Connecting Science to Practice
This study investigated whether the type of ifosfamide patients
receive—powder that is reconstituted versus premixed
solution—affects the risk for ifosfamide-related
neurotoxicity and encephalopathy (INT/E). We reviewed
records of >200 adult patients who received ifosfamide
and compared outcomes between the 2 formulations. We
found that patients who received the solution formulation
had a much higher rate of INT/E than those who received
the reconstituted powder. For everyday practice, this
means that pharmacists and providers should consider
drug formulation as a risk factor when selecting ifosfamide,
especially in patients who may already have other
risk factors for neurotoxicity.
Ifosfamide is an alkylating agent used in the treatment of hematologic and solid-tumor malignancies.1-4 In addition to the well-known class-related adverse events (AEs) of alkylating agents, including myelosuppression, nausea and vomiting, alopecia, infertility, and secondary malignancies, ifosfamide carries the additional notable AEs of hemorrhagic cystitis and neurotoxicity/encephalopathy.1,2 Although hemorrhagic cystitis has been well-described in the literature, with effective management strategies, including the coadministration of mesna,1,2 less is known about the risk factors and management of ifosfamide-associated neurotoxicity/encephalopathy (INT/E).
The presence of neurotoxicity is caused by the accumulation of the toxic metabolite chloroacetaldehyde, which readily crosses the blood–brain barrier.2 Chloroacetaldehyde is hypothesized to cause direct neurotoxic damage through the depletion of central nervous system glutathione and the inhibition of mitochondrial phosphorylation through disruption of long-chain fatty acid oxidation.2,5-8 The clinical presentation of INT/E can range from mild to severe and may include symptoms such as lethargy, confusion, hallucinations, and seizures.2 The incidence of INT/E ranges from 10% to 40%; although the symptoms of INT/E are reversible in most cases, there have been reports of severe complications, such as coma, and death.9 Although the optimal management strategies for the AEs related to INT/E are unknown, methylene blue, thiamine, and albumin have been used for the prevention and treatment of severe INT/E.2,10-13
Ifosfamide is available as a powder for reconstitution and as a solution for injection.1 Although all ifosfamide drugs have a boxed warning for INT/E, evidence regarding the risk factors for this AE is scarce.1,2 Previously, the proposed risk indicators for INT/E include hypoalbuminemia, renal impairment, hepatic insufficiency, and the coadministration of aprepitant.1,2,12 At our institution, Vanderbilt University Medical Center, we observed a greater incidence of INT/E with solution for injection ifosfamide than with reconstituted powder ifosfamide after a formulary change to the solution formulation. This drug interchange was a consequence of Hurricane Maria’s devastation of Puerto Rico in September 2017, which resulted in shortages of 0.9% sodium chloride small-volume bag and the subsequent increased and unsustainable demand for sterile water for injection, which is a critical component of the preparation of intravenous medications, including ifosfamide. As such, this limited access to sterile water for injection prompted a change to the ready-to-use solution of ifosfamide as the preferred formulation.
The results of a pediatric hematology/oncology study in France included 55 patients aged 3 months to 19 years who received a total of 237 courses of ifosfamide for sarcoma or acute lymphoblastic leukemia.14 The study’s results showed that the incidence of INT/E with the reconstituted powder formulation of ifosfamide was significantly lower than with the ifosfamide solution for injection (4.3% vs 21.1%, respectively; P<.001).14 In addition, Hillaire-Buys and colleagues published a case-control study on pediatric patients who received ifosfamide at 25 academic medical centers throughout France.15 The study showed an increased rate of encephalopathy with the reconstituted powder formulation compared with the solution formulation (adjusted odds ratio, 1.91; 95% confidence interval, 1.03-3.53).15 In a retrospective study of 191 French adults receiving ifosfamide, the rates of INT/E were significantly higher in those receiving the solution for injection formulation than the reconstituted powder formulation (1.9% vs 10.2%, respectively; P=.014).16 Other characteristics of patients with INT/E versus those without INT/E were identified in their study, including a longer infusion time (12 hours vs 3 hours, respectively; P<.001) and a more frequent poor performance status (54.5% vs 13.9%, respectively; P=.002).16
The objective of this study was to investigate the incidence of INT/E in association with the 2 available pharmaceutical formulations of ifosfamide in the United States and to dictate the selection of a preferred ifosfamide drug at our institution. Additional objectives of the study included evaluating select outcomes and the potential risk factors associated with the development of INT/E. To the best of our knowledge, this is the largest retrospective review of adults in the United States who had INT/E while receiving ifosfamide.
