Connecting Science to Practice
Published literature has shown the addition of bevacizumab to
trifluridine and tipiracil extended survival from 7.1 months to 10.8
months in patients with previously treated colorectal cancer. This
study represents a single-center experience with this combination
compared with trifluridine and tipiracil alone, reviewing previous
patients’ treatment at a single center over a 3-year timespan. Our
study showed that survival outcomes were not improved with combination
therapy in this trial, and no new adverse events were
noted. The additional intravenous therapy with bevacizumab to
trifluridine and tipiracil raises the question of cost versus benefit
and warrants a discussion among the care team.
Colorectal cancer (CRC) is the third leading cause of cancer-related death in the United States.1 The 5-year survival rate for patients with metastatic CRC is <20%, without treatment the estimated survival is 6 to 12 months. Treatment with intensive chemotherapy can increase patient survival time to 2 to 3 years.1 Fluorouracil-based chemotherapy regimens (ie, fluorouracil and oxaliplatin, fluorouracil and irinotecan) are recommended first-line therapies that may be administered with or without bevacizumab or EGFR-targeted agents (ie, panitumumab, cetuximab).1 Furthermore, data from a randomized, controlled trial demonstrated a survival benefit for continuing treatment with bevacizumab in the second-line setting, even after disease progression.2
After exhausting the fluorouracil-based chemotherapy options, there was no alternate approved therapy that showed improved progression-free survival (PFS) beyond 3 months.1 Regorafenib, fruquintinib, and trifluridine plus tipiracil are FDA-approved for the second- or third-line treatment of metastatic CRC in patients who received fluoropyrimidine-, oxaliplatin-, and irinotecan-based chemotherapy and an antivascular endothelial growth factor therapy or anti-EGFR therapy, if indicated, as an alternative to best supportive care or participation in clinical trials.1,3 Trifluridine plus tipiracil is a combination of a thymidine-based nucleoside analog and a thymidine phosphorylase inhibitor that interferes with DNA synthesis and cellular proliferation.4 In the phase 3 RECOURSE trial, trifluridine plus tipiracil resulted in improvement in overall survival (OS) when compared with placebo (median, 7.1 months vs 5.3 months, respectively; P<.001) in patients with metastatic CRC who previously received fluoropyrimidine-based chemotherapy.4
In recent years, 2 phase 2 trials have evaluated the benefit of adding bevacizumab to treatment with trifluridine and tipiracil.5,6 The C-TASK FORCE trial was a single-arm, phase 1/2 trial that evaluated 25 patients with unresectable, metastatic CRC that was refractory to treatment with or who were intolerant of first-line therapy.5 The patients received trifluridine and tipiracil (35 mg/m2 orally on days 1-5 and on days 8-12) with bevacizumab (5 mg/kg intravenously on days 1 and 15) every 28 days. At 16 weeks, the median PFS was 3.7 months (95% confidence interval [CI], 2-5.4 months) by central assessment, and the median OS was 11.4 months (95% CI, 7.6-13.9 months).5
A randomized phase 2 trial evaluated 93 patients with metastatic CRC that was refractory to treatment with or who were intolerant of fluorouracil-based therapy to receive trifluridine and tipiracil with or without bevacizumab in a similar fashion as the C-TASK FORCE regimen.6 Previous treatment with bevacizumab was allowed; approximately 80% of patients in each arm had received bevacizumab.6 The median PFS was 4.6 months with trifluridine and tipiracil plus bevacizumab and 2.6 months with trifluridine and tipiracil without bevacizumab (P=.001).6
In another prospective, single-arm phase 2 trial, the median PFS was 4.5 months with trifluridine and tipiracil plus bevacizumab (95% CI, 1.8-7.1 months).7 The phase 3 SUNLIGHT trial confirmed the previous trials’ findings and showed an improvement in OS with trifluridine and tipiracil plus bevacizumab therapy, with a median OS of 10.8 months versus 7.5 months with trifluridine and tipiracil without bevacizumab (hazard ratio, 0.61; 95% CI, 0.49-0.77; P<.001).8 There were no new safety signals with the addition of bevacizumab, but there were more dose delays, neutropenia (including grade ≥3), nausea, and hypertension in the bevacizumab-containing arm than in the arm that did not receive bevacizumab.8 The results from these trials led to a recommendation by the National Comprehensive Cancer Network for the use of trifluridine and tipiracil with or without the addition of bevacizumab in the third- and fourth-line treatment settings for CRC.3
To our knowledge, there are no studies that have included regorafenib in a previous line of therapy, and approximately 20% of patients in the trial by Pfeiffer and colleagues had not previously received bevacizumab.5-8 The differences from clinical trial to real-world exposure could include an increased use of bevacizumab or regorafenib. The use of bevacizumab should be considered, as well as that the incremental benefit of doing so is lower in later lines of therapy, although data support the continued use of bevacizumab in earlier lines of therapy.1 Previous treatment with regorafenib, which inhibits vascular endothelial growth factor among multiple kinases, may also contribute to treatment resistance.9
There are limited data describing the real-world experience of treatment with trifluridine and tipiracil plus bevacizumab in patients with metastatic CRC, especially in patients with a previous increased exposure to bevacizumab or those who previously received treatment with regorafenib. Thus, we sought to share our real-world experience with the effectiveness and safety of treatment with trifluridine and tipiracil plus bevacizumab in patients with metastatic CRC who previously received trifluridine and tipiracil without bevacizumab at a single academic medical center.
