Connecting Science to Practice
This study adapts the KEYNOTE-522 trial’s findings into
real-world treatment for triple-negative breast cancer (TNBC)
and highlights neoadjuvant chemoimmunotherapy with
pembrolizumab as the standard of care, emphasizing the
survival benefits of dose-dense doxorubicin plus cyclophosphamide
(ddAC) with granulocyte colony-stimulating factor
support. Although ddAC increases logistical challenges, it
offers improved outcomes such as a higher rate of pathologic
complete responses, which is a surrogate end point for overall
survival. These findings underline the need to balance
efficacy, adverse event management, and patient-centered
care while exploring strategies such as extended pembrolizumab
dosing intervals to ease scheduling. This iterative approach
ensures evidence-based, optimized care delivery for
patients with TNBC.
Triple-negative breast cancer (TNBC) is an aggressive subtype of breast cancer that lacks the expression of estrogen, progesterone, and HER2 receptors, making treatment with targeted therapeutic agents ineffective.1 The KEYNOTE-522 trial established neoadjuvant chemoimmunotherapy as the standard of care for patients with stage II or III TNBC after achieving improved pathologic complete response (pCR) rates with the addition of pembrolizumab to a taxane- and anthracycline-based chemotherapy regimen.2 Achieving pCR at the time of surgery is a reliable surrogate end point for overall survival in patients with TNBC.3 In a recent update, the investigators of KEYNOTE-522 announced statistically significant benefits in overall survival and event-free survival with neoadjuvant pembrolizumab plus chemotherapy followed by adjuvant pembrolizumab versus neoadjuvant chemotherapy alone.4
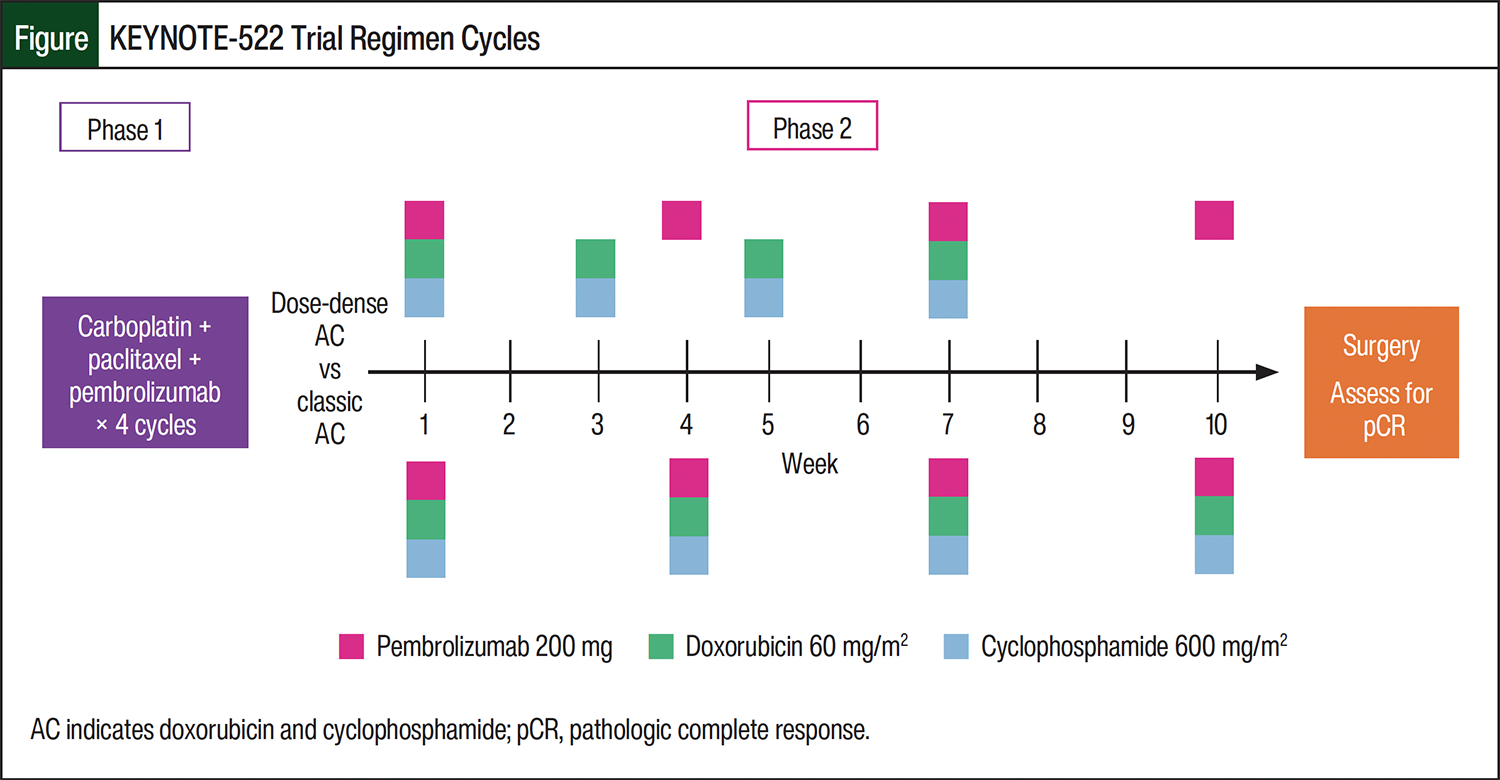
Although the KEYNOTE-522 trial regimen has been widely adopted across many centers since publication, the dosing schedule of doxorubicin and cyclophosphamide (AC) used in the trial differed from the current standard of care. AC administered every 2 weeks with growth factor support is termed dose-dense AC (ddAC) and has improved disease-free survival and overall survival with a reduced incidence of neutropenia when compared with a schedule of every 3 weeks (classic AC).5,6 The KEYNOTE-522 trial regimen consisted of 4 cycles of carboplatin weekly or