Methods
This was a single-center, institutional review board–approved, retrospective-cohort study at an academic medical center, Vanderbilt University Medical Center. Patients were identified for study eligibility based on the ifosfamide orders in the electronic medical record. The study patients were aged ≥18 years and received at least 1 dose of either of the available pharmaceutical formulations of ifosfamide from November 1, 2017, to September 30, 2021. The investigators received no financial support for research, authorship, or publication of this article.
The primary outcome of the study was the incidence of INT/E. The secondary outcome of the study was to identify the potential risk factors for INT/E.
Statistical Analysis
Statistical analyses were completed using R statistics software, version 3.6.1 (R Foundation for Statistical Computing; Vienna, Austria). Descriptive statistics were used to report the percentages, medians, and interquartile ranges. Univariate analyses were conducted using the Pearson test for nominal data and the Wilcoxon rank-sum test for continuous data, with an alpha significance level of 0.05 used for both tests.
Results
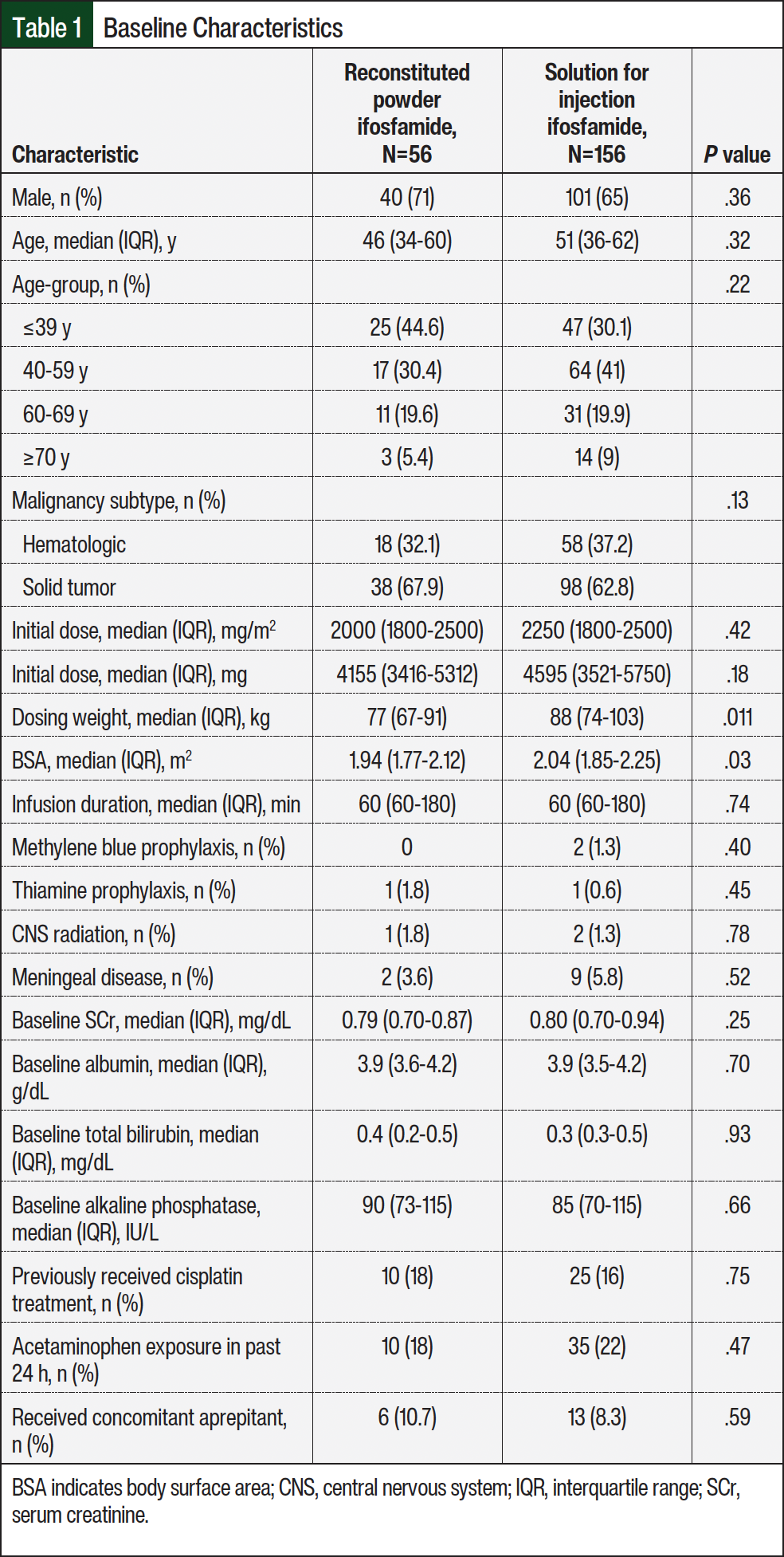
A total of 218 patients received ifosfamide within the specified time frame at our institution. In all, 6 patients were excluded for being aged <18 years. Of this remaining population of 212 adults, 156 (73.6%) patients received the solution for injection formulation of ifosfamide and 56 (26.4%) patients received the reconstituted powder formulation of ifosfamide.