Methods
This was a single-center, retrospective cohort study conducted at The James Comprehensive Cancer Center at The Ohio State University that compared treatment with trifluridine and tipiracil plus bevacizumab versus trifluridine and tipiracil without bevacizumab in patients who previously received treatment for metastatic CRC between July 1, 2018, and August 1, 2021. The patients received trifluridine and tipiracil (35 mg/m2 orally on days 1-5 and on days 8-12) with bevacizumab (5 mg/kg intravenously on days 1 and 15) every 28 days. Treatment with an FDA-approved biosimilar for bevacizumab was allowed if mandated by the payers. The study protocol was approved by the center’s institutional review board with a waiver of informed consent because of its retrospective nature. This research did not receive any specific funding from agencies in the public, commercial, or not-for-profit sectors.
Patients aged 18 to 89 years were included if they had histologically confirmed and previously treated metastatic CRC, as well as if they were receiving active treatment with trifluridine and tipiracil. Patients were excluded if they were pregnant, incarcerated, had open wounds or surgery within 4 weeks of initiating treatment with trifluridine and tipiracil, or had major bleeding, clinically relevant nonmajor bleeding, gastrointestinal perforation, myocardial infarction, or cerebrovascular accident in the 6 months before initiating therapy with trifluridine and tipiracil. Major bleeding and clinically relevant nonmajor bleeding were defined by the International Society on Thrombosis and Haemostasis.10,11
All data were abstracted from electronic medical records and were entered into a REDCap (Research Electronic Data CAPture) database hosted at The Ohio State University.12 The patients’ baseline characteristics included age, sex, ECOG performance status, primary tumor location, metastatic site(s), mismatch repair or microsatellite instability status, and the presence of RAS or RAF mutation. In addition, the number of lines of therapy received before treatment with bevacizumab, the number of cycles before receiving bevacizumab (28-day cycle), and the previous chemotherapy treatments received (eg, fluoropyrimidines, oxaliplatin, irinotecan, bevacizumab, EGFR inhibitors, regorafenib) were collected. For disease course, the dates of disease progression, treatment nonresponse, and last follow-up or death were collected. Regarding treatment, the duration of treatment after progression therapy, febrile neutropenia events, complete blood counts with differentials, blood pressure results, urine protein to creatinine ratios (only in patients who received bevacizumab), venous thromboembolism events, and dose delays and dose modifications were also collected.
The primary outcome was PFS, and OS was the secondary outcome. The exploratory secondary end points included PFS and OS that were stratified by previous lines of bevacizumab-containing therapy, previous regorafenib use, and RAS and RAF mutation status. A comparison of the progression after treatment with trifluridine and tipiracil versus with trifluridine and tipiracil plus bevacizumab, stratified by eligibility for further therapy, was also planned. A safety analysis included the occurrence of events from the start of the study treatment to 30 days after the end of the study treatment, including febrile neutropenia, grade 3 or 4 neutropenia, grade 3 or 4 thrombocytopenia, grade 3 or 4 anemia, major bleeding, clinically relevant nonmajor bleeding, hypertension, proteinuria, venous thromboembolism, and dose delays and dose modifications. Grade 3 or 4 cytopenias were defined by the Common Terminology Criteria for Adverse Events version 5.13
Statistical Analysis
Descriptive statistics were used to evaluate the patients’ baseline characteristics and other secondary outcomes, as appropriate. For the primary outcome, the patients who progressed or died during the study period were considered to have PFS. For the secondary outcome of OS, patients who did not die during the study period were considered to not have an OS event. Patients who did not have a confirmed death date were censored at their last date of contact or on the date of data abstraction (ie, December 31, 2021), whichever occurred first.
Kaplan-Meier curves were used to estimate the PFS, OS, and time to treatment nonresponse. The patient safety data were evaluated up to 30 days after the cessation of treatment. The study data were collected and managed using REDCap electronic data capture tools hosted at The Ohio State University.12,14 All statistical analyses were performed using SAS version 9.4 (SAS Institute; Cary, NC).
