This research was presented at the Annual Hematology/Oncology Pharmacy Association (HOPA) conference on March 31, 2023, as an affiliated poster.
Connecting Science to Practice
These findings support the practical approach of initiating
immunotherapy in the outpatient setting after stabilizing
patients with urgent chemotherapy during hospitalization.
This strategy balances the high responsiveness of small cell
lung cancer (SCLC) to chemotherapy, the delayed onset of
immunotherapy effects, and the financial and operational
constraints of inpatient immunotherapy. Future prospective
studies may further clarify the optimal timing of immunotherapy
initiation, but current evidence suggests that
delayed initiation is a viable and effective option in managing
extensive-stage SCLC.
Small cell lung cancer (SCLC) is an aggressive form of lung cancer that is characterized by a rapid doubling time, high growth fraction, and early metastatic dissemination.1 Accounting for <20% of all lung cancer cases worldwide, SCLC has an estimated 5-year overall survival (OS) rate of approximately 7%.2,3 SCLC is classified as either limited or extensive stage (ES), with most patients presenting with ES-SCLC at diagnosis.1 Limited-stage SCLC is confined to the ipsilateral hemithorax; whereas, ES-SCLC extends beyond the ipsilateral hemithorax or single radiation field and typically presents with widespread metastases.1 The goal of treatment for limited-stage disease is cure; however, the treatment goals for ES disease are focused on prolonging survival and symptom palliation.1
For more than 30 years, the first-line therapy for ES-SCLC consisted of a platinum agent plus etoposide.4-6 Given its aggressive nature, SCLC is highly responsive to radiation and chemotherapy; however, the ability for SCLC to rapidly develop chemoresistance leads to a median OS of 10 months after first-line chemotherapy.4-6 In recent years, clinical trials have evaluated the efficacy of adding immunotherapy to upfront chemotherapy, because of the high tumor mutational burden and genomic instability that characterizes the malignancy.4-6 The IMpower133 clinical trial evaluated the addition of the programmed death-ligand 1 inhibitor atezolizumab to carboplatin and etoposide as first-line therapy in patients with ES-SCLC.6 The patients received 4 cycles of chemoimmunotherapy followed by maintenance atezolizumab until disease progression or unacceptable adverse events (AEs). Atezolizumab significantly improved OS, reaching 12.3 months with chemoimmunotherapy compared with 10.3 months with chemotherapy alone.5,6 Similarly, the CASPIAN trial evaluated OS with the addition of durvalumab to a platinum agent and etoposide. The OS was significantly longer when durvalumab was added to chemotherapy, reaching 13 months compared with 10.3 months when receiving chemotherapy alone.3 These trials have expanded first-line therapy to include atezolizumab or durvalumab added to chemotherapy.
Patients with SCLC often present with cough, dyspnea, weight loss, bone pain, neurologic compromise, and debility.1 In addition, patients with ES-SCLC may present with oncologic emergencies, including spinal cord compression, superior vena cava syndrome, and other severe complications requiring hospitalization.1 For acutely symptomatic patients requiring hospitalization and emergent initiation of chemotherapy, there are limitations regarding the use of upfront immunotherapy in combination with a platinum drug and etoposide. The financial toxicity associated with immune checkpoint inhibitors is a significant barrier to administering such agents during admission.7,8 Unlike in the clinic setting, health systems are unable to mitigate large acquisition costs using 340B pricing and direct drug reimbursement from payers to justify their inpatient use.8 Admitted patients also require urgent tumor debulking to stabilize their clinical status and preserve organ function because of the rapid doubling time that characterizes SCLC.9 The delayed onset of action for immunotherapy and the high sensitivity of the malignancy to cytotoxic chemotherapy emphasizes the utility of the latter in the initial treatment of hospitalized patients.8,10 Furthermore, the use of inpatient immunotherapy in medically frail patients can worsen performance status and lead to an increased risk of immune-related adverse events (irAEs) or death.11
Traditional agents such as carboplatin and etoposide can be inexpensively administered and quickly temporize disease while providing time to transition patients to triple-agent therapy after discharge. At the study’s institution, immunotherapy is restricted to initiation in the outpatient setting, leading to the addition of atezolizumab or durvalumab to subsequent cycles after discharge. To determine if this practice impacts patient outcomes, this analysis evaluated the survival of patients receiving delayed initiation of immunotherapy with subsequent cycles compared with early initiation of immunotherapy with the first cycle of treatment.
Methods
The institutional investigational review board approved this single-center, multisite, pharmacist-led, retrospective cohort study conducted through electronic health record review. The study included adults diagnosed with ES-SCLC who received first-line treatment with a platinum drug and etoposide plus either atezolizumab or durvalumab between January 1, 2020, and July 1, 2022. Patients aged <18 years or actively enrolled in a clinical trial were excluded from the study.