every 3 weeks and paclitaxel weekly in a first phase, followed by 4 cycles of AC every 3 weeks in a second phase, with pembrolizumab administered every 3 weeks throughout and continued as adjuvant therapy (Figure). Administering AC every 3 weeks aligns with pembrolizumab infusions, reducing the number of infusion visits required and scheduling complexity.
Despite the scheduling asynchrony, the providers at our institution elect to prescribe a ddAC because of the demonstrated survival benefit and have created a modified KEYNOTE-522 regimen utilized in most patients at our institution. Few reports exist in the current literature that describe treatment with ddAC concomitantly with pembrolizumab.7 Our study sought to characterize the efficacy, safety, and scheduling logistics of ddAC within the KEYNOTE-522 regimen for TNBC, and to compare the outcomes between the small number of patients receiving AC every 3 weeks at our institution.
Methods
This was a retrospective cohort study that included adults with breast cancer who received at least 2 cycles of neoadjuvant AC concomitantly with pembrolizumab, carboplatin, and paclitaxel from October 1, 2021, to December 31, 2023, at Henry Ford Hospital in Detroit, MI. Patients were selected for the trial using a query of treatment plan titles entered into the electronic medical record. The modified KEYNOTE-522 treatment plan used at our hospital contains 4 cycles lasting 14 days each (except cycle 4), with AC received on day 1 of each cycle and pembrolizumab received on cycle 1 day 1, cycle 2 day 8, cycle 4 day 1, and cycle 4 day 22.
The prespecified patient characteristics for collection included age, sex, menopausal status, ECOG Performance Status, American Joint Committee on Cancer clinical stage group, nodal involvement, HER2 status score, and Ki-67 index. The study’s primary end point was the incidence of grade 4 neutropenia by Common Terminology Criteria for Adverse Events, version 5.0 at any point during the phase containing AC. The secondary end points included the pCR rate, incidences of anemia and thrombocytopenia, incidence of treatment delays, incidence of switching between AC schedules, sequence of neoadjuvant phases, total length of treatment, number of doses, and number of hospital admissions or emergency department visits during treatment with AC.