Results
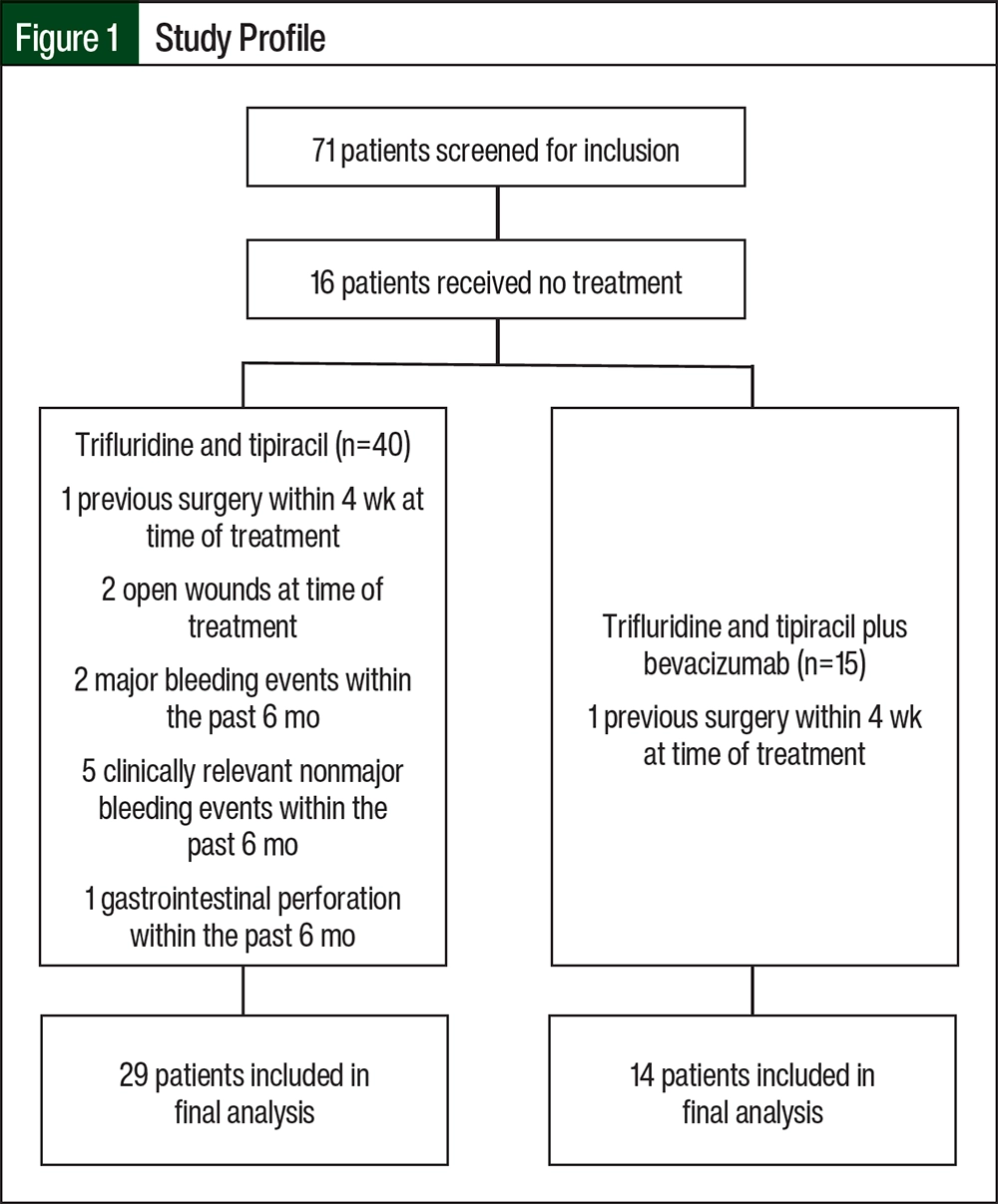
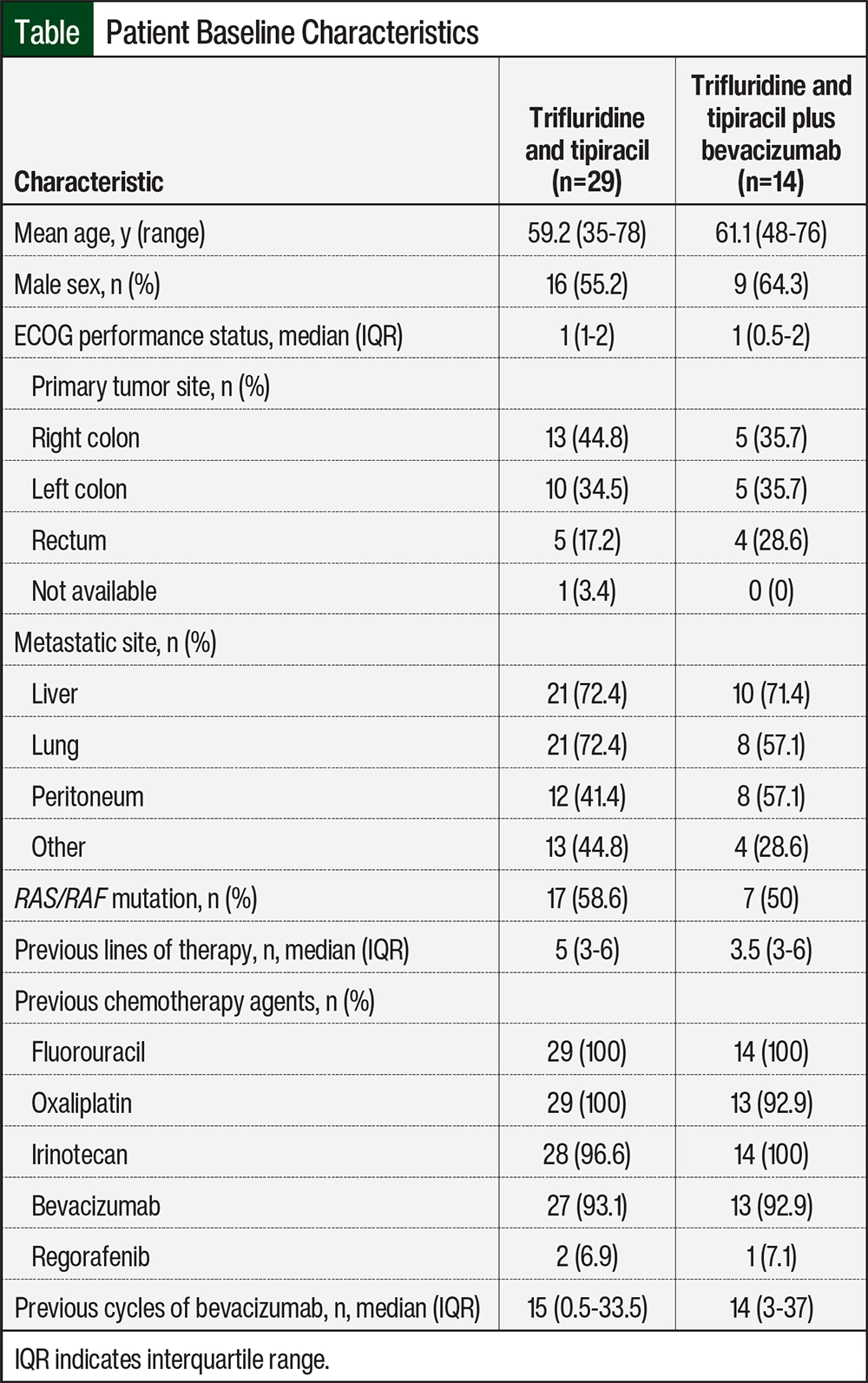
There were 71 patients who had trifluridine and tipiracil treatment prescribed and were screened for study inclusion. Ultimately, 43 patients were included in the study; most of the patients were excluded from the study because they did not start treatment (Figure 1). Most of the patients’ baseline characteristics were similar between the 2 treatment groups (Table). The median patient age was approximately 60 years, more than 50% of the patients were men, and the median ECOG Performance Status Score was 1. Right-sided tumors were more common in those receiving trifluridine and tipiracil, and rectal cancer was more common among those receiving trifluridine and tipiracil plus bevacizumab. The most common metastatic sites included the liver, lung, and peritoneum.