Early immunotherapy initiation was defined as patients who received atezolizumab or durvalumab in combination with a platinum drug and etoposide in the outpatient setting starting with the first cycle of treatment. Delayed immunotherapy initiation was defined as patients who received atezolizumab or durvalumab starting with the second or subsequent cycles of therapy for any reason and a platinum agent and etoposide alone with any previous cycles.
The primary end point of this comparative effectiveness study was OS, which was defined as the time from the start of initial therapy to death from any cause or loss to follow-up. Loss to follow-up was defined using a last-encounter schema, where the date of loss to follow-up was the latter of last treatment date or clinic visit. Patients who did not die during the study period were considered to not have an OS event. The patients who did not have a confirmed death date were censored at their date of loss to follow-up or on the date of data abstraction (ie, July 1, 2022), whichever occurred first.
Progression-free survival (PFS) was assessed secondarily and was defined as the time from the start of initial therapy to the date of disease progression, death from any cause in the absence of progression, or loss to follow-up. Disease progression was determined independently by study investigators through evaluating the presence of radiographic or pathologic progression or as described by the treating physician for tumor assessments. The additional secondary end points included the incidence of grade ≥3 hematologic AEs as well as select irAEs between the study groups. Hematologic AEs were categorized according to the Common Terminology Criteria for Adverse Events version 5.0,12 thyroid dysfunction was based on the National Comprehensive Cancer Network guideline definitions for subclinical or overt hypothyroidism,13 transaminitis was classified as grade ≥3 aspartate aminotransferase/alanine transaminase elevations per the NCCN’s guideline definitions,13 and all other irAEs were classified as those leading to treatment interruption or discontinuation. All safety end points were categorized by the researchers through retrospectively reviewing laboratory results when applicable or otherwise based on documentation in the providers’ notes, physical assessments, and diagnostic evaluations.
Statistical Analysis
