Pancreatic ductal adenocarcinoma (PDAC) accounts for more than 90% of cases of pancreatic cancer, the third leading cause of cancer-related deaths in men and women in the United States.1,2 An estimated 62,210 new cases of pancreatic cancer will be diagnosed and 49,830 deaths will occur from pancreatic cancer in the United States in 2022, comprising approximately 3.24% of all new cancer cases and 8.18% of all cancer-related deaths.1 PDAC is one of the most lethal cancers: the overall 5-year survival rate for pancreatic cancer is 11%, and 47% of new PDAC cases are diagnosed as metastatic disease, in which the 5-year survival rate is only 3%.1
PDAC is an epithelial tumor that originates in the exocrine compartment of the pancreas, which contains acinar cells that are responsible for the synthesis of digestive enzymes and the pancreatic duct functioning as a channel to the small intestines.3 PDAC disrupts the secretion of digestive enzymes, leading to maldigestion and consequently malnutrition.4 PDAC has an aggressive nature and metastasizes frequently, especially to the liver.5 The lack of reliable biomarker screening for pancreatic cancer, and the nonspecific symptoms in early disease, result in the insidious growth of malignant cells and late-stage clinical presentation.5
PDAC is known for its complex tumor biology, aggressive nature, and poor prognosis.6 Many investigational agents are currently in clinical development for PDAC, but only a select few show improvements in tumor response and survival benefit.
The goals of this review are to highlight the current treatment approaches in clinical practice, with a focus on the standards of therapy, approved targeted therapies, and approved immunotherapies, as well as present clinical data for the investigational agents currently being studied for the treatment of pancreatic cancer, particularly locally advanced or metastatic PDAC.
Chemotherapy Regimens for PDAC
For resectable or borderline resectable PDAC, the cornerstone of treatment involves surgical intervention, followed by adjuvant chemotherapy. The preferred chemotherapies are fluoropyrimidine-based or gemcitabine-based regimens.7,8
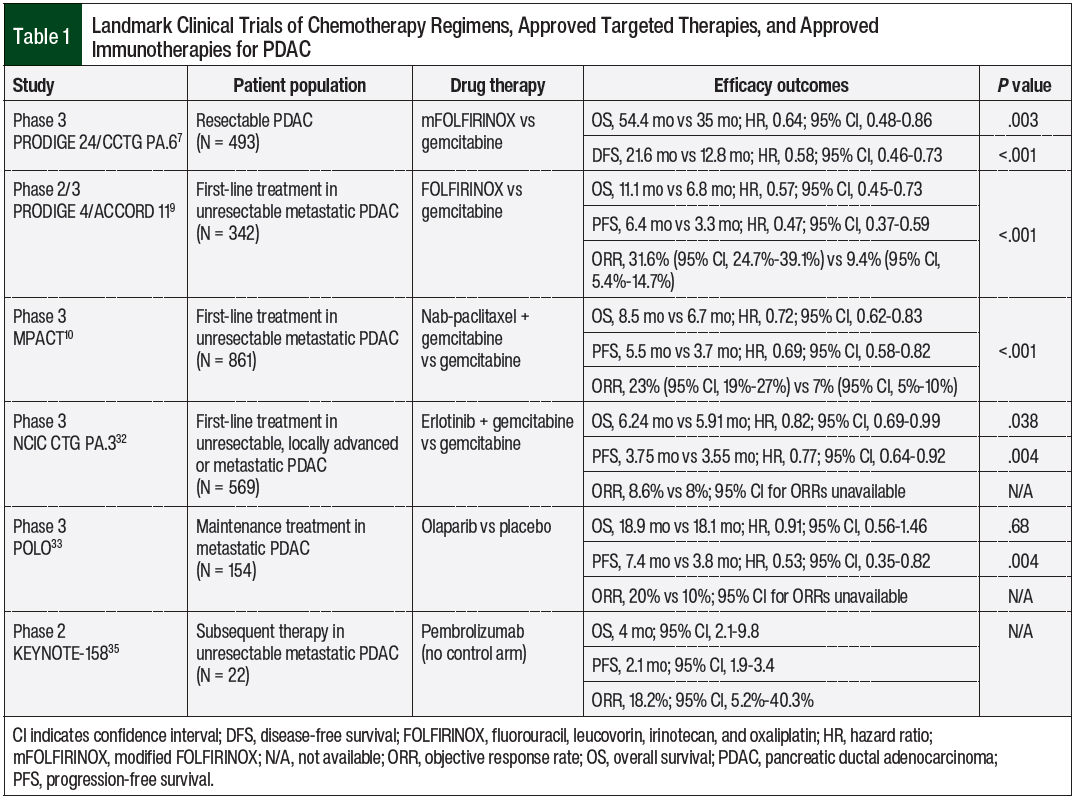
The multicenter phase 3 PRODIGE 24/CCTG PA.6 clinical trial included 493 patients with resectable PDAC and compared modified FOLFIRINOX (fluorouracil, leucovorin, irinotecan, and oxaliplatin) regimen and gemcitabine monotherapy as adjuvant chemotherapy.7 The modified FOLFIRINOX regimen removed bolus fluorouracil to decrease hematologic adverse events and reduced the irinotecan dose from 180 mg/m2 to 150 mg/m2 at an interim safety assessment.7
Improvements were seen with the modified FOLFIRINOX regimen compared with gemcitabine monotherapy in median overall survival (OS) of 54.4 months versus 35 months, respectively (hazard ratio [HR], 0.64; 95% confidence interval [CI], 0.48-0.86; P = .003) and median disease-free survival of 21.6 months versus 12.8 months, respectively (HR, 0.58; 95% CI, 0.46-0.73; P <.001).7 The cumulative incidence of grade 3 or 4 adverse events, including fatigue, diarrhea, nausea, vomiting, abdominal pain, paresthesia, sensory peripheral neuropathy, and mucositis, was higher with modified FOLFIRINOX than with gemcitabine monotherapy (75.9% vs 52.9%, respectively), but the incidence of thrombocytopenia was higher with gemcitabine monotherapy than with modified FOLFIRINOX (50.4% vs 47%, respectively).7
The National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Pancreatic Adenocarcinoma recommend systemic chemotherapy with or without radiation therapy as first-line treatment for locally advanced or metastatic PDAC.8 Because 47% of all pancreatic cancer cases present with unresectable metastatic disease, patients may not be candidates for radiation therapy.1 Chemotherapy regimens, such as FOLFIRINOX or nab-paclitaxel plus gemcitabine, are recommended by the NCCN as preferred therapies for patients with locally advanced or metastatic PDAC and good performance status.8
The phase 2/3 PRODIGE 4/ACCORD 11 landmark clinical trial compared FOLFIRINOX versus gemcitabine monotherapy for first-line treatment in 342 patients with unresectable metastatic PDAC.9 FOLFIRINOX showed significant advantages compared with gemcitabine in median OS (11.1 months vs 6.8 months, respectively; HR, 0.57; 95% CI, 0.45-0.73; P <.001) and median progression-free survival (PFS, 6.4 months vs 3.3 months, respectively; HR, 0.47; 95% CI, 0.37-0.59; P <.001).9 These data led to FOLFIRINOX becoming a preferred treatment regimen in unresectable PDAC, but these patients still had markedly reduced benefit versus those with resectable disease.7-9
In 2013, the US Food and Drug Administration (FDA) approved the combination of albumin-bound paclitaxel (ie, nab-paclitaxel) and gemcitabine as a first-line chemotherapy regimen for the treatment of metastatic PDAC. This approval was based on the phase 3 MPACT study of 861 patients with metastatic pancreatic cancer who were randomized to nab-paclitaxel plus gemcitabine or to gemcitabine monotherapy.10 The combination regimen showed improvement in median OS (8.5 months vs 6.7 months, respectively; HR, 0.72; 95% CI, 0.62-0.83; P <.001), median PFS (5.5 months vs 3.7 months, respectively; HR, 0.69; 95% CI, 0.58-0.82; P <.001), and overall response rate (ORR, 23% vs 7%, respectively; P <.001). However, the incidence of grade 3 or 4 peripheral neuropathy and myelosuppression were significantly higher with the combination than with gemcitabine alone.10
Disease relapse often occurs after treatment, regardless of surgical intervention. In patients with resectable PDAC who received adjuvant FOLFIRINOX in the PRODIGE 24/CCTG PA.6 study, the 3-year disease-free survival rate was 39.7% (95% CI, 32.8-46.6) and the 3-year OS rate was 63.4% (95% CI, 55.7-70.1).7
