Irinotecan is a topoisomerase I inhibitor that is prescribed alone or in combination with other cytotoxic chemotherapy and monoclonal antibodies for the treatment of recurrent or advanced gastrointestinal (GI) cancers.1 Irinotecan was originally FDA-approved for the treatment of metastatic colorectal cancer as a single agent dosed at 350 mg/m2 administered for 90 minutes,1 which is the infusion time used in many subsequent studies.2,3 Currently, the most common irinotecan regimen dosing is 125 to 180 mg/m2 intravenously (IV) infused over 90 minutes, which is half the dose used in the original trials.4,5
Irinotecan induces significant adverse events (AEs), which can be divided into acute (or “early”) and delayed (or “late”) phases.1 Acute AEs, occurring during or shortly after infusion, are linked to cholinergic syndrome, including symptoms such as diaphoresis, flushing, diarrhea, and bradycardia. Transient diarrhea can also occur. These acute AEs are mediated through the inhibition of acetylcholinesterase.1 In contrast, delayed AEs, which manifest days after the administration of irinotecan, involve delayed-onset diarrhea resulting from SN-38 reactivation, a metabolite of irinotecan.1,6 This reactivation of SN-38 in the intestines leads to mucosal damage, inflammation, and subsequent diarrhea.6 Understanding these mechanisms is crucial for the effective management and prevention of these AEs, which can be distressing for patients and nursing staff and cause delays in patient care as symptoms are managed.
Diarrhea can occur in the acute or delayed phases. When diarrhea occurs in the acute phase, it is usually transient, is infrequently severe, and may be accompanied by cholinergic symptoms.1 Acute diarrhea occurs in up to approximately 33% of patients receiving the FOLFIRI chemotherapy regimen.7 The prescribing information for irinotecan recommends that patients receive 0.25 mg to 1 mg of atropine IV or subcutaneously to antagonize the action of acetylcholine to treat or prevent these acute cholinergic symptoms.1 In the delayed phase, loperamide 4 mg should be received at the first onset of diarrhea and then at a dose of 2 mg every 2 hours until the patient is free of diarrhea for at least 12 hours.1
To focus on the severity of diarrhea, Saltz and colleagues evaluated irinotecan 125 mg/m2 IV administered for 90 minutes in patients with metastatic colorectal cancer.8 This dose is significantly less than the dose in the original phase 1 trial by Rowinsky and colleagues,9 suggesting that patients would have fewer AEs. However, the study results from Saltz and colleagues showed that grade 3 or 4 diarrhea, which occurred in 51 (22.7%) of the 225 study patients, was the main dose-limiting AE (Common Terminology Criteria for Adverse Events for diarrhea grade is referenced below).10 For the management of diarrhea, loperamide was employed, whereas atropine was administered for cholinergic syndrome.8 A study by Rothenberg and colleagues demonstrated the extent of diarrhea severity with irinotecan treatment, with 4 (44%) of 9 patients having grade 4 diarrhea when they received irinotecan at a dose of 150 mg/m2.11 This incidence decreased to 9 (23%) of 39 patients when the dose of irinotecan was lowered to 125 mg/m2. To alleviate this AE, loperamide was administered at the first sign of late diarrhea, whereas atropine was used during the acute phase.11 Notably, it’s important to highlight that atropine was not employed as a premedication in these studies.
Another AE that is rare but concerning to providers is dysarthria. Gunturu and colleagues revealed that 9 (25.7%) of 35 patients in their study had dysarthria during the irinotecan infusion.12 The study hypothesized that the order of drug administration in FOLFIRINOX, where oxaliplatin is routinely infused followed by irinotecan, might play a role in intensifying the cholinergic effects of irinotecan.12 Further research from Japan showed that 4 (44.4%) of 9 patients who were receiving FOLFIRINOX had dysarthria.13 In all cases, dysarthria occurred acutely during irinotecan infusion; the dysarthria resolved without any complications by either interrupting irinotecan infusion or administering anticholinergic agents, including atropine and/or diphenhydramine.13
In January 2021, Dana-Farber Cancer Institute (DFCI) changed the administration time for irinotecan doses of ≤180 mg/m2 from between 60 and 90 minutes to 30 minutes based on a phase 2 trial that evaluated a dose of 350 mg/m2 for 30 minutes in patients with advanced colorectal cancer and recommendations from the National Comprehensive Cancer Network (FOLFIRI template).14,15 The rationale behind combining the 60-minute and 90-minute infusion groups into a single group for comparison against the 30-minute infusion group was to ensure an adequate sample size for the comparison group, enhancing the statistical power and reliability of the study findings.
We aimed to include all patients with GI diagnoses who received irinotecan at a standard infusion time of >30 minutes. It is important to note that we did not anticipate a significant number of patients receiving irinotecan for 60 minutes. Hence, we incorporated them into the 90-minute group instead of creating a new comparator group. DFCI’s practice change also included shortening the administration time of concomitant leucovorin to 30 minutes. Leucovorin, a folinic acid, increases the effectiveness of fluorouracil, which is often given with irinotecan.16 In cases where leucovorin is received with irinotecan, it is typically given in an infusion treatment regimen.16
During the COVID-19 pandemic, this project was accelerated to reduce patient time spent in infusion chairs at our ambulatory care sites. After this practice change, the nursing and pharmacy departments made an anecdotal observation of an increase in acute irinotecan-related AEs with the shorter infusion. This study was conducted to assess whether there are more acute irinotecan-related AEs associated with a shorter irinotecan infusion time versus with a longer infusion time. Atropine on an as-needed administration schedule was used for the treatment of early cholinergic syndrome related to irinotecan infusion.
The primary objective of this study was to compare the median total atropine dose administered in patients receiving cycle-1, day-1 irinotecan for 60 to 90 minutes (the control arm) versus for 30 minutes (the intervention arm). The secondary objective was to evaluate the percentage of patients who tolerated irinotecan infusion over the intended administration time, the percentage of patients who had acute irinotecan-related AEs with the coadministration of oxaliplatin, and the percentage of patients who had acute irinotecan-related AEs with or without atropine premedication.
Methods
This was a descriptive, retrospective chart review of patients with a GI malignancy who received irinotecan for 60 to 90 minutes (the control arm) versus for 30 minutes (the intervention arm) between August 1, 2019, and October 31, 2021, at DFCI and its satellite locations.
The study eligibility criteria included age ≥18 years and receiving a dose of irinotecan of ≥150 mg/m2. Patients were excluded from the study if they previously received irinotecan. The patients were identified through a customized report of the electronic health records (EHRs). A list of patients receiving FOLFIRI, FOLFIRINOX, and FOLFOXIRI, with or without bevacizumab, cetuximab, or panitumumab, was generated via the Beacon module of the Epic EHR software to identify the eligible patients for analysis.
In our study, acute irinotecan-related AEs were defined as diarrhea, rhinitis, increased salivation, miosis, lacrimation, diaphoresis, flushing, or intestinal hyperperistalsis leading to abdominal cramping during and/or shortly after receiving an irinotecan infusion.
The irinotecan order sets at our institution specify the administration of atropine 0.2 mg IV as needed for the management of early cholinergic syndrome associated with irinotecan infusion. This may include symptoms such as severe diarrhea, emesis, diaphoresis, or abdominal cramping. The recommended dosing schedule allows for the administration of atropine every 15 minutes for up to 3 doses, after an initial dose of 0.4 mg. The cumulative maximum dose of atropine should not exceed 1 mg.
The data were collected from Epic reports and patient chart reviews and included age, sex, cancer diagnosis, treatment location, date of irinotecan infusion, irinotecan dose and administration time, as-needed atropine doses and administration times as premedication and/or for the management of irinotecan-related AEs, and irinotecan-related AE episodes during the infusion visit. Atropine administration was identified by reviewing the patient’s medication administration records on the day of their irinotecan infusion. Irinotecan tolerability was defined as no reports of irinotecan-related AEs from the nursing notes, atropine not administered to manage irinotecan-related AEs, and irinotecan infusion not paused, extended, or stopped for the management of irinotecan-related AEs.
Statistical Analysis
Wilcoxon rank-sum test and the Hodges-Lehmann estimator of location shift tests were used to analyze the primary objective. Descriptive statistics including percentages were used to analyze the secondary objectives. P<.05 was considered statistically significant.
Results
Between August 2019 and October 2021, a total of 400 patients were identified from DFCI and its satellite locations. Among the 319 patients who met the inclusion criteria, 151 patients were included in the 60- to 90-minute infusion group and 168 patients were included in the 30-minute infusion group.