Statistical Analysis
With an estimated 3:1 enrollment ratio, 52 patients were needed to yield 80% power in detecting a difference in the primary outcome between the treatment groups with an alpha value of 0.05. The categorical variables were compared with Pearson’s chi-square analysis or Fischer’s exact test if the expected cell count was <5. The ordinal and continuous data were compared with a Mann–Whitney U test. The statistics were computed in IBM SPSS Statistics version 28 (IBM Corporation). The differences were considered significant if the P value was <.05. The descriptive statistics, including mean, median, and percentages, were used to describe the patient population. This study was approved by our system’s institutional review board.
Results
A total of 64 patients met the inclusion criteria for the study and received qualifying treatment from November 3, 2021, to December 12, 2023. In all, 48 patients received ddAC. A total of 4 patients (n=1 for each of the following reasons) planned to receive the dose-dense schedule but switched to the classic schedule after receiving the first dose of AC as a result of bone pain, decreased appetite, febrile neutropenia, and nonmalignant spinal cord compression from degenerative disc disease, and were included in the analysis of the classic AC group, which totaled 16 patients. One patient received 2 cycles of AC 2 weeks apart, was admitted for septic shock, and received the remaining doses of AC 3 weeks apart; that patient was included in the adverse event (AE) and scheduling analysis of the ddAC group but was excluded from the efficacy analysis. One patient who received ddAC declined to proceed to surgery, and 1 patient in the classic AC group had stage IV disease, so these patients were also excluded from the efficacy analysis.
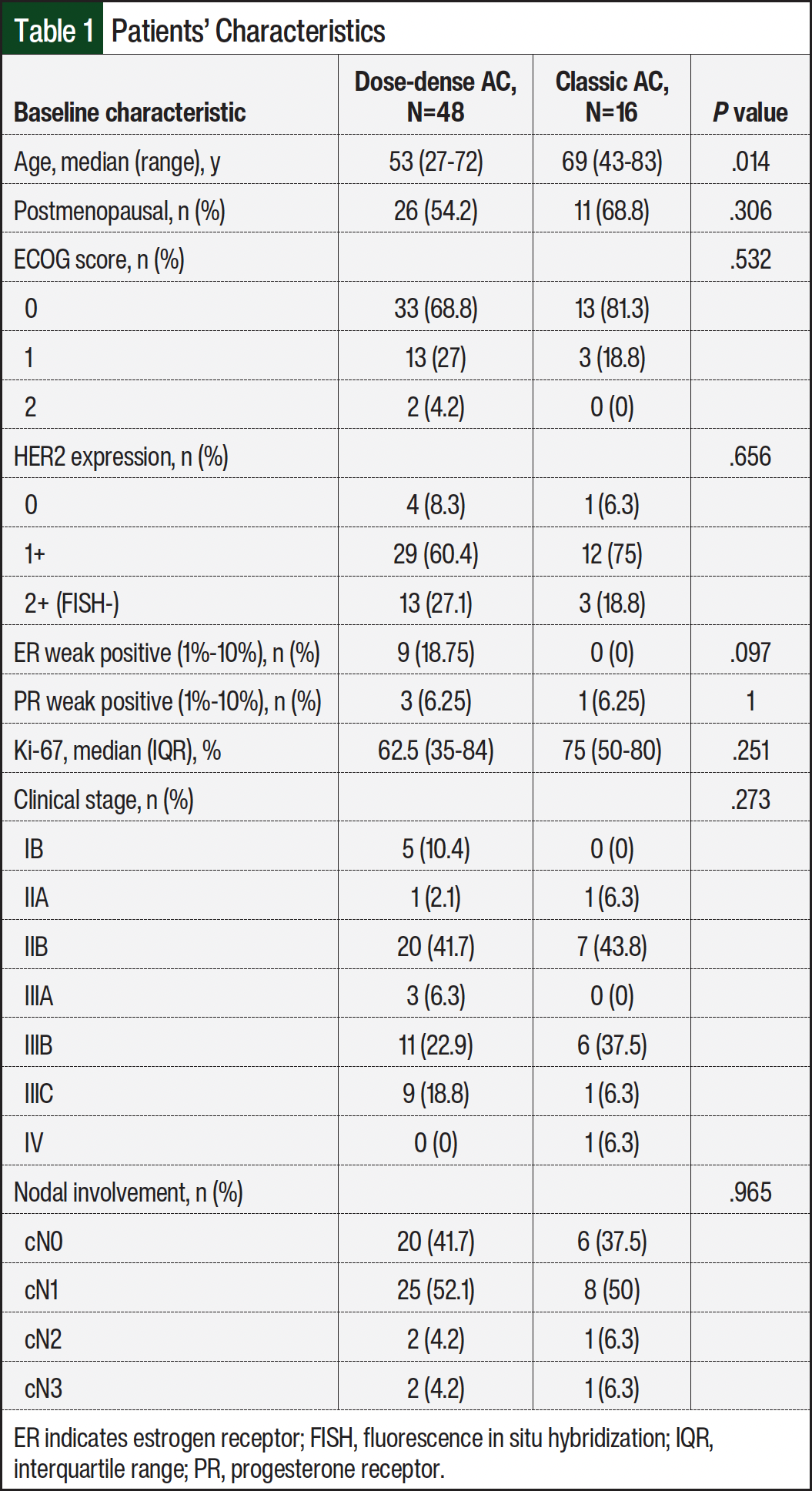