Patients with unresectable PDAC have a significantly increased risk of relapse, as was shown in the PRODIGE 4/ACCORD 11 study of patients with metastatic PDAC.9 The 18-month PFS and OS were 3.3% and 18.6%, respectively, in the FOLFIRINOX cohort versus 0% and 6%, respectively, with gemcitabine alone, demonstrating a markedly reduced time to disease recurrence in patients who presented with unresectable PDAC versus patients presenting with resectable disease.9
The NCCN Guidelines do not list any preferred chemotherapy regimen for subsequent therapy in recurrent PDAC.8 Gemcitabine-based regimens for patients whose disease progressed after first-line treatment with FOLFIRINOX may be considered, but the available data consist mostly of small, retrospective analyses.11-13 Conversely, subsequent treatment with FOLFIRINOX after first-line gemcitabine-based therapy may be considered.14-16
Of note, the standard infusion of gemcitabine 1000 mg/m2 given over 30 minutes weekly for 3 weeks every 28-day cycle is an NCCN Category 1 “other recommended regimen” for patients with recurrent disease and poor performance status.8 To reduce the adverse events with FOLFIRINOX, less-intensive fluoropyrimidine-based regimens are used. Different regimens or schedules combining oxaliplatin, fluorouracil, and leucovorin, such as the OFF and FOLFOX regimens, have been used but show conflicting efficacy data between the phase 3 CONKO-003 and PANCREOX trials.17,18
Fluorouracil plus leucovorin and irinotecan (FOLFIRI), nanoliposomal irinotecan, continuous fluorouracil infusion, and capecitabine (with or without oxaliplatin) have also been evaluated as salvage chemotherapies.8,19-22 In patients with metastatic PDAC who had received gemcitabine-based chemotherapy and had good performance status, nanoliposomal irinotecan plus fluorouracil and leucovorin is an NCCN Category 1 “other recommended regimen.”8
In the phase 3 NAPOLI-1 study, 417 patients with metastatic PDAC whose disease progressed after gemcitabine-based chemotherapy were randomized into 3 treatment arms to nanoliposomal irinotecan monotherapy, to fluorouracil plus leucovorin, or to the combination of nanoliposomal irinotecan, fluorouracil, and leucovorin.23 In the final analysis, nanoliposomal irinotecan plus fluorouracil and leucovorin demonstrated benefits over fluorouracil plus leucovorin in median OS (6.2 months vs 4.2 months, respectively; HR, 0.75; 95% CI, 0.57-0.99; P = .039) and median PFS (3.1 months vs 1.5 months, respectively; HR, 0.57; 95% CI, 0.43-0.76; P <.0001). Notably, nanoliposomal irinotecan monotherapy demonstrated no significant benefit over fluorouracil plus leucovorin in median OS or median PFS.23
The advantages of treatment with nanoliposomal irinotecan, fluorouracil, and leucovorin, however, pertain potentially to reduce the incidence of adverse events that lead to treatment discontinuation or dose reductions, because no substantial survival benefit is seen with this drug combination versus FOLFIRI.24 A retrospective analysis of FOLFIRI in 63 patients with metastatic PDAC resistant to gemcitabine plus platinum-based chemotherapy reported a median OS of 6.6 months (95% CI, 5.3-8.1).25 In addition, a prospective study of FOLFIRI in 50 patients with locally advanced or metastatic PDAC resistant to gemcitabine and platinum-based chemotherapy reported a similar median OS of 5 months and a median PFS of 3.2 months.19
No salvage chemotherapy regimen is clearly superior in terms of efficacy; hence, the NCCN Guidelines do not recommend any as preferred regimens and instead designate them as other recommended subsequent regimens.8 Therefore, there is a substantial unmet medical need in recurrent pancreatic cancer that targeted therapies and immunotherapies may potentially fulfill.
Targeted Therapies and Immunotherapies
Targeted therapy for patients who have biomarker-specific gene mutations or other alterations can be considered as first-line or as subsequent therapies.8 Among the oral tyrosine kinase inhibitors (TKIs), larotrectinib and entrectinib are FDA approved for patients with solid tumors and neurotrophic tyrosine receptor kinase (NTRK) gene fusions.26,27 The accelerated approvals of these 2 TKIs were based on pooled analyses of single-arm basket trials across multiple solid-tumor types in patients with confirmed NTRK mutation.26,27
NTRK mutations, however, are exceedingly rare, with an incidence of 0.31% (95% CI, 0.09-0.53) in patients with PDAC, as reported in a meta-analysis of NTRK gene fusions.28 In addition, clinical studies of larotrectinib and entrectinib have shown limited efficacy readout specifically for PDAC, because of the very small recruitment of NTRK-positive patients with PDAC.29,30 NTRK-targeted therapy is not an option for a vast majority of patients and is reserved for patients with metastatic PDAC and poor performance status or for those with recurrent disease.8
Erlotinib is another oral TKI targeting the epidermal growth factor receptor and is indicated for unresectable PDAC as well as non–small-cell lung cancer (NSCLC).31 In the phase 3 NCIC CTG PA.3 study, 569 patients with unresectable, locally advanced or metastatic PDAC received weekly gemcitabine, with or without once-daily erlotinib, as first-line treatment.32 The addition of erlotinib improved median OS versus gemcitabine alone (6.24 months vs 5.91 months, respectively; HR, 0.82; 95% CI, 0.69-0.99; P = .038) and the median PFS (3.75 months vs 3.55 months, respectively; HR, 0.77; 95% CI, 0.64-0.92; P = .004).32
Notably, the true survival benefit is rather small for erlotinib, and its use is therefore restricted to patients with locally advanced, metastatic, or recurrent PDAC and good performance status as an adjunctive agent to gemcitabine.8 Although the clinical benefit of erlotinib is limited in current practice, it is one of the first-generation TKIs, and few targeted therapies showed clinical benefit for PDAC when erlotinib was approved in 2005.
