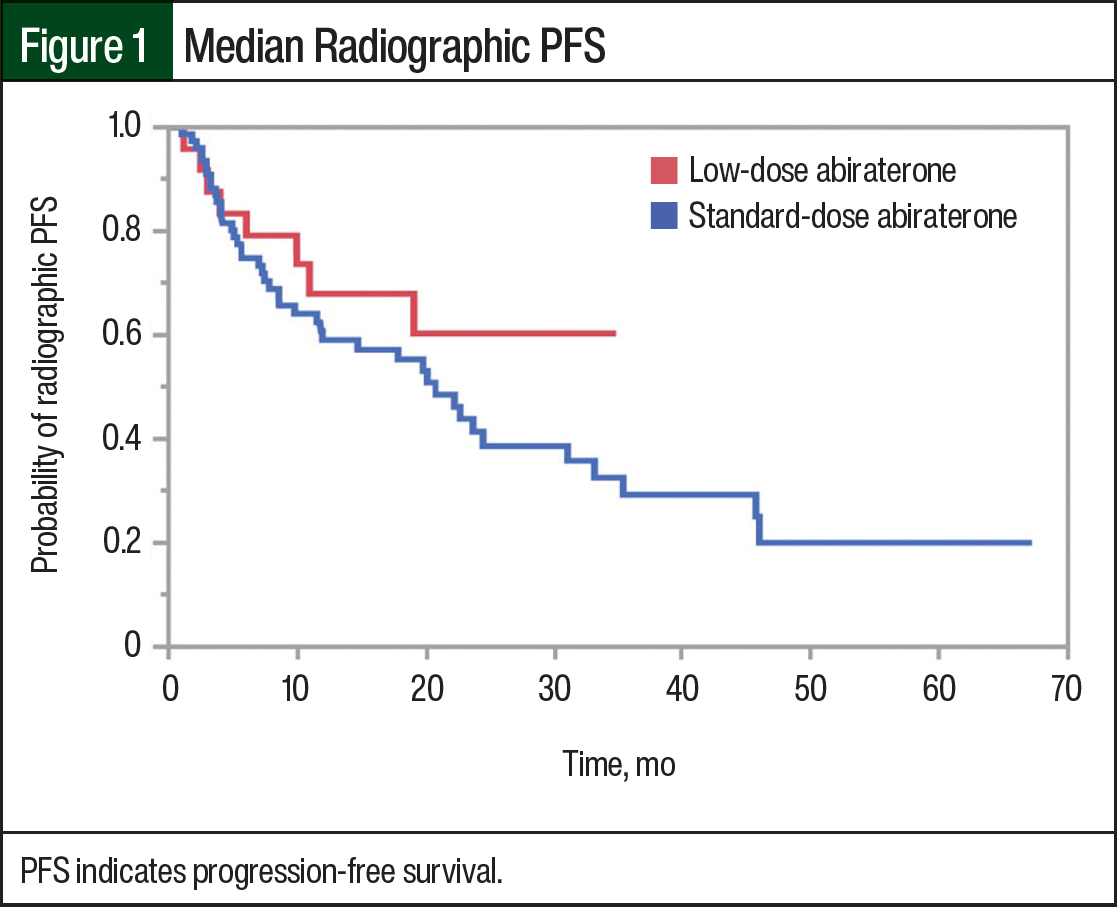
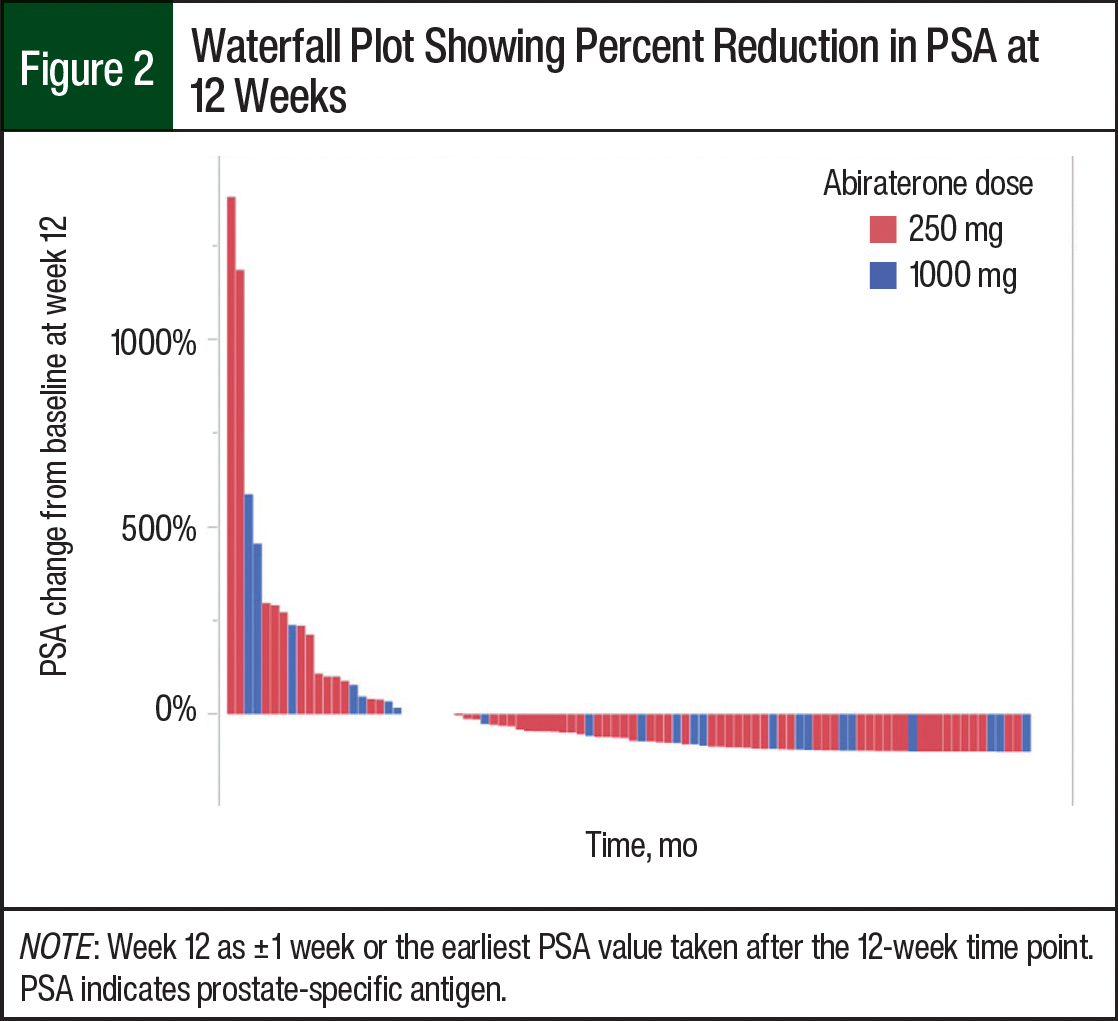
The median rPFS was 11.7 months in the low-dose abiraterone group and 11.6 months in the standard-dose abiraterone group (Figure 1). Of note, based on the sample sizes, a difference of 2 months in PFS (estimating a baseline rate of 12 months and a standard deviation of 3 months) was significant. The median PSA at week 12 was 0.9 ng/mL in the low-dose group and 1.09 ng/mL in the standard-dose group. In both groups, most of the patients had decreased or stable PSA levels at week 12 (Figure 2).