RAS or RAF mutations occurred in approximately 50% of the population, and only 1 patient in the trifluridine and tipiracil–only arm had mismatch repair–deficient or microsatellite instability–high disease. The median number of previous lines of therapy was 5 in the patients receiving trifluridine and tipiracil and 3.5 in those receiving trifluridine and tipiracil plus bevacizumab. All patients had previously received fluorouracil, and most patients had previously received treatment with oxaliplatin or irinotecan. More than 90% of the patients also had previously received treatment with bevacizumab, and the median number of cycles was approximately 15. Only 2 patients who received trifluridine and tipiracil and 1 patient who received trifluridine and tipiracil plus bevacizumab had previously received regorafenib (Table).
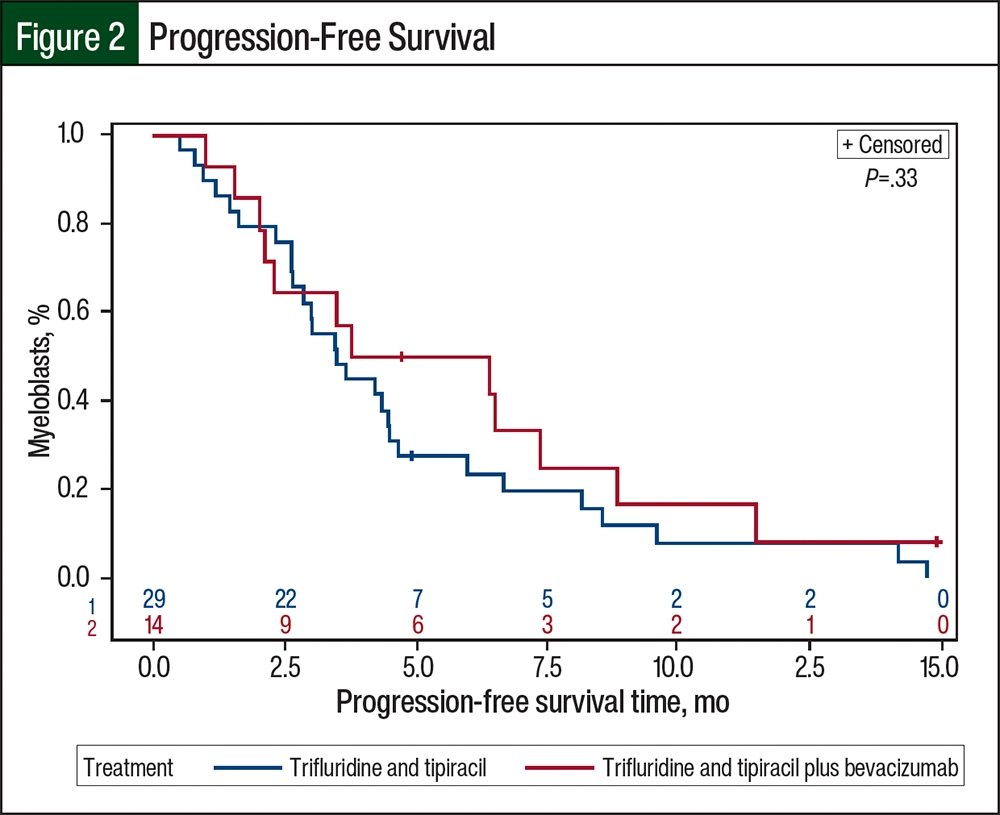
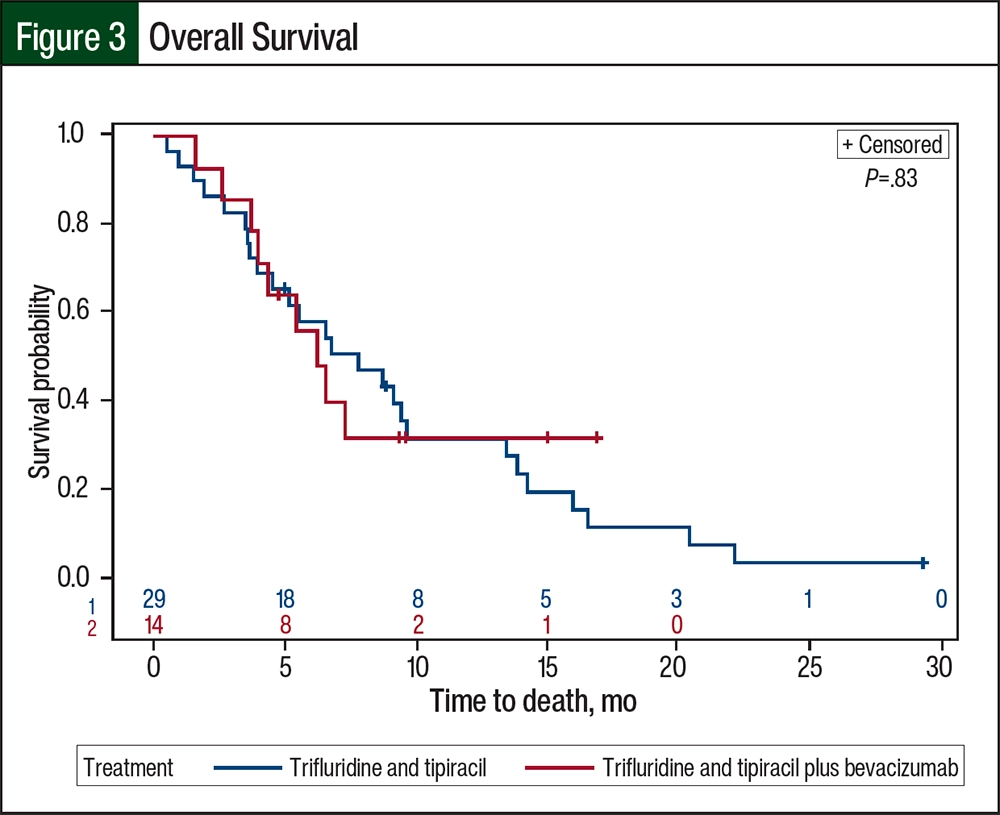
