These retrospective study findings were previously presented as an abstract and poster at the March 2022 American Pharmacists Association Annual Meeting and as an encore poster presentation at the September 2022 National Association of Specialty Pharmacy Annual Meeting. Prospective study findings were previously presented as an abstract and poster at the 2024 Hematology/Oncology Pharmacy Association Annual Conference.
Connecting Science to Practice
Patients who receive poly (ADP-ribose) polymerase (PARP)
inhibitors frequently have adverse events (AEs) after the start
of therapy that can result in changes in treatment. By identifying
and addressing these AEs early, patients’ adherence to
therapy can improve and fewer changes in therapy may be
needed. This study examined pharmacists who closely
watched for and managed AEs and how the AEs affected
medication adjustments, emergency department visits, and
hospital stays in the first 90 days of patients starting PARP
inhibitor therapy. This article highlights a novel strategy in
addressing AEs and demonstrates the utility of pharmacists
as part of the interdisciplinary healthcare team.
Poly (ADP-ribose) polymerase (PARP) inhibitors are used to treat various cancers including ovarian, breast, prostate, and pancreatic. Although each of the 4 PARP inhibitors (olaparib, niraparib, rucaparib, and talazoparib) have varying FDA-approved indications and adverse event (AE) profiles, the class shares common AEs, including fatigue, hematologic AEs, and gastrointestinal disorders.1-3 AEs often occur soon after the initiation of PARP inhibitor therapy and start to improve approximately 8 to 12 weeks after initiation.1,3 However, AEs are frequently the cause of PARP inhibitor dose interruptions and dose reductions, and severe AEs can lead to the discontinuation of therapy.2,3
Across clinical trials of PARP inhibitor treatment, AEs were responsible for up to 65% of dose interruptions,4,5 up to 55% of dose reductions,4 and up to 20% of treatment discontinuations.6 In a real-world study by Eakin and colleagues, AEs led to more dose reductions (63%) and treatment discontinuations (29%) than reported in clinical trials with more than half of the discontinuations occurring because of low-grade AEs.7 AEs may also contribute to nonadherence (ie, not taking medication as prescribed), which can negatively impact molecular response, result in a poorer prognosis, and lead to increased healthcare utilization.8,9 Nonadherence can lead to higher healthcare-related costs because of AE-related complications (eg, patient doubling a dose to make up for a missed dose or self-titrating dose), the need for additional testing, or worsening disease.8,9 In a matched cohort study using claims data to evaluate AE-related cost incurred across multiple AEs and cancer types, Wong and colleagues found incremental healthcare costs ranged from $576 to $24,633 for AEs of any severity.10
Oral anticancer medications provide patients with more dosing and administration flexibility.11 However, AEs and nonadherence are challenges that need to be managed to optimize patients’ outcomes. As part of an interdisciplinary team, pharmacists have the opportunity and knowledge required to manage oral anticancer therapy and coinciding AEs.12-15 Pharmacist-led oral anticancer monitoring programs have demonstrated improved adherence and response to therapy, better adherence to prescribing and monitoring guidelines, and high rates of patient learning and satisfaction.13-19 Further demonstrating AE challenges and optimization opportunity, Muluneh and colleagues reported that 57% of pharmacists’ interventions performed by an integrated, pharmacist-led oral oncolytic management program were related to AE management.18 AEs that result from treatment with a PARP inhibitor are managed using pharmacologic and nonpharmacologic interventions.20,21 Low-grade AEs can be managed using behavioral interventions whereas higher-grade AEs typically require supportive medications, such as antiemetics and antidiarrheal agents. Therapy interruptions (a purposeful hold of medication) are also used to manage more severe AEs with a poor response or limited supportive care options. In addition, dose reductions can ease persistent AEs that do not resolve with supportive therapy or therapy interruptions.20,21 Patients should be monitored closely for AEs during the first cycles of therapy, and a complete blood count should be monitored weekly to monthly for patients initiating treatment with a PARP inhibitor.20
Identifying and managing AEs early can help patients continue receiving oral anticancer treatment at the recommended starting dose. Frequent follow-up starting as early as 1 to 2 weeks after therapy initiation is recommended to manage AEs and promote medication adherence in patients receiving oral anticancer medications.9,12,22 The Hematology/Oncology Pharmacist Association Best Practices for the Management of Oral Oncolytic Therapy recommends monitoring symptoms and adherence at each clinical encounter and at least before each refill.12 Although frequent monitoring is recommended, there is a lack of literature describing the impact of tailoring monitoring strategies to the timing of specific medications’ AEs. PARP inhibitor clinical trial data provide insight into the timing of when specific AEs occur throughout a treatment course.
This study evaluated whether targeted patient monitoring at defined intervals reduced the rate of PARP inhibitor dose reductions, resulted in fewer treatment interruptions and therapy changes, and reduced healthcare resource utilization in the first 90 days of therapy.
Methods
This single-center, pre-/postintervention study included adults initiating PARP inhibitor therapy between November 2017 and October 2019 (preintervention) or between July 2021 and October 2022 (postintervention) at Vanderbilt University Medical Center with medication filled by the health-system specialty pharmacy (HSSP) or by a manufacturer’s assistance program (because these patients received the same monitoring calls from a pharmacist). Clinical trial participants and patients who were prescribed a PARP inhibitor for a non–FDA-approved use were excluded from the study. The study was approved by the center’s institutional review board.
This study was conducted at an integrated HSSP of Vanderbilt University Medical Center, a tertiary academic medical center in the United States, where specialty pharmacists are embedded in the outpatient clinics as part of the interdisciplinary patient care team. The oncology clinic has 3 full-time and 2 part-time oncology specialty pharmacists who manage medication access, evaluate patients for specialty medication safety and appropriateness, educate patients, and monitor and optimize patient response to oral anticancer medications. Patient monitoring frequency is determined by a clinic-specific protocol. In the oncology clinic, patients are evaluated monthly for adherence, AEs, and healthcare utilization, and annually for adherence to safety monitoring and therapeutic response. Pharmacist interventions (actions taken by the pharmacist) are documented in the specialty pharmacy patient management system, including reason for intervention and outcome. Pharmacist interventions could include providing additional patient education, performing medication reconciliation, recommending supportive over-the-counter or prescription medications, coordination of care for follow-up appointments or laboratory monitoring, or escalating care to the provider with a recommendation to hold or change a medication dose.