The poly (ADP) ribose polymerase inhibitor olaparib is reserved for maintenance therapy in PDAC for patients with a BRCA1 or BRCA2 mutation whose disease has not progressed within 4 to 6 months of receiving first-line platinum-based therapy.8
In the phase 3 POLO (Pancreas Cancer Olaparib Ongoing) study, 154 patients with metastatic PDAC and a germline BRCA1 or BRCA2 mutation whose disease had not progressed during first-line platinum-based therapy were randomized to oral olaparib 300 mg twice daily or to placebo.33 Although the median PFS was statistically significant with olaparib versus placebo (7.4 months vs 3.8 months, respectively; HR, 0.53; 95% CI, 0.35-0.82; P = .004), no substantial improvement was seen in median OS (18.9 months vs 18.1 months, respectively; HR, 0.91; 95% CI, 0.56-1.46; P = .68).33
For patients with mismatch repair-deficient (dMMR) or microsatellite instability-high (MSI-H) tumors, blocking the programmed cell death 1 (PD-1) receptor is a potential therapeutic option.8 Immunotherapy with pembrolizumab, a PD-1 inhibitor, was analyzed in 15 different solid-tumor types with dMMR or with MSI-H across 5 uncontrolled, single-arm clinical trials.34
In the phase 2 KEYNOTE-158 trial, 22 patients with unresectable or metastatic dMMR or MSI-H PDAC received a 200-mg infusion of pembrolizumab once every 3 weeks for approximately 2 years as subsequent therapy.35 The median OS was 4 months (95% CI, 2.1-9.8), and the median PFS was 2.1 months (95% CI, 1.9-3.4). Most adverse events were low grade in the entire study population.35
Pembrolizumab may be used as first-line therapy in patients with metastatic PDAC or as subsequent therapy in recurrent PDAC.8 Although the survival benefit is limited, pembrolizumab may be an alternative treatment option for patients who cannot tolerate the adverse events associated with fluoropyrimidine-based and gemcitabine-based chemotherapies.
Aside from the 3 TKIs (ie, larotrectinib, entrectinib, and erlotinib), olaparib, and pembrolizumab that are approved for PDAC, targeted therapy and immunotherapy have had limited success in the treatment of PDAC. Increased pharmaceutical interest in oncology and rare diseases allowed for notable advancements in the development of investigational combination and single-agent regimens.
Table 1 lists the landmark clinical trials of approved treatments for PDAC.7,9,10,32,33,35
Notable Combination Therapies Under Investigation
Combinations of pembrolizumab-based therapies are currently being studied in a variety of clinical trials. A phase 2 clinical trial conducted by the National Cancer Institute is investigating the addition of pembrolizumab to olaparib as maintenance therapy in 88 patients with metastatic PDAC and a BRCA1 or BRCA2 mutation.36 Another phase 2 study conducted by Sidney Kimmel Comprehensive Cancer Center is expected to recruit 38 patients with metastatic PDAC to receive low-dose gemcitabine, nab-paclitaxel, capecitabine, cisplatin, and irinotecan, followed by maintenance treatment with olaparib and pembrolizumab.37
Of interest, the combination of pembrolizumab, nab-paclitaxel, and gemcitabine with the investigational monoclonal antibody SEA-CD40, a CD40 agonist, is being studied as a first-line treatment in a phase 1 basket trial of 61 patients with metastatic PDAC.38
Dual immune checkpoint inhibition of PD-1 and cytotoxic T-lymphocyte–associated protein-4 (CTLA-4) enhances T-cell function more than single checkpoint blockade, resulting in improved responses in solid tumors, such as melanoma and renal-cell carcinoma.39 The combination of the PD-1 inhibitor nivolumab plus the CTLA-4 inhibitor ipilimumab is effective in dMMR or MSI-H colorectal cancer, but its efficacy is limited in microsatellite-stable solid tumors.39
To overcome intrinsic resistance to immune checkpoint blockade, a phase 2 study analyzed the addition of radiation therapy to nivolumab and ipilimumab in 25 patients with microsatellite-stable PDAC.40 The median OS was 4.2 months (95% CI, 2.1-6.4), the median PFS was 2.5 months (95% CI, 1.6-2.8), and the ORR was 12% (95% CI, 3-31).40 Although the survival benefit is limited, this combination is comparable to pembrolizumab monotherapy and may be beneficial as an alternative to salvage chemotherapy in the second-line or later setting.35
Of note, nivolumab monotherapy is inferior to pembrolizumab monotherapy and to the combination of nivolumab plus ipilimumab.41 In the phase 2 CheckPAC study, 84 patients with refractory metastatic PDAC were randomized 1:1 to stereotactic body radiotherapy (SBRT) plus nivolumab, with or without ipilimumab. PD ligand 1 (PD-L1) expression in the entire patient population was scarce, and approximately 52% of all patients had previously received second-line or later treatment. The group that received nivolumab plus ipilimumab and SBRT had a higher ORR than the group that received nivolumab monotherapy plus SBRT (14% vs 2.4%, respectively) and a higher 1-year OS (14% vs 7.3%, respectively).41
Nivolumab plus ipilimumab, however, did not demonstrate a clear benefit over nivolumab monotherapy in median OS (3.8 months for both) or median PFS (1.6 months vs 1.7 months, respectively).41 The investigators noted that because of the relatively small sample size and wide range in median time from diagnosis to study inclusion, a direct statistical comparison could not be made. It is also unclear if SBRT potentiated the immune checkpoint blockade, because SBRT was included in both treatment arms.41
Although the use of 2 immune checkpoint inhibitors can be more effective than 1 immune checkpoint inhibitor, the use of multiple drugs also results in more adverse events. Immune-mediated adverse events occurred more often with nivolumab plus ipilimumab than with nivolumab monotherapy in other disease states, including unresectable melanoma,42 NSCLC,43-45 renal-cell carcinoma,46,47 hepatocellular carcinoma,48,49 and colorectal cancer.50,51 Across multiple studies, the incidence of high-grade immune-mediated adverse events with combination therapy was more than double that of nivolumab monotherapy within the same disease state.42,45,47,49,51
The risk–benefit of nivolumab plus ipilimumab should be factored into the immunotherapy regimen selection, especially in patients who are susceptible to immune-mediated reactions. Follow-up studies in PDAC with a different treatment schedule are planned for nivolumab plus ipilimumab, including moving radiation therapy to earlier in the treatment course.40
Cellular therapy is being explored to enhance the effects of PD-L1 inhibition. Similar to chimeric antigen receptor T-cell therapy, engineered natural killer (NK) cells possess antineoplastic properties, with the added benefit of being an allogeneic, “off-the-shelf” ready-to-use immunotherapy.52 When combining PD-L1–targeting NK cells with an interleukin (IL)-15 superagonist, this drug cocktail effectively becomes a “cancer vaccine” that stimulates the immune system.52,53
This novel cancer vaccine, N-803, is currently being investigated in 328 patients across 3 cohorts in the phase 2/3 QUILT 88 study.54-56 Patients with unresectable, locally advanced or metastatic PDAC are randomized to standard chemotherapy, with or without N-803 plus aldoxorubicin (albumin-bound doxorubicin). Cohorts A, B, and C are testing the cancer vaccine combination in the maintenance, second-line induction, and third-line or later induction settings, respectively.55