The patients’ baseline characteristics were similar between the 2 treatment groups (Table 1). In the overall population, the median age was 48.5 years, and most patients were aged ≤39 years or aged between 40 years and 59 years. Approximately 68% of the patients were men. The most frequent indication for ifosfamide was a solid-tumor malignancy, which accounted for 64% of the patients. Overall, the most common solid-tumor diagnosis was sarcoma, and the most common hematologic cancer diagnosis was diffuse large B-cell lymphoma. A small percentage of patients in the reconstituted powder and solution groups (1.8% and 1.9%, respectively) received methylene blue or thiamine as prophylaxis for INT/E. Although the dosing weight and body surface area were statistically higher in the solution group, their initial dose of ifosfamide was similar to those receiving the reconstituted powder formulation.
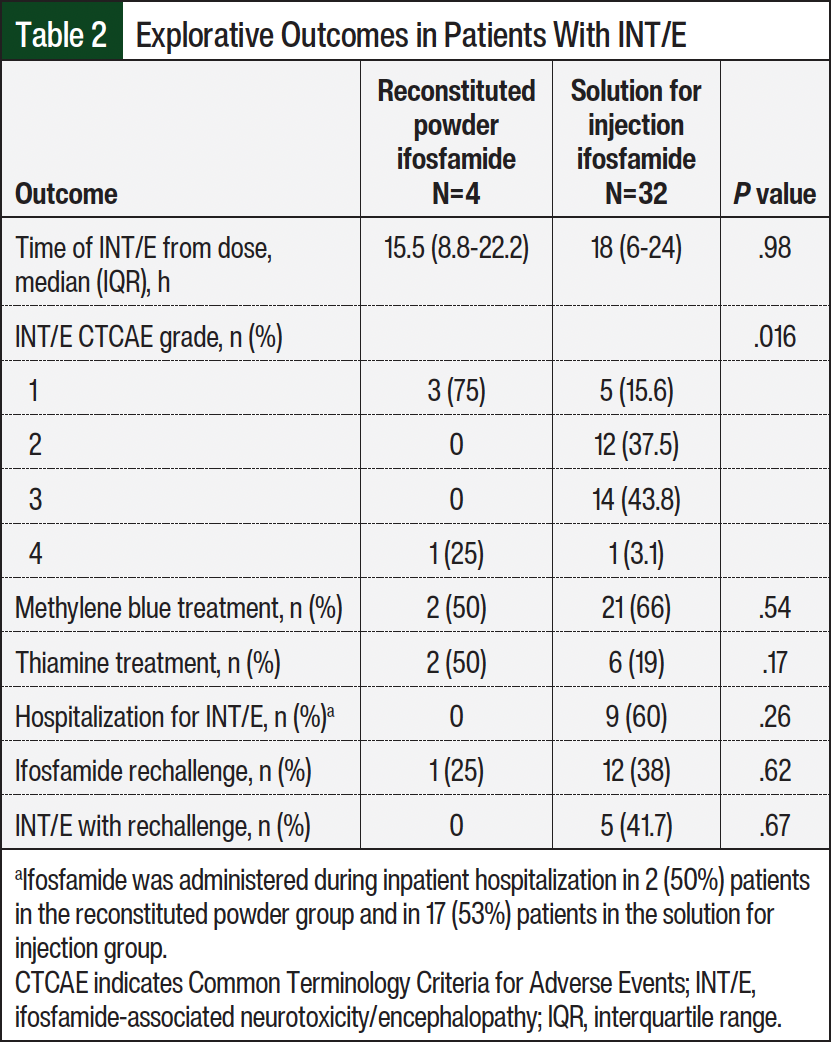
For the primary outcome, approximately 17% of patients in the overall population had INT/E. When broken down by cohort, the incidence of INT/E was significantly more common in the patients who received the solution for injection (n=32; 20.5%) compared with the powder for reconstitution (n=4; 7.1%), representing a number needed to harm of 7 (P=.022). When characterizing the exploratory outcomes in the patients who had INT/E, the time from the dose of ifosfamide to an AE was similar between the groups (15.5 hours vs 18 hours, respectively; P=.98; Table 2). Of the patients who had INT/E and received ifosfamide in the outpatient setting, 60% required hospitalization for AE monitoring and/or management, whereas none of the patients in the reconstituted powder group required inpatient admission. Grade 3 or 4 neurotoxic events (defined by the Common Terminology Criteria for Adverse Events, version 5.017) were significantly higher in the patients who received the solution formulation of ifosfamide versus the powder for reconstitution, with 15 (46.9%) of the 32 patients who had INT/E having grade 3 or 4 events (P=.016; Table 2).