Table 1 shows the patients’ baseline characteristics. All of the patients were women, the median age was 54 years, and most had an ECOG score of 0. The performance status score, HER2 expression, Ki-67 index, clinical stage, and nodal involvement did not differ significantly between the groups. The patients who received ddAC were younger and more likely to have weak estrogen receptor positivity than the patients in the classic AC group.
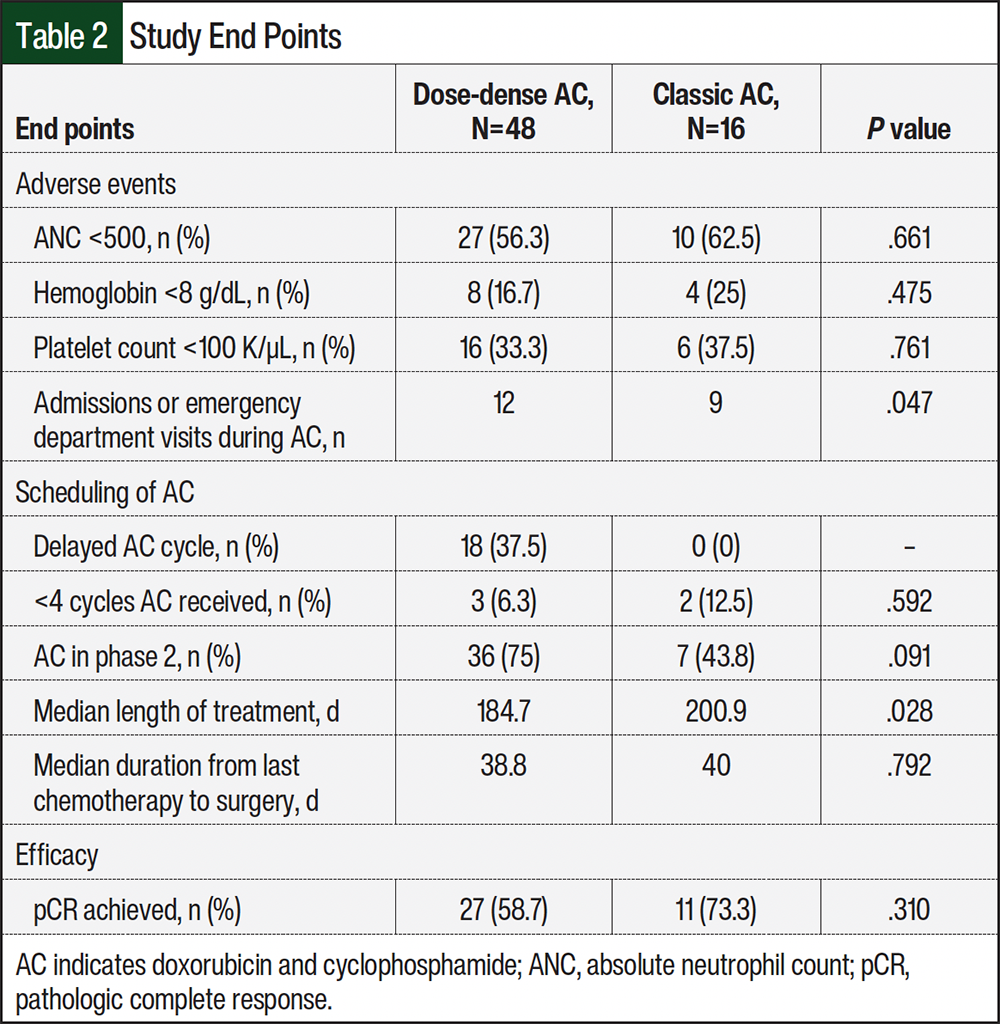
Grade 4 neutropenia during AC treatment occurred in 56.3% of patients in the ddAC group and in 62.5% of patients in the classic AC group (odds ratio [OR], 1.296; 95% confidence interval [CI], 0.406-4.142; P=.661; Table 2). Long-acting granulocyte colony-stimulating factor (G-CSF) was administered after all 4 doses of AC in all patients in the ddAC group. In the classic AC group, 7 (44%) patients received 4 doses of G-CSF, 5 patients received 3 doses (added after neutropenia occurred following cycle 1), 2 patients received 1 dose with cycle 1, and 2 patients did not receive any doses of G-CSF. Anemia (hemoglobin <8 g/dL) and thrombocytopenia (platelet count <100 K/µL) occurred in 16.7% and 33.3%, respectively, of patients in the ddAC group and in 25% and 37.5%, respectively, of patients in the classic AC group.
A total of 5 patients in the ddAC group and 2 patients in the classic AC group received carboplatin every 3 weeks with a target area under the curve (AUC) of 5, with the remainder receiving carboplatin once weekly with a target AUC of 1.5. A total of 75% of patients in the ddAC group and 43.8% of patients in the classic AC group received AC as the second phase of treatment (Table 2). Receiving AC in the second phase correlated with having a delay in AC treatment (OR, 0.22; 95% CI, 0.058-0.890; P=.009) in the overall study population. A cycle of AC was delayed in 18 (37.5%) patients in the ddAC group as a result of neutropenia (n=7), thrombocytopenia (n=2), COVID-19 (n=2), anemia (n=1), sepsis (n=1), abscess (n=1), diarrhea (n=1), bone pain (n=1), weight loss (n=1), and scheduling error (n=1). Of these patients, 16 continued to receive the remaining doses of AC in the dose-dense regimen. None of the patients in the classic AC group had a cycle of AC delayed. In all, 93.7% of patients received all 4 doses of AC in the ddAC group compared with 87.5% in the classic AC group (Table 2).
A total of 12 hospital admissions or emergency department visits occurred in the ddAC group as a result of febrile neutropenia (n=4), cough (n=3), septic shock (n=1), fever (n=1), abscess (n=1), pneumonia (n=1), and nausea/vomiting (n=1). In all, 9 occurred in the classic AC group as a result of febrile neutropenia (n=2), transient ischemic attack (n=1), chest pain (n=1), colitis (n=1), shortness of breath (n=1), nephritis (n=1), migraine (n=1), and spinal cord compression (n=1).
The mean and median pembrolizumab doses administered before surgery were 8 and did not differ between the treatment groups. A dose of pembrolizumab had been administered late (>25 days after the previous dose) in 11 (22.9%) patients in the ddAC group and in 4 (25%) patients in the classic AC group. A dose of pembrolizumab had been missed (>6 weeks in between doses) in 6 (12.5%) patients in the ddAC group and in 2 (12.5%) patients in the classic AC group. A dose of pembrolizumab was aligned to be given concomitantly with the first dose of chemotherapy in phase 2 (Figure) in 68.8% of patients in the ddAC group and in 87.5% of the classic AC group. A dose of pembrolizumab was given alone in between phase 1 and phase 2 of treatment in 27% of patients in the ddAC group and in 12.5% of patients in the classic AC group. A dose of pembrolizumab was given alone after the final dose of AC and before surgery in 79% of patients in the ddAC group and in 25% of patients in the classic AC group.