Recent data showed promising preliminary results in 65 patients with advanced PDAC in cohorts B and C, with 30 patients in the third-line treatment setting and 35 patients in the fourth-line or later treatment settings.56 In all 65 patients, the median OS was 5.8 months and the median PFS was 2.3 months (95% CI, 2-3.6). In the 30 patients in the third-line setting, the median OS was 6.3 months (95% CI, 5-9.8).56
The study sponsor intends to expand cohort C.57 The FDA accepted a Biologics License Application for N-803 in combination with intravesical Bacillus Calmette-Guérin in non–muscle-invasive bladder cancer on July 28, 2022, and granted it breakthrough therapy and fast track designations.58 The Prescription Drug User Fee Act (PDUFA) action date is scheduled for May 23, 2023.58
Notable Single Agents Under Investigation for PDAC
Adavosertib is an inhibitor of the WEE1 kinase, a regulator of the mitotic G2 checkpoint.59 By allowing malignant cells to bypass the G2 checkpoint, rapid replication occurs without repairs to damaged DNA and eventually induces apoptosis. A phase 1 dose-escalation study in 34 patients with unresectable, locally advanced PDAC analyzed the use of oral adavosertib as first-line treatment in combination with gemcitabine and radiation therapy.59
The patients received adavosertib on days 1, 2, 8, and 9 plus gemcitabine on days 1 and 8 for a maximum of eight 21-day cycles. Concurrent chemoradiation was delivered 5 times weekly during cycles 2 and 3, followed by a 3-week treatment break before cycle 4. The median OS was 21.7 months (90% CI, 16.7-24.8) and the median PFS was 9.4 months (90% CI, 8-9.9).59
KN046 is a novel recombinant PD-L1/CTLA-4 bispecific antibody fusion protein that binds to PD-L1 and CTLA-4 using a smart delivery system.60 The delivery mechanism is a nano-delivery agent formed from a metal–organic framework of zinc and imidazole that is rapidly degraded by glutathione in tumors, enabling a response in cancerous tissue while reducing off-target toxicities.60 A phase 2 study evaluated the use of KN046 infusion with nab-paclitaxel and gemcitabine as first-line treatment in 17 patients with unresectable, locally advanced or metastatic PDAC.61 Of the 9 patients included in the efficacy assessment, 5 had a partial response and 3 had stable disease, with an ORR of 55.6% (95% CI, 21.2-86.3).61
A phase 3 study of KN046 as first-line treatment in 408 patients with unresectable locally advanced or metastatic PDAC is expected to be completed by April 2024.62
Intratumoral injections of oncolytic adenoviruses represent a new frontier in immunotherapy that may be effective in solid tumors such as PDAC. VCN-01 is a genetically modified oncolytic adenovirus that selectively replicates within tumor cells that carry mutations in the retinoblastoma tumor suppressor protein.63 A phase 1 dose-escalation study in 8 patients with unresectable, locally advanced or metastatic PDAC analyzed an intratumoral injection of VCN-01 in conjunction with nab-paclitaxel plus gemcitabine as first-line treatment.63
An expansion cohort that evaluated the recommended phase 2 dose in 40 patients with locally advanced or metastatic PDAC showed measurable viral replication and an ORR of 40% to 45%.64 Based on the dose-escalation study data, a phase 2 study of VCN-01 as first-line treatment in PDAC is planned across multiple centers in the United States and Europe.65
Similarly, another oncolytic adenovirus called LOAd703 may show promise in the treatment of PDAC through the sensitization of tumors to immune checkpoint inhibitors.66 Positive results were reported in a phase 1/2 trial of 21 patients with unresectable or metastatic PDAC who received LOAd703.67 Patients received up to 6 intratumoral injections of LOAd703 in combination with nab-paclitaxel and gemcitabine as first-line or subsequent therapy. Among all the response-evaluable patients, the median OS was 8.7 months and the ORR was 44%. A follow-up phase 1/2 study of this regimen and the immune checkpoint inhibitor atezolizumab is underway.67
All-trans retinoic acid (ATRA) is an oral vitamin A derivative used for the induction of remission of acute promyelocytic leukemia.68 Its mechanism of action involves promoting cytodifferentiation and decreased proliferation of leukemia cells, but in PDAC it also reduces the number of activated stromal fibroblasts and the rate of cancer cell proliferation in mouse models.69
In the STARPAC phase 1b dose-escalation study, 27 patients with locally advanced or metastatic PDAC received ATRA in combination with nab-paclitaxel and gemcitabine as first-line treatment.70 For the 15 patients who received the maximum tolerated dose, the median OS was 11.7 months (95% CI, 8.6-15.7) and the median PFS was 6.4 months (95% CI, 3.5-not reached).70 The STARPAC2 phase 2b study will analyze the addition of ATRA to nab-paclitaxel plus gemcitabine in 170 patients with unresectable, locally advanced pancreatic cancer and is estimated to be completed by February 2024.71
The Kirsten rat sarcoma virus (KRAS) proto-oncogene is implicated in the tumorigenesis of many cancers, and KRAS gene mutations are present in 94.1% of patients with PDAC.72 The KRAS G12C mutation activates the KRAS protein in a GTP-bound state, promoting oncogenesis through downstream effector pathways.73 Adagrasib is a small-molecule inhibitor of KRAS G12C that irreversibly binds to the KRAS G12C mutation, stabilizing the inactive GDP-bound form of the KRAS protein and inhibiting downstream tumor proliferation.73
In the ongoing phase 1/2 KRYSTAL-1 study, oral adagrasib monotherapy is being studied as subsequent treatment in a phase 2 cohort of 30 patients with unresectable or metastatic solid tumors and KRAS G12C mutation, including 12 patients with PDAC.74,75 The interim data showed an ORR of 50% and a median PFS of 6.6 months (95% CI, 1-9.7) in the 10 evaluable patients with PDAC.74 The study is expected to be completed by December 2022.75 Of note, adagrasib is pending FDA review of its New Drug Application for the treatment of NSCLC harboring KRAS G12C mutation, with a PDUFA target date of December 14, 2022.76
Sotorasib is a small-molecule inhibitor targeting the KRAS G12C mutation that received FDA accelerated approval on May 28, 2021, for the treatment of locally advanced or metastatic NSCLC with KRAS G12C mutation.77 As the first FDA-approved KRAS inhibitor, sotorasib has a robust clinical development program analyzing its use in multiple solid tumors, including PDAC.78
In the CodeBreaK100 phase 1/2 clinical trial in patients with solid tumors and KRAS G12C mutation, 38 patients with metastatic PDAC received sotorasib 960 mg once daily as subsequent therapy.79 In the combined phase 1/2 analysis, 79% of the patients were in the third-line or later treatment setting and the median treatment duration was 4.1 months. With a median follow-up of 16.8 months, 8 patients had confirmed partial responses, with an ORR of 21.1% (95% CI, 9.55-37.32). The median OS was 6.87 months (95% CI, 4.96-9.13) and the median PFS was 3.98 months (95% CI, 2.79-5.59).79