The patients’ characteristics, outcomes, and safety were summarized using frequencies and percent for categorical variables or as median and range for continuous variables. PFS and OS were estimated by the Kaplan-Meier analysis and differences were assessed using a log-rank (Mantel–Cox) test. The Cox proportional hazards model was used to evaluate the effect of delayed immunotherapy initiation, including hazard ratios (HRs) and 95% confidence intervals (CIs). All tests were 2-sided, and P values ≤.05 were considered statistically significant. A priori power analysis was not conducted to determine the sample size necessary to ensure adequate power for detecting a statistically significant effect between the groups. Thus, all statistics should be interpreted accordingly. Analyses were performed using GraphPad Prism 9 version 9.5.1 (GraphPad Software, LLC; Boston, MA).
Results
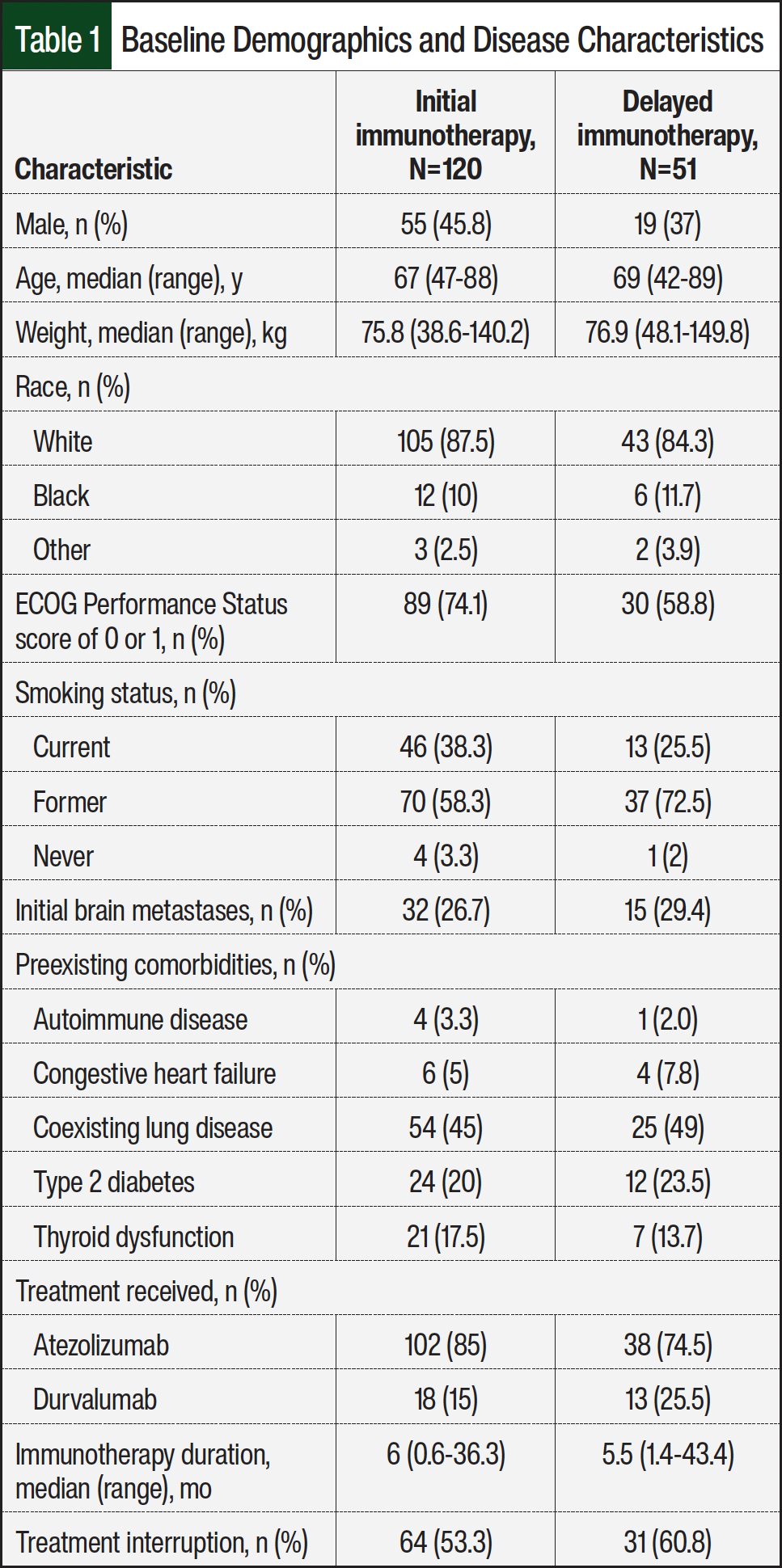
Throughout the 2.5-year study period, 188 patients with ES-SCLC who received first-line treatment with platinum doublet therapy plus immunotherapy were identified, and 171 patients met the full inclusion criteria. Among the excluded patients, 6 received part of their treatment at another institution, 5 had limited-stage disease, 5 received maintenance immunotherapy only, and 1 did not respond to their first cycle of treatment. The patients’ median age was 68 years (range, 42-89 years), 74 (43.3%) of the patients were men, 166 (97%) were current or former smokers, and 47 (27.5%) had baseline brain or central nervous system (CNS) metastases (Table 1). In all, 69.5% of the patients had an ECOG Performance Status score of 0 or 1; however, a larger portion of patients in the delayed immunotherapy group had an ECOG score of ≥2. The additional baseline characteristics were balanced between the study groups (Table 1).
A platinum drug plus etoposide plus atezolizumab was the most frequently prescribed regimen, administered to 140 (81.9%) patients, whereas durvalumab was the immunotherapy of choice for 31 (18.1%) patients (Table 1). A total of 120 (70.2%) patients had early initiation of immunotherapy with the first cycle of therapy, and 51 (29.8%) patients had delayed initiation of immunotherapy with subsequent cycles. The cause of delay in immunotherapy initiation to the second or later cycle was most often because of hospitalization during the first cycle of chemotherapy (78.4%). Additional reasons for immunotherapy delay included concomitant whole-brain radiation therapy with steroids (7.8%), lack of treatment authorization and insurance approval (7.8%), and active transaminitis (5.9%). The median duration of immunotherapy was 6 months (range, 0.6-36.3 months) in the early-initiation group and 5.5 months (range, 1.4-43.4 months) in the delayed-initiation group (Table 1).