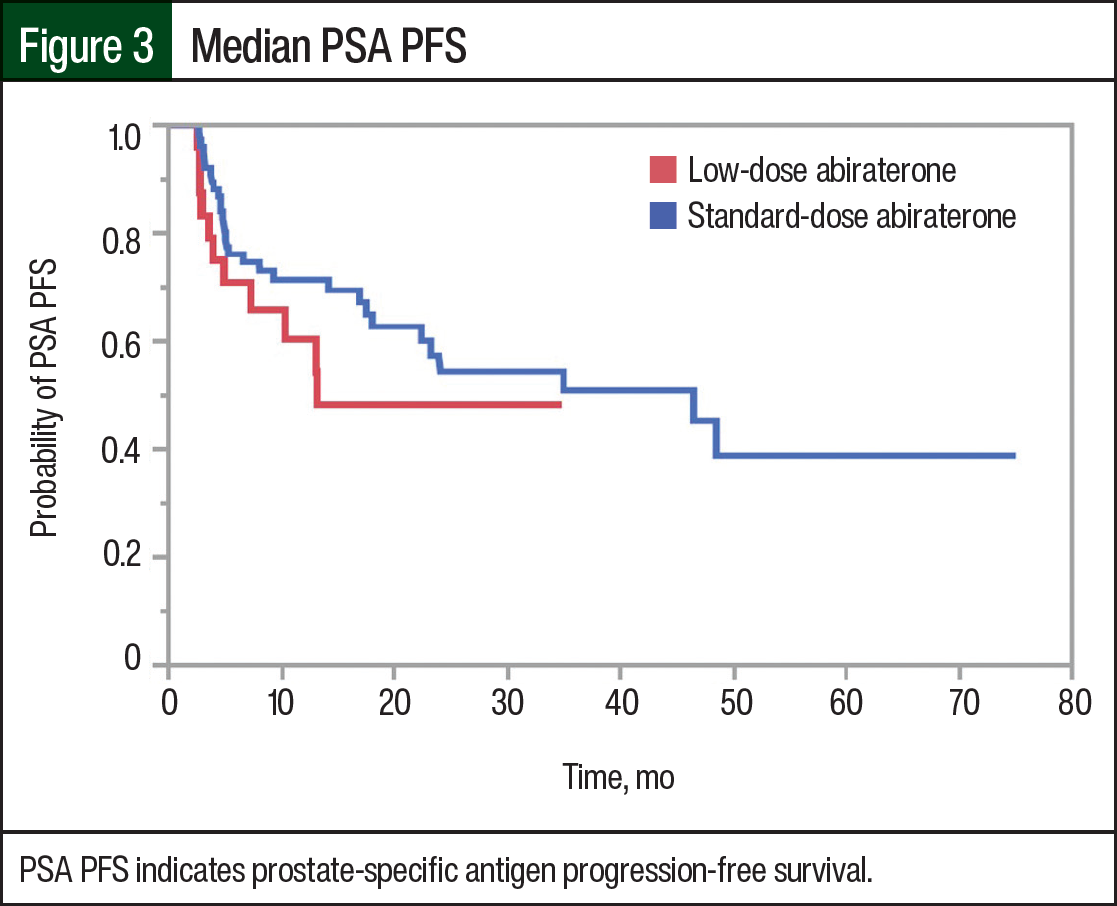
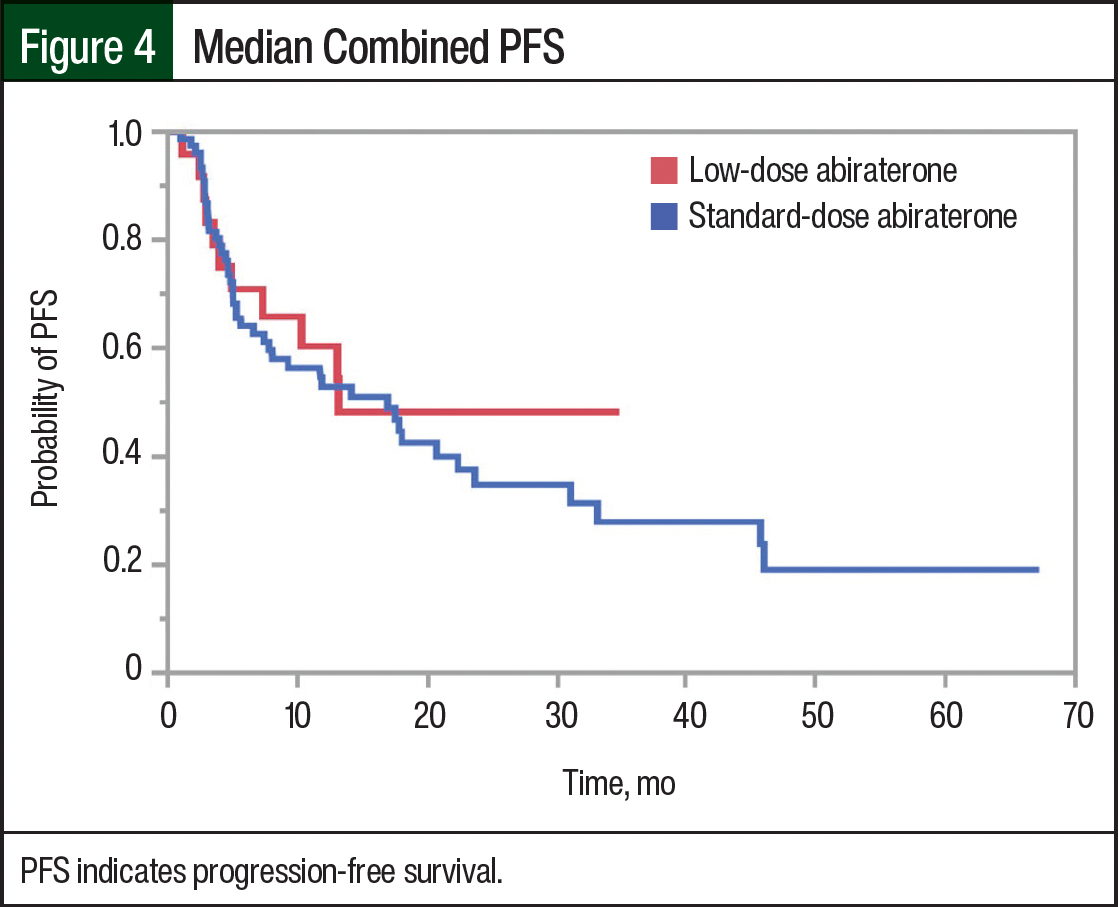
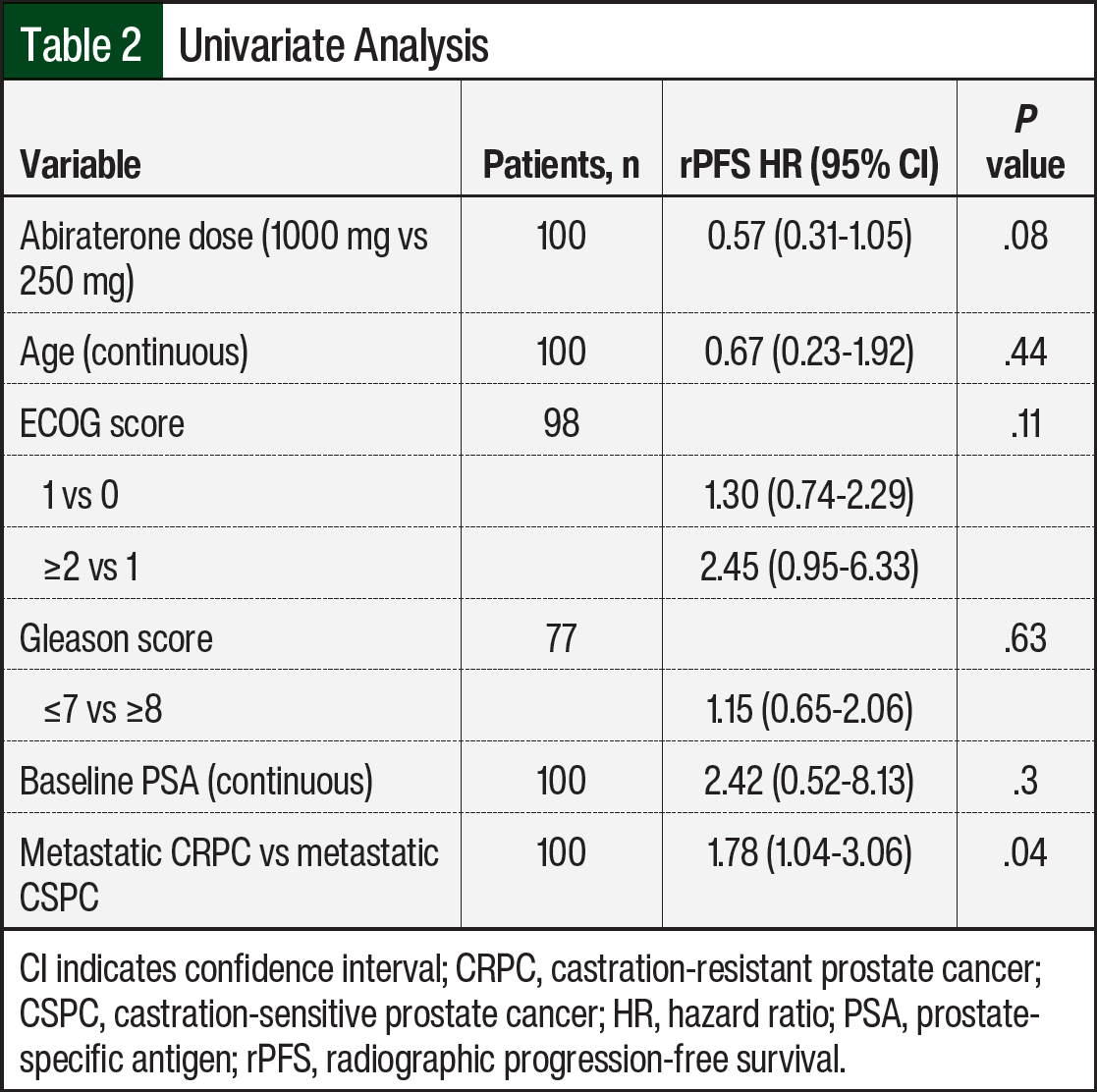
The median PSA PFS was 9.5 months in the low-dose abiraterone group and 13.1 months in the standard-dose abiraterone group (Figure 3). The median PFS encompassing radiographic and PSA progression was 9.5 months in the low-dose group and 7.95 months in the standard-dose group (Figure 4). The univariate analysis, including potential confounders and outcomes that may influence PFS, lacked statistical significance, except for metastatic CRPC versus metastatic CSPC status (Table 2).