The median time from the first dose of chemotherapy to the day of surgery was 26.4 weeks in the ddAC group and 28.7 weeks in the classic AC group (P=.028). The median number of days from the final dose of chemotherapy to the day of surgery was 38.8 days in the ddAC group and 40 days in the classic AC group (P=.792; Table 2).
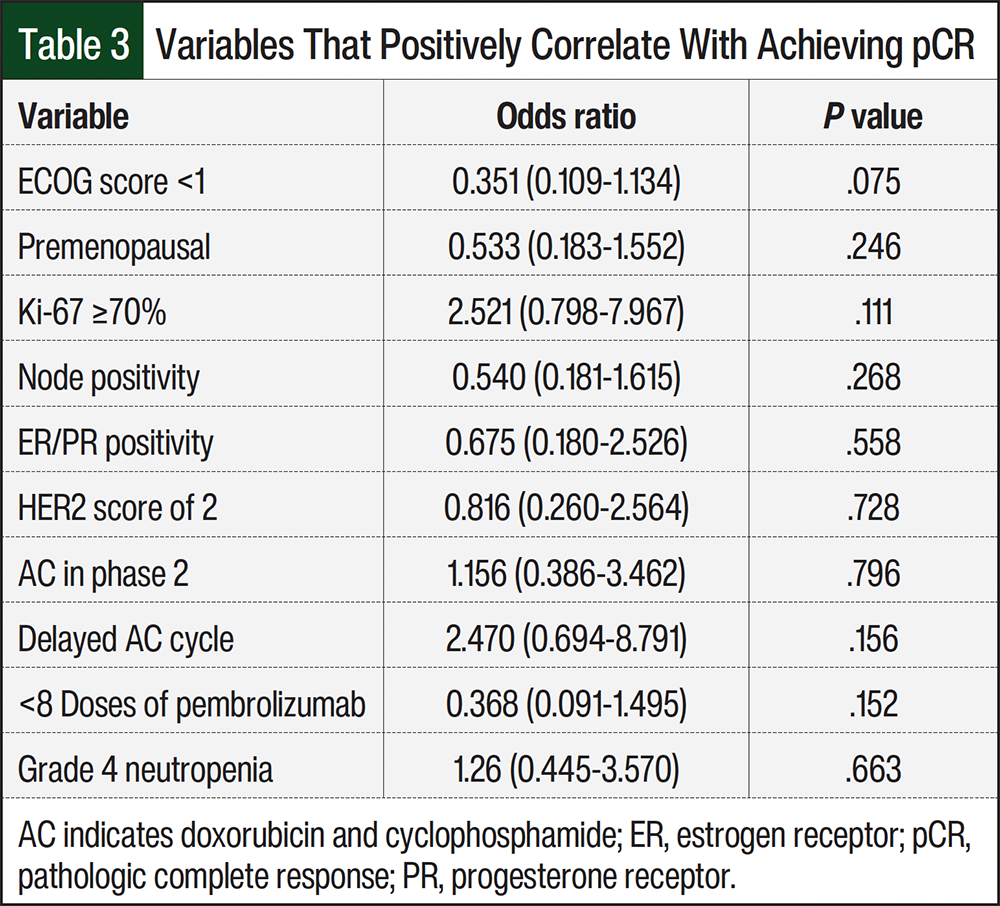
A total of 27 (58.7%) patients in the ddAC group achieved pCR compared with 11 (73.3%) patients in the classic AC group (OR, 1.935; 95% CI, 0.535-7.003; P=.310; Table 2). The variables in the entire study population that positively correlate with achieving pCR (P<0.2) were an ECOG score <1, Ki-67 of ≥70%, receiving fewer doses of pembrolizumab, and having a cycle of AC delayed (Table 3). Due to low enrollment in the classic AC group, a logistical regression incorporating these variables was not possible. The factors that did not correlate with pCR were premenopausal status, receiving AC in phase 2, node positivity, estrogen receptor/progesterone receptor positivity, a HER2 score of 2, and grade 4 neutropenia.