In the preintervention cohort, patients received thorough patient education before initiating PARP inhibitor treatment. Unless patients reached out to the clinic or pharmacists, they were contacted by the HSSP 3 times after initial counseling in the first 90 days of therapy for a refill assessment. Specialty pharmacy technicians contacted patients by phone or through the electronic health record (EHR) patient message portal to schedule medication refills approximately 7 days before the patient was expected to finish their medication per the clinic’s protocol. A specialty pharmacist was alerted if the patient reported missing any doses of medication, having any AEs, or having any treatment-related concerns. The pharmacist would then contact the patient or physician when needed to provide patient education or make a recommendation for supportive therapy, medication adjustments, or additional healthcare service (eg, laboratory monitoring, urgent care visit, or clinic visit).
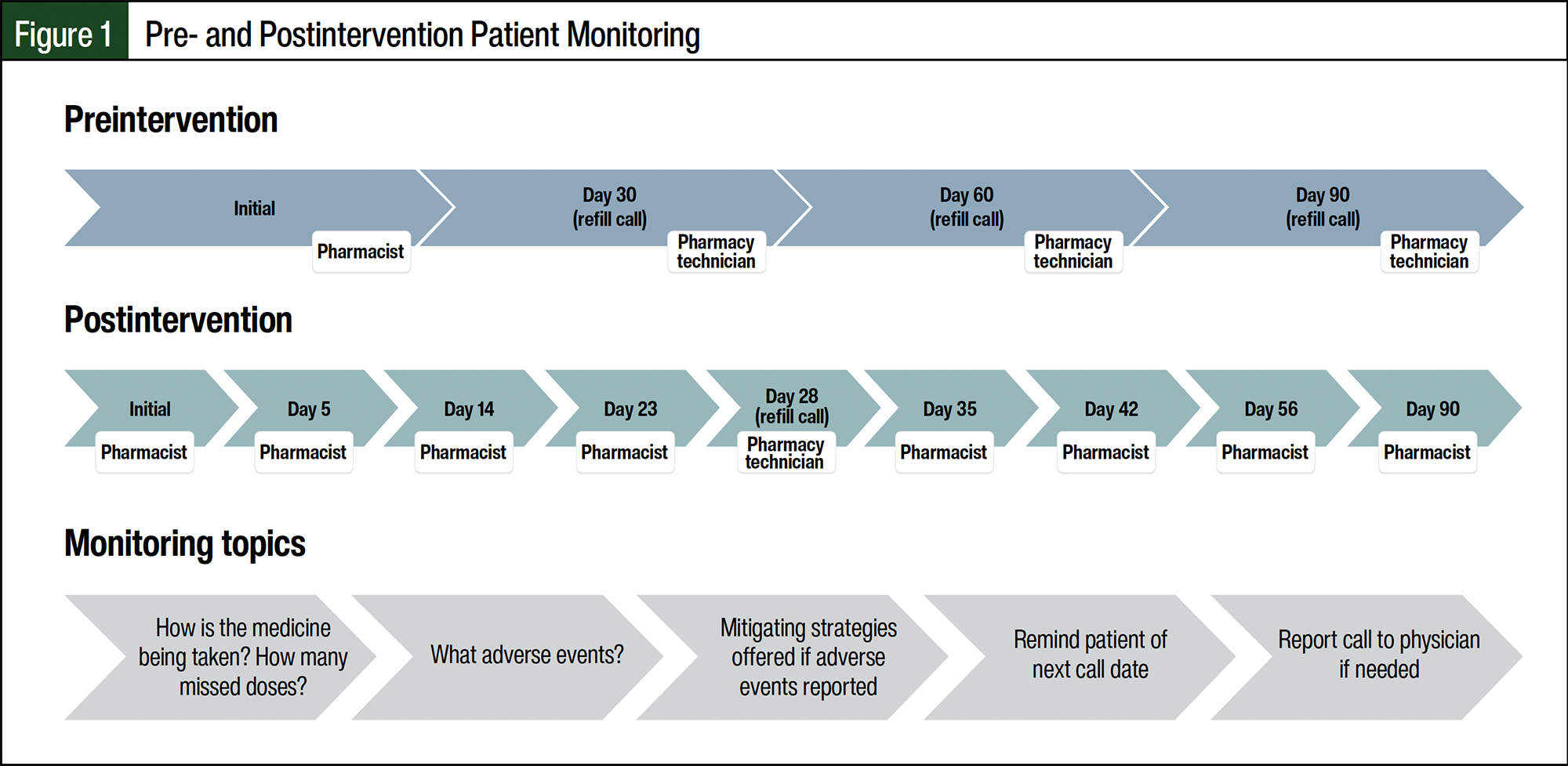
In July 2021, a tailored early treatment monitoring program was implemented for patients initiating PARP inhibitors that were filled through the HSSP or the manufacturer’s assistance program. Patients initiating PARP inhibitor therapy received comprehensive counseling and a welcome kit at therapy initiation. The welcome kit contained items useful for managing the anticipated AEs of PARP inhibitor therapy and contact information for the HSSP. In addition to pharmacy technician outreach to schedule refills and patients being able to contact the pharmacist at any time, the specialty pharmacist proactively contacted the patient at 7 designated times during the 90 days after starting their medication. The call schedule and general topics reviewed are shown in Figure 1. Appendix Table S1 provides a detailed list of questions addressed by the pharmacist at each monitoring call.