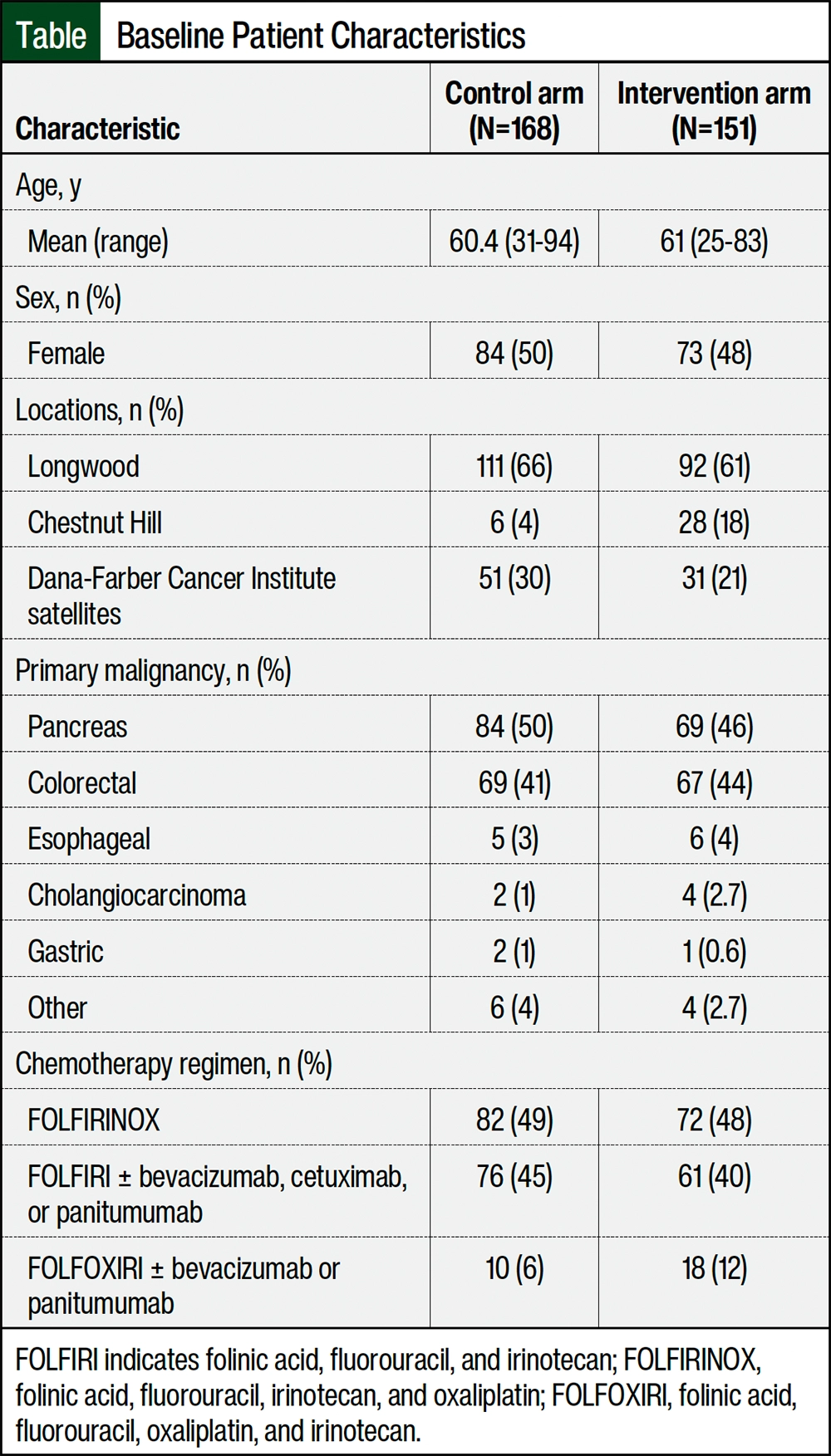
The patients’ demographics and baseline disease characteristics were similar between the 2 treatment groups (Table). The majority of patients had pancreatic cancer followed by colorectal cancer in both arms, and the most frequent chemotherapy regimens received were FOLFIRINOX and FOLFIRI with or without monoclonal antibodies.
The total median atropine dose was 0.4 mg (interquartile range, 0.4-0.4) during cycle 1, day 1 for both arms (95% confidence interval, 0-0; P=.8).
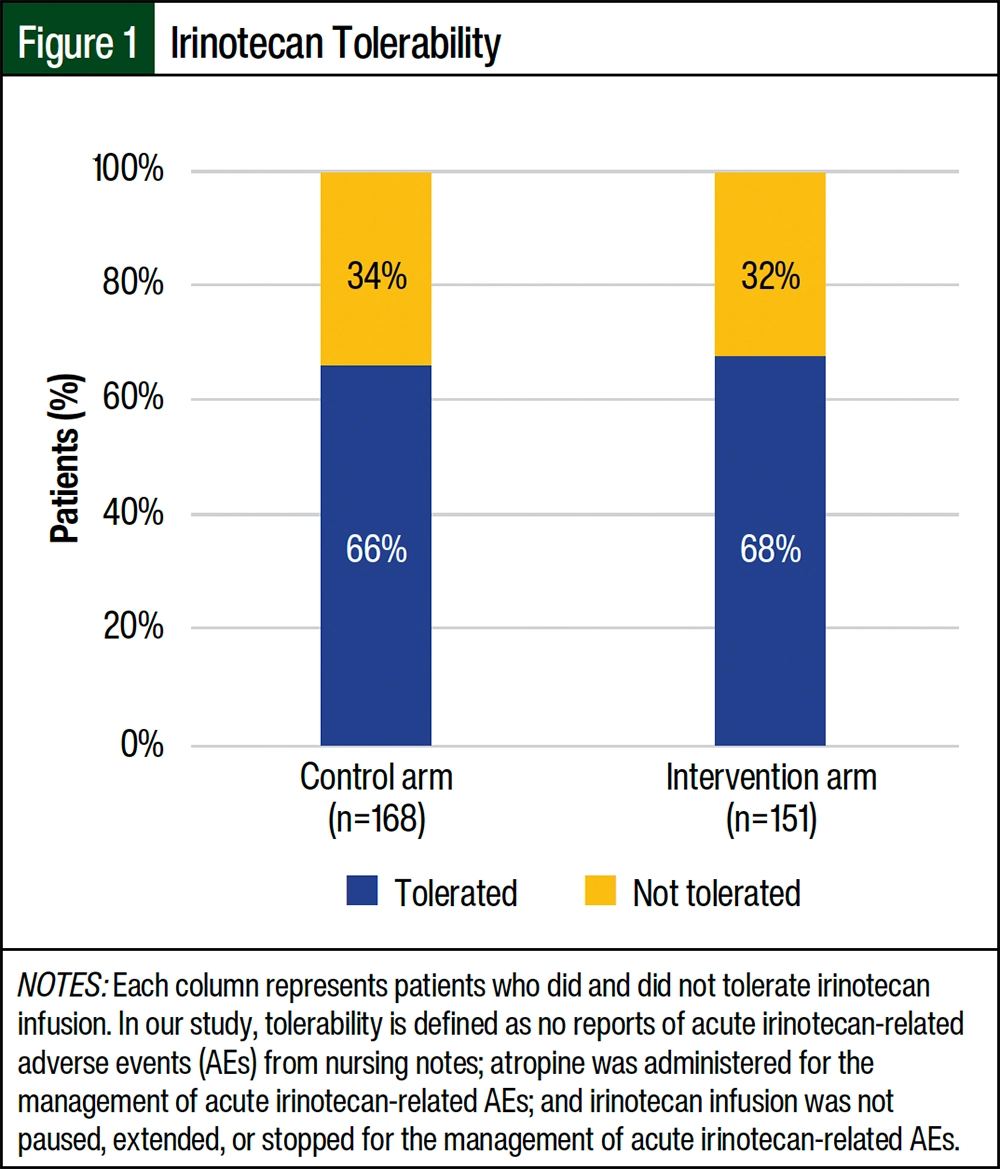
Irinotecan tolerability was assessed in our study, with each column representing patients who tolerated or did not tolerate the irinotecan infusion. Among the patients in the 60- to 90-minute arm, 111 (66%) of the 168 patients tolerated the irinotecan infusion (Figure 1). In the 30-minute arm, 102 (68%) of the 151 patients tolerated the irinotecan infusion.
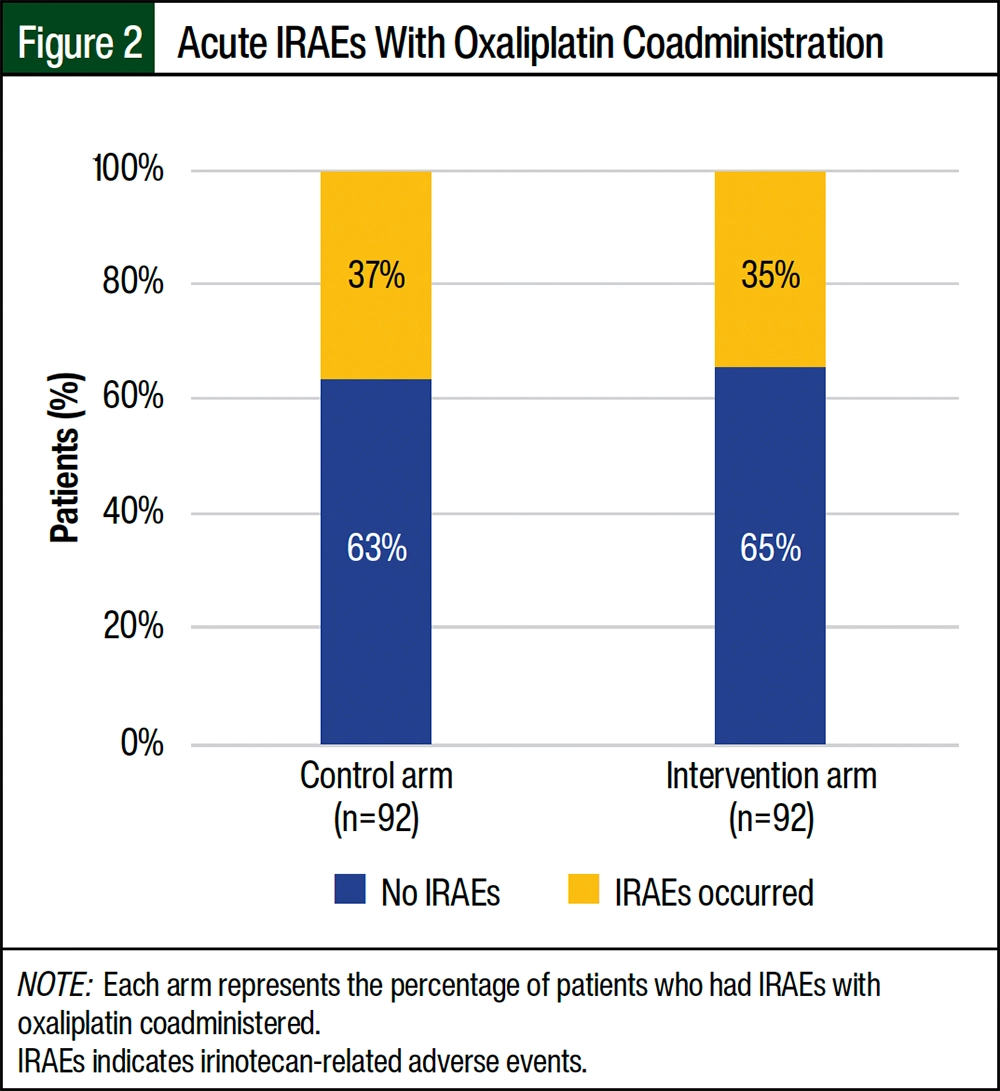
The results revealed that among the patients in the control arm, 34 (37%) of the 92 patients who received oxaliplatin coadministered with irinotecan had irinotecan-related AEs (Figure 2). Similarly, in the intervention arm, 32 (35%) of the 92 patients who received oxaliplatin coadministered with irinotecan also had irinotecan-related AEs.
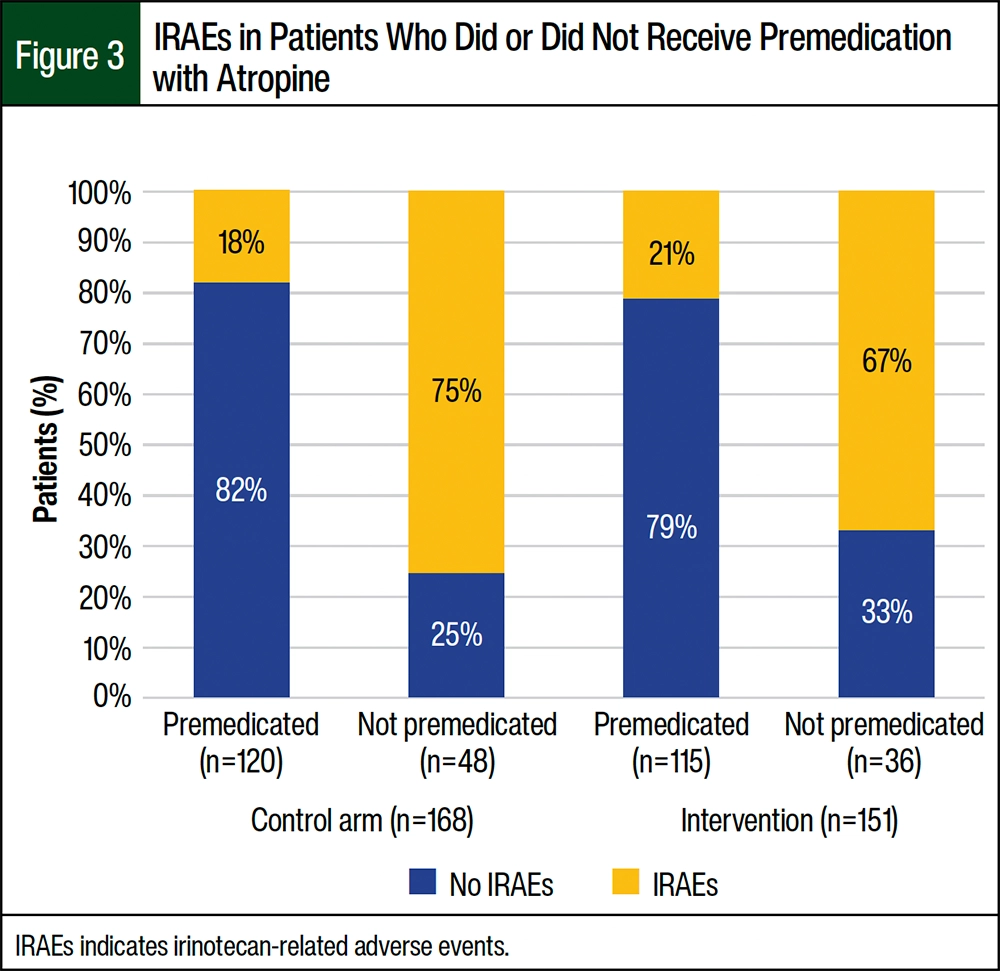
Of the 120 patients in the control arm who received premedication with atropine, 21 (18%) patients had acute irinotecan-related AEs whereas 36 (75%) of the 48 patients who did not receive atropine premedication had acute irinotecan-related AEs (Figure 3). Of the 115 patients in the intervention arm who received premedication with atropine, 24 (21%) patients had acute irinotecan-related AEs whereas 24 (67%) of the 36 patients who did not receive atropine premedication had acute irinotecan-related AEs. It is interesting to note that the patients who did not receive premedication with atropine in both arms had a higher occurrence of irinotecan-related AEs.
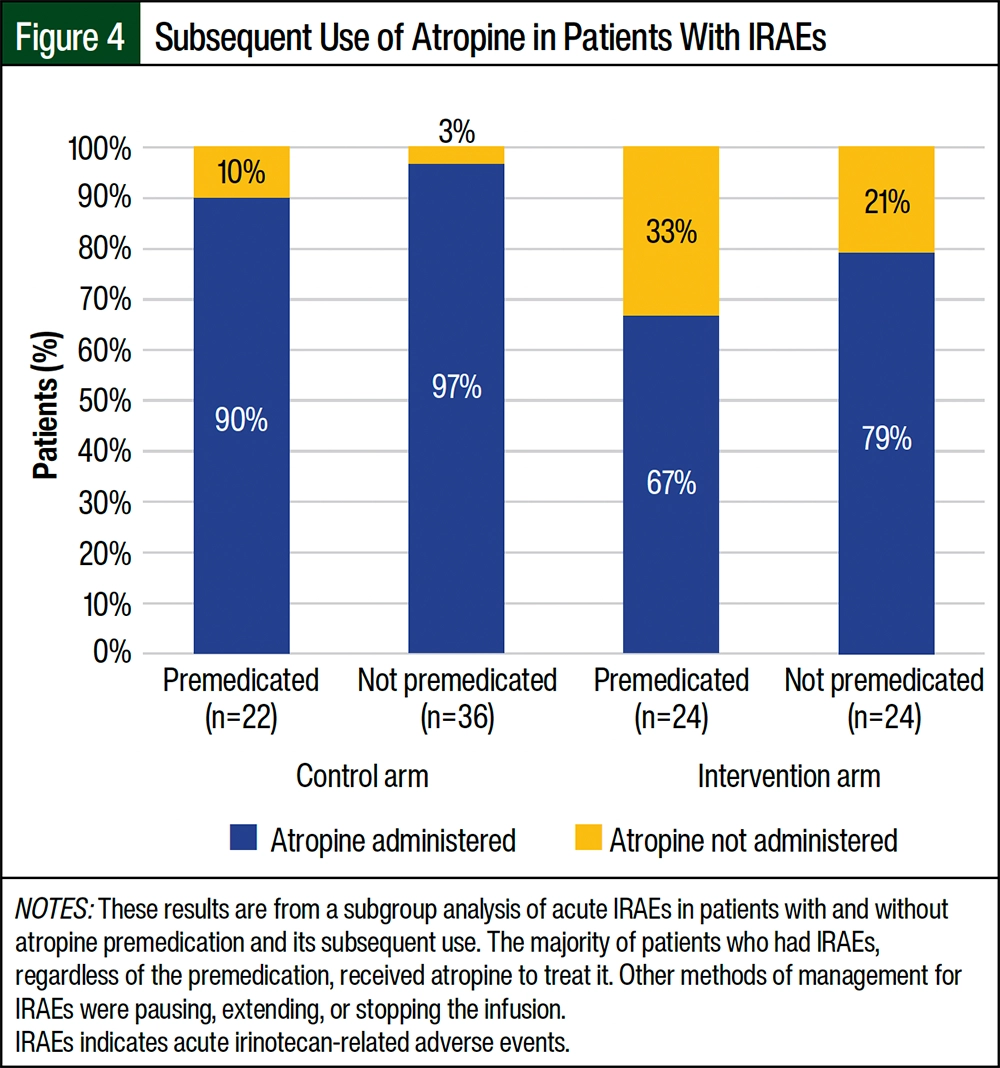
In the group that received atropine premedication in the control arm, of the 22 patients who had acute irinotecan-related AEs, 19 (90%) received atropine as a treatment (Figure 4). Conversely, in the group that did not receive atropine premedication, of the 36 patients who had acute irinotecan-related AEs, 35 (97%) received atropine as treatment. In the control group that received atropine premedication, 3 (10%) patients required additional management strategies, such as pausing, extending, or stopping the infusion, as a result of the severity of the irinotecan-related AEs. Similarly, in the control group that did not receive atropine premedication, 1 (3%) patient necessitated additional management methods.
Within the intervention arm, the results of the subgroup analysis demonstrated similar trends. In the group that received atropine premedication, 16 (67%) of the 24 patients who had acute irinotecan-related AEs received atropine as a treatment. Among these patients, 8 individuals (33%) required additional management interventions, such as pausing, extending, or stopping the infusion. In the group that did not receive atropine premedication in the intervention arm, 19 (79%) of the 24 patients who had acute irinotecan-related AEs received atropine as a treatment (Figure 4).
These findings indicate that atropine was frequently employed as a management strategy for acute irinotecan-related AEs in the patients who did and did not receive premedication. In addition, a small proportion of patients required alternative management approaches when atropine alone was insufficient in controlling irinotecan-related AEs.
Discussion
This single-institution, retrospective study validates the safety and tolerability of shortened irinotecan infusions in a large population of patients with GI cancer, with a specific focus on atropine premedication, in the hope that patients will spend less time in the infusion clinic and that nurses will therefore have more time to care for a larger number of patients.
Despite reports from the nursing and pharmacy staff that indicated a possible increase in acute irinotecan-related AEs with the shorter infusion, this study’s results showed that the total median atropine dose administered during cycle 1, day 1 was 0.4 mg, regardless of its use for premedication or for the treatment of acute irinotecan-related AEs. There was no significant difference in atropine dose between the control and intervention arms (P=.80), indicating comparable atropine administration practices.
The results for the secondary objective indicated that irinotecan tolerability was similar between the study arms, with 66% of patients in the control arm and 68% of patients in the intervention arm tolerating the irinotecan infusion. These findings show that the shorter infusion duration in the intervention arm did not compromise irinotecan’s tolerability.
Among the patients who received oxaliplatin in addition to irinotecan, 37% of patients in the control arm and 35% of those in the intervention arm had irinotecan-related AEs. These results indicate that the incidence of irinotecan-related AEs associated with oxaliplatin coadministration was similar between the 2 arms, and that the infusion’s duration did not significantly impact the prevalence of oxaliplatin-related AEs.
The subgroup analysis revealed that atropine administration was the primary method of managing acute irinotecan-related AEs in both arms, regardless of the patient’s atropine premedication status. These results show that atropine premedication may have a protective effect against acute irinotecan-related AEs, regardless of the infusion duration. Thus, premedication may be prudent in patients without contraindications. Contraindications to atropine premedication include glaucoma, prostatic hypertrophy or urinary retention, cardiac arrhythmias or tachycardia, myasthenia gravis, intestinal obstruction, or severe inflammatory bowel disease, and known hypersensitivity or allergy to atropine or any related medications.1
At our institution, premedication with atropine before a cycle-1, day-1 infusion of irinotecan was not standard practice; however, the data collection revealed that the vast majority of patients received atropine premedication, with rates of 71% in the control arm and 76% in the intervention arm. This high use of atropine premedication may be attributed to several factors. First, the experienced staff members at DFCI are well-versed in the management of acute irinotecan-related AEs, and they have implemented atropine premedication based on their clinical expertise to prevent cholinergic symptoms. This use reflects the institution’s commitment to the proactive management of irinotecan-related AEs and optimizing patient care. To date, this study is the first to compare the tolerability of irinotecan infusion for 30 minutes versus 60 to 90 minutes using atropine administration.
Limitations
The study had several potential limitations, including its retrospective study design and the potential for confounding factors. The data collected were limited to cycle 1, day 1 of treatment, and no data were documented in the EHR on irinotecan-related AEs occurring after discharge from the infusion center within 24 hours. Moreover, the analysis was based on data from medical records, including the incomplete data and delays in time stamps relative to the actual time of care. Last, the severity of these AEs was also not assessed.
During the time of this research, giving atropine as a premedication before cycle-1, day-1 irinotecan was not routine practice. The outcomes of this study have led our institution to introduce atropine premedication orders for irinotecan-based treatment plans on cycle 1, day 1, especially when the dose is ≥150 mg/m2. This integration of atropine premedication has been established as a standard protocol within our institution, as guided by the findings of this study.
Conclusion
In conclusion, the findings of this study show that the administration of a shorter infusion (30 minutes vs 60-90 minutes) of irinotecan in our patients with GI malignancy was well-tolerated and was not associated with an increase in irinotecan-related acute AEs. In addition, the study results show that atropine premedication is effective in reducing the incidence of acute irinotecan-related AEs, regardless of the infusion’s duration.
These results provide valuable insights for clinicians to optimize the administration protocols for irinotecan, potentially improving patient experiences and treatment outcomes in the management of solid tumors. The results of this study support continuing the practice of administering irinotecan at an infusion time of 30 minutes.
Acknowledgments
We would like to thank Chao Ma for the help with data analysis and Yun Man for her help with the institutional review board application.
Author Disclosure Statement
Dr Bui, Dr Hammam, Dr Schlechter, and Dr Blouin have no conflicts of interest to report.
References
- Camptosar (irinotecan hydrochloride) injection, for intravenous use [prescribing information]. Pfizer; January 2022. Accessed April 15, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2022/020571Orig1s053lbl.pdf
- Masi G, Falcone A, Paolo A, et al. A phase I and pharmacokinetic study of irinotecan given as a 7-day continuous infusion in metastatic colorectal cancer patients pretreated with 5-fluorouracil or raltitrexed. Clin Cancer Res. 2004;10 (5):1657-1663.
- Bouzid K, Khalfallah S, Tujakowski J, et al. A randomized phase II trial of irinotecan in combination with infusional or two different bolus 5-fluorouracil and folinic acid regimens as first-line therapy for advanced colorectal cancer. Ann Oncol. 2003;14:1106-1114.
- André T, Louvet C, Maindrault-Goebel F, et al. CPT-11 (irinotecan) addition to bimonthly, high-dose leucovorin and bolus and continuous-infusion 5-fluorouracil (FOLFIRI) for pretreated metastatic colorectal cancer. GERCOR. Eur J Cancer. 1999;35(9):1343-1347.
- Assersohn L, Brown G, Cunningham D, et al. Phase II study of irinotecan and 5-fluorouracil/leucovorin in patients with primary refractory or relapsed advanced oesophageal and gastric carcinoma. Ann Oncol. 2004;15:64-69.
- Koselke E, Kraft S. Chemotherapy-induced diarrhea: options for treatment and prevention. J Hematol Oncol Pharm. 2012;2:143-151.
- Salehifar E, Gheibi S, Janbabaei G, Mousavi K. Adverse effects of chemotherapy regimens used in colorectal cancer patients in a referral cancer center in north of Iran, 2008-2014. J Pharm Care. 2016;4:9-13.
- Saltz LB, Cox JV, Blanke C, et al. Irinotecan plus fluorouracil and leucovorin for metastatic colorectal cancer. N Engl J Med. 2000;343:905-914.
- Rowinsky EK, Grochow LB, Ettinger DS, et al. Phase I and pharmacological study of the novel topoisomerase I inhibitor 7-ethyl-10-[4-(1-piperidino)-1] carbonyloxycamptothecin (CPT-11) administered as a ninety-minute infusion every 3 weeks. Cancer Res. 1994;54:427-436.
- National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE). Version 4.03. June 14, 2010. Accessed April 15, 2024. https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03/CTCAE_4.03_2010-06-14_QuickReference_8.5x11.pdf
- Rothenberg ML, Eckardt JR, Kuhn JG, et al. Phase II trial of irinotecan in patients with progressive or rapidly recurrent colorectal cancer. J Clin Oncol. 1996;14:1128-1135.
- Gunturu KS, Yao X, Cong X, et al. FOLFIRINOX for locally advanced and metastatic pancreatic cancer: single institution retrospective review of efficacy and toxicity. Med Oncol. 2013;30:361.
- Matsuoka A, Maeda O, Inada-Inoue M, et al. FOLFIRINOX-induced reversible dysarthria: a case report and review of previous cases. Oncol Lett. 2015;10:2662-2664.
- Rougier P, Bugat R, Douillard JY, et al. Phase II study of irinotecan in the treatment of advanced colorectal cancer in chemotherapy-naive patients and patients pretreated with fluorouracil-based chemotherapy. J Clin Oncol. 1997;15:251-260.
- National Comprehensive Cancer Network. Colon Cancer (FOLFIRI Chemotherapy Order template. COL14). August 2, 2021. Accessed October 20, 2021. [Requires subscription to access.]
- DeLap RJ. The effect of leucovorin on the therapeutic index of fluorouracil in cancer patients. Yale J Biol Med. 1988;61:23-34.