Sotorasib has a rigorous clinical development program for PDAC, including potential expansion cohorts in CodeBreaK100 and a phase 1b/2 study combining sotorasib with liposomal irinotecan plus 5-fluorouracil and leucovorin or with nab-paclitaxel plus gemcitabine as second-line therapy.80
siG12D-LODER is a miniature biodegradable polymeric matrix that contains small interfering RNA (siRNA) targeting the KRAS G12D mutation.81 This novel therapy is designed to provide a slow and prolonged local drug release within the tumor over 4 months and protects the siRNA from degradation. In a dose-escalation phase 1/2a study of 15 patients with unresectable, locally advanced PDAC, patients received chemotherapy (FOLFIRINOX or gemcitabine-based regimen) in conjunction with intratumoral siG12D-LODER across 3 doses as first-line treatment. The median OS was 15.1 months (95% CI, 10.2-18.4) and the 18-month OS was 38.5%.81
siG12D-LODER is currently being analyzed as neoadjuvant chemotherapy in a phase 2 study of 39 patients with resectable, locally advanced pancreatic cancer.82 All patients were expected to receive modified FOLFIRINOX or nab-paclitaxel plus gemcitabine and up to 3 doses of siG12D-LODER once every 12 weeks.82 The study was anticipated to be completed in October 2022.83
The PanCAN Precision Promise Consortium
The Pancreatic Cancer Action Network (PanCAN) is the first and largest national nonprofit organization dedicated to fighting pancreatic cancer. In 2019, PanCAN launched the Precision Promise adaptive clinical trial platform to accelerate new treatment options for PDAC.84 This large phase 2/3 clinical trial is designed to cross-analyze several new PDAC treatments in multiple experimental and control arms across more than 20 sites in the United States.84
In addition, the platform allows for multiple drug candidates to be tested (such as a patient receiving second-line treatment with a different agent if first-line therapy is not effective), expedites studies to be completed in as few as 3 years, and reduces study expenses by as much as 50%.85 The investigational agents participating in the Precision Promise Consortium currently include pamrevlumab and nadunolimab.86,87
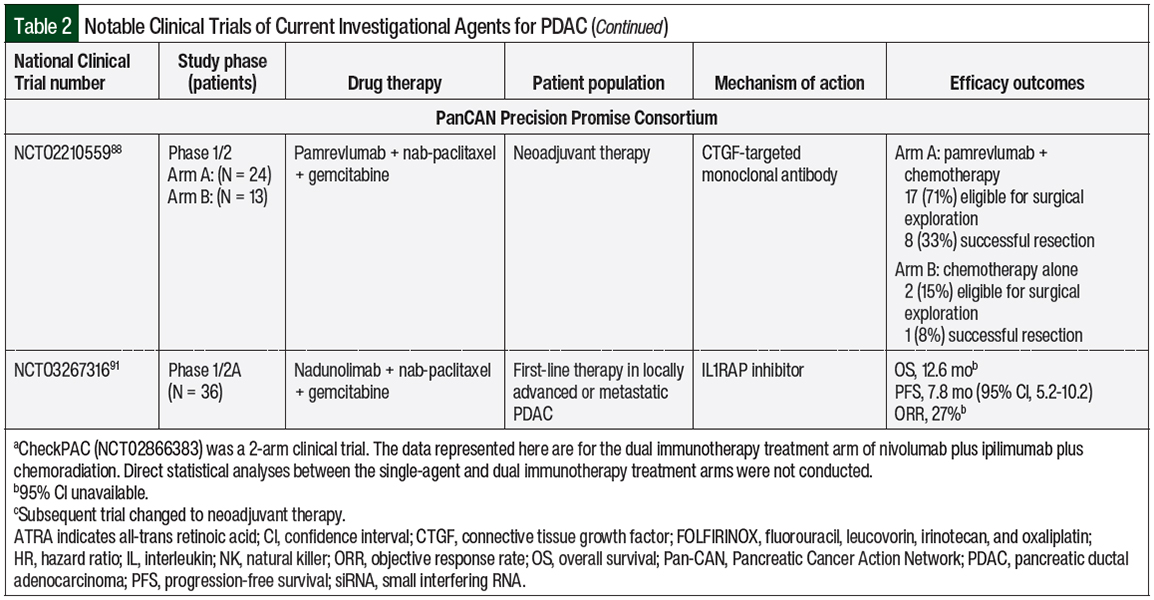
Pamrevlumab is a monoclonal antibody targeting connective tissue growth factor, which is implicated in the tissue adhesion capabilities of cancerous pancreatic cells.88 A phase 1/2 study of 37 patients with baseline unresectable, locally advanced PDAC evaluated the addition of pamrevlumab to nab-paclitaxel plus gemcitabine as neoadjuvant chemotherapy. A greater proportion of patients who received pamrevlumab were eligible for surgical exploration and had resection compared with those who received only standard chemotherapy.88
In the Precision Promise Consortium, patients with metastatic PDAC are to receive pamrevlumab in combination with nab-paclitaxel and gemcitabine in an experimental treatment arm.86 A separate, unassociated phase 3 trial will also evaluate the use of pamrevlumab with FOLFIRINOX or with nab-paclitaxel plus gemcitabine as neoadjuvant therapy in 284 patients with initially unresectable, locally advanced PDAC.89
IL-1 is an inflammatory cytokine that mediates local and systemic immune responses.90 Nadunolimab is an IL-1 receptor accessory protein (IL1RAP)-targeted monoclonal antibody that inhibits IL-1 downstream signaling, which is often implicated in cancer and inflammatory diseases.90 In the phase 1/2a CANFOUR trial, nadunolimab was administered with nab-paclitaxel and gemcitabine to 36 patients with unresectable, locally advanced or metastatic PDAC as first-line treatment.91 The interim data showed that in the 33 evaluable patients with PDAC, the median OS was 12.6 months (95% CI, not estimable), the median PFS was 7.8 months (95% CI, 5.2-10.2), and the ORR was 27%.91 In the Precision Promise Consortium, up to 175 patients with metastatic PDAC are to receive nadunolimab in combination with nab-paclitaxel and gemcitabine in an experimental treatment arm.87
Table 2 outlines the ongoing clinical trials for investigational agents for the treatment of PDAC.36-38,40,41,54-56,59,61,64,67,70,74,75,79,81,88,91
Conclusion
PDAC is a particularly lethal cancer that is difficult to screen for and is often diagnosed as late-stage disease. There is an unmet medical need in recurrent PDAC in which only select populations are eligible to receive the preferred treatment, and salvage chemotherapy has mixed or limited data. Immunotherapy and targeted therapy for PDAC are limited, with pembrolizumab as the only FDA-approved monoclonal antibody, and small-molecule inhibitors reserved for niche populations with a specific genetic mutation.
Innovative approaches are being investigated in PDAC, such as cellular therapy, nucleotide-based delivery systems, and immunotherapy combinations. Monoclonal antibodies are increasingly of interest in solid tumors, and PDAC can serve as its own cohort in basket trials. Intratumoral injection may be a viable route of administration for increasing efficacy, decreasing toxicities, and introducing more biologically complex therapies, such as oligonucleotides or adenoviruses.
Despite the challenges with PDAC treatment, many agents are being explored. The PanCAN Precision Promise Consortium has facilitated the acceleration and reduced cost of clinical trials and encourages cross-collaboration between pharmaceutical companies. We hope for future data that will signal improved survival benefits for patients with PDAC and await a new breakthrough therapy to change clinical practice.
Author Disclosure Statement
The authors have no conflicts of interest to report.
References
- Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72:7-33.
- Hu JX, Zhao CF, Chen WB, et al. Pancreatic cancer: a review of epidemiology, trend, and risk factors. World J Gastroenterol. 2021;27:4298-4321.
- Anatomy. In: Pandol SJ. The Exocrine Pancreas. San Rafael, CA: Morgan & Claypool Life Sciences; 2011. Colloquium Series on Integrated Systems Physiology: from Molecule to Function to Disease, Lecture #14. www.ncbi.nlm.nih.gov/books/NBK54134/. Accessed March 19, 2022.
- Vujasinovic M, Valente R, Del Chiaro M, et al. Pancreatic exocrine insufficiency in pancreatic cancer. Nutrients. 2017;9:183. doi: 10.3390/nu9030183.
- Das S, Batra SK. Pancreatic cancer metastasis: are we being pre-EMTed? Curr Pharm Des. 2015;21:1249-1255.
- Le Large TYS, Bijlsma MF, Kazemier G, et al. Key biological processes driving metastatic spread of pancreatic cancer as identified by multi-omics studies. Semin Cancer Biol. 2017;44:153-169.
- Conroy T, Hammel P, Hebbar M, et al; for the Canadian Cancer Trials Group and the Unicancer-GI–PRODIGE Group. FOLFIRINOX or gemcitabine as adjuvant therapy for pancreatic cancer. N Engl J Med. 2018;379:2395-2406.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Pancreatic Adenocarcinoma. Version 1.2022. February 24, 2022. www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf. Accessed March 19, 2022.
- Conroy T, Desseigne F, Ychou M, et al; for the Groupe Tumeurs Digestives of Unicancer and the PRODIGE Intergroup. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011;364:1817-1825.
- Von Hoff DD, Ervin T, Arena FP, et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med. 2013;369:1691-1703.