Discussion
Our study did not observe a significant difference in the incidence of neutropenia, anemia, or thrombocytopenia between the patients who received AC every 2 weeks and the patients who received AC every 3 weeks, but the inconsistent use of G-CSF in the classic AC group may greatly confound this finding. It is well known that the hematologic tolerability of the dose-dense approach is contingent on the use of growth factor with each cycle. Meanwhile, the use of G-CSF within the classic AC schedule at our institution varied among the providers’ preferences at the time of this study, with many providers waiting until neutropenia occurred after cycle 1 to initiate prophylaxis with subsequent cycles. The lack of consistency makes it difficult to conclude whether the observed hematologic outcomes are the result of receiving G-CSF or the dosing intervals themselves. Although there were significantly more hospital admissions and emergency department visits in the classic AC group than in the ddAC cohort, they were for a wide variety of indications that are unlikely to be attributed to AEs from the AC regimen, aside from the 2 patients who were admitted for febrile neutropenia.
The patients who received ddAC were more likely to have a cycle of AC delayed than the patients who received classic AC, but in nearly all cases the remainder of cycles were administered in the dose-dense method, indicating that AEs were usually not long-lasting. There was no difference in the number of patients who received all 4 doses of AC. Having a delay in AC was also associated with achieving pCR, suggesting that increased AEs may correlate with better disease response. A recent retrospective study of 128 patients at Memorial Sloan Kettering (MSK) who received a modified regimen similar to the KEYNOTE-522 regimen showed no significant difference in pCR rate among those who had a delay in receiving AC versus those who did not have a delay, although clinical importance approached the threshold.8 Delays in therapy are typically undesirable and thought to compromise outcomes, but in our study we did not observe any detrimental effects from treatment delays.
It is unknown if the sequence in which AC and carboplatin plus paclitaxel are administered has an impact on outcomes. Some of our providers chose to give AC first, perceiving it as the stronger component of the regimen. Both our study and MSK’s study did not find a significant correlation between sequence and pCR rate,8 but receiving AC in phase 2 (Figure) was associated with a higher chance of having a delay in AC (OR, 6.840; 95% CI, 1.412-33.141; P=.009). AC was received in phase 2 by most patients in the ddAC group, but providers were more likely to use AC upfront in the classic AC group. The total length of treatment was an average of 2 weeks longer in the classic AC group than in the ddAC group, and there was no difference in the amount of time between the last dose of chemotherapy and surgery, so giving a final dose of pembrolizumab alone in the ddAC group did not prolong the time to surgery.
When a dose of pembrolizumab is delayed, treatment plans must be manipulated within the electronic medical record to reschedule future doses, introducing the potential for error in the form of late or missed doses. We did not find any difference in this outcome between the groups, but our definitions of late and missed doses of pembrolizumab were based on the length of time between treatments and did not discriminate between intentional or unintentional delays, so an absence of scheduling error is not proven. Furthermore, the dose-dense approach requires a greater number of infusion center visits than when received classically, inflicting a greater time burden on patients and an increased financial burden on facilities. The incidence in which pembrolizumab was aligned to be received concomitantly with the first dose of chemotherapy in phase 2 was assessed to further characterize scheduling efficiency, with patients needing more infusion visits if not aligned. The misalignment of infusion visits occurred more frequently in the ddAC group than in the classic AC group, but its presence in the classic AC group altogether suggests multifactorial causes. The use of pembrolizumab in an extended interval of every 6 weeks would eliminate 2 infusion visits if properly aligned with AC treatment, which is a potential strategy to mitigate this complexity in future patients.
Limitations
Our study had some limitations. This study had an overall pCR rate of 59.4%, which was similar to the pCR rate in KEYNOTE-5222 and other trials8-10 of neoadjuvant chemoimmunotherapy for TNBC, and a numerically higher rate of pCR in the classic AC group. However, because of the anticipated modest enrollment in the classic AC group, the pCR rate was not selected as the primary outcome and our study was not powered to detect differences in efficacy.