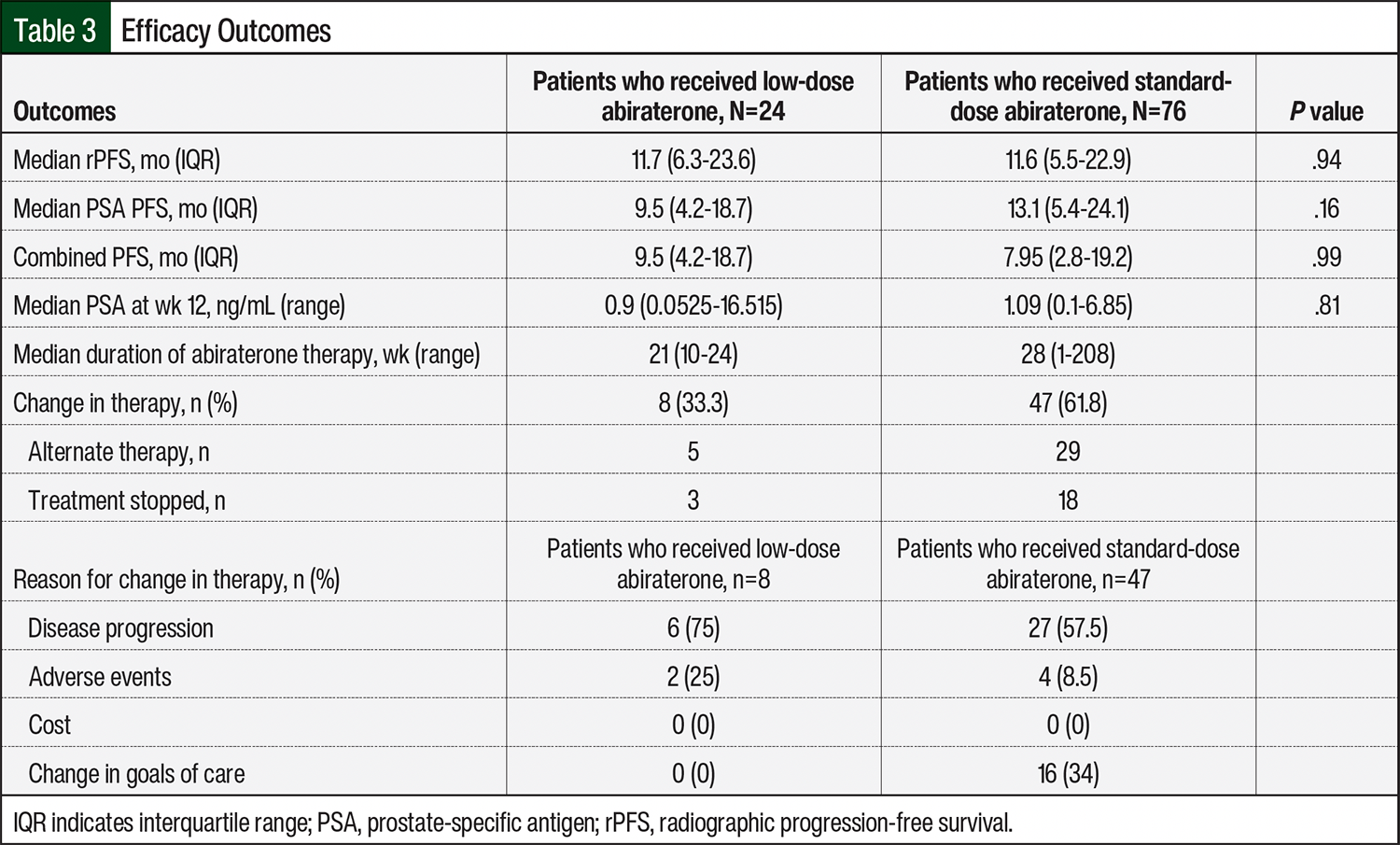
The median duration of abiraterone therapy was 21 weeks (range, 10-24 weeks) in the low-dose group and 28 weeks (range, 1-208 weeks) in the standard-dose group (Table 3). A total of 33.3% of the patients in the low-dose group had a change in therapy during the defined study period compared with 61.8% of patients in the standard-dose group. The remaining patients continued receiving treatment, and the data were censored at the study’s end date. Of the patients who changed therapy, the primary reason was disease progression, which occurred in 75% of the low-dose cohort and in 58.7% of the standard-dose cohort. Therapy was changed or discontinued as a result of a change in the goals of care in none of the patients in the low-dose group and in 32.6% of patients in the standard-dose group (Table 3).