Outreach was scheduled to coincide with the expected onset of AEs based on data from clinical trials4-6 and the manufacturer. Follow-up monitoring was performed by the pharmacist and was conducted by phone. During monitoring calls, patients were asked specific questions related to the anticipated AEs and were provided mitigation strategies. The specialty pharmacist intervened as needed, depending on patient-reported missed doses, AEs, or other concerns, including changes to the medication list, administration questions, new food or drug allergies, or insurance/financial issues.
A retrospective chart review was conducted to obtain data for the preintervention cohort before intervention implementation. In addition to AEs that were reported from refill assessments, AEs that were documented in the EHR from clinic visits or other patient contacts were included. Postintervention data were prospectively entered during survey calls, at refill assessments, and when patients contacted the pharmacy to report AEs. The data were maintained in the specialty pharmacy patient management system and the EHR as part of normal clinical practice and were collected using Research Electronic Data Capture hosted at Vanderbilt University.23,24
The primary outcome of this study was the number and duration of treatment interruptions, dose reductions, and medication discontinuations before and after the intervention’s implementation. The secondary outcomes included the number of drug- or cancer-related emergency department visits and hospitalizations during the first 90 days of treatment in each cohort. The type and frequency of AEs reported and pharmacist interventions provided in each cohort were also described.
Statistical Analysis
Descriptive statistics were used to compare the data in the pre- and postintervention cohorts. Continuous variables were reported as median and interquartile ranges (IQRs), and categorical variables were reported as frequencies and percentages. Statistical analysis was performed using R version 4.3.1.
Results
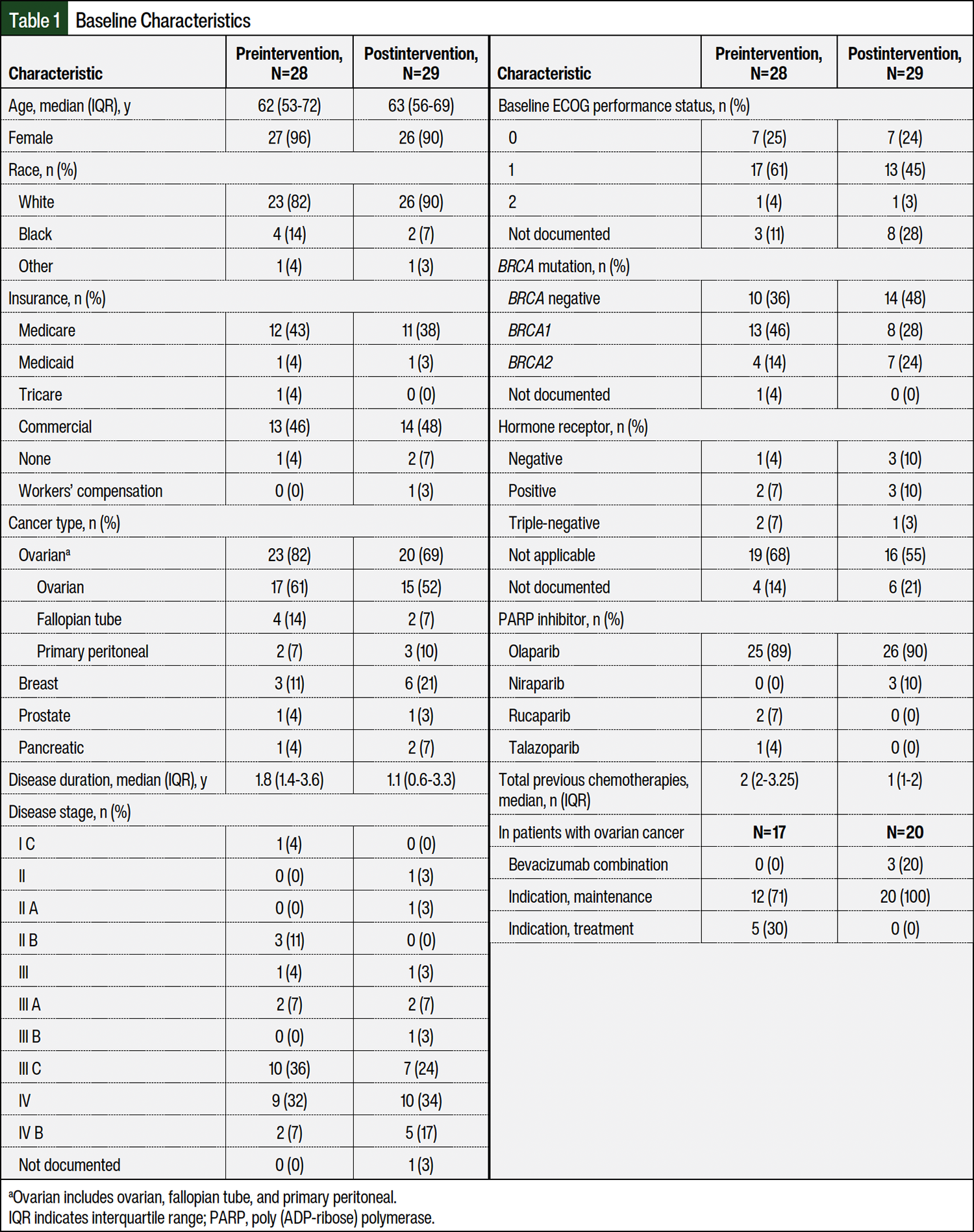
The preintervention (n=28) and postintervention (n=29) populations were similar and were mostly white (n=23 [82%] vs n=26 [90%]) and female (n=27 [96%] vs n=26 [90%]). The median patient age was 62 years (IQR, 53-72 years) in the preintervention group and 63 years (IQR, 56-69 years) in the postintervention group, and the median disease duration was 1.8 years (IQR, 1.4-3.6 years) and 1.1 years (IQR, 0.6-3.3 years), respectively (Table 1). Olaparib was the most frequently prescribed PARP inhibitor (n=25 [89%] preintervention vs n=26 [90%] postintervention), and ovarian cancer was the most frequent cancer type (n=23 [82%] preintervention vs n=20 [69%] postintervention).