The study’s small sample size precluded the ability to perform a linear regression to account for intergroup variations because of a lack of randomization. Additional limitations to the comparative AE analysis include the inconsistent use of G-CSF in the classic AC group, inconsistent phasing of AC and of carboplatin plus paclitaxel, and the introduction of bias by including patients who had at first intended to receive ddAC in the classic AC group.
Because the main objective of this study was to characterize feasibility in scheduling, as many patients were included as possible, and conclusions regarding AEs and efficacy should be interpreted with caution. The collection of residual cancer burden scoring metrics in patients who did not achieve a pCR would also have been desirable to further characterize efficacy and may be pursued in future studies.
Conclusion
Administering AC in the dose-dense approach within a modified KEYNOTE-522 regimen is feasible at our institution with a low incidence of missed or late doses of pembrolizumab. We did not observe greater AEs with a ddAC regimen compared with a classic AC regimen, but confounding variables attenuate the conclusions that can be drawn from our study. Our pCR rate is consistent with pCR rates in the previous literature, and future studies are needed to determine the differences in efficacy between ddAC and classic AC regimens.
Author Disclosure Statement
Dr Hodges, Dr Patterson, and Dr Ljucovic have no conflicts of interest to report.
References
- Foulkes WD, Smith IE, Reis-Filho JS. Triple-negative breast cancer. N Engl J Med. 2010;363:1938-1948. doi:10.1056/NEJMra1001389
- Schmid P, Cortes J, Pusztai L, et al. Pembrolizumab for early triple-negative breast cancer. N Engl J Med. 2020;382:810-821. doi:10.1056/NEJMoa1910549
- Cortazar P, Zhang L, Untch M, et al. Pathological complete response and long term clinical benefit in breast cancer: the CTNeoBC pooled analysis. Lancet. 2014;384:164-172. Erratum in: Lancet. 2019;393:986. doi:10.1016/S0140-6736(13)62422-8
- Schmid P, Cortes J, Dent R, et al. Overall survival with pembrolizumab in early-stage triple-negative breast cancer. N Engl J Med. 2024;391:1981-1991. doi:10.1056/NEJMoa2409932
- Citron ML, Berry DA, Cirrincione C, et al. Randomized trial of dose-dense versus conventionally scheduled and sequential versus concurrent combination chemotherapy as postoperative adjuvant treatment of node-positive primary breast cancer: first report of intergroup trial C9741/cancer and leukemia group B trial 9741. J Clin Oncol. 2003;21:1431-1439. Erratum in: J Clin Oncol. 2003;21:2226. doi:10.1200/JCO.2003.09.081
- Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Increasing the dose intensity of chemotherapy by more frequent administration or sequential scheduling: a patient-level meta-analysis of 37 298 women with early breast cancer in 26 randomised trials. Lancet. 2019;393:1440-1452. doi:10.1016/S0140-6736(18)33137-4
- Santa-Maria CA, O’Donnell M, Nunes R, et al. Integrating immunotherapy in early-stage triple-negative breast cancer: practical evidence-based considerations. J Natl Compr Canc Netw. 2022;20:738-744. doi:10.6004/jnccn.2022.7025
- Mai N, Myers S, Shen S, et al. Dose dense doxorubicin plus cyclophosphamide in a modified KEYNOTE522 regimen for triple negative breast cancer. NPJ Breast Cancer. 2024;10:39. doi:10.1038/s41523-024-00643-5
- Mittendorf EA, Zhang H, Barrios CH, et al. Neoadjuvant atezolizumab in combination with sequential nab-paclitaxel and anthracycline-based chemotherapy versus placebo and chemotherapy in patients with early-stage triple-negative breast cancer (IMpassion031): a randomised, double-blind, phase 3 trial. Lancet. 2020;396:1090-1100. doi:10.1016/S0140-6736(20)31953-X
- Gianni L, Huang CS, Egle D, et al. Pathologic complete response (pCR) to neoadjuvant treatment with or without atezolizumab in triple-negative, early high-risk and locally advanced breast cancer: NeoTRIP Michelangelo randomized study. Ann Oncol. 2022;33:534-543. doi:10.1016/j.annonc.2022.02.004