Discussion
Although limited by a relatively small and heterogeneous patient population, there was no significant difference in radiographic or PSA PFS in this real-world comparison of low-dose abiraterone taken with food compared with standard-dose abiraterone taken on an empty stomach. These data provide an important addition to the current body of knowledge of abiraterone dosing in patients with prostate cancer.
Although a lower-dose abiraterone regimen can be appealing to patients and providers, clinical efficacy data that support this alternative dosing strategy for abiraterone remain limited in the literature.9 Meenu and colleagues conducted an open-label, crossover pharmacokinetic study that evaluated 17 patients with metastatic CRPC who received 250-mg abiraterone with milk (7% fat) compared with abiraterone 1000 mg in the fasted state.10 The patients’ Cmax and AUC were similar among the 2 dosing strategies, with the investigators concluding that the pharmacokinetics of abiraterone 250 mg with milk were similar to 1000-mg abiraterone in a fasted state and is a cost-effective alternative for future study.10
Szmulewitz and colleagues evaluated the alternative dosing regimen of 250-mg abiraterone with food in a phase 2 clinical study and concluded that this alternative dosing regimen was noninferior to standard 1000-mg abiraterone with fasting with respect to the primary outcome of change in PSA at 12 weeks.6 Despite the investigators admitting that this study was not powered for a practice-changing, clinically validated end point,6 the compelling pharmacoeconomic benefits of these data have led to prescribing changes in some settings.7,8 Although a randomized phase 3 trial would be ideal, no additional clinical studies have been undertaken. Therefore, there is a need for centers that are using this alternative abiraterone dosing strategy to share data to further elucidate its place in therapy.
Similar outcomes were observed in rPFS or PSA PFS among the different dosing regimens in this analysis. Given our cohort size, a PFS difference of at least 2 months would have been required to demonstrate statistical significance. Although PSA PFS was longer in our study than the 8.6 months with this dosing regimen in the study by Szmulewitz and colleagues,6 our analysis also included patients with CSPC. Patients with metastatic CSPC have a longer median PFS than patients with metastatic CRPC.2,11 The standard-dose group had a slightly higher percentage of patients with metastatic CRPC than the low-dose group, which is a potential confounder (P=.04).
As with all large phase 3 clinical studies that have been conducted with abiraterone 1000 mg in the fasting state, it would be prudent to use standard-dose abiraterone in patients with adequate insurance coverage and no adherence concerns. However, the financial AEs of anticancer therapy can be significant, and this lower-dose regimen for abiraterone may provide a feasible path for affordable medication access. A patient-assistance program is no longer offered for abiraterone from the manufacturer, which leaves patients with fewer options for affordable therapy.
One resource for affordable medication access is Mark Cuban Cost Plus Drug Company (ie, Cost Plus Drugs). At the time of this writing, abiraterone is offered through Cost Plus Drugs at a price of approximately $27 plus shipping and taxes for a 1-month, 30-tablet supply of the low-dose (250-mg) regimen, whereas the cost of a 1-month, 120-tablet supply of the standard-dose (250-mg) regimen is approximately $98.12 The use of this program could result in an annual savings of $800, and for patients who may be receiving treatment with abiraterone for many months to years, this alternative regimen may significantly alleviate the financial barriers to treatment. Therefore, in patients where cost is prohibitive to receiving this therapy, the previously mentioned available data and the findings of this study do not show a notable concern regarding the use of an alternative dosing regimen of 250-mg abiraterone taken with a low-fat meal.6,9,10
Limitations
Although the best-available data were used, provider documentation was variable, which is a limitation inherent to this study’s retrospective design. The frequency of follow-up appointments, laboratory testing, and imaging were also variable, which may affect PFS outcomes.