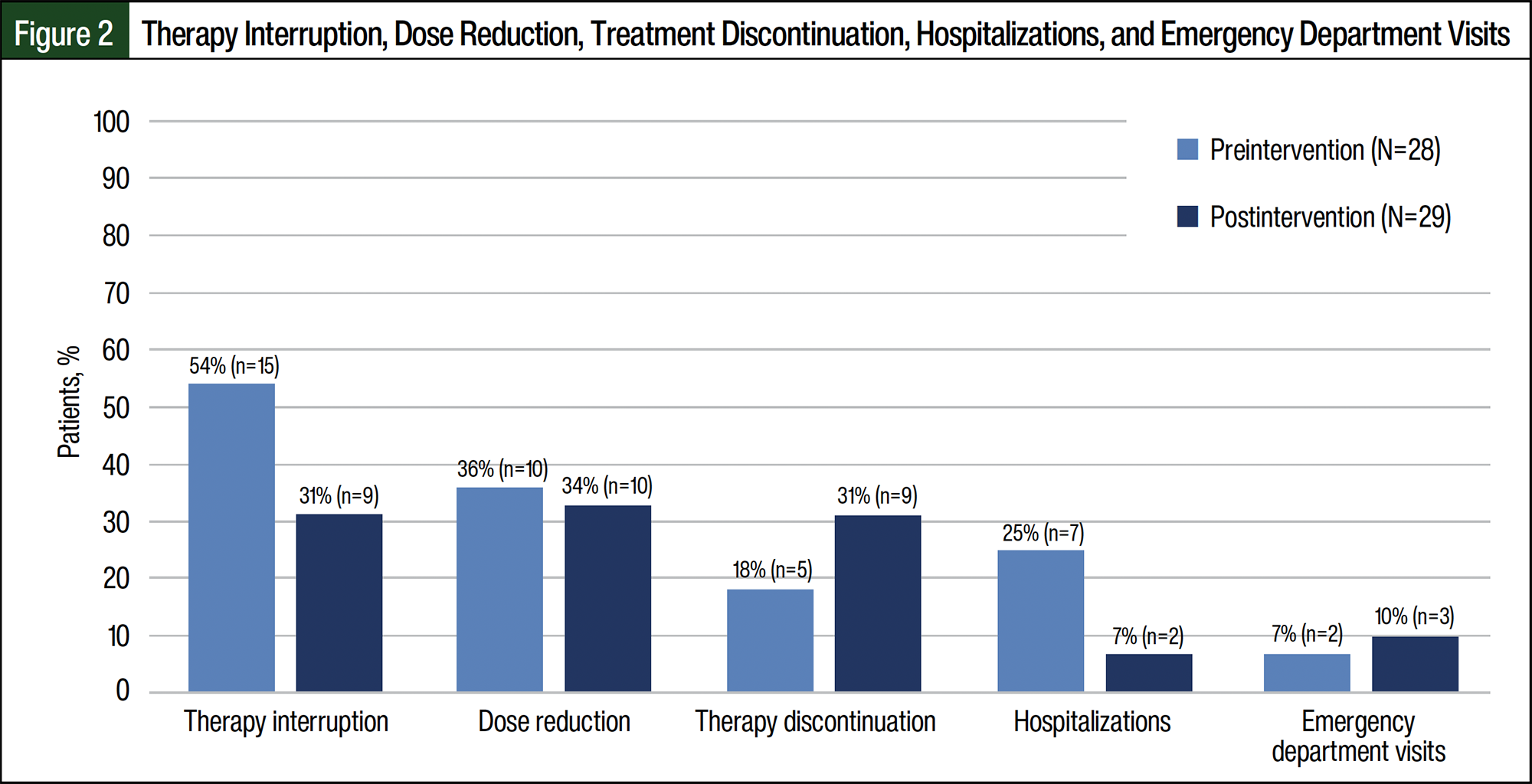
Postintervention, fewer patients had therapy interruptions during the first 90 days of treatment (n=9 [31%] postintervention vs n=15 [54%] preintervention; Figure 2). Preintervention, 11% of patients had more than 1 therapy interruption, whereas none of the patients had more than 1 interruption in the postintervention cohort. The duration of therapy interruptions was shorter in the postintervention group than in the preintervention group (median, 7 days with IQR of 6-21 days vs 17 days with IQR of 7-24 days, respectively). The dose reductions were similar between the preintervention and postintervention cohorts (n=10 [36%] vs n=10 [34%], respectively). More patients discontinued therapy in the postintervention cohort than in the preintervention cohort (n=9 [31%] vs n=5 [18%], respectively); however, disease progression drove discontinuations in both cohorts (n=4 [80%] preintervention vs n=8 [89%] postintervention). Fewer hospitalizations occurred in the postintervention group than in the preintervention group (n=2 [7%] vs n=7 [25%], respectively), although emergency department visits were similar between the groups (n=3 [10%] vs n=2 [7%], respectively; Figure 2).