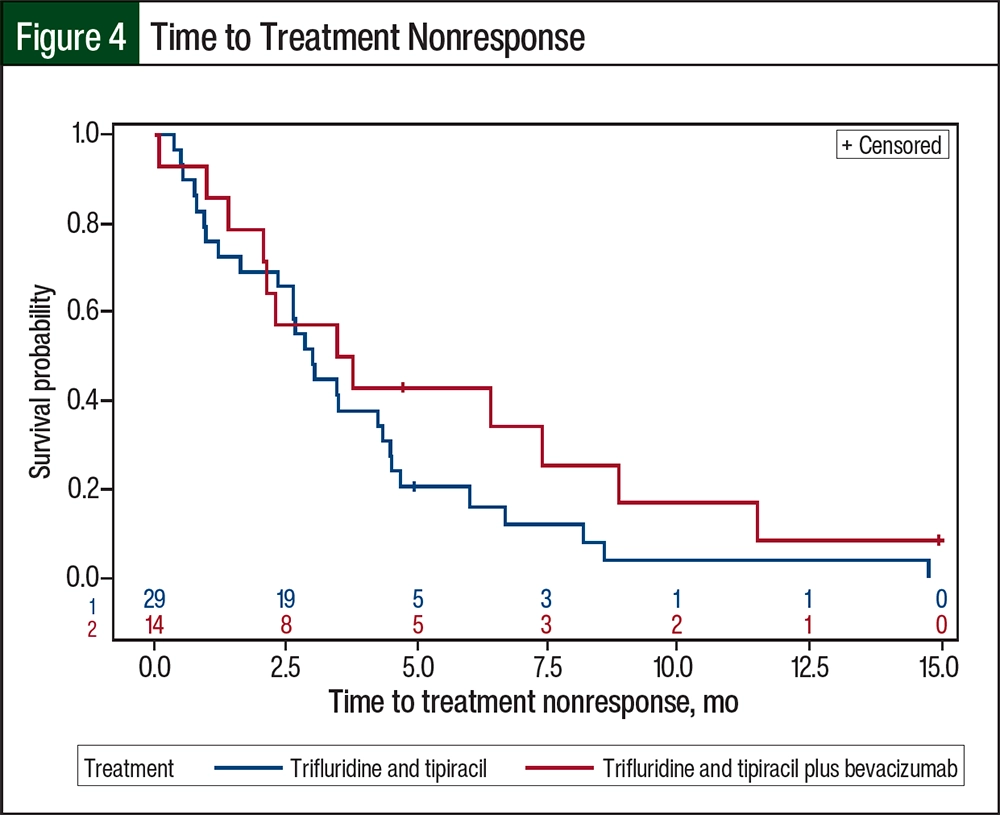
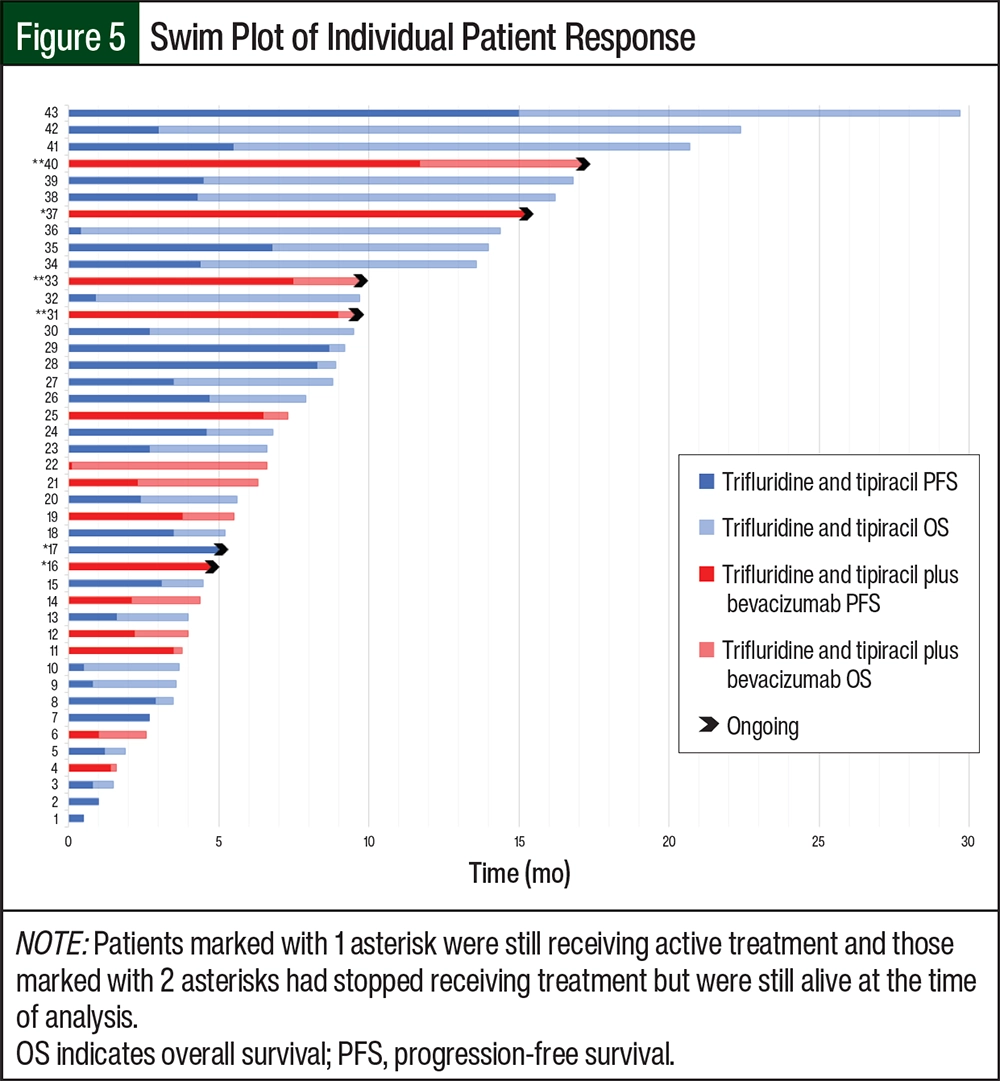
The median PFS was 3.48 months (95% CI, 2.63-4.50 months) for the patients who received trifluridine and tipiracil compared with 5.09 months (95% CI, 2.04-7.36 months) for those who received trifluridine and tipiracil plus bevacizumab (P=.33; Figure 2). The median OS for patients receiving trifluridine and tipiracil was 7.75 months (95% CI, 3.91-9.59 months) compared with 6.21 months for patients receiving trifluridine and tipiracil plus bevacizumab (95% CI, 3.71 months-not estimable; the upper limit could not be obtained because the confidence curve did not pass the median; P=.83; Figure 3). The median time to treatment nonresponse was 2.99 months (95% CI, 1.61-4.34 months) for the patients who received trifluridine and tipiracil and 3.63 months (95% CI, 1.38-8.84 months) for those who received trifluridine and tipiracil plus bevacizumab (Figure 4). The final disposition of the individual patients is shown in Figure 5.
After disease progression, a larger proportion of patients who received trifluridine and tipiracil went on to receive additional therapy than those who received trifluridine and tipiracil plus bevacizumab (58.6% vs 35.7%, respectively). Regorafenib was the most frequent subsequent treatment choice after disease progression. In contrast, more patients who received trifluridine and tipiracil plus bevacizumab were deemed ineligible for further therapy (35.7%) than those who received trifluridine and tipiracil alone (20.7%) based on having no further treatment options available, on Performance Status Score, and on earlier treatment tolerance. A similar proportion of patients elected to receive best supportive care (17.2% vs 14.3%, respectively).