For the treatment of INT/E, all patients in the reconstituted powder formulation cohort received methylene blue or thiamine, whereas 15% of those in the solution formulation group did not receive treatment with methylene blue or thiamine. Of the patients who had INT/E and continued ifosfamide therapy, 41.7% of those receiving the solution formulation had INT/E with rechallenge whereas none of the patients who received the reconstituted powder formulation did (Table 2).
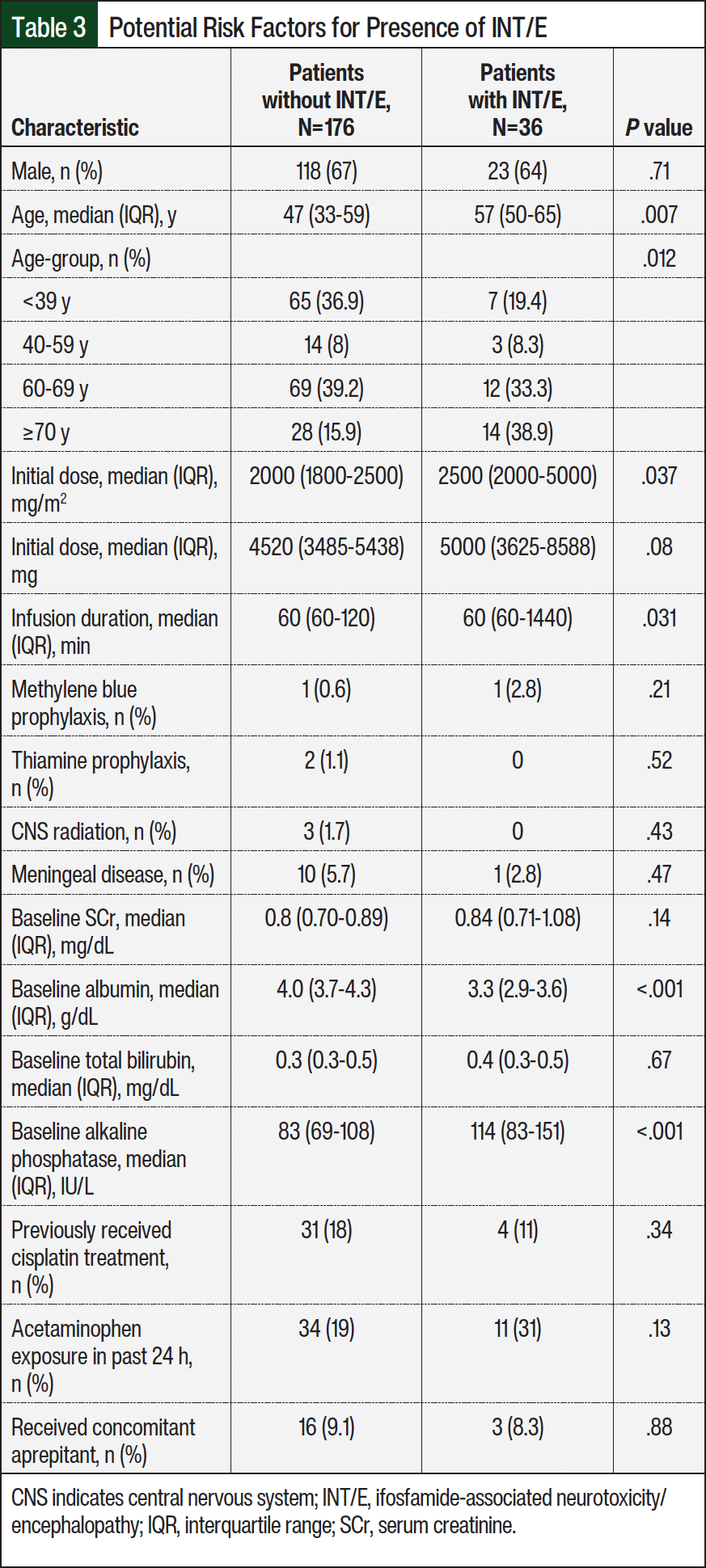
The secondary outcomes or additional risk factors for the presence of INT/E are shown in Table 3. The median age of the patients with INT/E was 57 years versus 47 years in those without INT/E (P=.007). Patients aged <39 years were less likely to have INT/E whereas patients aged >70 years were more likely to have INT/E (P=.012). Receiving higher daily doses of ifosfamide was also associated with increased rates of INT/E (P=.037). Other risk factors that resulted in a significant difference in the cohort with INT/E were longer infusion duration (P=.031), lower baseline albumin levels (3.3 g/dL vs 4 g/dL, respectively; P<.001), and higher baseline alkaline phosphatase levels (114 U/L vs 83 U/L, respectively; P<.001) with the solution for injection. The between-group differences noted for all other variables tested were not statistically significant, including other baseline demographics, the use of prophylaxis, central nervous system radiation, having meningeal disease, baseline serum creatinine and bilirubin, previous cisplatin exposure, recent acetaminophen use, or concomitant aprepitant administration.
During the aftermath of Hurricane Maria, a formulary change was implemented and the ifosfamide solution was chosen as the preferred drug formulation because of the shortage of sterile water for injection and the need for conservation strategies. With the switch to the solution, there were cost and operational benefits to the pharmacy workflow. Although the ifosfamide solution vials are ready to use straight from the manufacturer, the powder formulation requires reconstitution by the pharmacy team, which can take a significant amount of time and can negatively impact productivity. After this formulary change, clinicians noticed an increase in the episodes of INT/E in patients with hematologic malignancies.