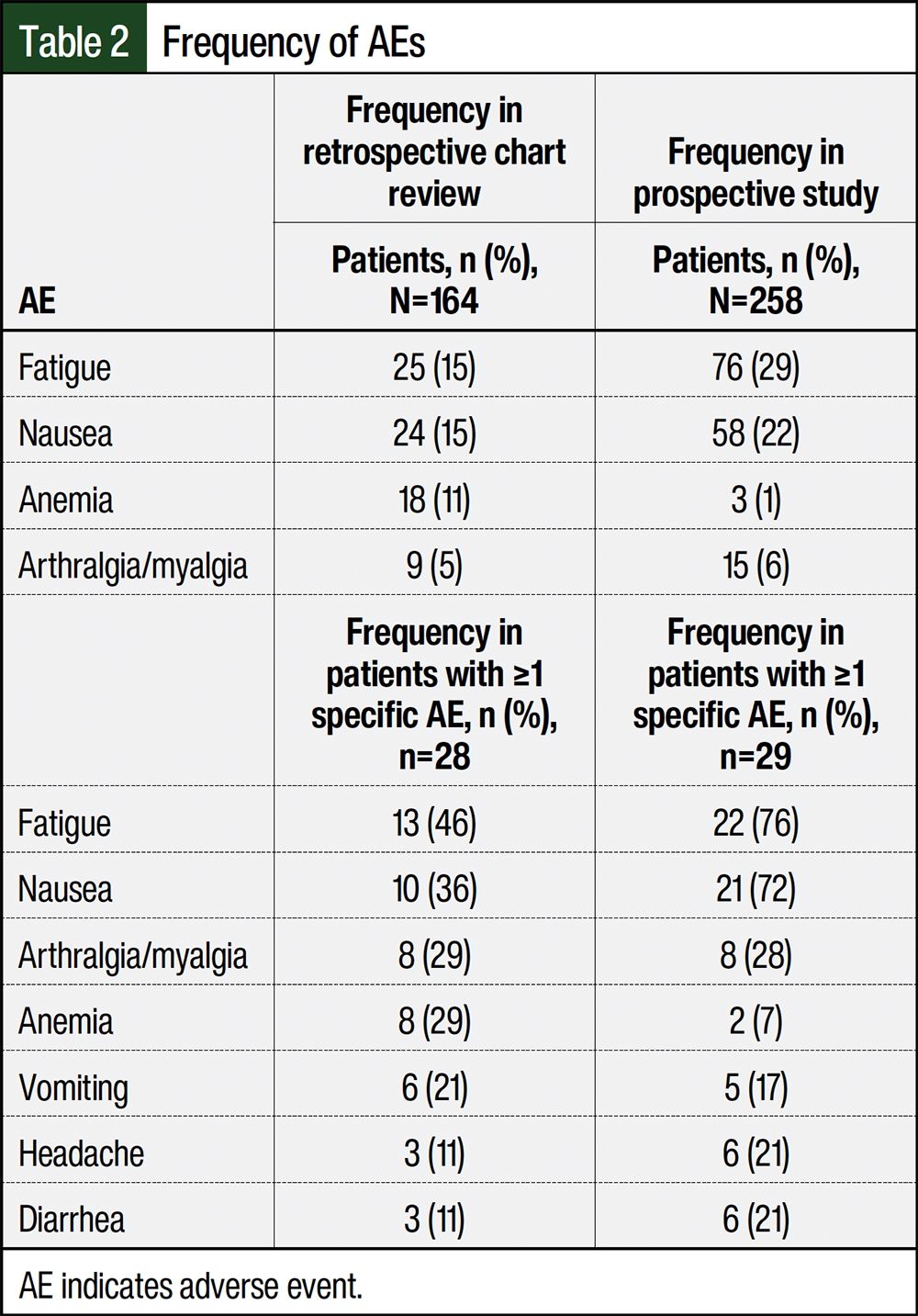
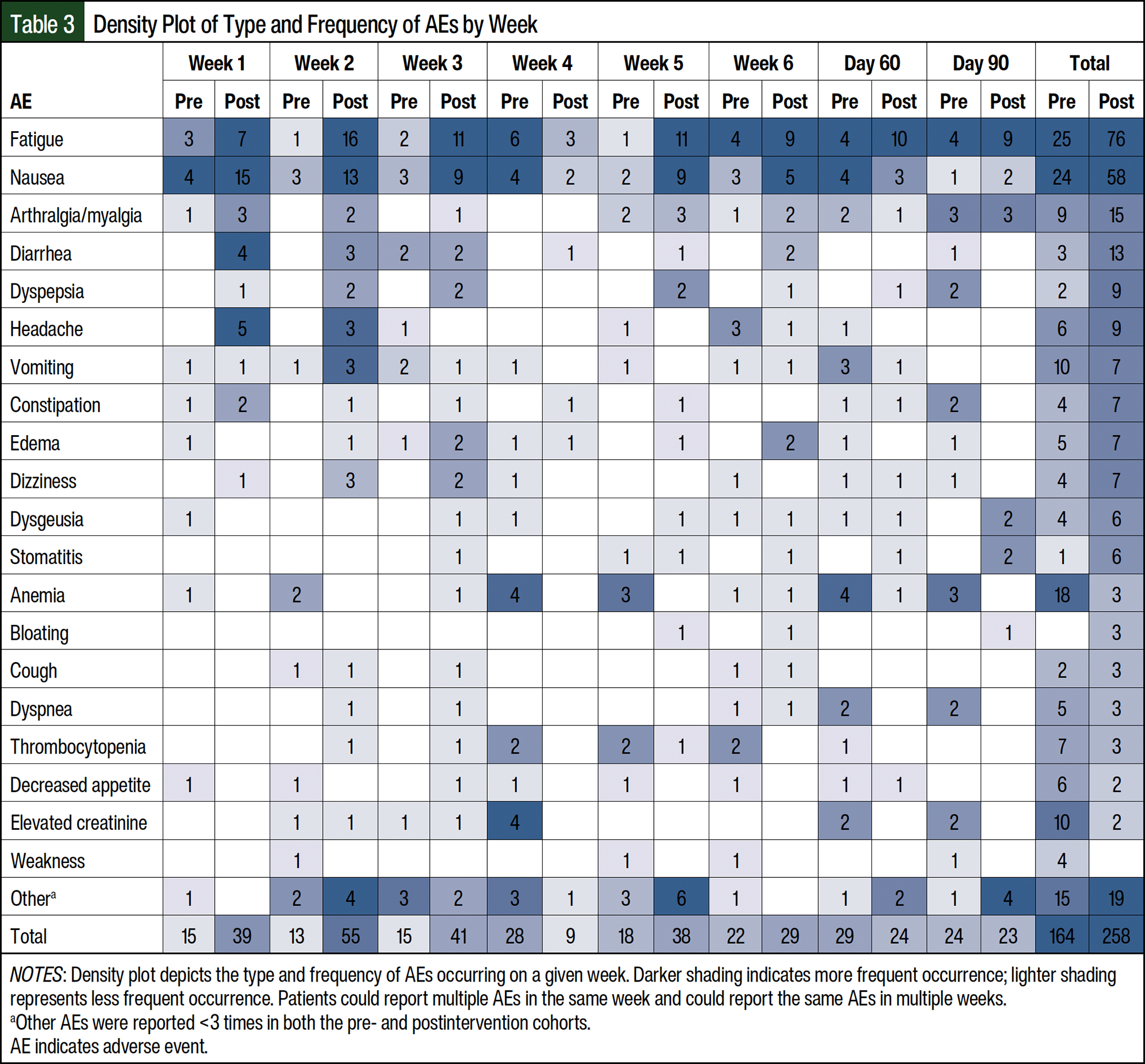
Patients could report multiple AEs at 1 time and the same AE at different times during treatment. When evaluating the most common AEs, of the 28 preintervention patients, 13 (46%) had fatigue and 10 (36%) had nausea at least once (Table 2). Of the 29 postintervention patients, fatigue and nausea were reported at least once by 22 (76%) and 21 (72%) patients, respectively. There were more AEs in the postintervention cohort than in the preintervention cohort (258 vs 164 patients, respectively; Table 2). In the first month of treatment, 71 AEs were reported in the preintervention group compared with 144 in the postintervention group (Table 3). Fatigue (15% vs 29%, respectively) and nausea (15% vs 22%, respectively) were the most frequent AEs in the preintervention (n=164) and postintervention (n=258) cohorts.