The patients’ adverse events (AEs) were consistent with previously published literature5-8 on trifluridine and tipiracil plus bevacizumab or trifluridine and tipiracil alone. There was a higher prevalence of neutropenia among patients who received trifluridine and tipiracil alone (grade 3, 41.4%; grade 4, 13.8%) than those who received trifluridine and tipiracil plus bevacizumab (grade 3, 28.6%; grade 4, 7.1%). There were more incidents of grade 3 anemia in the cohort that received trifluridine and tipiracil alone than in those who received trifluridine and tipiracil plus bevacizumab (31% vs 21.4%, respectively). There was a higher proportion of grade 3 thrombocytopenia after treatment with trifluridine and tipiracil plus bevacizumab than after treatment with trifluridine and tipiracil (14.3% vs 6.9%, respectively). Grade 2 hypertension was the most frequent AE, occurring in 44.8% of patients who received trifluridine and tipiracil and in 57.1% of those who received trifluridine and tipiracil plus bevacizumab.
The rates of venous thromboembolism were higher in the trifluridine and tipiracil plus bevacizumab arm than in the arm without bevacizumab (21.4% vs 10.3%, respectively). There were no cases of major bleeding events noted in either group, but there were 3 cases of clinically nonrelevant major bleeding in the patients receiving trifluridine and tipiracil alone. There were no cases of proteinuria in those receiving trifluridine and tipiracil plus bevacizumab. A dose delay occurred in 72.4% of patients who received trifluridine and tipiracil and similarly in the group that received trifluridine and tipiracil plus bevacizumab (71.4%). A total of 8 (57.1%) patients in the trifluridine and tipiracil plus bevacizumab arm and 13 (44.8%) patients in the trifluridine and tipiracil arm required a dose modification. The most frequent reason for dose delays or modifications included neutropenia and fatigue.
Discussion
The purpose of this single-center, retrospective study was to share the real-world effectiveness and safety of treatment with trifluridine and tipiracil with or without bevacizumab. There was no marked improvement in PFS or OS with the addition of bevacizumab to trifluridine and tipiracil. Patients who received trifluridine and tipiracil plus bevacizumab had a PFS of 5.1 months, which is similar to previously published literature of 3.7 to 5.6 months.5-8 Because of the smaller-than-expected sample size, we were unable to assess the survival outcome by potentially important baseline factors, such as previous treatments and the duration of previous treatment with bevacizumab.
Almost 70% of the patients in each treatment group required a dose delay, which was similar to the delay in the SUNLIGHT trial.8 Comparatively, dose reductions in the SUNLIGHT trial were required in 16.3% of patients in the trifluridine and tipiracil plus bevacizumab group whereas 50% in each group in our trial required a modification, highlighting the tolerability of trifluridine and tipiracil.8 One method for aiming to improve treatment tolerability is to administer trifluridine and tipiracil on a biweekly schedule (days 1-5 and days 15-19) instead of on days 1 to 5 and days 8 to 12, as in the BiTS study by Satake and colleagues.15 In that study, patients had fewer cases of grade ≥3 neutropenia (15.9%) than in our study, and there were no cases of grade 4 neutropenia.15 There was also longer OS, at 10.86 months, with biweekly dosing than with the dosing regimen in our study.15 In addition, there is a correlation between neutropenia after receiving trifluridine and tipiracil and longer PFS.16
The results of a Japanese study on the cost-effectiveness of trifluridine and tipiracil plus bevacizumab showed that combination treatment with bevacizumab was cost-effective and was below the willingness-to-pay threshold, but that trifluridine and tipiracil alone was less expensive than trifluridine and tipiracil with bevacizumab.17 The cost of bevacizumab in Japan versus in the United States was significantly different ($1.92 per mg vs $6.46 per mg, respectively), which may affect the study’s data extrapolation to patients in the United States.17 Depending on the patient’s clinical and payer statuses, a discussion among the care team is warranted to determine the cost-benefit risk of patients receiving additional intravenous therapy and the extra adverse financial impact. Other factors to consider are the AEs that accompany treatment with bevacizumab and the fitness level of the patient after receiving multiple lines of therapy. Higher rates of neutropenia, hypertension, and venous thromboembolism were observed, and the added risks for AEs after receiving bevacizumab should be taken into consideration.
Limitations
Our study had limitations. At the time of the data analysis, 5 patients were still alive or were receiving treatment with trifluridine and tipiracil plus bevacizumab, which may not have allowed enough time to show the effect of the addition of bevacizumab. The smaller-than-expected sample size also did not meet the study’s power to evaluate the primary or secondary outcomes. Dose modifications or the decision to hold treatment were physician-specific and may have reflected the individual practices rather than practices at large. Also, we were unable to verify adherence to trifluridine and tipiracil because of the retrospective nature of this study.
It is likely that our study’s results differed from other published reports because of differences in the study populations. The patients who received trifluridine and tipiracil alone in this study may have been more physically fit at baseline or may have had more favorable biology, as suggested by the higher number of earlier treatments, when compared with the patients who received trifluridine and tipiracil plus bevacizumab (5 vs 3.5, respectively).
