Connecting Science to Practice
Drug repository programs are designed to accept unused
medications and redistribute them to patients in need.
Given the high costs of cancer medications, the use of repository
programs to redispense oral cancer drugs may play
a role in decreasing the financial burdens of care. This observational
study shows how a new cancer drug repository
service was created at a community cancer center and characterizes
the results from the first 22 months of operation.
The results indicate that, even in smaller or resource-constrained
areas, drug repositories are feasible, may prevent
drug waste, and may help bridge gaps in medication coverage
for at-risk patients. A statewide cancer drug repository
service has since been created in Michigan to broaden the
impact of these programs.
The annual US spending on oral cancer drugs has surpassed $947 million1 and is expected to increase 12% to 15% annually through 2027.2 In many cases, a significant portion of these funds come directly from patients.3 The out-of-pocket costs for the top 54 oral chemotherapy agents in 2019 were projected to exceed $10,000 per fill annually for patients enrolled in Medicare Part D plans.3
The consequences of such costs are felt in various ways, including directly and indirectly. Approximately 49% of patients who are receiving treatment for cancer in the United States have financial burden of any kind, with wide-ranging and disparate manifestations, depending on critical factors such as age, ethnicity, insurance coverage, and marital status.4 Patients who have financial toxicity have more than twice the odds of worse self-reported physical, mental, emotional, and social functioning, as well as increased rates of depression and anxiety, compared with patients without financial toxicity.5-9 In a single US study of more than 230,000 patients (all cancers except nonmelanoma skin cancer), financial toxicity, as evidenced by bankruptcy, was an independent risk factor for early mortality.10
The high cost of chemotherapy is exacerbated by excess drug waste within the US healthcare system.11 It is estimated that 25% of annual US healthcare spending (approximately $765 billion) is wasted each year, and approximately 740 tons of unused medications may be unnecessarily discarded by US nursing homes on an annual basis.11,12 An analysis of drug waste from a regional cancer center concluded that, over a 3-year period, 41% of patients who received prescriptions for everolimus, sunitinib, axitinib, or vemurafenib had wasted at least some of their drug supply.13 The reasons for waste were cancer progression (69%), adverse events (13%), patient death (13%), and dose changes (5%). Wasted drug was calculated to have a combined average wholesale price of approximately $250,000 and required more than $2500 in copays for a drug that was not used.13
To ameliorate the cycle of increasing costs and waste generation, state and regional governments have enacted legislation that permits the reuse of prescription medication within drug repository programs. As of September 2024, 45 states, Washington DC, and Guam had adopted such legislation, with 29 states having operational programs.14 ASCO endorses the use of repository programs to mitigate financial toxicity,15 and emerging data support the viability and benefit of cancer drug repository (CDR) operation.16,17 Despite this progress, however, notable obstacles, such as burdensome requirements and administration, lack of financial or operational resources, and a lack of public awareness, may impede the routine implementation of these programs.18
In November 2021, Cowell Family Cancer Center Infusion Pharmacy became one of the only active CDR programs in Michigan. The goal of this study was to characterize the preliminary results of a newly implemented CDR at a community cancer center, share a framework for repository implementation, and disseminate lessons broadly to accelerate participation in repositories regionally and nationally. The primary objective was to characterize the number of patients served through the repository and share the corresponding US dollar value of dispensed medications. A secondary objective was to characterize the US dollar value of medication saved from the unnecessary disposal of drugs through the use of the repository.
Methods
This single-center, observational study was conducted at Cowell Family Cancer Center in Traverse City, MI, from November 2021 through September 2023. The study received approval as exempt research by the Munson Healthcare Institutional Review Board to evaluate the impacts made by participation in the Michigan Cancer Drug Repository Program.
Cowell Family Cancer Center is a community cancer center operating within the Munson Healthcare System to serve patients throughout the northwest region of Michigan’s lower peninsula. A total of 14 clinical oncology pharmacists work alongside 9 medical oncologists, 1 gynecologic oncologist, and several advanced practice providers to service approximately 3000 new patients annually.
As a participating site in the Pharmacists Optimizing Oncology Care Excellence in Michigan collaborative quality initiative,19 2 of the 14 clinical oncology pharmacists within Munson Healthcare are embedded within ambulatory clinics that provide patient education for each new oral chemotherapy regimen. Each of these education encounters includes brief information about the CDR, criteria for participation, and instructions on how to donate and receive medications through the repository, if applicable. In addition, printed materials were created for patient distribution and are included in every new patient information packet.
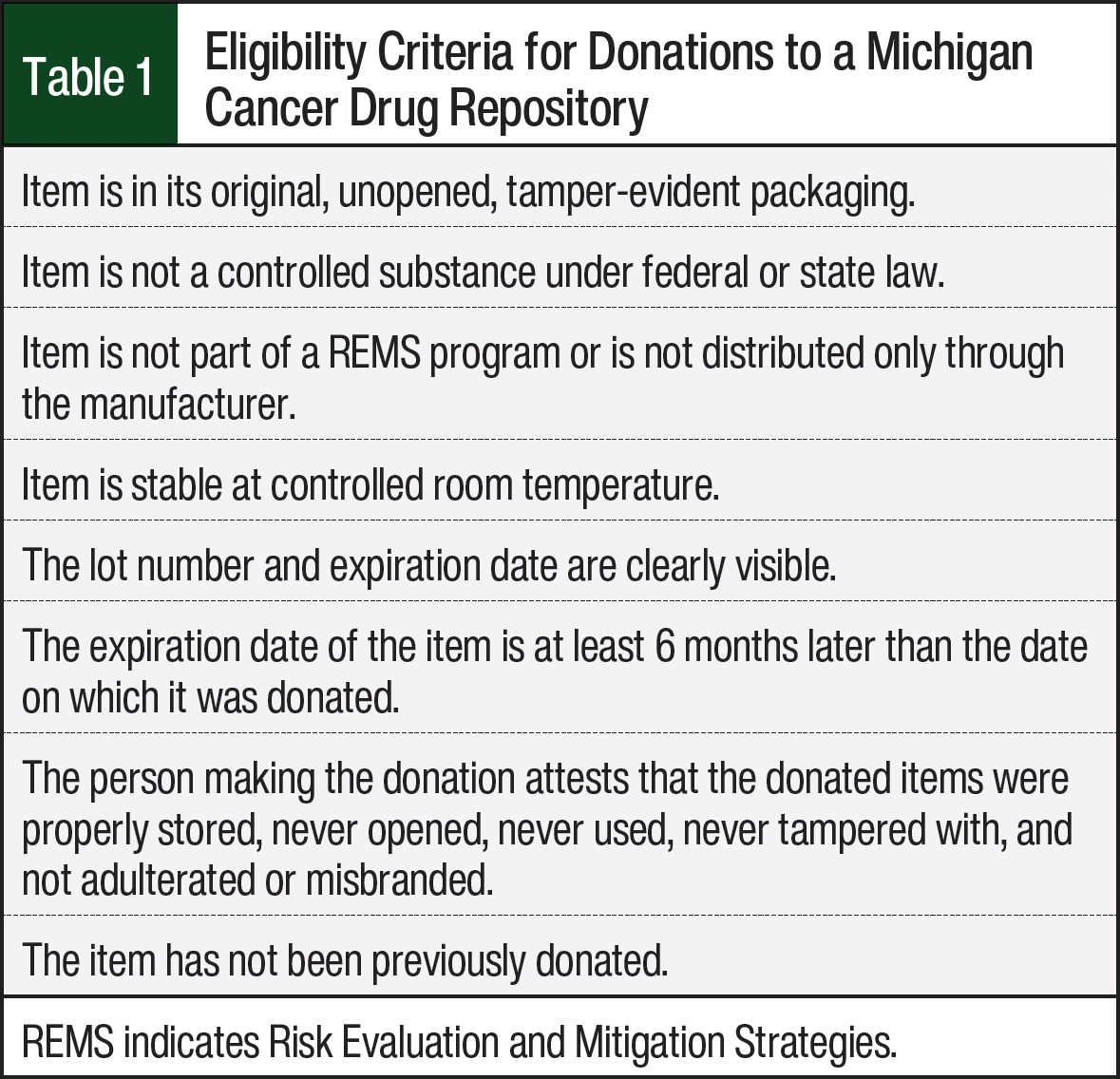
Adopted by the Michigan Legislature in 2006, the Michigan Cancer Drug Repository Program allows for the donation of previously dispensed medications and supplies used for the treatment of cancer or its side effects, as long as the donated items meet the criteria listed in Table 1. The donated medications and supplies may be redispensed by a pharmacist pursuant to a prescription for patients with cancer who have permanent residency status in the state of Michigan. There are no residency requirements for the donors of the medication. All participants in the program (those who donate and those who receive donations) must sign documentation agreeing to the program’s requirements.
The institutional infrastructure for the CDR was developed and implemented over a 6-month period. Before the CDR’s implementation, the study team reviewed guidance from the ASCO 2020 and ASCO 2022 position statements on drug repository programs and the requirements from the Michigan Department of Licensing and Regulatory Affairs20 to create new standard operating procedures related to the receipt, storage, and dispensing of eligible donated medications. To leverage the stakeholders’ buy-in and disseminate pertinent information, meetings were held with select teams (ie, biomedical ethics, financial navigation, patient safety and quality, medical oncology, legal services, pharmacy clinical council) throughout the healthcare system. Because CDR prescriptions are not a substitute for a long-term supply of medication, intentional and transparent communication between the pharmacy and financial navigation services was a special priority to ensure that there is a viable plan for therapy after each CDR dispense.
An electronic CDR inventory was created using the site’s existing inventory management software, which allows separate tracking of all CDR items. CDR medications are kept in existing storage areas and are physically separated from the general inventory of drugs. CDR prescriptions are dispensed similar to non-CDR prescriptions with standard procedures and dating. The notable differences between CDR and non-CDR prescriptions include a disclosure on the CDR label that identifies the medication as previously dispensed and donated, a note within the electronic medical record that documents the patient’s receipt of a CDR prescription, and designation on the recipient patient’s home medication list that the medication originated from the CDR. State-required documentation of the intake and dispensing of the donated drug is stored in a hard-copy format. Records are kept for source verification and reference for at least 5 years.
The CDR does not operate with separately designated full time–equivalent employees. The initial policies and procedures were developed as part of a PGY-1 residency project, and relevant updates to policy will be overseen by the infusion pharmacy manager. Inventory management, administrative duties, and staff training are primarily assigned to 1 pharmacy technician who balances the CDR needs with other duties. All pharmacists, technicians, and pharmacy residents assist with the receipt of donations as the donations are brought to the pharmacy or ambulatory clinic. Dispensing from the repository and the associated documentation are facilitated by any licensed pharmacist who is available. Two clinical pharmacists divide the remainder of the CDR tasks (committee meetings, collaboration with financial navigation services, patient outreach) as a component of their overall responsibilities.
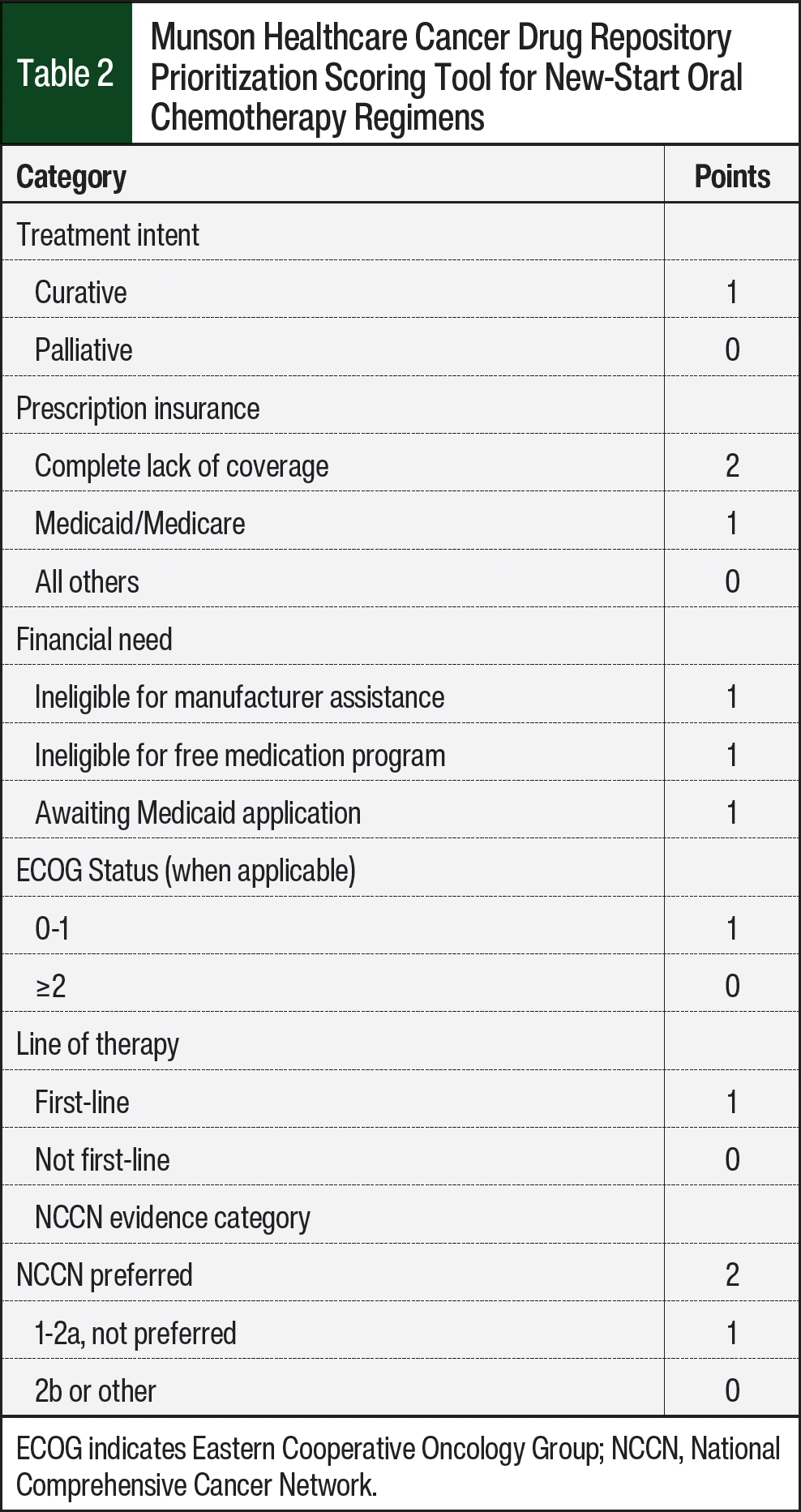
Michigan law requires that allocations through a CDR be prioritized by certain criteria. The first priority is that the drug goes to individuals who are uninsured or who lack coverage for the medication, including patients who do not qualify for manufacturer or institutional copay assistance or who cannot afford their medication despite such copay assistance. The second priority is to individuals who are enrolled in Medicaid, Medicare, or any other public assistance healthcare program. The third priority is to all others who have been diagnosed with cancer. The study team believed that additional criteria would be beneficial to operate the CDR ethically and with just principles of scarce resource allocation, consistent with previous strategies for CDR operation.16
Table 2 shows the current iteration of a scoring tool used by the site to objectively evaluate candidates for new regimens, with higher scores indicating a greater priority for dispensing. The scoring tool is designed to be used in collaboration with the medical oncologist and pharmacist when needing to allocate scarce drug among multiple patients who are not otherwise prioritized by state law (eg, when there is supply for 1 patient, but 2 patients with the same prescription insurance express financial need for the drug). Anticipating ambiguity in such decisions, a standing ethics committee has also been created to guide the allocation of the donated drugs. This committee meets on an as-needed basis and consists of at least 1 member from the system’s biomedical ethics committee, 1 clinical pharmacist, and 1 medical oncologist. Notably, this approach to CDR operation attempts to minimize the use of a first-come, first-served approach, which is thought to further advantage those patients with more privilege at baseline.21,22
Results
Cowell Family Cancer Center received regulatory approval for CDR operation from the Michigan Board of Pharmacy in November 2021 and received its first donation of oral chemotherapy in December 2021. By November 2023, 6 additional sites within Munson Healthcare received the Board of Pharmacy’s approval for CDR operation, for a total of 7 CDR sites spread throughout the regional Munson Healthcare System. All sites share centralized inventory and tracking of CDR medications.
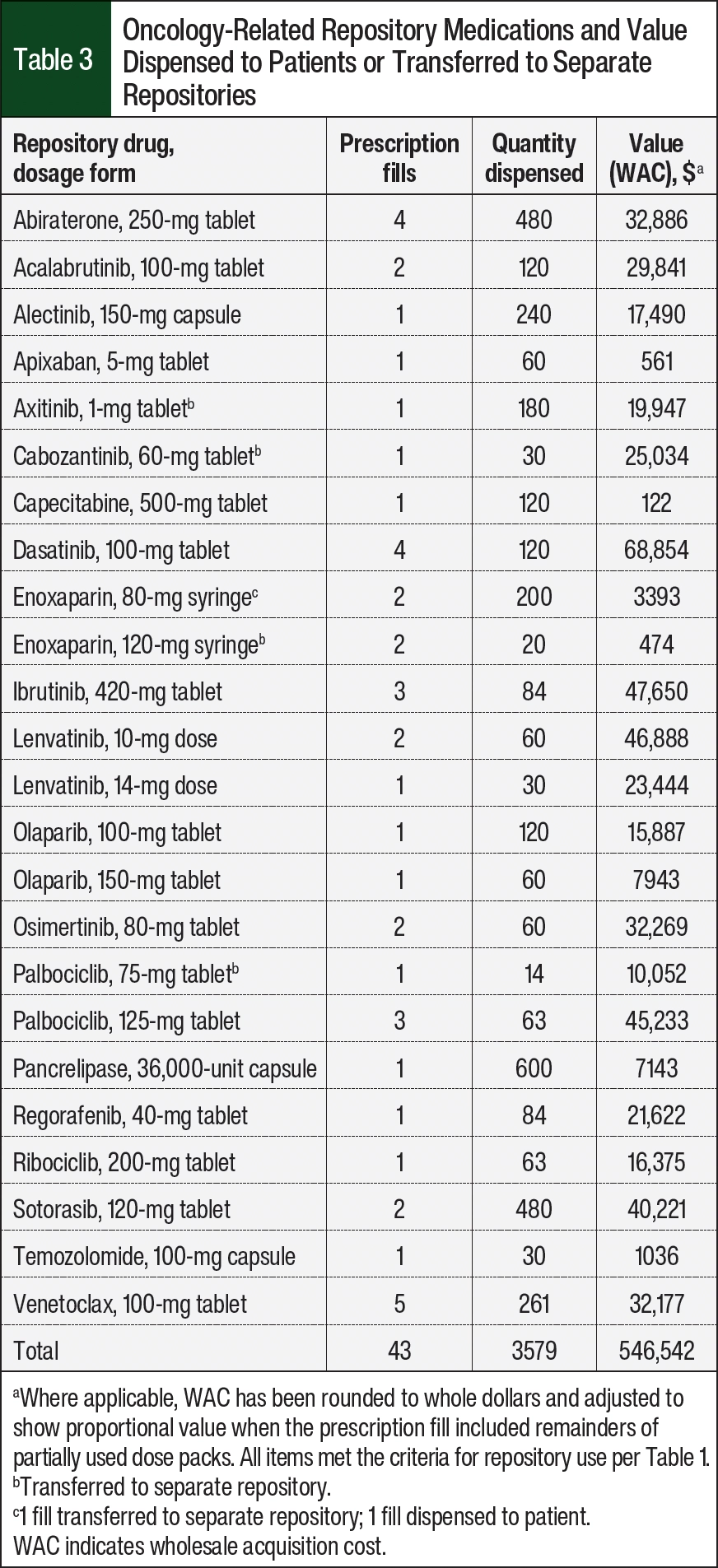
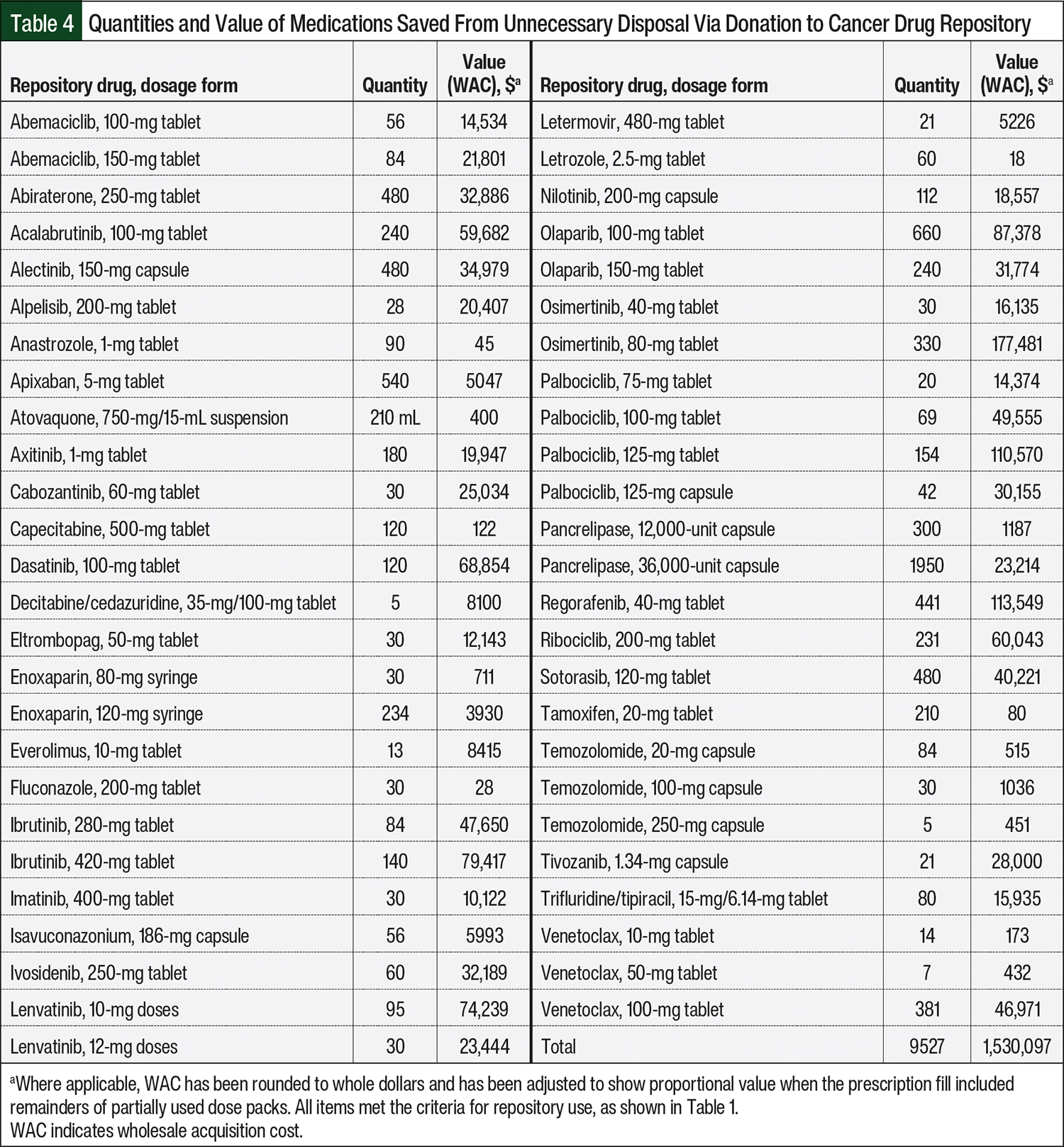
As of September 30, 2023, the system repository accepted a total of 96 drug donations, consisting of 56 unique medications, valued at approximately $1.64 million. The repository has dispensed 38 prescriptions that were valued at more than $490,000 to 35 patients and transferred medications with a value of $55,800 to separate repositories for an additional 5 patients. All medication values are reported in US dollar wholesale acquisition cost (WAC) at the time of dispensing per Micromedex’s RED BOOK data. Table 3 summarizes the CDR medications that have been distributed for patient use, and Table 4 summarizes the total value of medication saved from unnecessary disposal by using the repository operation.
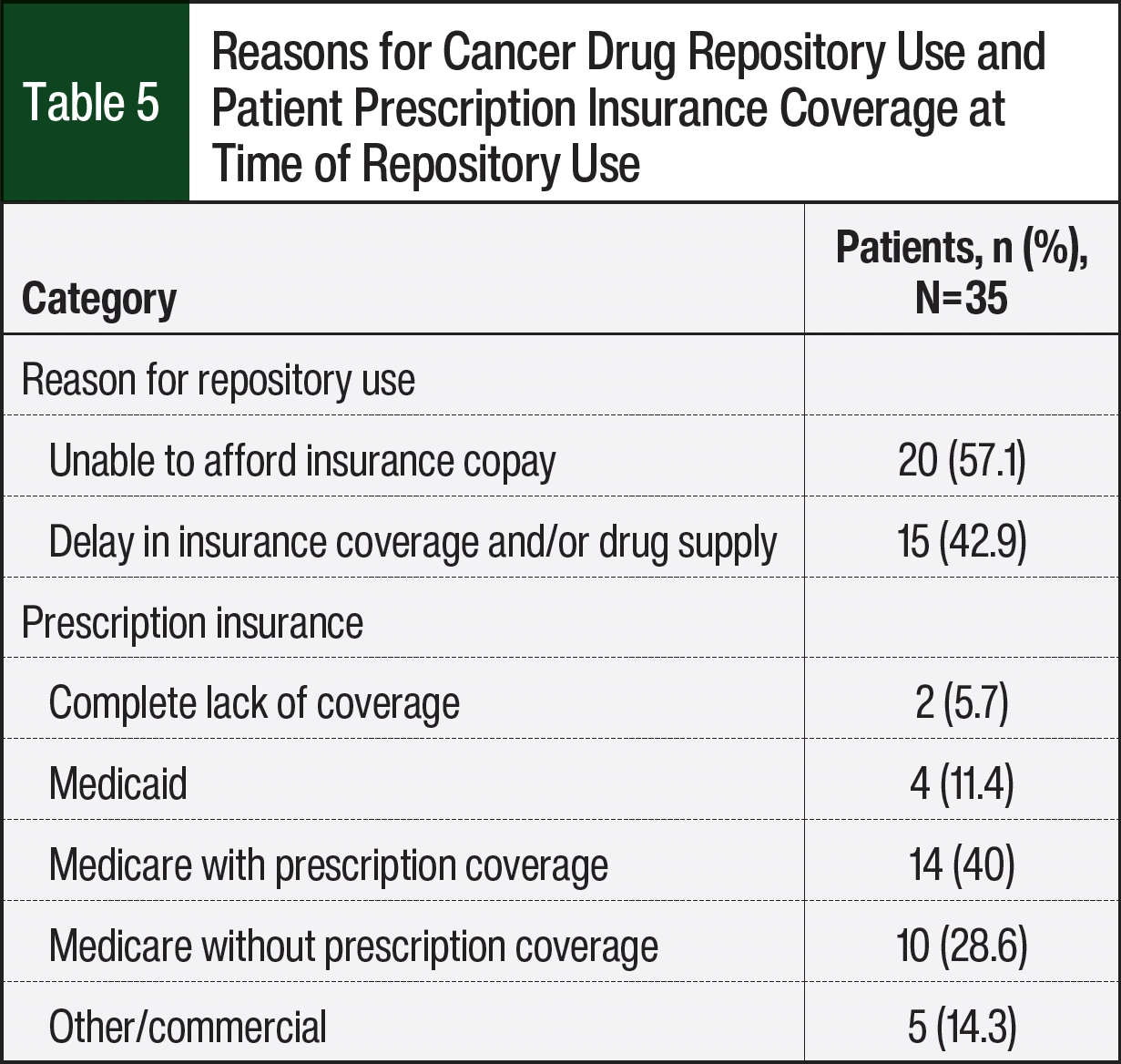
Table 5 summarizes the patients’ reasons for the use of the repository and their prescription insurance coverage status. Most (n=20; 57.1%) of the 35 patients who received drugs through the repository were unable to afford the copay for their medication and were seeking alternate payment methods for coverage. The remainder of the 35 patients (n=15; 42.9%) were having difficulty acquiring a supply of medication because of insurance or processing delays. Of the 35 patients served, most (n=28; 80%) received health insurance through Medicare or Medicaid, although 10 patients with Medicare did not have active prescription drug coverage (Table 5). A total of 2 patients expressed a need for CDR services and were ineligible for the program because of out-of-state residency. All other identified patients were eligible for CDR services. Further, the collaboration between pharmacy and financial navigation ensured that all applicable CDR recipients had an affordable, long-term supply of medication before running out of supply from the CDR.
Discussion
Despite a lack of dedicated pharmacy resources within our community setting, we were able to establish a systemwide CDR service using guidance from the Michigan Board of Pharmacy, ASCO, and previous publications. As of September 30, 2023, the CDR had dispensed prescription fills for 20 patients who had difficulty affording or accessing their cancer-related medications. Before the availability of the CDR in our system, these patients would have had to wait until existing measures (eg, financial navigation, manufacturer assistance programs) were operational, pay unaffordable copays, or forego therapy altogether. The CDR provides a bridge supply of medication to patients while a more sustainable supply is secured. For example, venetoclax tablets were dispensed to 5 patients with newly diagnosed acute myeloid leukemia at hospital discharge, and each patient was able to continue therapy until their outpatient supply of venetoclax was available. In this manner, the CDR bridged a potential gap in care for an acute disease state that would not have been available otherwise.
The RED BOOK data suggest that CDR operation may have prevented the unnecessary disposal of approximately $1.53 million of medication. WAC pricing is not indicative of actual consumer cost, but using more accurate methods of cost comparison, such as prescription copays, was outside the scope of this study. As a bridge to sustainable therapy, CDR medications also decreased the aspects of opportunity cost by providing direct medication supply, simplifying the supply chain process, and minimizing wait times.
During the first 18 months of the CDR’s operation, more medications were donated to the CDR than were dispensed to patients, which resulted in a considerable inventory of drugs. Although such surplus may allow the CDR to care for a diverse patient population, many of the medications have rare indications, short expiration dates at donation, or both. Together, these factors have required the CDR to dispose of 6 donated medications (alpelisib, imatinib, lenvatinib, pancrelipase, regorafenib, and sunitinib), which were valued at approximately $114,000, and there was more than $91,000 of medication that was going to expire by the end of 2023.
To prevent waste, CDR stakeholders have begun to work on multiple fronts to maximize patients’ benefit from the CDR. In select circumstances when the medications have less than a 90-day shelf life, the repository personnel may proactively contact patients and offer the CDR’s supply of a drug. This occurs primarily when the CDR has sufficient quantity of a drug on hand to also cover urgent fills or new starts of the same regimen. When medications have less than a 45-day shelf life, the CDR does not require that a drug’s inventory be kept on hand to address urgent fill requests. For example, the CDR had 120-mg tablets of sotorasib that were expiring in 60 days. By analyzing a report generated through the electronic medical records, it was discovered that 3 patients in the community were currently maintained on this regimen. Of the 3 patients, 1 patient did not wish to pursue therapy, 1 patient had a $0 copay, and 1 patient self-reported financial distress from their copay for sotorasib. A 30-day supply of sotorasib was dispensed through the CDR to this patient. In cases when more than 1 patient may be eligible for an excess drug supply, the CDR’s ethics committee may be convened for consultation.
An additional countermeasure for drug waste includes collaboration and program growth. Encouraged in part by the early success of Munson Healthcare’s CDR, Michigan oncology stakeholders and leaders have since collaborated to create YesRx, a statewide charitable service organization that centralizes CDR operations throughout Michigan and aims to advance health equity in underserved communities. The services provided through YesRx may decrease CDR drug waste as well as increase the breadth and impact of repository operations statewide.
Separately, it is relevant to discuss external changes that may alter the operation of a future CDR. As part of the 2022 Inflation Reduction Act, the 5% coinsurance requirement within the catastrophic phase of Medicare Part D plans was eliminated in 2024.23 This change is expected to have the largest effect for patients who use costly brand-name drugs, such as oral chemotherapy. Further, out-of-pocket prescription spending will be capped at $2000 beginning in 2025.23 Because most patients served within our CDR were Medicare beneficiaries, these changes may alter the demographic that needs or receives CDR medications.
Limitations
The results of the CDR program must be viewed simultaneously with known study and site limitations, most notably that results reflect practices and the data gathered retrospectively at a single site with a small sample size. Centrally important to the success of the Munson CDR is the existence of a closed-loop model between the patient, prescribing oncologist, and the ambulatory oncology specialist. All patients who are starting to receive oral chemotherapy meet with the same pharmacy team and are provided education on the CDR, creating a durable system for CDR donation and dispensing. Therefore, the potential for the success of a repository program that operates without this clinical infrastructure cannot be determined.
Second, the creation, maintenance, and operation of even a small CDR program requires dedicated staff, resources, and institutional support. Such resources are scarce and may not be available at many sites, especially larger sites where the operation of a CDR may necessitate dedicated full time–equivalent employees or storage space.
Last, the accurate reporting of drug costs and financial impact is challenging without patient-level insurance information. Although referencing WAC may provide a comparison of drug value between the different entities, reporting the cost of medications in meaningful ways remains challenging. As such, our reported values of repository medications should be seen only as estimates.
Conclusion
The preliminary results from this single-center study show that drug repositories may function to prevent drug waste and provide a bridge supply of medications to high-risk patients who are underinsured. Cancer drug repositories may be successful even in rural communities, but further work is needed to characterize the best-practice models of a repository’s operation. A centralized, integrated, and statewide repository now exists in Michigan with the goal of increasing health equity and maximizing patient benefit. This work is needed alongside the equally important work of increasing access to affordable cancer care in our communities.
Author Disclosure Statement
Ms Palmer is a consultant to YesRx; Dr Haroney, Dr Kogelman, Dr Cloud, and Dr Kroll have no conflicts of interest to report.
References
- Fu M, Naci H, Booth CM, et al. Real-world use of and spending on new oral targeted cancer drugs in the US, 2011-2018. JAMA Intern Med. 2021;181:1596-1604.
- Aitken M, Kleinrock M, Connelly N, Pritchett J. Global Oncology Trends 2023-Outlook to 2027. IQVIA Institute for Human Data Science. May 2023. Accessed February 17, 2025. www.iqvia.com/insights/the-iqvia-institute/reports-and-publications/reports/the-global-use-of-medicines-2023
- Dusetzina SB, Huskamp HA, Keating NL. Specialty drug pricing and out-of-pocket spending on orally administered anticancer drugs in Medicare part D, 2010 to 2019. JAMA. 2019;321:2025-2027. Erratum in: JAMA. 2019;322:174.
- Smith GL, Lopez-Olivo MA, Advani PG, et al. Financial burdens of cancer treatment: a systematic review of risk factors and outcomes. J Natl Compr Canc Netw. 2019;17:1184-1192.
- Ell K, Xie B, Wells A, et al. Economic stress among low-income women with cancer: effects on quality of life. Cancer. 2008;112:616-625.
- Kale HP, Carroll N V. Self-reported financial burden of cancer care and its effect on physical and mental health-related quality of life among US cancer survivors. Cancer. 2016;122:1283-1289.
- de Souza JA, Yap BJ, Wroblewski K, et al. Measuring financial toxicity as a clinically relevant patient-reported outcome: the validation of the Comprehensive Score for financial Toxicity (COST). Cancer. 2017;123:476-484.
- Zafar SY, McNeil RB, Thomas CM, et al. Population-based assessment of cancer survivors’ financial burden and quality of life: a prospective cohort study. J Oncol Pract. 2015;11:145-150.
- Fenn KM, Evans SB, McCorkle R, et al. Impact of financial burden of cancer on survivors’ quality of life. J Oncol Pract. 2014;10:332-338.
- Ramsey SD, Bansal A, Fedorenko CR, et al. Financial insolvency as a risk factor for early mortality among patients with cancer. J Clin Oncol. 2016;34:980-986.
- Smith MD, Saunders R, Stuckhardt L, McGinnis JM, eds. Best Care at Lower Cost: The Path to Continuously Learning Health Care in America. Washington, DC: The National Academies Press; 2013.
- Allen M. America’s other drug problem. ProPublica; April 27, 2017. Accessed February 7, 2025. www.propublica.org/article/americas-other-drug-problem
- Monga V, Meyer C, Vakiner B, Clamon G. Financial impact of oral chemotherapy wastage on society and the patient. J Oncol Pharm Pract. 2019;25:824-830.
- National Conference of State Legislatures. State prescription drug repository programs. 2024. Accessed February 14, 2025. www.ncsl.org/health/state-prescription-drug-repository-programs
- American Society of Clinical Oncology. Position Statement on Drug Repository Programs. ASCO; 2022. Accessed February 10, 2025. https://cdn.bfldr.com/KOIHB2Q3/as/c5sccm86fvg48sb7wbc859/2022-Drug-Repository-Statement
- Stanz L, Ulbrich T, Yucebay F, Kennerly-Shah J. Development and implementation of an oral oncology drug repository program. JCO Oncol Pract. 2021;17:e426-e432.
- Kuhn A, Elsey R, Lockhorst R, Toennies M. Pharmacist impact on reducing medication costs for patients and decreasing medication waste: implementation and expansion of the South Dakota Drug Repository Pilot Program. HOPA News. 2021;18:12-13.
- POEM: Pharmacists Optimizing Oncology Care Excellence in Michigan. Michigan Oncology Quality Consortium. Accessed February 6, 2025. https://moqc.org/initiatives/clinical/poem/
- Smale EM, Verkerk EW, Heerdink ER, et al. Barriers and facilitators to implement the redispensing of unused oral anticancer drugs in clinical care: a hybrid-effectiveness type I study. Explor Res Clin Soc Pharm. 2024;15:100493.
- Cancer Drug Repository Program. Michigan Department of Licensing and Regulatory Affairs. Accessed February 17, 2025. www.michigan.gov/lara/bureau-list/bpl/resources/special-programs/Cancer-Drug-Repository-Program
- Persad G, Wertheimer A, Emanuel EJ. Principles for allocation of scarce medical interventions. Lancet. 2009;373:423-431.
- Krütli P, Rosemann T, Törnblom KY, Smieszek T. How to fairly allocate scarce medical resources: ethical argumentation under scrutiny by health professionals and lay people. PLoS One. 2016;11:e0159086.
- Cubanski J, Neuman T. Changes to Medicare Part D in 2024 and 2025 under the Inflation Reduction Act and how enrollees will benefit. KFF; 2023. Accessed February 17, 2025. www.kff.org/medicare/issue-brief/changes-to-medicare-part-d-in-2024-and-2025-under-the-inflation-reduction-act-and-how-enrollees-will-benefit/