- da Rocha Lino A, Abrahão CM, Brandão RM, et al. Role of gemcitabine as second-line therapy after progression on FOLFIRINOX in advanced pancreatic cancer: a retrospective analysis. J Gastrointest Oncol. 2015;6:511-515.
- Viaud J, Brac C, Artru P, et al. Gemcitabine as second-line chemotherapy after Folfirinox failure in advanced pancreatic adenocarcinoma: a retrospective study. Dig Liver Dis. 2017;49:692-696.
- Nguyen KT, Kalyan A, Beasley HS, et al. Gemcitabine/nab-paclitaxel as second-line therapy following FOLFIRINOX in metastatic/advanced pancreatic cancer—retrospective analysis of response. J Gastrointest Oncol. 2017;8:556-565.
- Sawada M, Kasuga A, Mie T, et al. Modified FOLFIRINOX as a second-line therapy following gemcitabine plus nab-paclitaxel therapy in metastatic pancreatic cancer. BMC Cancer. 2020;20:449. doi: 10.1186/s12885-020-06945-8.
- Kobayashi N, Shimamura T, Tokuhisa M, et al. Effect of FOLFIRINOX as second-line chemotherapy for metastatic pancreatic cancer after gemcitabine-based chemotherapy failure. Medicine (Baltimore). 2017;96:e6769. doi: 10.1097/MD.0000000000006769.
- Lee MG, Lee SH, Lee SJ, et al. 5-Fluorouracil/leucovorin combined with irinotecan and oxaliplatin (FOLFIRINOX) as second-line chemotherapy in patients with advanced pancreatic cancer who have progressed on gemcitabine-based therapy. Chemotherapy. 2013;59:273-279.
- Oettle H, Riess H, Stieler JM, et al. Second-line oxaliplatin, folinic acid, and fluorouracil versus folinic acid and fluorouracil alone for gemcitabine-refractory pancreatic cancer: outcomes from the CONKO-003 trial. J Clin Oncol. 2014;32:2423-2429.
- Gill S, Ko YJ, Cripps C, et al. PANCREOX: a randomized phase III study of fluorouracil/leucovorin with or without oxaliplatin for second-line advanced pancreatic cancer in patients who have received gemcitabine-based chemotherapy. J Clin Oncol. 2016;34:3914-3920.
- Zaniboni A, Aitini E, Barni S, et al. FOLFIRI as second-line chemotherapy for advanced pancreatic cancer: a GISCAD multicenter phase II study. Cancer Chemother Pharmacol. 2012;69:1641-1645.
- Wang-Gillam A, Li CP, Bodoky G, et al; for the NAPOLI-1 study group. Nanoliposomal irinotecan with fluorouracil and folinic acid in metastatic pancreatic cancer after previous gemcitabine-based therapy (NAPOLI-1): a global, randomised, open-label, phase 3 trial. Lancet. 2016;387:545-557. Erratum in: Lancet. 2016;387:536.
- Maisey N, Chau I, Cunningham D, et al. Multicenter randomized phase III trial comparing protracted venous infusion (PVI) fluorouracil (5-FU) with PVI 5-FU plus mitomycin in inoperable pancreatic cancer. J Clin Oncol. 2002;20:3130-3136.
- Xiong HQ, Varadhachary GR, Blais JC, et al. Phase 2 trial of oxaliplatin plus capecitabine (XELOX) as second-line therapy for patients with advanced pancreatic cancer. Cancer. 2008;113:2046-2052.
- Wang-Gillam A, Hubner RA, Siveke JT, et al. NAPOLI-1 phase 3 study of liposomal irinotecan in metastatic pancreatic cancer: final overall survival analysis and characteristics of long-term survivors. Eur J Cancer. 2019;108:78-87.
- Tossey JC, Reardon J, VanDeusen JB, et al. Comparison of conventional versus liposomal irinotecan in combination with fluorouracil for advanced pancreatic cancer: a single-institution experience. Med Oncol. 2019;36:87. doi: 10.1007/s12032-019-1309-6.
- Neuzillet C, Hentic O, Rousseau B, et al. FOLFIRI regimen in metastatic pancreatic adenocarcinoma resistant to gemcitabine and platinum-salts. World J Gastroenterol. 2012;18:4533-4541.
- Vitrakvi (larotrectinib) capsules, for oral use/oral solution [prescribing information]. Bayer HealthCare Pharmaceuticals; March 2021. https://labeling.bayerhealthcare.com/html/products/pi/vitrakvi_PI.pdf. Accessed March 19, 2022.
- Rozlytrek (entrectinib) capsules, for oral use [prescribing information]. Genentech; July 2022. www.gene.com/download/pdf/rozlytrek_prescribing.pdf. Accessed October 25, 2022.
- Forsythe A, Zhang W, Strauss UP, et al. A systematic review and meta-analysis of neurotrophic tyrosine receptor kinase gene fusion frequencies in solid tumors. Ther Adv Med Oncol. 2020;12:1758835920975613. doi: 10.1177/1758835920975613.
- Drilon A, Laetsch TW, Kummar S, et al. Efficacy of larotrectinib in TRK fusion–positive cancers in adults and children. N Engl J Med. 2018;378:731-739.
- Doebele RC, Drilon A, Paz-Ares L, et al; for the trial investigators. Entrectinib in patients with advanced or metastatic NTRK fusion-positive solid tumours: integrated analysis of three phase 1–2 trials. Lancet Oncol. 2020;21:271-282. Errata in: Lancet Oncol. 2020;21:e70; Lancet Oncol. 2020;21:e341; Lancet Oncol. 2020;21:e372; Lancet Oncol. 2021;22:e428.
- Tarceva (erlotinib) tablets, for oral use [prescribing information]. Astellas Pharma US; Genentech; October 2016. www.gene.com/download/pdf/tarceva_prescribing.pdf. Accessed March 19, 2022.
- Moore MJ, Goldstein D, Hamm J, et al. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol. 2007;25:1960-1966.
- Golan T, Hammel P, Reni M, et al. Maintenance olaparib for germline BRCA-mutated metastatic pancreatic cancer. N Engl J Med. 2019;381:317-327.
- Keytruda (pembrolizumab) injection, for intravenous use [prescribing information]. Merck & Co; August 2022. www.merck.com/product/usa/pi_circulars/k/keytruda/keytruda_pi.pdf. Accessed October 25, 2022.
- Marabelle A, Le DT, Ascierto PA, et al. Efficacy of pembrolizumab in patients with noncolorectal high microsatellite instability/mismatch repair–deficient cancer: results from the phase II KEYNOTE-158 study. J Clin Oncol. 2020;38:1-10.
- Testing the addition of pembrolizumab, an immunotherapy cancer drug to olaparib alone as therapy for patients with pancreatic cancer that has spread with inherited BRCA mutations. NLM identifier: NCT04548752. Updated October 28, 2022. https://clinicaltrials.gov/ct2/show/NCT04548752. Accessed October 28, 2022.
- Multi-agent low dose chemotherapy gax-ci followed by olaparib and pembro in metastatic pancreatic ductal cancer. NLM identifier: NCT04753879. Updated March 16, 2022. https://clinicaltrials.gov/ct2/show/NCT04753879. Accessed October 28, 2022.
- Bajor DL, Gutierrez M, Vaccaro GM, et al. Preliminary results of a phase 1 study of sea-CD40, gemcitabine, nab-paclitaxel, and pembrolizumab in patients with metastatic pancreatic ductal adenocarcinoma (PDAC). J Clin Oncol. 2022;40(4 suppl):Abstract 559.
- Wei SC, Anang NAAS, Sharma R, et al. Combination anti–CTLA-4 plus anti–PD-1 checkpoint blockade utilizes cellular mechanisms partially distinct from monotherapies. Proc Natl Acad Sci U S A. 2019;116:22699-22709.
