Connecting Science to Practice
Treatment with biosimilars is an effective way to expand patients’
access to care and lower healthcare costs. This study
demonstrates the real-life experiences of adding biosimilars
into practice at National Cancer Institute–designated comprehensive
cancer centers through examining the results of a
40-question survey that was developed to gather information
on biosimilar use at these healthcare organizations. The
survey’s results highlight the advantages and disadvantages of
how biosimilars are managed compared with reference
drugs, and it can ultimately affect healthcare policy as it
pertains to the safe and effective use of biosimilars.
Biosimilars are succinctly described as a copy of a biologic drug for which data protection has expired.1 These biosimilar drugs can include therapeutic classes, such as vaccines and proteins. The parent compounds that these biosimilars are based on have rapidly developed over the past 20 years and have been associated with a high cost; the average cost of cancer treatment in 2020 was approximately $150,000 per person.2 These costs strain the healthcare system in the United States. Compared with similar high-income European countries, cancer drug costs increased higher and faster in the United States, outpacing inflation.3 One way to help offset these high healthcare costs is market competition with biosimilar drugs, which, similar to generic drugs, can help adjust treatment costs downward.2 A review of 23 studies showed that most physicians consider the use of biosimilars to be financially advantageous.4
Legislation for a biosimilar approval pathway was defined by the Biologics Price Competition and Innovation Act of 2009 and was enacted in 2010.5 This act established that biosimilars may not be chemically identical to an approved reference biologic agent, but they are highly similar and have no clinically meaningful differences.4 Producing biologics is a complicated process that introduces the potential for clinically insignificant variations in structure.4 It is interesting to note that much of this act is similar in concept to the Drug Price Competition and Patent Term Restoration Act of 1984, which provided an abbreviated pathway for the approval of generic drugs.2 In law, there are a lot of similarities between generic drugs and biosimilars, but their relationship in clinical practice is much more complex. Although substitutions with generic drugs are generally well-accepted, pharmacist-led substitutions with biosimilars do not have the same level of acceptance with physicians.4
One major challenge to biosimilars is how they are manufactured.1 Unlike generic drugs, which have a well-established chemical structure that allows for a reproducible manufacturing process, biologic drugs are produced from living organisms. This makes the molecules more difficult to characterize and allows for small amounts of variability. For example, there are batch-to-batch variations in glycosylation that can be seen within the originator drugs, such as rituximab and darbepoetin alfa,6 and evidence shows some variations can have clinical impact.7 There can also be other differences, such as posttranslational protein modifications and formulations within biosimilars and across batches of originator drugs.7 On the safety side, biologic drugs are also complex. For example, among other reasons, epoetin-associated pure red blood cell aplasia has been linked to changes in the formulation in epoetin alfa and the use of uncoated rubber stoppers in prefilled syringes.8 This complexity contributes to uncertainty among pharmacists, payers, physicians, and patients.
The financial advantage may also be more complicated than originally thought. Although biosimilars are a more economical choice for health systems and payers, the anticipated financial impact is projected to be less than expected.9 For example, biosimilars were only preferred by US commercial payers in 14% of cases in 1 survey.10 Biosimilar use was restricted by payers in many cases and the utilization of originator drugs has continued after biosimilar approval. Although oncology drugs face fewer restrictions for biosimilars, the impact of these restrictions on health systems is less known when dealing with multiple payers and their preferences.
The management of biosimilars at the organizational level is also an important consideration. For most healthcare systems, formulary management starts with a Pharmacy & Therapeutics (P&T) committee.11 The P&T committee assesses the safety, efficacy, and economic considerations of drugs.11 Even though the regulatory pathway parallels generic drugs, efficacy and safety data are limited for the consideration of biosimilar drugs. In addition, reimbursement for drugs by payers also has a considerable impact on formulary decisions and is subject to change at the payers’ discretion.11 Even when data and finances are in order and support biosimilar adoption, physicians’ preferences and uncertainty continue to also be a hindrance to the use of biosimilars.4,12
After the approval of biosimilars, plans for implementation, storage, and medication safety may be different for biosimilars.11 For example, there are different administration methods for drugs such as rituximab, trastuzumab, and pegfilgrastim. Specifically, rituximab can be administered intravenously and subcutaneously, depending on the formulation. The administration techniques can also vary; for example, in patients who tolerate previous infusions well, rituximab can be administered as a rapid infusion.13 Payers’ requirements to move from a biosimilar to a biosimilar or from an originator drug to a biosimilar could have safety implications. Having a cohesive medication use system is crucial for the safe, effective implementation of biosimilar treatment, and this can vary from system to system.
Overall, literature about the successes, challenges, and best practices of biosimilar adoption in clinical practice is limited. Practitioners identified cost as a barrier to treatment with biologics, and biosimilars as a potential solution to increase access to these drugs.14,15 However, according to a survey of managed care and specialty pharmacy professionals, pricing and contract issues and state laws and regulations were 2 major barriers to oncology biosimilar utilization.16 In addition to the persistent reimbursement issues, other legal and ethical concerns regarding the adoption of biosimilars have arisen.16 According to another survey, 94.8% of oncology practitioners would substitute a reference drug with an FDA-approved biosimilar; however, in the same survey, practitioner responses varied about the importance of informing patients of biosimilar drug use and its involvement in treatment.17 The actual implementation of biosimilars comes down to organizational preferences and comforts.
Although the benefits of biosimilar use are enticing, the identified obstacles highlight the importance of closely considering the standard policies and procedures for the optimal and safe use of biosimilars across the United States. Currently, there are no data available that qualify the current state of oncology biosimilar use and the challenges faced by institutions. Acquiring knowledge about barriers to the successful adoption of biosimilars will aid in making informed decisions when integrating oncology biosimilars into clinical practice. Therefore, we conducted a comprehensive survey to describe the current state of biosimilars in oncology practices and the barriers to the adoption of these drugs.
Methods
To gather information regarding biosimilar use, a 40-item survey was developed by the Department of Pharmacy at a major cancer center (Roswell Park Comprehensive Cancer Center). The online survey was designed to take approximately 30 minutes to complete, with a recommendation to collaborate within each respective institution to provide accurate answers. An email was sent that contained the survey’s purpose and rationale along with the hyperlink to the Research Electronic Data Capture (REDCap) survey. The study data were collected and managed using REDCap, which is a secure, web-based application designed to support data capture for research studies, providing an intuitive interface for validated data entry, audit trails for tracking data manipulation and export procedures, automated export procedures for seamless data downloads to common statistical packages, and procedures for importing data from external sources.
The survey was conducted in accordance with the Declaration of Helsinki and was approved by the organization’s institutional review board. Oncology pharmacy professionals were identified using the Hematology/Oncology Pharmacy Association (HOPA) membership email list that included 3767 individuals. Using this HOPA membership list, the survey was distributed via email in January 2022 and remained open for 1 month. The survey items included questions on formulary management, drug use, payers, policies, technology, safety, education, and overall considerations. In addition, the use of the biosimilars bevacizumab, filgrastim, epoetin, infliximab, pegfilgrastim, rituximab, and trastuzumab was evaluated.
The responses to each question were sorted and analyzed to draw conclusions and associations between the institution’s demographics and oncology biosimilar adoption and implementation strategies. The centers’ characteristics and survey responses were summarized in the overall sample and by center type using the appropriate descriptive statistics and graphical summaries. The means for quantitative responses and percentages for qualitative responses were estimated with 95% confidence intervals, as appropriate. For the open-text responses, the qualitative descriptions were summarized. If multiple respondents came from the same organization, the surveys were stratified by role, favoring higher-ranked pharmacists.
Associations between the survey responses and centers’ characteristics (number of full-time oncology pharmacists, oncology inpatient beds, and infusion visits) were evaluated using the appropriate generalized linear models. Univariate and multivariate models were considered where variables were selected for multivariate models using stepwise selection. Model assumptions were verified graphically, and transformations were applied to the data as appropriate.
Results
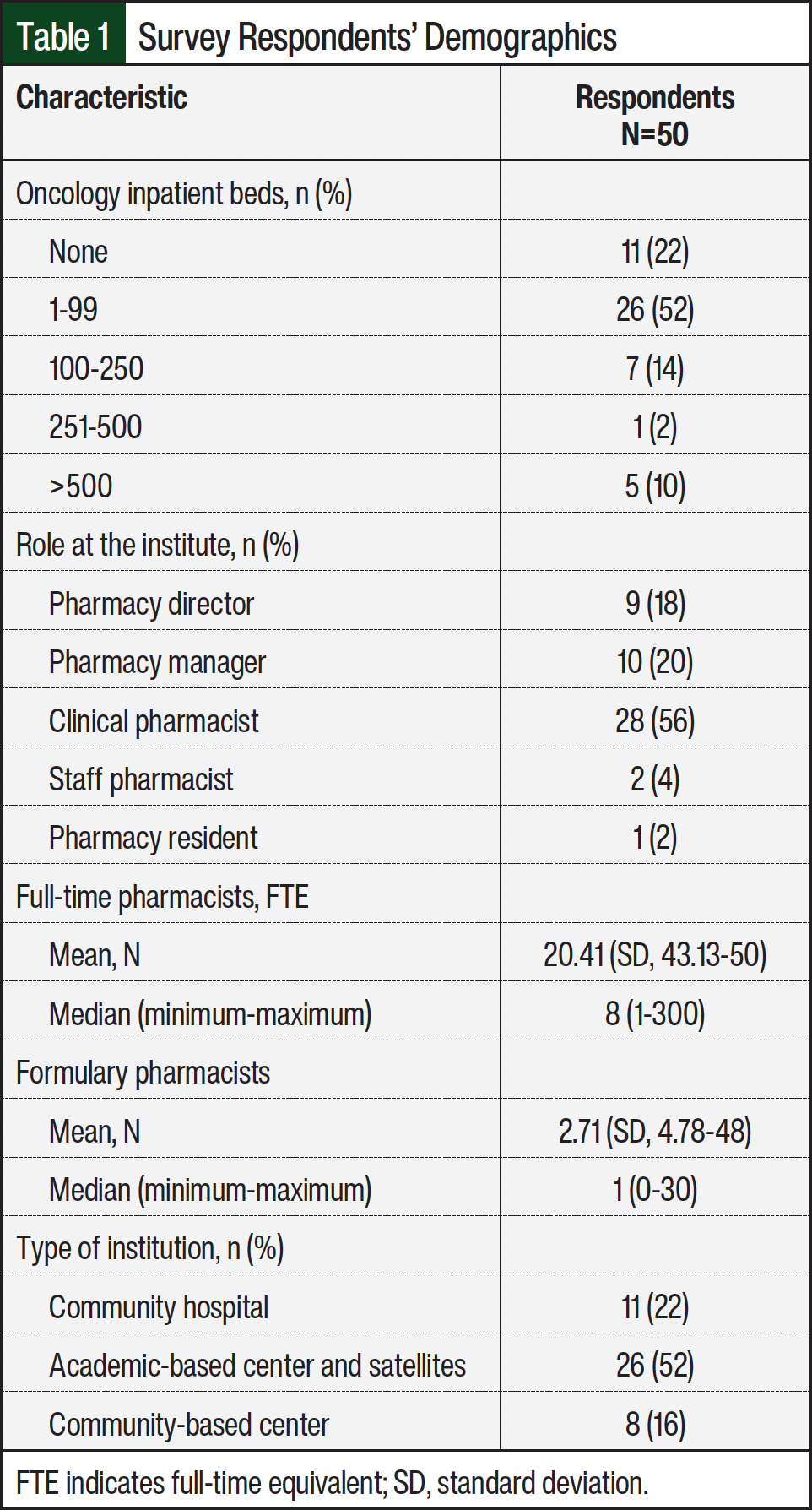
Overall, the survey was sent to 3767 oncology pharmacy professionals. A total of 179 surveys were initiated, and of those who started the survey, only 31% completed the survey entirely. Of those complete responses, 6 were removed because there were multiple respondents from the same organization, resulting in 50 unique surveys with 21 (42%) of those responses coming from National Cancer Institute (NCI)-designated comprehensive cancer centers.
Approximately 38% of the respondents were pharmacy managers or pharmacy directors. In all, 30% of the respondents documented >10,000 outpatient oncology infusion visits annually. Most of the responses were from academic medical centers (Table 1). Of the responding population, the median number of pharmacists dedicated to oncology services was 8 full-time equivalents (FTEs), and most did not have a designated FTE responsible for formulary management.
The responses were split for how biosimilars are added to a formulary; all respondents used P&T committees to accomplish this, of whom 50% maintained a formal P&T approval and the remaining respondents used abbreviated administrative approval. Among all of the respondents, >90% of institutions had a preferred biosimilar on formulary. The inpatient formulary decisions ranked acquisition cost as the highest priority (80.5%) followed by reimbursement (67.6%). In the outpatient setting, reimbursement was the highest priority (40.9%) followed by acquisition cost (37%). In both settings, provider preference/clinical practice was ranked as the lowest priority. To assess for patterns based on practice size, formulary decision priorities were stratified against infusion visits in the 3 categories of unknown, ≤10,000 visits, and >10,000 visits. There were no significant correlations to formulary priorities in the inpatient or outpatient services based on the number of infusion visits. There were also no significant correlations with the number of beds or the number of FTE pharmacists.
A total of 66% of the institutions in the survey had a biosimilar interchangeability policy in place and 32% restricted biosimilars to their FDA-approved indication only. The perspectives of the respondents on interchangeability did not differ based on the number of infusion visits (P=.098). Regarding interchangeability policies, most respondents stated that biosimilar interchanges were rarely rejected by providers and that substitution during treatment was only allowed if it was required by the payer. There were no patterns of acceptance based on size, inpatient and outpatient visits, or number of pharmacists.
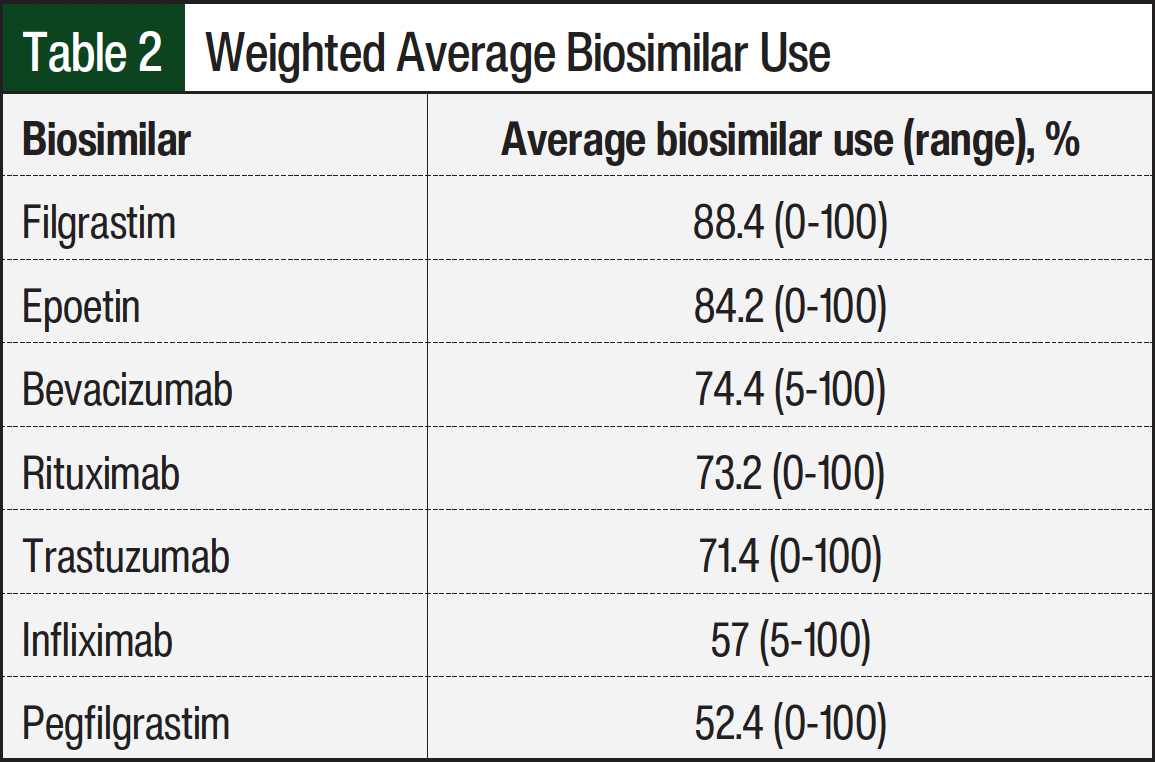
For the corresponding reference drugs, the weighted average utilization of biosimilars was highest for filgrastim, followed by epoetin, bevacizumab, rituximab, trastuzumab, infliximab, and pegfilgrastim (Table 2). Epoetin was more frequently administered in hospitals with >100 oncology inpatient beds (P=.031) and in centers with >10,000 visits (P=.024), but this was not based on the number of FTEs. None of the other drugs had significant correlations.
Most responding institutions were using a fee-for-service payment model (67.3%) followed by outcomes-based models (24%) and then other models (4%). The payment model did not differ based on the number of infusion visits or the hospital size. Insurance coverage varied among the respondents and included commercial, Medicare, Medicaid, and federal government insurance coverage. When asked about payer involvement, 72% of the respondents stated that payers specified the selection of biosimilars, and 76% stated that payer reimbursement limited the ability to participate in contract pricing. Payer selection of the preferred biosimilar was problematic, regardless of the number of oncology inpatient beds or the infusion clinic volume.
In addition, prior authorizations required by payers have imposed an additional workflow operation. Most (56%) respondents stated that prior authorization requests are initiated via a linked order set for the respective drug in the electronic health record (EHR). Moreover, patient medication access teams or office staff were the most utilized to obtain the prior authorization, with pharmacists most often responsible for confirming that the prior authorization matches the ordered biosimilar. Finally, 72% of respondents stated that treatment delays resulting from reimbursement or payment issues occurred weekly to monthly.
Regarding technical support for biosimilars, most organizations used Epic Systems (58%) as the EHR followed by Cerner (12%) and had the capability to order biosimilars using either the reference drug or biosimilar nomenclature. Most respondents stated that they used only the generic reference drug name for the smart pump libraries. A total of 66% of the respondents stated that they used an automatic default to the institute’s preferred biosimilar when orders were placed for biosimilars. In all, 34% of the respondents utilized oncology treatment pathways, with most having biosimilars on pathway. However, in the same question, 82% of the respondents stated that prior authorization is still required for the biosimilars that are on pathway.
Most respondents stated that biosimilar education is provided across the departments (ie, nursing, providers, pharmacy). Approximately 33% of the respondents stated that patients are educated on the general use of biosimilars, as well as on the specific biosimilar in their treatment regimen. Further, 38% of the respondents stated that patients are notified if they receive an alternative biosimilar instead of the previous treatment. Consent for treatment is required by 86% of the respondents, with 37% utilizing specific language that addresses biosimilar use.
Medication safety was an important consideration for all respondents. In terms of safety measures, gravimetric analysis (88%), barcode scanning (84%), segregated storage (50%), and image capturing (38%) are utilized by organizations. Despite the safety measures in place, 26% of the respondents stated that there was a medication error when a patient received a biosimilar or reference drug that was not the intended drug, with 18% attributing the event to a communication error. Human factors, namely confusion and drug labeling, were also attributed to errors. Safety technology was also assessed against the number of infusion visits. Most centers do not use image capturing (P=.39). No other patterns were identified. Centers individualized the medication safety requirements based on institutional needs.
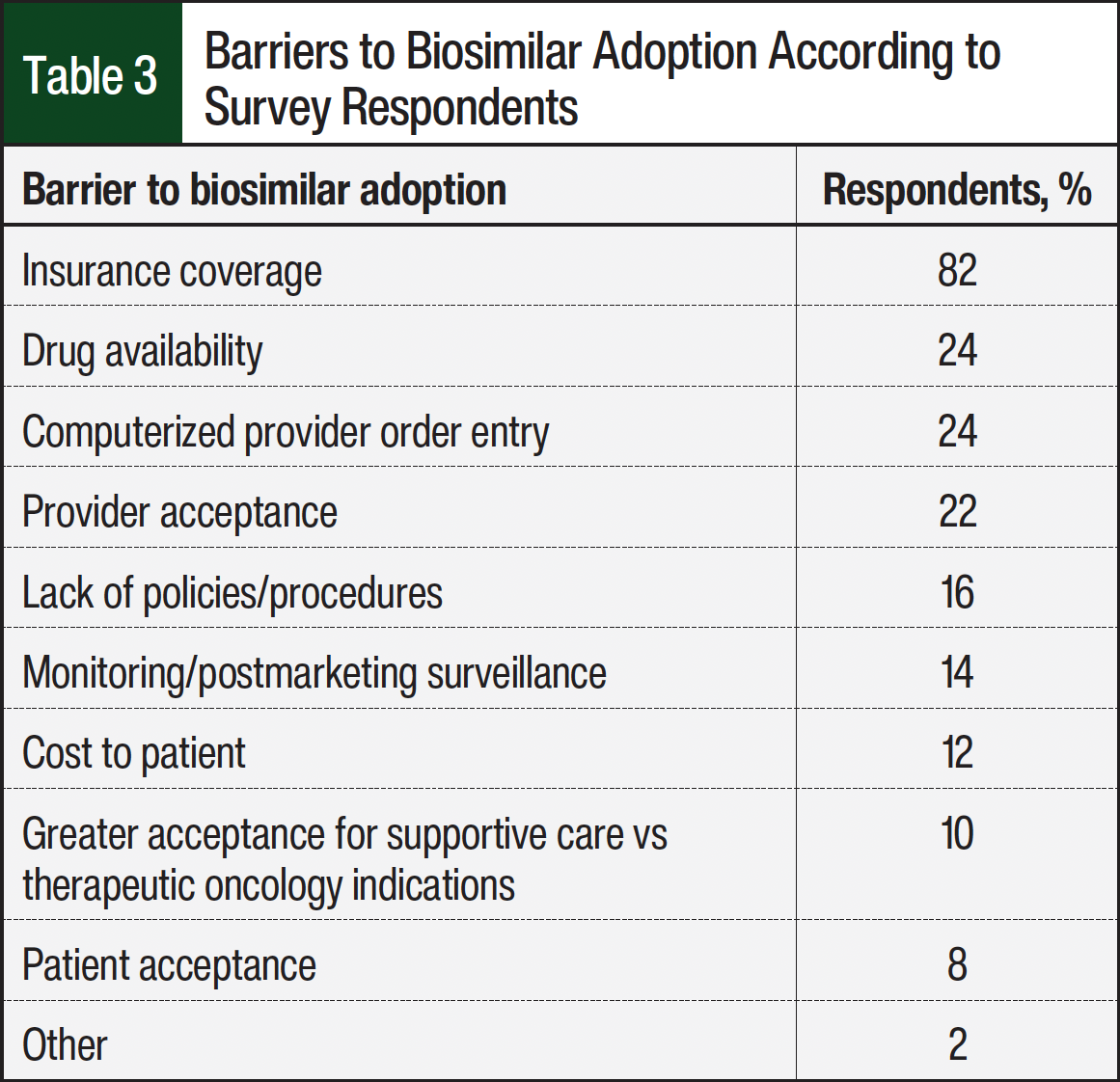
When provided a list of possible barriers to biosimilar adoption, the respondents recognized insurance coverage as the main barrier regardless of inpatient or outpatient considerations (Table 3). Drug availability and computerized provider order entry were tied as the second most common barriers to biosimilar adoption. Drug availability was problematic when assessed against the number of oncology inpatient beds (P=.46), whereas <100 beds was associated with less drug availability. Other respondents listed provider acceptance and lack of policies and procedures as challenges to the adoption of biosimilars. The respondents stated that payer requirements, price, and the availability of contract pricing were the most relevant considerations when deciding which biosimilar to prescribe. There were blank boxes for respondents to list additional barriers to biosimilar adoption that were not listed in the survey. Some of the respondents included a lack of adoption from organizations such as the American College of Rheumatology, forced “white bagging,” and nononcologic indications having limited “champions.”
Discussion
The goal of this survey was to gain an understanding of biosimilar practices within the United States. Based on the survey’s responses, there were a variety of practices represented, with only 42% of centers being NCI-designated. This diversity of respondents allows for some insight into practice styles and practical approaches that various organizations are using. Also, many sites had several oncology pharmacists on staff to support the service lines and corresponding populations. Although the number of responses to the survey was low, some of the surveys were completed by pharmacy directors and managers who play a role in payer relations, formulary management, drug use, and other factors in this survey.
Unsurprisingly, biosimilar usage across all institutions was high, and most respondents had identified preferred biosimilar options driven mostly by acquisition costs and reimbursement. One identified statistically significant barrier was drug availability in organizations with <100 oncology inpatient beds, perhaps indicating a smaller formulary or budgetary constraints.
In cost-avoidance situations such as inpatient usage, acquisition costs were more important in making decisions. Reimbursement was an important consideration in inpatient and outpatient settings, and many centers cited that biosimilar choices were dictated by insurance providers; this limited their ability to take advantage of special pricing of these high-cost drugs. Although not entirely assessed in this survey, the promoted and perceived financial advantage of using biosimilars is complicated by the restraints placed by the volatility of reimbursement rules from insurance carriers.
Despite previous reported negative feelings about being removed from decision-making, provider preferences were ranked least important in driving decisions regarding biosimilar utilization. The lack of consideration of provider preferences also suggests that the framework regarding interchangeability has largely been minimized in practice and clinical applicability is debatable. Most responding organizations had interchange policies, despite not having an interchangeable biosimilar approved in the United States. In addition, substitutions and interchanges of biosimilars with reference drugs or each other were rarely rejected by providers. Many centers also adopted abbreviated pathways for formulary additions of biosimilars, which allows centers to move dynamically with changing payer requirements and formulary adjustments.
To complicate matters for biosimilar utilization, there are important education and safety considerations in their implementation. According to the survey responses, 26% of the respondents reported administering the wrong drug, citing communication as a common contributor. This was despite education being provided by the staff and the use of EHR systems that were able to select or default to the preferred biosimilar drug. Many centers also used checking aids, such as gravimetric analyses, barcode scanning, image capturing, and other methods. These results also point to important considerations that technology integration is complicated and a possible error source. The nomenclature of biosimilars and reference drugs can be similar, even if they are segregated, and because insurance carriers mandate different drugs, technicians and pharmacists can dispense different brand-name agents in the same day. The effect of medication errors was not assessed in this survey; however, many biosimilars are known for resulting in allergic-like reactions, and therefore the risks associated with these errors can be pretty high.
Last, but more important, few centers educate patients on their exposure to biosimilars. Often in the community pharmacy setting, patients are counseled and notified with warnings when generic drug manufacturers change their medications; only 37% of centers have included language for the consent of treatment of cancer therapy, and 38% notify patients when the biosimilar is going to change. Because insurance reimbursement drives the choice of biosimilar drug and concerns of interchangeability are minimized through workflows, patient awareness and education are important considerations. For example, during a course of therapy, a patient could have several treatment switches based on various factors, such as drug affordability, formulary requirements, or the patient relocating.18 Although this practice of switching biosimilars has been safe in practice so far, it has not been adopted by any regulatory bodies.18
Limitations
Our study’s findings should be viewed in consideration of some common limitations when utilizing a survey. Our actual response rate was lower than estimates, especially because 179 surveys were initiated and only 56 surveys were fully completed. This lack of completion was most likely a result of the voluntary nature of the survey and the limited window of response time (ie, 4 weeks). Furthermore, the low response rate restricted our ability to evaluate the associations between the survey responses and the various centers’ characteristics.
An additional limitation is the variable participants’ interpretation of questions and the subjective nature of the responses. Last, this survey was shared in 2022, and therefore the rates of biosimilar use (eg, with pegfilgrastim) may have changed.
Conclusion
Payers’ preferences and reimbursement pose strong influences on and affect the overall utilization of oncology biosimilars. Although biosimilars were created to minimize healthcare costs and provide greater healthcare access, payers’ preferences require institutions to add multiple biosimilar drugs to the formulary for 1 reference drug and add several complexities to the medication use process. This dilutes the ability to use an institution’s own preferred biosimilar, limiting the ability for contract pricing and increasing the potential for delays and medication errors.
Overall, these survey results highlight the need to build efficiency and uniformity in biosimilar adoption across all institutions. Collaboration among oncology pharmacy professionals at various institutions may provide the opportunity to identify and create best practices in healthcare. Furthermore, this study highlights the opportunities that exist in the collaboration of healthcare systems and payers to align formularies and promote safe and cost-effective healthcare for their members.
Funding Source
This survey was supported and funded by the National Comprehensive Cancer Center and Pfizer Global Medical Grant, Adoption of Biosimilars in Oncology.
Author Disclosure Statement
Dr Przespolewski is on the Advisory Board of Fresenius Kabi, Genentech, and Sobi; Dr Walsh is a consultant to Precision Value & Health; Dr Amsler was previously employed at Johnson & Johnson and Merck; Dr Reed, Dr Forbes, Dr Attwood, and Dr Riebandt have no conflicts of interest to report.
References
- Weise M, Bielsky MC, De Smet K, et al. Biosimilars: what clinicians should know. Blood. 2012;120:5111-5117.
- Green AK, Ohn JA, Bach PB. Review of current policy strategies to reduce US cancer drug costs. J Clin Oncol. 2019;38:372-379.
- Vokinger KN, Hwang TJ, Daniore P, et al. Analysis of launch and postapproval cancer drug pricing, clinical benefit, and policy implications in the US and Europe. JAMA Oncol. 2021;7:e212026.
- Sarnola K, Merikoski M, Jyrkkä J, Hämeen-Anttila K. Physicians’ perceptions of the uptake of biosimilars: a systematic review. BMJ Open. 2020;10:e034183.
- Biologics Price Competition and Innovation Act of 2009. Meetings on user fee program for biosimilar and interchangeable biological product applications; request for notification of stakeholder intention to participate. Fed Reg. 2010;75:76472-76473.
- Schiestl M, Stangler T, Torella C, et al. Acceptable changes in quality attributes of glycosylated biopharmaceuticals. Nat Biotechnol. 2011;29:310-312.
- Lyman GH, Zon R, Harvey RD, Schilsky RL. Rationale, opportunities, and reality of biosimilar medications. N Engl J Med. 2018;378:2036-2044.
- McKoy JM, Stonecash RE, Cournoyer D, et al. Epoetin-associated pure red cell aplasia: past, present, and future considerations. Transfusion. 2008;48:1754-1762.
- Dolan C. Opportunities and challenges in biosimilar uptake in oncology. Am J Manag Care. 2018;24(11 suppl):S237-S243.
- Yu T, Jin S, Li C, et al. Factors associated with biosimilar exclusions and step therapy restrictions among US commercial health plans. BioDrugs. 2023;37:531-540.
- Cuellar S, McBride A, Medina P. Pharmacist perspectives and considerations for implementation of therapeutic oncology biosimilars in practice. Am J Health Syst Pharm. 2019;76:1725-1738.
- Leonard E, Wascovich M, Oskouei S, et al. Factors affecting health care provider knowledge and acceptance of biosimilar medicines: a systematic review. J Manag Care Spec Pharm. 2019;25:102-112.
- Sehn LH, Donaldson J, Filewich A, et al. Rapid infusion rituximab in combination with corticosteroid-containing chemotherapy or as maintenance therapy is well tolerated and can safely be delivered in the community setting. Blood. 2007;109:4171-4173.
- Lammers P, Criscitiello C, Curigliano G, Jacobs I. Barriers to the use of trastuzumab for HER2+ breast cancer and the potential impact of biosimilars: a physician survey in the United States and emerging markets. Pharmaceuticals (Basel). 2014;7:943-953.
- Monk BJ, Lammers PE, Cartwright T, Jacobs I. Barriers to the access of bevacizumab in patients with solid tumors and the potential impact of biosimilars: a physician survey. Pharmaceuticals (Basel). 2017;10:19.
- Greene L, Singh RM, Carden MJ, et al. Strategies for overcoming barriers to adopting biosimilars and achieving goals of the biologics price competition and innovation act: a survey of managed care and specialty pharmacy professionals. J Manag Care Spec Pharm. 2019;25:904-912.
- Cook JW, McGrath MK, Dixon MD, et al. Academic oncology clinicians’ understanding of biosimilars and information needed before prescribing. Ther Adv Med Oncol. 2019;11:1758835918818335.
- Cohen HP, Hachaichi S, Bodenmueller W, et al. Switching from one biosimilar to another biosimilar of the same reference biologic: a systematic review of studies. BioDrugs. 2022;36:625-637.