The lack of data regarding whether any patients in our cohort received docetaxel with abiraterone and androgen-deprivation therapy based on the PEACE-1 trial is also a limitation.13 This triplet regimen was not yet widely used during the study period, but its use cannot be fully ruled out.
Another limitation of this study was the ability to confirm patients’ adherence to the instructed recommendations to take abiraterone with food, given the retrospective nature of the study. In the previous analysis of this dosing regimen, patients in the low-dose group were instructed to receive abiraterone with a conventional low-fat breakfast, and they kept food diaries to monitor adherence.6 What constitutes a low-fat meal when left up to a patient’s discretion also may vary. However, the inclusion of patients regardless of their diet adherence expands the external validity of the study and feasibility of the regimen in standard practice.
Finally, despite attempts to control for potential confounding, the small sample size of this study remains a limitation, and the results should be interpreted with caution, because the cohort size provides insufficient power to use a noninferiority design.
Conclusion
rPFS, PSA PFS, and combined PFS were similar in the low-dose abiraterone group versus the standard-dose abiraterone group. The cost of anticancer therapies remains a great patient concern that this alternative dosing regimen for abiraterone could help mitigate. Although this study is exploratory in nature because of its limited sample size and heterogeneous patient population, these results provide additional clinical evidence to support treatment with low-dose abiraterone as a reasonable alternative to standard-dose abiraterone if cost or compliance limit the patient’s ability to adhere to standard-dose abiraterone.
Author Disclosure Statement
Dr Hulterstrum, Dr Grindeland, and Dr Czmowski have no conflicts of interest to report.
References
- Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72:7-33.
- Fizazi K, Tran N, Fein L, et al. Abiraterone plus prednisone in metastatic, castration-sensitive prostate cancer. N Engl J Med. 2017;377:352-360.
- Zytiga (abiraterone acetate) tablets, for oral use [prescribing information]. Janssen Biotech; November 2024. Accessed January 14, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2021/202379s035lbl.pdf
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): prostate cancer. Version 4. 2024. May 17, 2024. Accessed September 4, 2024. www.nccn.org/professionals/physician_gls/pdf/prostate.pdf
- Chi KN, Spratlin J, Kollmannsberger C, et al. Food effects on abiraterone pharmacokinetics in healthy subjects and patients with metastatic castration-resistant prostate cancer. J Clin Pharmacol. 2015;55:1406-1414.
- Szmulewitz RZ, Peer CJ, Ibraheem A, et al. Prospective international randomized phase II study of low-dose abiraterone with food versus standard dose abiraterone in castration-resistant prostate cancer. J Clin Oncol. 2018;36:1389-1395.
- Premnath N, Sedhom R, Gupta A. Potential cost savings with low-dose abiraterone in the United States. JCO Glob Oncol. 2020:743-744.
- Patel A, Tannock IF, Srivastava P, et al. Low-dose abiraterone in metastatic prostate cancer: is it practice changing? Facts and facets. JCO Glob Oncol. 2020:382-386.
- Dey T, Goyal S, Periasamy K, Madan R. Is low-dose abiraterone for prostate cancer an attractive strategy for limited resource settings? Ind J Med Paediatr Oncol. 2022;43:40-46.
- Meenu M, Sahoo RK, Seth A, et al. Food enhanced pharmacokinetics for clinical translation of low dose abiraterone acetate in metastatic castration-resistant prostate cancer. J Cancer Sci Clin Ther. 2020;4:314-324.
- de Bono JS, Logothetis CJ, Molina A, et al. Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med. 2011;364:1995-2005.
- Abiraterone acetate. Costplusdrugs.com. Accessed September 9, 2024. costplusdrugs.com/medications/abiraterone-acetate-250mg-tablet/
- Fizazi K, Foulon S, Carles J, et al. Abiraterone plus prednisone added to androgen deprivation therapy and docetaxel in de novo metastatic castration-sensitive prostate cancer (PEACE-1): a multicentre, open-label, randomised, phase 3 study with a 2×2 factorial design. Lancet. 2022;399:1695-1707.