- Parikh AR, Szabolcs A, Allen JN, et al. Radiation therapy enhances immunotherapy response in microsatellite stable colorectal and pancreatic adenocarcinoma in a phase II trial. Nat Cancer. 2021;2:1124-1135.
- Chen IM, Johansen JS, Theile S, et al. Randomized phase II study of nivolumab with or without ipilimumab combined with stereotactic body radiotherapy for refractory metastatic pancreatic cancer (CheckPAC). J Clin Oncol. 2022;40:3180-3189.
- Wolchok JD, Chiarion-Sileni V, Gonzalez R, et al. Overall survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med. 2017;377:1345-1356. Erratum in: N Engl J Med. 2018;379:2185.
- Brahmer J, Reckamp KL, Baas P, et al. Nivolumab versus docetaxel in advanced squamous-cell non–small-cell lung cancer. N Engl J Med. 2015;373:123-135.
- Borghaei H, Paz-Ares L, Horn L, et al. Nivolumab versus docetaxel in advanced nonsquamous non–small-cell lung cancer. N Engl J Med. 2015;373:1627-1639.
- Hellmann MD, Paz-Ares L, Bernabe Caro R, et al. Nivolumab plus ipilimumab in advanced non–small-cell lung cancer. N Engl J Med. 2019;381:2020-2031.
- Motzer RJ, Escudier B, McDermott DF, et al; for the CheckMate 025 investigators. Nivolumab versus everolimus in advanced renal-cell carcinoma. N Engl J Med. 2015;373:1803-1813.
- Motzer RJ, Tannir NM, McDermott DF, et al; for the CheckMate 214 investigators. Nivolumab plus ipilimumab versus sunitinib in advanced renal-cell carcinoma. N Engl J Med. 2018;378:1277-1290.
- El-Khoueiry AB, Sangro B, Yau T, et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 2017;389:2492-2502.
- Yau T, Kang YK, Kim TY, et al. Efficacy and safety of nivolumab plus ipilimumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib: the CheckMate 040 randomized clinical trial. JAMA Oncol. 2020;6:e204564. doi: 10.1001/jamaoncol.2020.4564. Erratum in: JAMA Oncol. 2021;7:140.
- Overman MJ, McDermott R, Leach JL, et al. Nivolumab in patients with metastatic DNA mismatch repair-deficient or microsatellite instability-high colorectal cancer (CheckMate 142): an open-label, multicentre, phase 2 study. Lancet Oncol. 2017;18:1182-1191. Erratum in: Lancet Oncol. 2017;18:510.
- Lenz HJ, Van Cutsem E, Limon ML, et al. First-line nivolumab plus low-dose ipilimumab for microsatellite instability-high/mismatch repair-deficient metastatic colorectal cancer: the phase II CheckMate 142 study. J Clin Oncol. 2022;40:161-170.
- Fabian KP, Padget MR, Donahue RN, et al. PD-L1 targeting high-affinity NK (t-haNK) cells induce direct antitumor effects and target suppressive MDSC populations. J Immunother Cancer. 2020;8:e000450. doi: 10.1136/jitc-2019-000450.
- Knudson KM, Hicks KC, Alter S, et al. Mechanisms involved in IL-15 superagonist enhancement of anti-PD-L1 therapy. J Immunother Cancer. 2019;7:82. doi: 10.1186/s40425-019-0551-y.
- Combination immunotherapy plus standard-of-care chemotherapy versus standard-of-care chemotherapy for the treatment of locally advanced or metastatic pancreatic cancer. NLM identifier: NCT04390399. Updated October 28, 2022. https://clinicaltrials.gov/ct2/show/NCT04390399. Accessed November 1, 2022.
- Seery TE, Nangia CS, Sender LS, et al. Trial in progress: open-label, randomized, comparative phase 2/3 study of combination immunotherapy plus standard-of-care chemotherapy and SBRT versus standard-of-care chemotherapy for the treatment of locally advanced or metastatic pancreatic cancer. J Clin Oncol. 2021;39(15 suppl):Abstract TPS4174.
- Seery TE, Nangia CS, McKean HA, et al. Phase 2 Quilt 88 trial of DAMP inducers combined with IL15 superagonist, N-803, and anti–PD-L1 NK cell therapy more than doubles historical overall survival in patients with third- to sixth-line advanced pancreatic cancer. J Clin Oncol. 2022;40(16 suppl):Abstract 4147.
- ImmunityBio. ImmunityBio announces results of phase 2 metastatic pancreatic cancer trial at ASCO GI with median overall survival of 6.3 months in patients with third-line disease, more than doubling historical survival. January 18, 2022. https://immunitybio.com/immunitybio-announces-results-of-phase-2-metastatic-pancreatic-cancer-trial-at-asco-gi-with-median-overall-survival-of-6-3-months-in-patients-with-third-line-disease-more-than-doubling-historical-sur/. Accessed March 19, 2022.
- ImmunityBio. ImmunityBio announces FDA acceptance of Biologics License Application for N-803 in BCG-unresponsive non-muscle-invasive bladder cancer carcinoma in situ. July 28, 2022. https://immunitybio.com/immunitybio-announces-fda-acceptance-of-biologics-license-application-for-n-803-in-bcg-unresponsive-non-muscle-invasive-bladder-cancer-carcinoma-in-situ/. Accessed November 1, 2022.
- Cuneo KC, Morgan MA, Sahai V, et al. Dose escalation trial of the Wee1 inhibitor adavosertib (AZD1775) in combination with gemcitabine and radiation for patients with locally advanced pancreatic cancer. J Clin Oncol. 2019;37:2643-2650.
- Jiang C, Zhang L, Xu X, et al. Engineering a smart agent for enhanced immunotherapy effect by simultaneously blocking PD-L1 and CTLA-4. Adv Sci (Weinh). 2021;8:2102500. doi: 10.1002/advs.202102500.
- Jin G, Guo S, Zhang Y, et al. Efficacy and safety of KN046 plus nab-paclitaxel/gemcitabine as first-line treatment for unresectable locally advanced or metastatic pancreatic ductal adenocarcinoma (PDAC). J Clin Oncol. 2021;39(15 suppl):Abstract 4138.
- KN046 in subjects with advanced pancreatic ductal adenocarcinoma. NLM identifier: NCT05149326. Updated October 28, 2022. https://clinicaltrials.gov/ct2/show/NCT05149326. Accessed November 2, 2022.
- Bazan-Peregrino M, Garcia-Carbonero R, Laquente B, et al. VCN-01 disrupts pancreatic cancer stroma and exerts antitumor effects. J Immunother Cancer. 2021;9:e003254. doi: 10.1136/jitc-2021-003254.
- Garcia-Carbonero R, Gil Martín M, Alvarez Gallego R, et al. Systemic administration of the hyaluronidase-expressing oncolytic adenovirus VCN-01 in patients with advanced or metastatic pancreatic cancer: first-in-human clinical trial. Ann Oncol. 2019;30(suppl 5):v271-v272.
- VCNBiosciences. Synthetic Biologics announces planned transformative acquisition of VCN Biosciences; developer of a novel oncolytic virus platform targeting pancreatic and other solid tumors. December 14, 2021. www.vcnbiosciences.com/index.php/news/97-synthetic-biologics-announces-planned-transformative-acquisition-of-vcn-biosciences-developer-of-a-novel-oncolytic-virus-platform-targeting-pancreatic-and-other-solid-tumors-2. Accessed March 19, 2022.