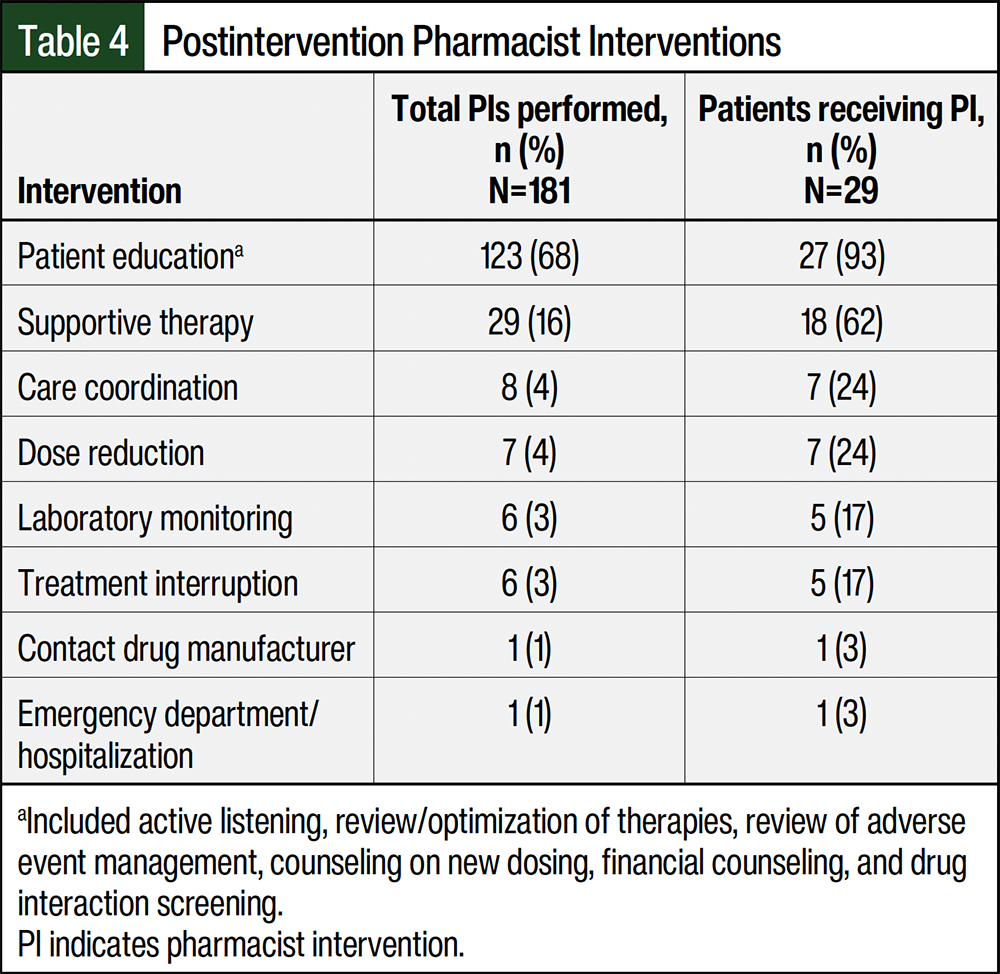
Pharmacists completed 181 interventions in the postintervention cohort (Table 4). Patient education was the most prevalent intervention, with 93% of patients receiving education at least once during the first 90 days of treatment. In all, 18 (62%) of the 29 patients received supportive therapy. Fewer patients had interventions related to care coordination (n=7 [24%]), dose reduction (n=7 [24%]), laboratory monitoring (n=5 [17%]), treatment interruption (n=5 [17%]), manufacturer communication (n=1 [3%]) and emergency department/hospitalization (n=1 [3%]) interventions.
Discussion
This study shows that patients who received pharmacist monitoring calls at targeted intervals after PARP inhibitor initiation had fewer and shorter dose interruptions. Previous research shows that dose reductions and interruptions because of chemotherapy-related AEs can affect patient outcomes,25 and that dose reductions in certain chemotherapy regimens lead to inferior survival outcomes26; however, other literature shows that dose reductions and interruptions for the management of AEs within the first 12 weeks of treatment do not impact progression-free survival.27,28 More studies are needed to determine the effect of dose reductions and interruptions on long-term outcomes, because the degree of dose reduction, the length of interruption, the type of cancer, and the line of treatment (eg, primary, adjuvant, or maintenance) may impact the results. A persistent point throughout current research and in this study is that AEs are one of the most common reasons for dose reductions and interruptions.29
The most frequent AEs were fatigue and nausea, and the time to presentation of these AEs was within the first 2 to 4 weeks after the initiation of treatment. This confirms the results of previous research12 and is consistent with what was observed in the preintervention cohort for patients initiating treatment with PARP inhibitors. The tailored monitoring strategy implemented resulted in more AEs in the postintervention cohort than in the preintervention cohort. In addition, there were more AEs earlier in therapy (weeks 1-3) postintervention, rather than a bolus of AEs at the week 4 refill call; however, this higher number of AEs did not affect treatment discontinuations, because these were similar between the cohorts, with most discontinuation of treatment resulting from disease progression rather than AEs.