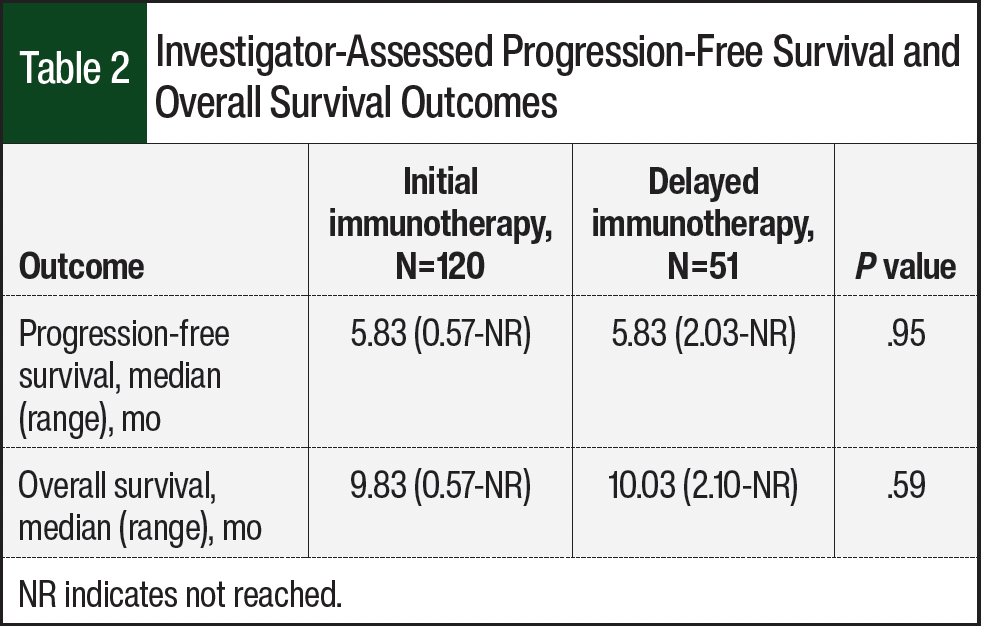
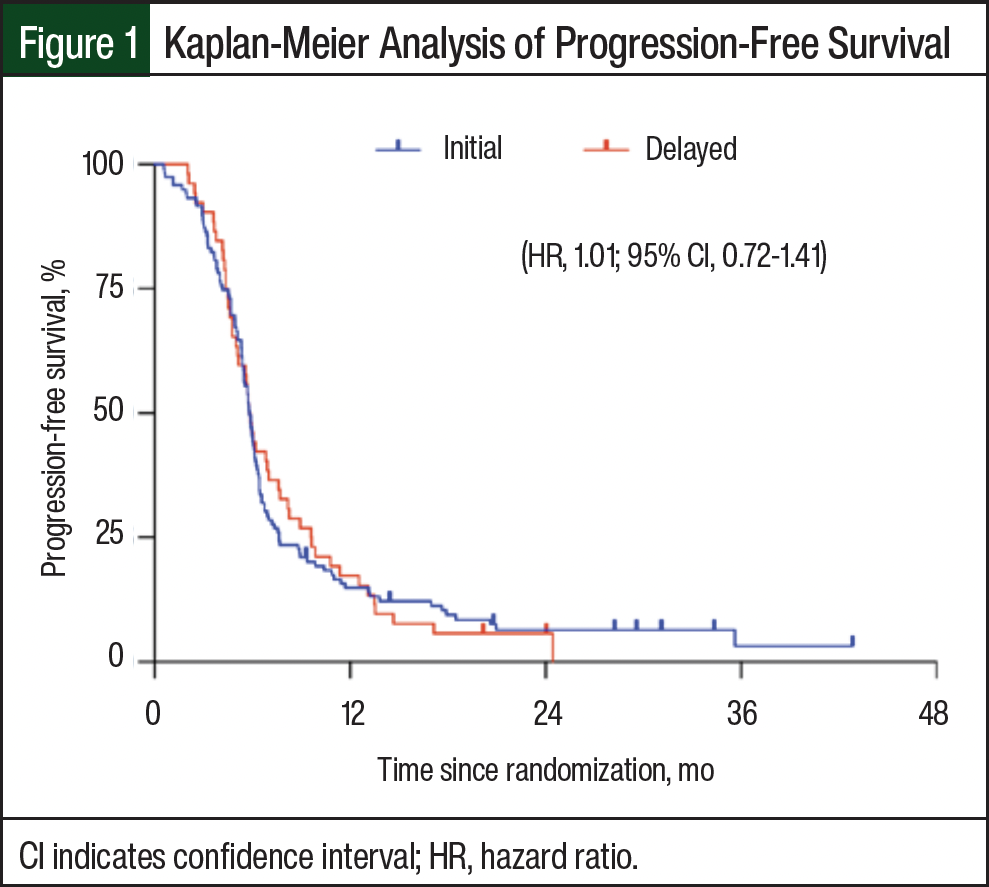
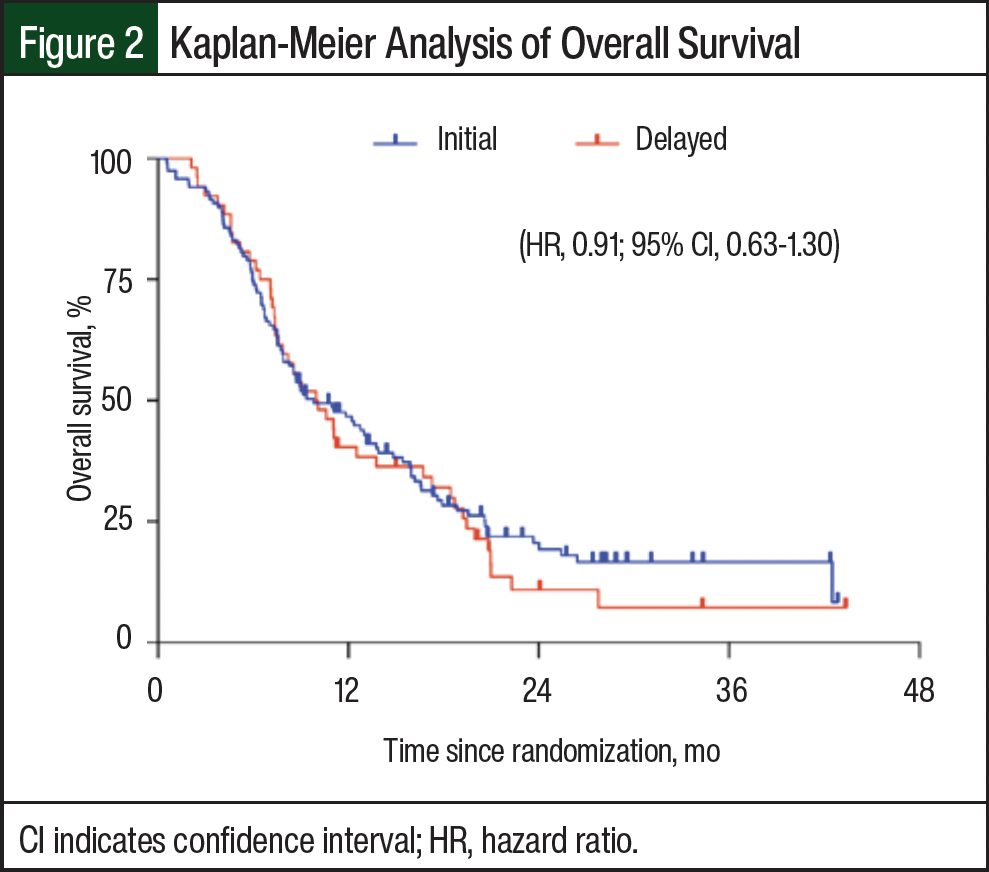
The median PFS was 5.83 months (range, 0.57 months-not reached [NR]) in the early immunotherapy group and 5.83 months (range, 2.03-NR) in the delayed immunotherapy group (Table 2). The HR for disease progression was 1.01 (95% CI, 0.72-1.41; Figure 1). The 6-month PFS rates were 44.5% and 44.2%, and the 12-month PFS rates were 14.7% and 17% in the early and delayed immunotherapy groups, respectively. There were 145 (84.8%) deaths in the early immunotherapy initiation group and 44 (86.3%) deaths in the delayed initiation group. The median OS was 9.83 months (range, 0.57-NR) and 10.03 months (range, 2.10-NR) for patients in the early and delayed immunotherapy groups, respectively (Table 2), with an HR for death of 0.91 (95% CI, 0.63-1.30; Figure 2). The 12-month OS rates were 46.5% in the early immunotherapy initiation group and 40.2% in the delayed immunotherapy initiation group.