Our work supports the current published evidence with the incidence of INT/E being significantly more common in patients who receive the solution for injection formulation. As suggested in other reports, this may be mechanistically related to drug stability and the rate of conversion to metabolites, including chloroacetaldehyde, and another neurotoxic metabolite, chloroethylamine.6,8 It is postulated that as the solution degrades over time, the formation of chloroethylamine is increased, resulting in a heightened frequency of INT/E.16 The results of this study may also help identify patients who have a higher risk for INT/E, particularly older (aged ≥60 years) patients who are receiving higher doses of ifosfamide over a longer (>1 hour) duration. In response to these data, the ifosfamide powder for reconstitution formulation has been reinstated as the preferred drug at our institution.
Limitations
This study has limitations. This is a single-center study with a relatively small sample size, which may have prevented some comparisons from reaching statistical significance. In addition, this was a retrospective review of the electronic medical records that relied on the documentation of INT/E as well as on clinical manifestations and grading, which may have been missed if not thoroughly recorded. Last, because of the retrospective nature of this study, knowledge of specific manufacturers of each drug were unknown and thus were not collected, analyzed, nor reported in this article.
Conclusion
Neurotoxicity and encephalopathy are well-known, concerning AEs associated with ifosfamide treatment, but without well-defined prevention and management strategies. These AEs can potentially be dangerous and life-threatening and can lead to therapy discontinuation and impact the ability to effectively treat a patient’s malignancy. This study sought to reduce the risk for INT/E, improve patient safety, and identify if the ifosfamide drug formulation may be implicated in AE development after anecdotal evidence suggests it may be a risk factor.
During the aftermath of Hurricane Maria, a formulary change was implemented and the ifosfamide solution was chosen as the preferred drug formulation because of the shortage of sterile water for injection and the need for conservation strategies After this change, clinicians noticed an increase in the episodes of INT/E in patients with hematologic malignancies.
Our work supports the current published evidence with the incidence of INT/E being significantly more common in patients who receive the solution for injection formulation. These results may also help identify patients who have a higher risk for INT/E, particularly patients aged >60 years who are receiving higher doses of ifosfamide over a longer duration (>1 hour). In response to these data, the ifosfamide powder for reconstitution formulation has been reinstated as the preferred drug at our institution.