Furthermore, although it was meant to minimize internal selection bias, the exclusion of patients with potential contraindications to bevacizumab therapy (eg, having previous surgery within 4 weeks, open wounds, major bleeding within the past 6 months) may have resulted in the selection of a more favorable population than in other studies. This could explain why patients who received trifluridine and tipiracil monotherapy in this study had a longer median OS than patients in the RECOURSE study or in the study by Pfeiffer and colleagues.4,6
Conclusion
In our analysis, the addition of bevacizumab to trifluridine and tipiracil did not improve PFS or OS versus trifluridine and tipiracil alone in patients with previously treated metastatic CRC. Most patients had a dose delay or dose modification, which highlights the tolerability of trifluridine and tipiracil plus bevacizumab. Although there may be clinical benefit to the addition of bevacizumab to trifluridine and tipiracil, factors to consider include Performance Status Score, the patient’s physical fitness after receiving previous lines of treatment, the patient’s ability to tolerate further bevacizumab treatment, and payer status.
Author Disclosure Statement
Dr Arango is on the Advisory Board of Astellas Pharmaceuticals; Dr Kendzierski is employed at McKesson; Dr Hays is on the Advisory Boards of AADI Biosciences, Alkermes, AstraZeneca, Eisai, GOG Foundation, ImmunoGen, Merck, and Mersana Therapeutics, and is a Speaker for Curio Science and MJH Life Sciences, and travels for Regeneron Pharmaceuticals, Curio Science, and MJH Life Sciences; Dr Musson and Rachel Smith have no conflicts of interest to report.
References
- Biller LH, Schrag D. Diagnosis and treatment of metastatic colorectal cancer: a review. JAMA. 2021;325:669-685.
- Bennouna J, Sastre J, Arnold D, et al. Continuation of bevacizumab after first progression in metastatic colorectal cancer (ML18147): a randomised phase 3 trial. Lancet Oncol. 2013;14:29-37.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): colon cancer. Version 5.2024. August 22, 2024. Accessed September 7, 2024. www.nccn.org/professionals/physician_gls/pdf/colon.pdf
- Mayer RJ, Van Cutsem E, Falcone A, et al. Randomized trial of TAS-102 for refractory metastatic colorectal cancer. N Engl J Med. 2015;372:1909-1919.
- Kuboki Y, Nishina T, Shinozaki E, et al. TAS-102 plus bevacizumab for patients with metastatic colorectal cancer refractory to standard therapies (C-TASK FORCE): an investigator-initiated, open-label, single-arm, multicentre, phase 1/2 study. Lancet Oncol. 2017;18:1172-1181.
- Pfeiffer P, Yilmaz M, Möller S, et al. TAS-102 with or without bevacizumab in patients with chemorefractory metastatic colorectal cancer: an investigator-initiated, open-label, randomised, phase 2 trial. Lancet Oncol. 2020;21:412-420.
- Yoshida Y, Yamada T, Kamiyama H, et al. Combination of TAS-102 and bevacizumab as third-line treatment for metastatic colorectal cancer: TAS-CC3 study. Int J Clin Oncol. 2021;26:111-117.
- Prager GW, Taieb J, Fakih M, et al. Trifluridine-tipiracil and bevacizumab in refractory metastatic colorectal cancer. N Engl J Med. 2023;388:1657-1667.
- Dasari A, Lonardi S, Garcia-Carbonero R, et al. Fruquintinib versus placebo in patients with refractory metastatic colorectal cancer (FRESCO-2): an international, multicentre, randomised, double-blind, phase 3 study. Lancet. 2023;402:41-53.
- Schulman S, Angerås U, Bergqvist D, et al. Definition of major bleeding in clinical investigations of antihemostatic medicinal products in surgical patients. J Thromb Haemost. 2010;8:202-204.
- Kaatz S, Ahmad D, Spyropoulos AC, et al. Definition of clinically relevant non-major bleeding in studies of anticoagulants in atrial fibrillation and venous thromboembolic disease in non-surgical patients: communication from the SSC of the ISTH. J Thromb Haemost. 2015;13:2119-2126.
- Harris PA, Taylor R, Thielke R, et al. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377-381.
- National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE). Version 5.0. November 27, 2017. Accessed September 1, 2021. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_5x7.pdf
- Harris PA, Taylor R, Minor BL, et al. The REDCap consortium: building an international community of software partners. J Biomed Inform. 2019;95:103208.
- Satake H, Kato T, Oba K, et al. Phase Ib/II study of biweekly TAS-102 in combination with bevacizumab for patients with metastatic colorectal cancer refractory to standard therapies (BiTS Study). Oncologist. 2020;25:e1855-e1863.
- Hamauchi S, Yamazaki K, Masuishi T, et al. Neutropenia as a predictive factor in metastatic colorectal cancer treated with TAS-102. Clin Colorectal Cancer. 2017;16:51-57.
- Sugiura K, Seo Y, Takahashi T et al. Cost-effectiveness of TAS-102 plus bevacizumab versus TAS-102 monotherapy in patients with metastatic colorectal cancer. BMC Gastroenterol. 2021;21:184.