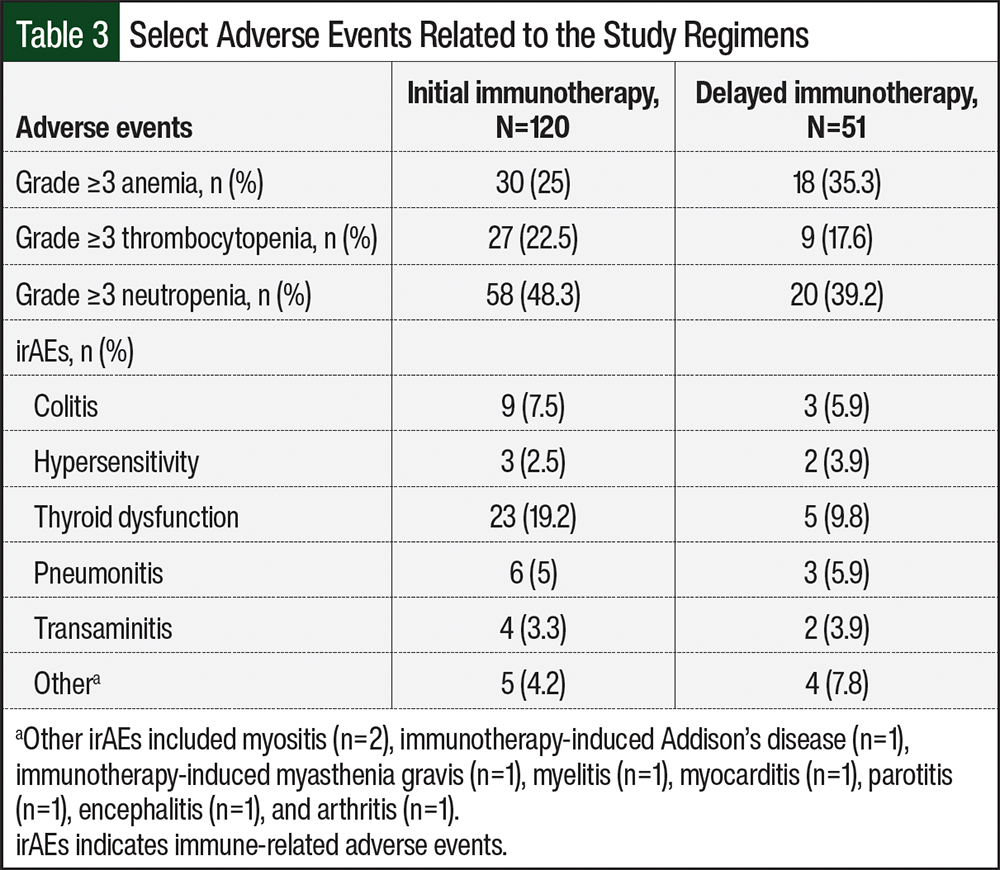
Grade ≥3 AEs of any cause occurred in 75% of patients in the early immunotherapy group and in 74.5% of patients in the delayed immunotherapy group (Table 3). The most common hematologic AE in both groups was neutropenia, which occurred in 48.3% and 39.2% of patients in the early and delayed immunotherapy groups, respectively. Grade ≥3 irAEs were reported in 33.3% of patients in the early immunotherapy group and in 27.5% of patients in the delayed immunotherapy group. The most common irAE in each group was thyroid dysfunction, which occurred in 19.2% of patients in the early immunotherapy group and in 9.8% of patients in the delayed immunotherapy group. AEs led to an interruption in therapy for 53.3% of patients in the early immunotherapy group and for 60.8% of patients in the delayed immunotherapy group. The most common causes of treatment delay included fatigue, neutropenia, and irAEs requiring steroids. Deaths resulting from irAEs occurred in 3 patients, all in the early immunotherapy group; the cause of death was pneumonitis in all 3 events.
Discussion
The findings of this retrospective review show that delaying the start of immunotherapy to subsequent outpatient cycles in patients requiring inpatient initiation of first-line treatment for ES-SCLC resulted in no apparent changes in survival outcomes. The patients who delayed immunotherapy beginning with the second treatment cycle or later had numerically similar OS to those who received immunotherapy starting with their first cycle of treatment. Consistent with the results for OS, PFS was identical between the 2 groups.
Literature published before the addition of immunotherapy to the first-line treatment of ES-SCLC concluded that patients with SCLC who initially received treatment in the acute care setting, especially those with a poor performance status, achieve shorter PFS and OS than those who received treatment in the ambulatory care setting.14 In our study and the review by Rittberg and colleagues, patients with delayed immunotherapy initiation had a 12-month OS rivaling that of patients who began receiving treatment in the outpatient setting. The equalizing effect on survival in our study may be in part attributed to the addition of immunotherapy to previous standard-of-care platinum-based chemotherapy regimens, because despite the differences in initial immunotherapy start times in our patients, the median durations of immunotherapy received in both groups were similar.
These similar survival outcomes also contradict that the delayed immunotherapy group had an appropriately worse initial ECOG score than the patients in the early immunotherapy group, given that the diminished efficacy of immunotherapy in patients with poor performance is well documented.15,16 It could be postulated that receiving chemotherapy in the inpatient setting improves performance status for these already sicker patients, allowing for immunotherapy once initiated with subsequent cycles to provide a more robust therapeutic effect, similar to that of fitter patients starting in the outpatient setting. This hypothesis is being investigated in the as yet unpublished phase 2 REACTION trial, where patients with ES-SCLC who achieve a complete or partial response after receiving 2 cycles of platinum-based chemotherapy, including patients who enrolled with an ECOG performance score of 2 and then upgrade to an ECOG score of 1 or 0 with treatment, are then randomized to continue receiving chemotherapy or to combination chemoimmunotherapy with subsequent cycles.17
The general survival results of our 2 patient populations have mixed consistency with those of the phase 3 IMpower133 and CASPIAN trials.3,6 The median PFS of each group in our study aligns with what was observed in the chemoimmunotherapy arms of the IMpower133 and CASPIAN trials.3,6 In addition, the 12-month PFS rates for the early and delayed immunotherapy groups are between those in the chemoimmunotherapy arms of the 2 trials. There was a larger difference of almost 3 months, however, when comparing the OS of patients receiving chemoimmunotherapy in the phase 3 trials with those of the real-world population in our review.3,6 This discrepancy in OS may be a result of the heterogeneous patient population that we included, which more closely represents clinical practice.
In the IFCT-1905 CLINATEZO study, a nationwide, retrospective chart review of 518 patients in France that mimicked the inclusion criteria of the IMpower133 trial using real-world patients, the median PFS was 5.2 months and the median OS was 11.3 months, resembling our findings.18 Unlike the landmark phase 3 trials, our study did not exclude patients with an ECOG score of ≥2, patients with active or untreated CNS metastases or leptomeningeal disease, or patients with a history of autoimmune disease. Furthermore, the median age at diagnosis, the proportion of patients with brain metastases at enrollment, and current or former smoker status were all higher in our study than in the chemoimmunotherapy arms of the IMpower133 and CASPIAN trials.3,6 Each of these baseline characteristics has been associated with poorer prognosis and may explain the overall lower OS in our population.19,20
Two recent phase 3b trials, MAURIS and IMfirst, sought to evaluate the efficacy of frontline chemoimmunotherapy in a broader ES-SCLC population closer to clinical practice by including these patients with an ECOG score of 2, untreated asymptomatic brain metastases, or underlying autoimmune diseases.21,22 The median real-world PFS was 5.5 months and 6.2 months, respectively, whereas the median OS was 10.7 months and 10 months, respectively, for the 2 trials.21,22 These results also align with our study and highlight the challenges in providing durable outcomes in patients with a poorer overall status in the real world.
Although the overall incidence of grade ≥3 AEs was higher in our study than in the previously mentioned phase 3 trials,21,22 it was lower than the incidences in the other real-world retrospective reviews.3,6,15,17 There was no large disparity in AEs between the patients who received immunotherapy with the first cycle and those who received immunotherapy with delayed initiation, with the exception of the incidence of immune-mediated subclinical or primary hypothyroidism. This discrepancy could be attributed to the numerically higher percentage of patients with thyroid dysfunction before treatment initiation in the initial immunotherapy group versus in the delayed immunotherapy group (Table 1).
Limitations
This study has several limitations, including its single-center, retrospective, nonrandomized chart review design. Accuracy for determining disease progression, survival, and drug-related AEs relied on documentation in the electronic health record that spans multiple sites within our study institution and is not as detailed as those in clinical trials. The timeline of this retrospective review also took place during the COVID-19 pandemic, which could have reduced the number of patient visits and increased the administrative burdens, potentially altering the frequency of monitoring and the reporting rates of disease status and treatment-related AEs.
Given the lack of difference in outcomes between the 2 patient cohorts, a post-hoc analysis was conducted. With an expected power value of 0.8, the minimum detectable effect on survival that could be detected based on this study population is 1.04 months. Consequently, a sample size of more than 3100 patients would be required to provide sufficient power to validate our observed difference in survival between the groups. The lack of statistical power necessary to clearly determine a significant nonequivalent difference margin in survival limits the ability to propose any definitive claims on outcomes, and all findings must be taken with caution and can only be hypothesis generating.
Conclusion
To our knowledge, this is the first study of the potential impact on survival of delaying immunotherapy to subsequent outpatient cycles in patients with ES-SCLC who are hospitalized for the first cycle of treatment. Taken together, our survival results are also consistent with recent literature assessing the real-world efficacy of first-line chemoimmunotherapy.
Delaying the initiation of immunotherapy to subsequent cycles in patients with ES-SCLC did not result in a numerically shorter PFS or OS; however, future prospective studies that assess the timing of immunotherapy initiation could provide more definitive evidence to this claim. Despite the relatively limited sample of our study, this study offers valuable insights for institutions to assess the restriction criteria of inpatient immunotherapy. As a result of our findings, it may be possible to safely limit the use of atezolizumab or durvalumab to the outpatient setting in patients receiving a concomitant platinum agent and etoposide without negatively impacting clinical outcomes.
Editor’s Note: The views expressed in this article are those of the researchers and do not necessarily reflect the position or policy of the Department of Veterans Affairs or the US government.
References
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): small cell lung cancer. Version 2.2026. September 16, 2025. Accessed December 9, 2025. www.nccn.org/professionals/physician_gls/pdf/sclc.pdf
- Giunta EF, Addeo A, Rizzo A, Banna GL. First-line treatment for advanced SCLC: what is left behind and beyond chemoimmunotherapy. Front Med (Lausanne). 2022;9:924853.
- Paz-Ares L, Dvorkin M, Chen Y, et al. Durvalumab plus platinum-etoposide versus platinum-etoposide in first-line treatment of extensive stage small-cell lung cancer (CASPIAN): a randomized, controlled, open-label, phase 3 trial. Lancet. 2019;394:1929-1939.
- Meijer JJ, Leonetti A, Airò G, et al. Small cell lung cancer: novel treatments beyond immunotherapy. Semin Cancer Biol. 2022;86:376-385.
- Pacheco JM. Immunotherapy for extensive stage small cell lung cancer. J Thorac Dis. 2020;12:6212-6224.
- Horn L, Mansfield AS, Szczesna A, et al. First-line atezolizumab plus chemotherapy in extensive-stage small-cell lung cancer. N Engl J Med. 2018;379:2220-2229.
- Riaz F, Vaugh JL, Zhu H, et al. Inpatient immunotherapy outcomes study: a multicenter retrospective analysis. JCO Oncol Pract. 2025;21:1165-1173.
- Patel AK, Duperreault MF, Pandya CJ, et al. Outcomes of immune checkpoint inhibitor administration in hospitalized patients with solid tumor malignancies. JCO Oncol Pract. 2023;19:e298-e305.
- Shrestha P, Kao S, Cheung VK, et al. Circulating tumor cells: advancing personalized therapy in small cell lung cancer patients. Mol Oncol. 2025;19(7):1998-2013.
- O’Sullivan H, Collins D, O’Mahony D, et al: End-of-life care and immune checkpoint inhibitors. J Clin Oncol. 2019;37(suppl 15):Abstr 11531.
- Glisch C, Hagiwara Y, Gilbertson-White S, et al. Immune checkpoint inhibitor use near the end of life is associated with poor performance status, lower hospice enrollment, and dying in the hospital. Am J Hosp Palliat Care. 2020;37:179-184.
- US Department of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE). Version 5.0. November 27, 2017. Accessed December 9, 2025. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): management of immunotherapy-related toxicities. Version 1.2025. December 20, 2024. Accessed December 9, 2025. www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf
- Rittberg R, Green S, Aquin T, et al. Effect of hospitalization during first chemotherapy and performance status on small-cell lung cancer outcomes. Clin Lung Cancer. 2020;21:e388-e404.
- Kubo S, Kobayashi N, Matsumoto H, et al. Atezolizumab addition to platinum doublet: evaluating survival outcomes for patients with extensive disease small cell lung cancer. J Cancer Res Clin Oncol. 2023;149:17419-17426.
- Belluomini L, Calvetti L, Inno A, et al. SCLC treatment in the immuno-oncology era: current evidence and unmet needs. Front Oncol. 2022;12:840783.
- ClinicalTrials.gov. REACTION: a phase II study of etoposide and cis/carboplatin with or without pembrolizumab in untreated extensive small cell lung cancer. NLM identifier: NCT02580994. Updated December 11, 2023. Accessed September 25, 2024. https://clinicaltrials.gov/study/NCT02580994
- Falchero L, Guisier F, Darrason M, et al. Long-term effectiveness and treatment sequences in patients with extensive stage small cell lung cancer receiving atezolizumab plus chemotherapy: results of the IFCT-1905 CLINATEZO real-world study. Lung Cancer. 2023;185:107379.
- Ma X, Zhang Z, Chen X, et al. Prognostic factor analysis of patients with small cell lung cancer: real-world data from 988 patients. Thorac Cancer. 2021;12:1841-1850.
- Huang LL, Hu XS, Wang Y, et al. Survival and pretreatment prognostic factors for extensive-stage small cell lung cancer: a comprehensive analysis of 358 patients. Thorac Cancer. 2021;12:1943-1951.
- Bria E, Morgillo F, Garassino MC, et al. Atezolizumab plus carboplatin and etoposide in patients with untreated extensive-stage small-cell lung cancer: interim results of the MAURIS phase IIIb trial. Oncologist. 2024;29:e690-e698.
- Garcia Campelo MR, Domine Gomez M, De Castro Carpeno J, et al. Primary results from IMfirst, a phase IIIb open label safety study of atezolizumab (ATZ) + carboplatin (CB)/cisplatin (CP) + etoposide (ET) in an interventional real-world (RW) clinical setting of extensive-stage small cell lung cancer (ES-SCLC) in Spain. Ann Oncol. 2022;33(15 suppl):S1246-S1247.