Connecting Science to Practice
In patients with AML, the initial treatment selection of traditional
7+3 chemotherapy or a hypomethylating agent
(HMA)+venetoclax depends on the patient’s eligibility for
intensive induction therapy. Recent guidelines recommend
the consideration of intensive therapy as a function of overall
fitness rather than age; however, what factors determine
this fitness in the real-world setting are not clearly defined.
Treatment selection may be particularly difficult in middle-
aged patients and those who are younger but have comorbidities.
In this single-center, retrospective, cohort study, we
sought to evaluate potential drivers in treatment selection as
well as outcomes in patients with AML between 50 and 70
years old who received 7+3 versus HMA+venetoclax. The
findings of this study emphasize the importance of a patient-
specific approach with thoughtful discussion amongst
the interdisciplinary team.
In patients with acute myeloid leukemia (AML) without targetable mutations, the initial treatment selection depends on the patient’s eligibility for intensive induction therapy.1 Eligibility is determined based on factors such as performance status, comorbid conditions, cytogenetic risk factors, and previous exposure to chemotherapy or radiation therapy. Although age previously played a major role in the determination of induction intensity, the NCCN’s treatment guidelines now recommend the consideration of intensive induction therapy as a function of overall fitness rather than age.1 Previously, age >60 years was considered a therapeutic divergence point to not pursue intensive induction based on a higher prevalence of unfavorable genetics and increased comorbidities.1 However, the NCCN’s guidelines now favor the consideration of intensive cytarabine-based therapy, regardless of age, in patients who have de novo AML without unfavorable cytogenetics, with minimal comorbidities, and an ECOG score of 0 to 2.1
For patients who are eligible for intensive induction therapy, the first-line treatment is standard 7+3 chemotherapy.1 This regimen consists of a continuous infusion of cytarabine for 7 days in combination with an anthracycline on days 1 to 3. In patients who are ineligible for intensive induction chemotherapy but who want aggressive chemotherapeutic intervention, a regimen of a hypomethylating agent (HMA), such as decitabine or azacitidine, in combination with venetoclax is preferred.1
The approval of the HMA+venetoclax regimen in 2018 provided practitioners with a new treatment option for patients whose risk factors or comorbidities may make traditional chemotherapy agents a greater risk than a benefit.2 This combination was FDA approved for patients aged ≥75 years who have comorbidities precluding the use of intensive induction chemotherapy.2-4 As a result of this approval, the selection of therapy has historically been based on arbitrary cutoffs of age; however, older patients may have better overall fitness and performance status than expected, whereas younger patients may have worse overall fitness for intensive chemotherapy because of factors beyond age, such as comorbidities and type and burden of disease. In such patients, treatment selection may be a more nuanced discussion between the patient and a multidisciplinary team of providers.5-7
Although some studies have examined the use of more intensive chemotherapy regimens in patients aged >60 years, there is little literature to provide guidance on younger patients with AML who may be poor candidates for intensive induction.4,5 The purpose of this study was to assess the efficacy and adverse events (AEs) of 7+3 chemotherapy versus HMA+venetoclax in patients who are marginal candidates for either regimen by examining the diagnostic and comorbidity profiles of patients aged 50 to 70 years who received either 7+3 chemotherapy or HMA+venetoclax as induction therapy.
Methods
This single-center, retrospective-cohort study assessed the efficacy and safety of HMA+venetoclax versus traditional 7+3 chemotherapy in patients aged between 50 and 70 years at the time of treatment. The patients in this age-group who had a new diagnosis of AML and initiated first-line treatment between January 1, 2017, and December 21, 2022, were included, and the outcomes were evaluated for 1 year from the date of treatment initiation. The time range was selected to align with the FDA’s approval of the venetoclax combination and to allow for a full year of patient follow-up. Patients were excluded from the study if they were pregnant or incarcerated at the time of treatment, were diagnosed with acute promyelocytic leukemia or mixed-lineage leukemia, or if they received any additional agent as part of induction therapy (eg, gemtuzumab ozogamicin, midostaurin, ivosidenib, or liposomal daunorubicin plus cytarabine). The data were collected through retrospective chart review using Research Electronic Data Capture (REDCap) tools. REDCap is a secure, web-based software platform that is designed to support data capture for research studies.8,9
The primary end point was 1-year event-free survival (EFS). The events included death from any cause within 1 year of treatment initiation, nonresponse after receiving the first treatment after bone marrow biopsy, or relapse within 1 year of treatment initiation. The secondary end points included 1-year overall survival (OS), response after the first bone marrow biopsy (performed at approximately day 14 for 7+3 chemotherapy and at day 21 for HMA+venetoclax), and documented recurrence within 1 year.