The management of AEs at the time when they occur is important, because these events clearly impact the patients’ ability to adhere to and continue receiving medication.29 Formulating a monitoring schedule, using the frequency and anticipated timing of AEs, resulted in pharmacists providing 181 interventions to 29 patients during the first 3 months of treatment, with the provision of patient education and use of supportive therapies being the most common interventions. Extending pharmacist-targeted outreach to 90 days after the initiation of treatment allowed for the management of more persistent AEs associated with receiving PARP inhibitors (eg, fatigue). The results from this study further support the conclusion that a pharmacist-led monitoring program can affect patients’ outcomes and provide novel insight that tailored monitoring based on medication class AE timing can be beneficial.14
The national costs for cancer care were projected to be $200.6 billion in 2020, including the costs for medical services (eg, hospitalizations, physician services, outpatient hospital services) and oral prescription medications.30 Wong and colleagues showed that the costs related to AEs were substantial and contributed to a large economic burden.10 Our study showed a lower number of hospitalizations in the postintervention cohort, confirming earlier studies that demonstrate the value that pharmacists provide to the healthcare team by safely and effectively helping patients manage lower-grade AEs, such as nausea and vomiting, at home and thereby preventing the escalation of AEs that could lead to hospitalization.31,32 With the incidence of cancer increasing and the estimated shortage of oncology healthcare professionals, previous studies have shown the importance of implementing pharmacists into the oncology team to aid oncologists and nurses by sharing the workload.33,34
Tailored monitoring calls were time-intensive and alternative approaches to patient outreach may be considered, such as using electronic patient-reported outcomes or the EHR patient portal.35,36 Furthermore, the tailored monitoring approach may not be beneficial for all oral anticancer medications. Additional studies are needed to validate this method in other oral anticancer medication classes before allocating pharmacist and technician resources.
Limitations
This study has limitations, including being conducted in a single-center, oncology outpatient clinic and having a small sample size. Although the preintervention and postintervention populations were similar, the sample size was too small to detect if differences in population characteristics (eg, indication) had an impact on the results. The implementation of pharmacist-led tailored monitoring was delayed because of the COVID-19 pandemic, which had the potential to impact the patients’ characteristics because additional indications for PARP inhibitors were also approved during the delay.
More studies are needed on a larger scale with a more diverse patient population. Although this study evaluated patient-reported AEs, it did not use a validated patient-reported outcome monitoring tool, which would have been useful when comparing the results to clinical trial data.
Conclusion
Specialty pharmacies should consider tailored early monitoring for PARP inhibitor therapy based on the positive impact on dose interruptions and hospitalizations demonstrated by this monitoring approach. The high rate of AEs reported through tailored early monitoring of patients offers important insight into the patient experience and provides an opportunity for better support.
Acknowledgment
Special thanks to Carson S. Lamb, PharmD, CSP, for assisting in tailored monitoring calls. The project described was supported by Clinical and Translational Science Awards UL1 TR002243 and UL1 TR000445 from the National Center for Advancing Translational Sciences (NCATS). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of NCATS or the National Institutes of Health.
Funding Source
This study was supported by a grant from AstraZeneca and Merck Sharp &; Dohme, LLC, a subsidiary of Merck & Co, Rahway, NJ, who are codeveloping olaparib.
Author Disclosure Statement
Dr Looney received research support from ASHP Foundation, AstraZeneca, and Pfizer, and has been a speaker for AstraZeneca; Dr White received research support from AstraZeneca, BeiGene, and Pfizer; Dr Zuckerman received research support from ASHP Foundation, AstraZeneca, BeiGene, Pfizer, and Sanofi; Dr Hueseman is an employee of and owns stock in AstraZeneca; Dr Shah received research support from AstraZeneca and Pfizer and owns stock in Genentech; Dr Whelchel received research support from ASHP Foundation; Mr Moore and Dr Choi have no conflicts of interest to report.
References
- LaFargue CJ, Dal Molin GZ, Sood AK, Coleman RL. Exploring and comparing adverse events between PARP inhibitors. Lancet Oncol. 2019;20:e15-e28.
- Arend RC, O’Malley DM, Banerjee S, et al. Utilization of poly(ADP-ribose) polymerase inhibitors in ovarian cancer: a retrospective cohort study of US Healthcare Claims Data. Adv Ther. 2022;39:328-345.
- Hao J, Liu Y, Zhang T, et al. Efficacy and safety of PARP inhibitors in the treatment of advanced ovarian cancer: an updated systematic review and meta-analysis of randomized controlled trials. Crit Rev Oncol Hematol. 2021;157:103145.
- Ledermann JA, Oza AM, Lorusso D, et al. Rucaparib for patients with platinum-sensitive, recurrent ovarian carcinoma (ARIEL3): post-progression outcomes and updated safety results from a randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2020;21:710-722.
- Litton JK, Hurvitz SA, Mina LA, et al. Talazoparib versus chemotherapy in patients with germline BRCA1/2-mutated HER2-negative advanced breast cancer: final overall survival results from the EMBRACA trial. Ann Oncol. 2020;31:1526-1535.
- Ray-Coquard I, Pautier P, Pignata S, et al. Olaparib plus bevacizumab as first-line maintenance in ovarian cancer. N Engl J Med. 2019;381:2416-2428.