In addition to a patient safety benefit, there may be decreased supportive care and lower admission-related costs to the institution with use of ifosfamide. This could be cost avoidance associated with the medication treatment utilized for INT/E, including methylene blue and thiamine, plus a reduction in urgent admissions for disease management. In the patients who receive the solution for injection formulation of ifosfamide and have low-grade INT/E, our work potentially supports the interchange to the reconstituted powder formulation, so the patient may continue to safely receive the intended therapy.
At our institution, patients with hematologic malignancies who received ifosfamide had significantly higher rates of INT/E with the solution for injection compared with the reconstituted powder formulation. These findings support the use of reconstituted powder as the preferred ifosfamide drug formulation as a result of increased patient safety and potential cost avoidance.
Author Disclosure Statement
Dr Perciavalle is currently employed and owns stock in Legend Biotech; Dr Keedy is a consultant to Epizyme and Deciphera; Dr Abernathy, Dr McCabe, Dr Cortez, Dr Miller, and Dr Yelvington have no conflicts of interest to report.
References
- Ifex (ifosfamide) injection, for intravenous use [prescribing information]. Baxter Healthcare Corporation; December 2024. Accessed February 19, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2024/019763s021lbl.pdf
- Ajithkumar T, Parkinson C, Shamshad F, Murray P. Ifosfamide encephalopathy. Clin Oncol (R Coll Radiol). 2007;19:108-114.
- Hagemeister FB. The role of ifosfamide in the treatment of lymphomas. Leuk Lymphoma. 1999;34:433-441.
- Frustaci S, De Paoli A, Bidoli E, et al. Ifosfamide in the adjuvant therapy of soft tissue sarcomas. Oncology. 2003;65(suppl 2):80-84.
- Nicolao P, Giometto B. Neurological toxicity of ifosfamide. Oncology. 2003;65(suppl 2):11-16.
- Küpfer A, Aeschlimann C, Cerny T. Methylene blue and the neurotoxic mechanisms of ifosfamide encephalopathy. Eur J Clin Pharmacol. 1996;50:249-252.
- McVay JI, Wood AM. Suspected ifosfamide-induced neurotoxicity. Pharmacotherapy. 1999;19:1450-1455.
- Chatton JY, Idle JR, Vågbø CB, Magistretti PJ. Insights into the mechanisms of ifosfamide encephalopathy: drug metabolites have agonistic effects on alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA)/kainate receptors and induce cellular acidification in mouse cortical neurons. J Pharmacol Exp Ther. 2001;299:1161-1168.
- Sweiss KI, Beri R, Shord SS. Encephalopathy after high-dose ifosfamide. Drug Safety. 2008;31:989-996.
- Pelgrims J, De Vos F, Van den Brande J, et al. Methylene blue in the treatment and prevention of ifosfamide-induced encephalopathy: report of 12 cases and a review of the literature. Br J Cancer. 2000;82:291-294.
- Abahssain H, Moukafih B, Essangri H, et al. Methylene blue and ifosfamide-induced encephalopathy: myth or reality? J Oncol Pharm Pract. 2021;27:143-149.
- Müngen E, Yaman Bajin, I, Öz S, et al. Ifosfamide-induced encephalopathy with rapid response to thiamine: a pediatric case. J Pediatr Hematol Oncol. 2022;44:402-404.
- Kataria PS, Kendre PP, Patel AA. Ifosfamide-induced encephalopathy precipitated by aprepitant: a rarely manifested side effect of drug interaction. J Pharmacol Pharmacother. 2017;8:38-40.
- Filhon B, Lacarra B, Hervouet C, et al. Ifosfamide-induced encephalopathy due to a novel formulation of ifosfamide. Pediatr Blood Cancer. 2016;63:372-373.
- Hillaire-Buys D, Mousset M, Allouchery M, et al. Liquid formulation of ifosfamide increased risk of encephalopathy: a case-control study in a pediatric population. Therapies. 2020;75:471-480.
- Chambord J, Henny F, Salleron J, et al. Ifosfamide-induced encephalopathy: brand-name (Holoxan) vs generic formulation (Ifosfamide eg). J Clin Pharm Ther. 2019;44:372-380.
- US Department of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE). Version 5.0. November 27, 2017. Accessed April 10, 2023. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf