Acute myeloid leukemia (AML) is the most common type of acute leukemia in adults, with incidence rates increasing with age.1 Standard treatment for AML consists of intensive induction therapy to deplete the bone marrow of leukemic cells, followed by consolidation therapy to achieve disease control.1 This regimen produces a high risk for infection as a result of prolonged (duration of >7 days) and profound neutropenia (absolute neutrophil count [ANC] ≤100 cells/mm3).2 The risk for infection increases as the depth and duration of neutropenia increase.3 During periods of neutropenia, the signs and symptoms of infection may be absent, aside from a fever. Patients with neutropenia who present with sepsis or septic shock may have mortality rates as high as 50%.4 Thus, febrile neutropenia (FN) is considered an oncologic emergency and requires prompt evaluation and the initiation of broad-spectrum antibiotics.
The initial treatment regimens for AML are dependent on the patient’s age, performance status, cytogenetics, the presence of comorbid conditions, and preexisting myelodysplasia.1 A standard regimen for intensive induction candidates includes a combination of intravenous (IV) cytarabine daily for 7 days, with concomitant IV daunorubicin daily for the first 3 of the 7 days (frequently referred to as 7+3).1 Data also exist to support an induction regimen known as FLAG-ida, which consists of fludarabine and cytarabine on days 2 to 6, idarubicin on days 4 to 6, and a granulocyte colony-stimulating factor (G-CSF) on days 1 to 7.1 The rate of complete remission with cytarabine plus daunorubicin is approximately 60%; however, synergistic multidrug regimens (such as FLAG-ida) have shown complete remission rates up to 80%.5 The potential for increased adverse events with FLAG-ida (ie, delayed recovery of neutrophils) has limited its use compared with 7+3,6 which has been the standard-of-care treatment for many years. Patients aged >60 years typically benefit less from standard induction regimens because of a higher rate of comorbidities, a higher prevalence of unfavorable cytogenetics, and a higher incidence of multidrug resistance.1 Lower-intensity regimens can be used in these patients when intensive induction is not possible, which generally consist of a hypomethylating agent, such as azacitidine or decitabine, combined with venetoclax.7
Venetoclax acts by decreasing the survival of AML blasts, because it is an oral inhibitor of BCL-2 protein.7 The adverse events related to treatment with venetoclax include prolonged cytopenia, metabolic and electrolyte abnormalities, increased liver aminotransferases, and abdominal pain.8 So far, venetoclax has been limited to use in lower-intensity chemotherapy regimens.1 However, a 2021 study by Dinardo and colleagues demonstrated an improvement in complete remission rates for the induction of AML with FLAG-ida plus venetoclax.5 In this single-arm study of 68 patients, FLAG-ida plus venetoclax demonstrated a composite complete response rate of 90% for patients with newly diagnosed AML and a 12-month event-free survival rate of 85%. The adverse events in the study were primarily infections, with observed rates of FN similar to induction regimens across other types of AML, particularly secondary AML or relapsed or remitting AML, at approximately 50%. Despite G-CSF utilization, delayed count recovery was common, and cycle lengths exceeded 40 days in approximately 60% of patients.5
To date, the impact of adding venetoclax to intensive AML induction has not been thoroughly evaluated. With the added neutropenic effects of venetoclax in a myeloablative regimen, there is concern for an increased risk for FN incidence in this population. The aim of this study was to evaluate if the addition of venetoclax to intensive AML induction regimens results in an increased incidence of FN compared with regimens without venetoclax.
Methods
In this single-center, retrospective cohort study, adults (aged ≥18 years) with AML who completed induction chemotherapy between January 2018 and October 2021 at Sanford Medical Center in Fargo, ND, were included. Patients were excluded if they died or transitioned to comfort care before completing induction chemotherapy or initiated and/or completed induction therapy as an outpatient. Approval was obtained by the institution’s investigational review board before the study’s initiation.
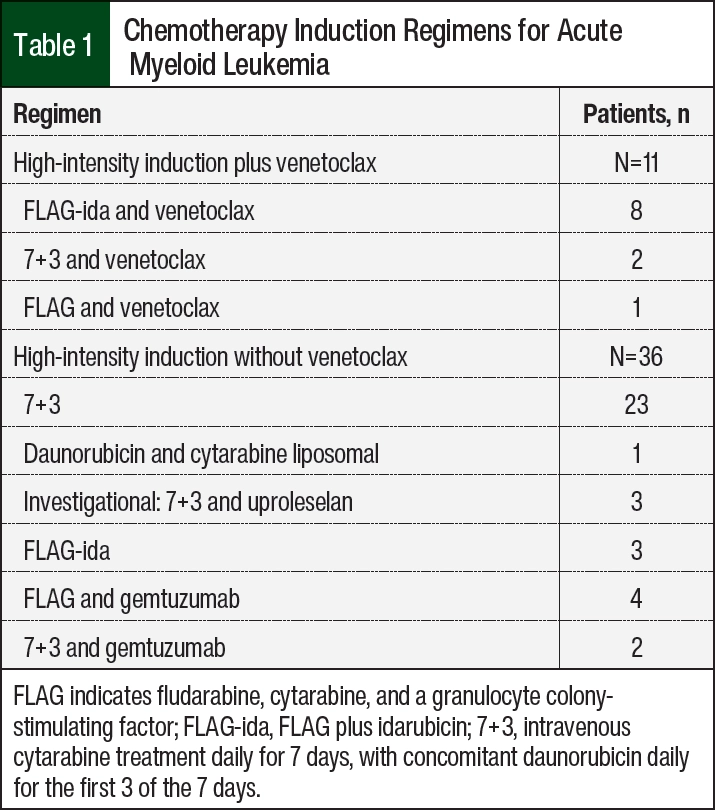
Induction chemotherapy was separated into high intensity and low intensity, aligning with National Comprehensive Cancer Network guidelines.1 The characteristics of patients who are candidates for high-intensity inductions include AML with actionable mutations, therapy-related AML, age <60 years, good performance and functional status, and no comorbid conditions.1 The choice of induction chemotherapy was determined by the treating physician. The high-intensity regimens that were evaluated are shown in Table 1. In addition, the choice of agent and timing of the initiation of antimicrobial prophylaxis were determined by the treating physician. Most often, levofloxacin, valacyclovir, and posaconazole were initiated after a decrease in ANC <500 cells/mm3. In patients with FN (defined as a temperature of >100.4°F and ANC <500 cells/mm3), culture microbiology, previous infection with an antimicrobial-resistant organism, utilization of and indication for vancomycin, and the duration of IV antibiotics were collected. The outcomes analyzed included the length of hospital stay, remission after induction, intensive care unit (ICU) admission, sepsis during hospitalization (as defined by life-threatening organ dysfunction caused by a dysregulated host response to infection), and mortality during hospitalization.9
The primary study outcome was the incidence of FN in patients receiving intensive AML induction regimens containing venetoclax compared with patients receiving AML induction regimens without venetoclax. The secondary outcomes for the high- and low-intensity AML induction regimens included the rate of ICU admission; length of hospital/ICU stay; duration of IV antibiotics; incidence of sepsis; bacteremia and/or refractory fever despite receiving IV antibiotics for 48 hours; use of G-CSF; the duration and degree of neutropenia; and in-hospital mortality. A variety of additional infectious-related outcomes were also collected and were compared between the groups, including the time to first fever, culture positivity, incidence and type of antimicrobial prophylaxis, and escalation of prophylaxis. The patients were followed until hospital discharge.
Statistical Analysis
The categorical variables were assessed using chi-square test or Fisher’s exact test, as appropriate. The continuous variables were assessed using the Wilcoxon rank-sum test because of a non-normal distribution of data. The data were analyzed using JMP software, version 15.1.0 (SAS Institute, Inc; Cary, NC), with a P value of <.05 considered statistically significant.
Results
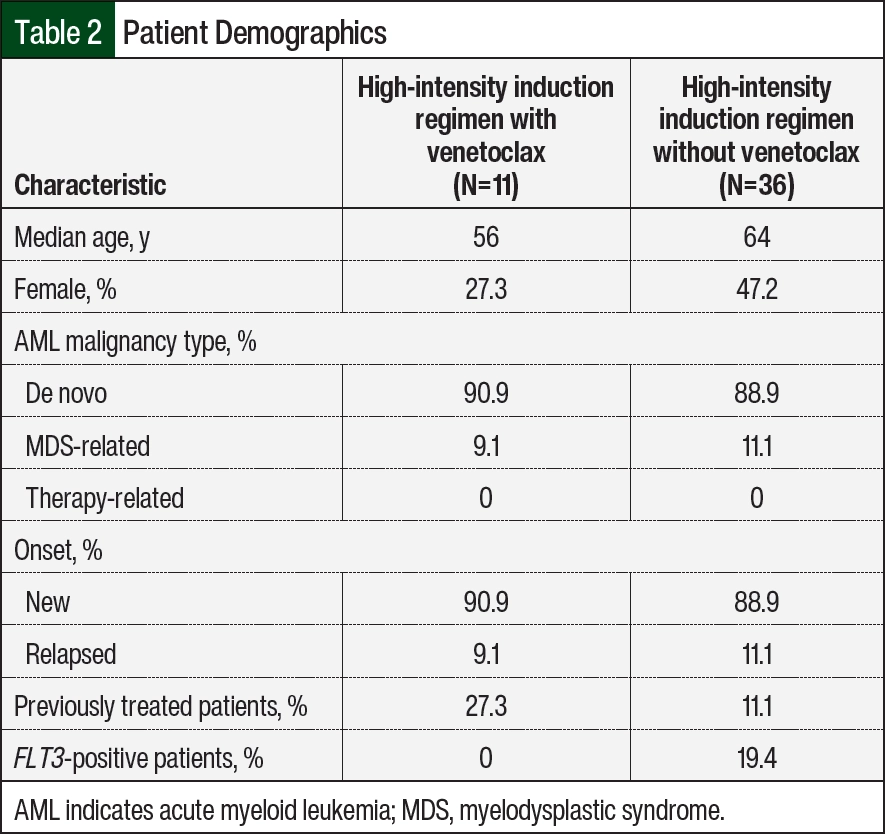
A report was run from January 1, 2018, to October 1, 2021, that identified patients who had received venetoclax, cytarabine, or the combination of daunorubicin and cytarabine. A total of 233 charts were evaluated, with 59 patients meeting the study inclusion criteria. A total of 174 patients were excluded from the study, primarily as a result of the medications included in the report not being used for induction chemotherapy or because of discharge or transition to comfort care before the induction chemotherapy was complete. Of the 59 individual patients included in the study, 47 patients received high-intensity induction regimens and 12 patients received low-intensity induction regimens. Because of the small number of low-intensity induction regimens that met the inclusion criteria and the already available evidence for the use of venetoclax in this population, low-intensity induction regimens were not included in the analysis. Of the 47 high-intensity induction regimens, 11 included venetoclax. The patients’ demographic information is summarized in Table 2. A significant difference in age was observed between the high-intensity induction treatment groups, with the patients receiving a regimen containing venetoclax being younger than the patients who did not receive venetoclax (56 years vs 64 years, respectively). Notably, patients with a positive FLT3 biomarker (19.4%) were only present in the high-intensity induction regimens without venetoclax. There were no significant differences in AML malignancy type, onset of disease, or prevalence of previous therapy received between the groups.
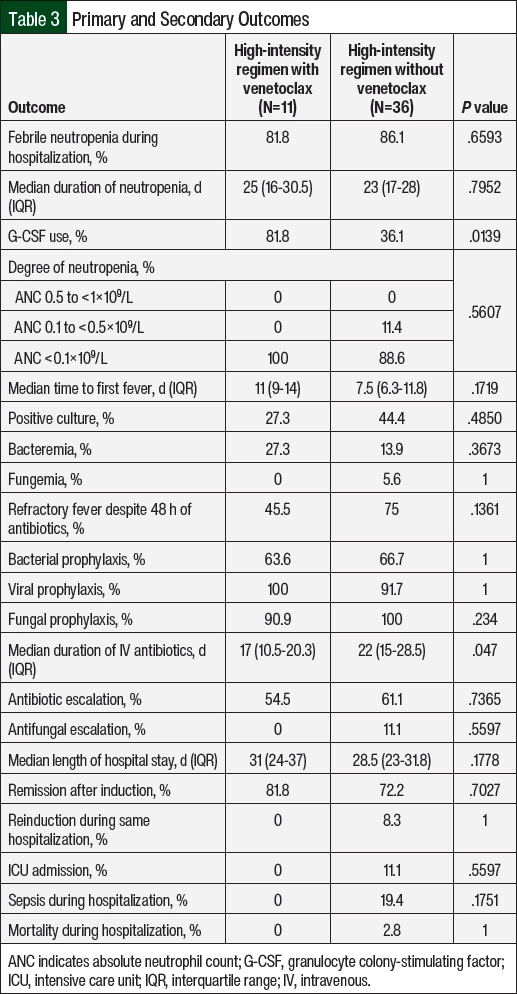
The primary and secondary study outcomes are shown in Table 3. For the primary outcome, the rate of FN in the high-intensity induction regimens containing venetoclax was 81.8% compared with 86.1% in the high-intensity induction regimens without venetoclax (P=.6593).
The secondary outcomes were not significantly different between the cohorts, except that the median duration of IV antibiotics was shorter for regimens containing venetoclax than for those that did not include venetoclax (17 days vs 22 days, respectively; P=.047). Notably, the median duration of neutropenia was 25 days for the high-intensity induction regimens with venetoclax and 23 days in the regimens without venetoclax (P=.7952). All high-intensity induction regimens with venetoclax had an ANC nadir of <0.1 cells/mm3, whereas 11.4% of regimens without venetoclax had an ANC nadir of <0.5 cells/mm3 but >0.1 cells/mm3. The use of G-CSF was 81.8% in the high-intensity induction regimens containing venetoclax compared with 36.1% in the regimens without venetoclax (P=.0139). The patients who had refractory fever despite receiving the appropriate antimicrobial treatment prompted the escalation of antibiotics and/or antifungals to medications with broader antimicrobial coverage, such as the addition of vancomycin or the escalation from cefepime to meropenem or fluconazole to micafungin, as aided by infectious disease consultation. This escalation of care did not differ significantly between the groups.
Discussion
There is a paucity of published literature regarding the safety and efficacy of venetoclax added to high-intensity induction therapy for AML. Because of concern for additional neutropenic effects of venetoclax on an already myeloablative regimen, this study sought to evaluate whether this combination led to increased rates of FN. Overall, the incidence of FN remained similar between the 2 groups, at 81.8% in the regimens containing venetoclax and 86.1% in the regimens without venetoclax.
In contrast to the hypothesized prolonged duration of neutropenia in the venetoclax group, we observed similar durations of neutropenia between the 2 groups, with regimens containing venetoclax being slightly longer at 25 days compared with 23 days without venetoclax, although this was not significant. The degree of neutropenia when venetoclax was added was more profound, with all patients demonstrating an ANC <0.1 cells/mm3, whereas a few patients who did not receive venetoclax maintained an ANC of <0.5 cells/mm3 but >0.1 cells/mm3. We hypothesize that the increased use of G-CSF in the venetoclax cohort was likely a result of the high use of the FLAG-ida regimen (which contains G-CSF as part of the regimen), but this presents a potential confounder for a comparison of neutropenia depth and duration between the groups. An additional finding was the significant difference in the duration of IV antibiotics received between the 2 groups. The 5-day shorter duration in the venetoclax group than in the group that did not receive venetoclax (17 days vs 22 days, respectively) could be attributed to lower rates of refractory fever, fewer positive microbiologic cultures, none of the patients meeting the sepsis criteria, or changes to antimicrobial stewardship practices in excess of the study time frame.
The patients’ outcomes, including remission after induction, ICU admission, and sepsis or mortality during hospitalization, were all numerically improved in the venetoclax-containing regimens, although they were not statistically significant. The lengths of hospital stay were also similar between the groups, although it was numerically shorter in the regimens without venetoclax than in the regimens with venetoclax (28.5 days vs 31 days, respectively).
Limitations
The limitations of this study include its retrospective design, single-center population, and small sample size. In addition, our institution did not have a protocol regarding antimicrobial prophylaxis, and the agent selection and timing varied by the physicians and patient scenarios. Because of the retrospective nature of the study, data may have been missing from the medical records, and the inclusion of regimens that may have used investigational agents could have influenced the results.
The patients’ ages were significantly lower in the regimens containing venetoclax, which is potentially a confounder indicating that fitter patients were selected for a more intense treatment regimen. There were differences in G-CSF use between the groups, which we were unable to adjust for, thereby potentially confounding the results. In addition, this study population had higher rates of FN than was previously reported in the literature, with an approximate increase in the incidence of FN of 30% compared with the outcomes from Dinardo and colleagues’ study.5 We hypothesized that this finding may coincide with our small sample size and/or differences in defining FN among the different institutions. Future prospective studies with larger sample sizes are needed to support the results of this trial.
Conclusion
This study’s results show that the inclusion of venetoclax into high-intensity induction chemotherapy regimens for AML does not have a significant impact on the incidence of FN. Although there was a higher complete response rate in the patients who received venetoclax than in those who did not receive venetoclax, this was not a significant finding. This study highlights the need for further investigation of the inclusion of venetoclax into induction chemotherapy regimens for the treatment of AML with the potential to improve patients’ response rates and outcomes.
Author Disclosure Statement
Dr DeWitt, Dr Grindeland, and Dr Schulze have no conflicts of interest to report.
References
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): acute myeloid leukemia. Version 2.2024. March 22, 2024. Accessed April 26, 2024. www.nccn.org/professionals/physician_gls/pdf/aml.pdf
- Freifeld AG, Bow EJ, Sepkowitz KA, et al. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2011;52:e56-e93.
- Taplitz RA, Kennedy EB, Bow EJ, et al. Outpatient management of fever and neutropenia in adults treated for malignancy: American Society of Clinical Oncology and Infectious Diseases Society of America clinical practice guideline update. J Clin Oncol. 2018;36:1443-1453.
- Legrand M, Max A, Peigne V, et al. Survival in neutropenic patients with severe sepsis or septic shock. Crit Care Med. 2012;40:43-49.
- DiNardo CD, Lachowiez CA, Takahashi K, et al. Venetoclax combined with FLAG-IDA induction and consolidation in newly diagnosed and relapsed or refractory acute myeloid leukemia. J Clin Oncol. 2021;39:2768-2778.
- Burnett AK, Russell NH, Hills RK, et al. Optimization of chemotherapy for younger patients with acute myeloid leukemia: results of the medical research council AML15 trial. J Clin Oncol. 2013;31:3360-3368.
- DiNardo CD, Pratz K, Pullarkat V, et al. Venetoclax combined with decitabine or azacitidine in treatment-naïve, elderly patients with acute myeloid leukemia. Blood. 2019;133:7-17.
- Venclexta (venetoclax) tablets, for oral use [prescribing information]. AbbVie; June 2022.
- Evans L, Rhodes A, Alhazzani W, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021;47:1181-1247.