- Eakin CM, Ewongwo A, Pendleton L, et al. Real world experience of poly (ADP-ribose) polymerase inhibitor use in a community oncology practice. Gynecol Oncol. 2020;159:112-117.
- Salgado TM, Mackler E, Severson JA, et al. The relationship between patient activation, confidence to self-manage side effects, and adherence to oral oncolytics: a pilot study with Michigan oncology practices. Support Care Cancer. 2017;25:1797-1807.
- McCue DA, Lohr LK, Pick AM. Improving adherence to oral cancer therapy in clinical practice. Pharmacotherapy. 2014;34:481-494.
- Wong W, Yim YM, Kim A, et al. Assessment of costs associated with adverse events in patients with cancer. PLoS One. 2018;13:e0196007.
- Aisner J. Overview of the changing paradigm in cancer treatment: oral chemotherapy. Am J Health Syst Pharm. 2007;64(suppl 5):S4-S7.
- Mackler E, Segal EM, Muluneh B, et al. 2018 Hematology/Oncology Pharmacist Association Best Practices for the Management of Oral Oncolytic Therapy: Pharmacy Practice Standard. J Oncol Pract. 2019;15:e346-e355.
- Wong SF, Bounthavong M, Nguyen CP, Chen T. Outcome assessments and cost avoidance of an oral chemotherapy management clinic. J Natl Compr Canc Netw. 2016;14:279-285.
- Patel JM, Holle LM, Clement JM, et al. Impact of a pharmacist-led oral chemotherapy-monitoring program in patients with metastatic castrate-resistant prostate cancer. J Oncol Pharm Pract. 2016;22:777-783.
- Parsons LB, Edwards K, Perez A, et al. Positive outcomes associated with a pharmacist-driven oral chemotherapy program. J Hematol Oncol Pharm. 2015;5:99-108.
- Holle LM, Bilse T, Alabelewe RM, et al. International Society of Oncology Pharmacy Practitioners (ISOPP) position statement: role of the oncology pharmacy team in cancer care. J Oncol Pharm Pract. 2021;27:785-801.
- Conliffe B, Figg L, Moffett P, et al. Impact of a formal pharmacist-run oral antineoplastic monitoring program: a pilot study in an adult genitourinary oncology clinic. J Oncol Pharm Pract. 2019;25:777-786.
- Muluneh B, Schneider M, Faso A, et al. Improved adherence rates and clinical outcomes of an integrated, closed-loop, pharmacist-led oral chemotherapy management program. J Oncol Pract. 2018;14:e324-e334.
- Lam MS, Cheung N. Impact of oncology pharmacist-managed oral anticancer therapy in patients with chronic myelogenous leukemia. J Oncol Pharm Pract. 2016;22:741-748.
- Gunderson CC, Matulonis U, Moore KN. Management of the toxicities of common targeted therapeutics for gynecologic cancers. Gynecol Oncol. 2018;148:591-600.
- Moore KN, Monk BJ. Patient counseling and management of symptoms during olaparib therapy for recurrent ovarian cancer. Oncologist. 2016;21:954-63.
- Zerillo JA, Goldenberg BA, Kotecha RR, et al. Interventions to improve oral chemotherapy safety and quality: a systematic review. JAMA Oncol. 2018;4:105-117.
- Harris PA, Taylor R, Minor BL, et al. The REDCap consortium: building an international community of software platform partners. J Biomed Inform. 2019;95:103208.
- Harris PA, Taylor R, Thielke R, et al. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377-381.
- Nielson CM, Bylsma LC, Fryzek JP, et al. Relative dose intensity of chemotherapy and survival in patients with advanced stage solid tumor cancer: a systematic review and meta-analysis. Oncologist. 2021;26:e1609-e1618.
- Denduluri N, Lyman GH, Wang Y, et al. Chemotherapy dose intensity and overall survival among patients with advanced breast or ovarian cancer. Clin Breast Cancer. 2018;18:380-386.
- Francis KE, Kim SI, Friedlander M, et al. The impact of olaparib dose reduction and treatment interruption on treatment outcome in the SOLO2/ENGOT-ov21 platinum-sensitive recurrent ovarian cancer. Ann Oncol. 2022;33:593-601.
- Berek JS, Matulonis UA, Peen U, et al. Safety and dose modification for patients receiving niraparib. Ann Oncol. 2018;29:1784-1792. Erratum in: Ann Oncol. 2019;30:859.
- Deutsch S, Koerner P, Miller RT, et al. Utilization patterns for oral oncology medications in a specialty pharmacy cycle management program. J Oncol Pharm Pract. 2016;22:68-75.
- Mariotto AB, Enewold L, Zhao J, et al. Medical care costs associated with cancer survivorship in the United States. Cancer Epidemiol Biomarkers Prev. 2020;29:1304-1312.
- Nhean S, Kostoff D, Yang JJ, et al. Impact of oral chemotherapy management program on capecitabine toxicity management. JCO Oncol Pract. 2021;17:e1021-e1029.
- Khandelwal N, Duncan I, Ahmed T, Rubinstein E, Pegus C. Impact of clinical oral chemotherapy program on wastage and hospitalizations. J Oncol Pract. 2011;7(3 suppl):e25s-9s.
- Avery M, Williams F. The importance of pharmacist providing patient education in oncology. J Pharm Pract. 2015;28:26-30.
- Sessions JK, Valgus J, Barbour SY, Iacovelli L. Role of oncology clinical pharmacists in light of the oncology workforce study. J Oncol Pract. 2010;6:270-272.
- Doolin JW, Berry JL, Forbath NS, et al. Implementing electronic patient-reported outcomes for patients with new oral chemotherapy prescriptions at an academic site and a community site. JCO Clin Cancer Inform. 2021;5:631-640.
- Saadeh C, Hill M, Snowaert S. Patient-reported outcomes for oral oncolytic therapy: a pilot study utilizing an electronic patient portal in a community cancer center. J Oncol Pharm Pract. 2023;29:1974-1981.