- Wenthe J, Eriksson E, Sandin L, et al. Inflaming advanced solid tumors including pancreatic cancer using LOAd703, a TMZ-CD40L/4-1BBL-armed oncolytic virus. Cancer Res. 2021;81(22 suppl):Abstract PO-018.
- Musher BL, Smaglo BG, Abidi W, et al. A phase I/II study of LOAd703, a TMZ-CD40L/4-1BBL-armed oncolytic adenovirus, combined with nab-paclitaxel and gemcitabine in advanced pancreatic cancer. J Clin Oncol. 2022;40(16 suppl):Abstract 4138.
- Mandelli F, Diverio D, Avvisati G, et al; for the Gruppo Italiano-Malattie Ematologiche Maligne dell’ Adulto and Associazione Italiana di Ematologia ed Oncologia Pediatrica cooperative groups. Molecular remission in PML/RARα-positive acute promyelocytic leukemia by combined all-trans retinoic acid and idarubicin (AIDA) therapy. Blood. 1997;90:1014-1021.
- Sunami Y, Böker V, Kleeff J. Targeting and reprogramming cancer-associated fibroblasts and the tumor microenvironment in pancreatic cancer. Cancers (Basel). 2021;13:697. doi: 10.3390/cancers13040697.
- Kocher HM, Basu B, Froeling FEM, et al. Phase I clinical trial repurposing all-trans retinoic acid as a stromal targeting agent for pancreatic cancer. Nat Commun. 2020;11:4841. doi: 10.1038/s41467-020-18636-w.
- Phase IIb randomised trial of ATRA in a novel drug combination for pancreatic cancer (STARPAC2). NLM identifier: NCT04241276. Updated January 27, 2020. https://clinicaltrials.gov/ct2/show/NCT04241276. Accessed November 3, 2022.
- Waters AM, Der CJ. KRAS: the critical driver and therapeutic target for pancreatic cancer. Cold Spring Harb Perspect Med. 2018;8:a031435. doi: 10.1101/cshperspect.a031435.
- Hallin J, Engstrom LD, Hargis L, et al. The KRASG12C inhibitor MRTX849 provides insight toward therapeutic susceptibility of KRAS-mutant cancers in mouse models and patients. Cancer Discov. 2020;10:54-71.
- Bekaii-Saab TS, Spira AI, Yaeger R, et al. KRYSTAL-1: updated activity and safety of adagrasib (MRTX849) in patients (Pts) with unresectable or metastatic pancreatic cancer (PDAC) and other gastrointestinal (GI) tumors harboring a KRASG12C mutation. J Clin Oncol. 2022;40(4 suppl):Abstract 519.
- Phase 1/2 Study of MRTX849 in patients with cancer having a KRAS G12C mutation KRYSTAL-1. NLM identifier: NCT03785249. Updated February 22, 2022. https://clinicaltrials.gov/ct2/show/NCT03785249. Accessed November 4, 2022.
- Mirati Therapeutics. U.S. Food and Drug Administration (FDA) accepts Mirati Therapeutics’ New Drug Application for adagrasib as treatment of previously treated KRASG12C-mutated non-small cell lung cancer. February 15, 2022. https://ir.mirati.com/press-releases/press-release-details/2022/U.S.-Food-and-Drug-Administration-FDA-Accepts-Mirati-Therapeutics-New-Drug-Application-for-Adagrasib-as-Treatment-of-Previously-Treated-KRASG12C-Mutated-Non-Small-Cell-Lung-Cancer/default.aspx. Accessed March 19, 2022.
- Lumakras (sotorasib) tablets, for oral use [prescribing information]. Amgen; May 2021. www.pi.amgen.com/-/media/Project/Amgen/Repository/pi-amgen-com/Lumakras/lumakras_pi_hcp_english.pdf. Accessed June 24, 2022.
- Hong DS, Fakih MG, Strickler JH, et al. KRASG12C inhibition with sotorasib in advanced solid tumors. N Engl J Med. 2020;383:1207-1217.
- Strickler JH, Satake H, Hollebecque A, et al. First data for sotorasib in patients with pancreatic cancer with KRAS p.G12C mutation: a phase I/II study evaluating efficacy and safety. J Clin Oncol. 2022;40(36 suppl):Abstract 360490.
- Mahalingam D, Burns MC, Kalyan A, et al. A phase Ib/II study of sotorasib combined with chemotherapy for second-line treatment of KRAS p. G12C–mutated advanced pancreatic cancer. J Clin Oncol. 2022;40(16 suppl):Abstract TPS4194.
- Golan T, Khvalevsky EZ, Hubert A, et al. RNAi therapy targeting KRAS in combination with chemotherapy for locally advanced pancreatic cancer patients. Oncotarget. 2015;6:24560-24570.
- Varghese AM, Ang C, Dimaio CJ, et al. A phase II study of siG12D-LODER in combination with chemotherapy in patients with locally advanced pancreatic cancer (PROTACT). J Clin Oncol. 2020;38(15 suppl):Abstract TPS4672.
- A phase 2 study of siG12D LODER in combination with chemotherapy in patients with locally advanced pancreatic cancer (PROTACT). NLM identifier: NCT01676259. Updated July 2, 2021. https://clinicaltrials.gov/ct2/show/NCT01676259. Accessed November 4, 2022.
- Pancreatic Cancer Action Network. Precision Promise. www.pancan.org/research/precision-promise/. Accessed March 19, 2022.
- Scholten J. Pancreatic Cancer Action Network announces groundbreaking clinical trial platform transforming development of treatment options for world’s toughest cancer. October 13, 2020. www.pancan.org/press-releases/pancreatic-cancer-action-network-announces-groundbreaking-clinical-trial-platform-transforming-development-of-treatment-options-for-worlds-toughest-cancer/. Accessed March 19, 2022.
- FibroGen. Pamrevlumab included in Pancreatic Cancer Action Network’s adaptive clinical trial platform. June 16, 2021. https://investor.fibrogen.com/news-releases/news-release-details/pamrevlumab-included-pancreatic-cancer-action-networks-adaptive. Accessed March 19, 2022.
- Cantargia. Cantargia advances nadunolimab against pancreatic cancer in the PanCAN Precision Promise phase 2/3 clinical trial. January 3, 2022. https://cantargia.com/en/press-releases/cantargia-advances-nadunolimab-against-pancreatic-cancer-in-the-pancan-precision-promisesm-phase-2-3-clinical-trial. Accessed March 19, 2022.
- Picozzi V, Alseidi A, Winter J, et al. Gemcitabine/nab-paclitaxel with pamrevlumab: a novel drug combination and trial design for the treatment of locally advanced pancreatic cancer. ESMO Open. 2020;5:e000668. doi: 10.1136/esmoopen-2019-000668.
- Evaluation of efficacy and safety of neoadjuvant treatment with pamrevlumab in combination with chemotherapy (either gemcitabine plus nab-paclitaxel or FOLFIRINOX) in participants with locally advanced pancreatic cancer. NLM identifier: NCT03941093. Updated March 16, 2022. https://clinicaltrials.gov/ct2/show/NCT03941093. Accessed October 28, 2022.
- Robbrecht D, Jungels C, Sorensen MM, et al. First-in-human phase 1 dose-escalation study of CAN04, a first-in-class interleukin-1 receptor accessory protein (IL1RAP) antibody in patients with solid tumours. Br J Cancer. 2022;126:1010-1017.
- Awada AH, Zematis M, Ochsenreither S, et al. Nadunolimab (CAN04), a first-in-class monoclonal antibody against IL1RAP, in combination with chemotherapy in subjects with pancreatic cancer (PDAC) and non-small cell lung cancer (NSCLC). Ann Oncol. 2021;32(suppl 5):S602-S603.