Additional safety and efficacy end points comprised an evaluation of AEs within 30 days of treatment, including documented infection by positive culture or imaging; the duration of neutropenia; hepatic dysfunction with liver function tests >5 times the upper limit of normal; documented mucositis; a cardiac event, including myocardial infarction, myocarditis, or new-onset atrial fibrillation; as well as transfer to an intensive care unit (ICU) at any time during the induction hospitalization.
Statistical Analysis
Descriptive statistics for the demographic and clinical characteristics were reported by treatment group and overall. Continuous variables were summarized with mean, quantiles, and range, and categorical variables were summarized using frequency and percentages. The differences in the patient characteristics and treatment groups were evaluated using appropriate univariate tests, such as chi-square test, Wilcoxon rank-sum test, and Fisher’s exact test.
All statistical tests were calculated with a fixed type-I error rate of 0.05, and all analyses were completed using SAS version 9.4 (SAS Institute, Inc; Cary, NC).
Results
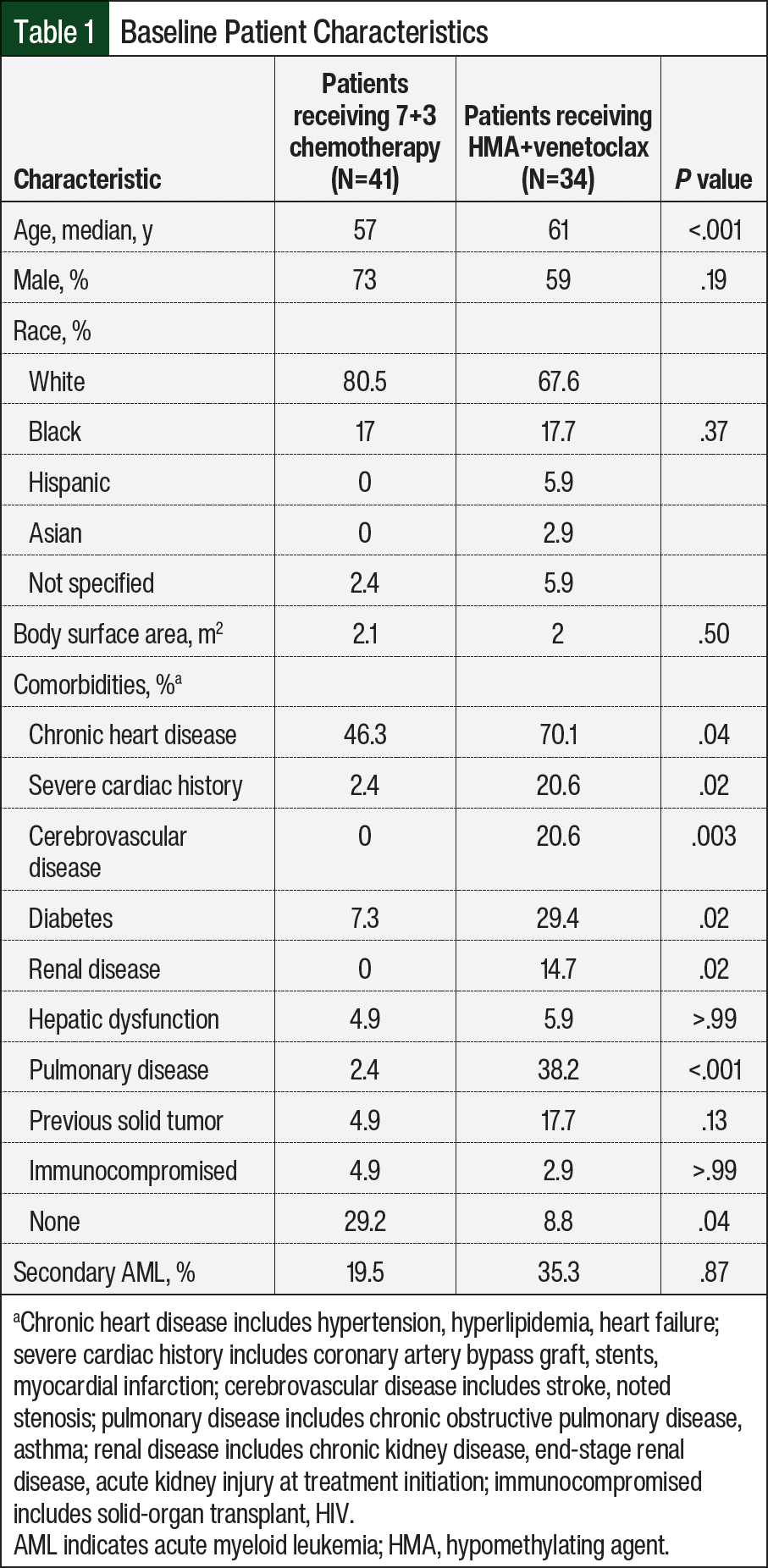
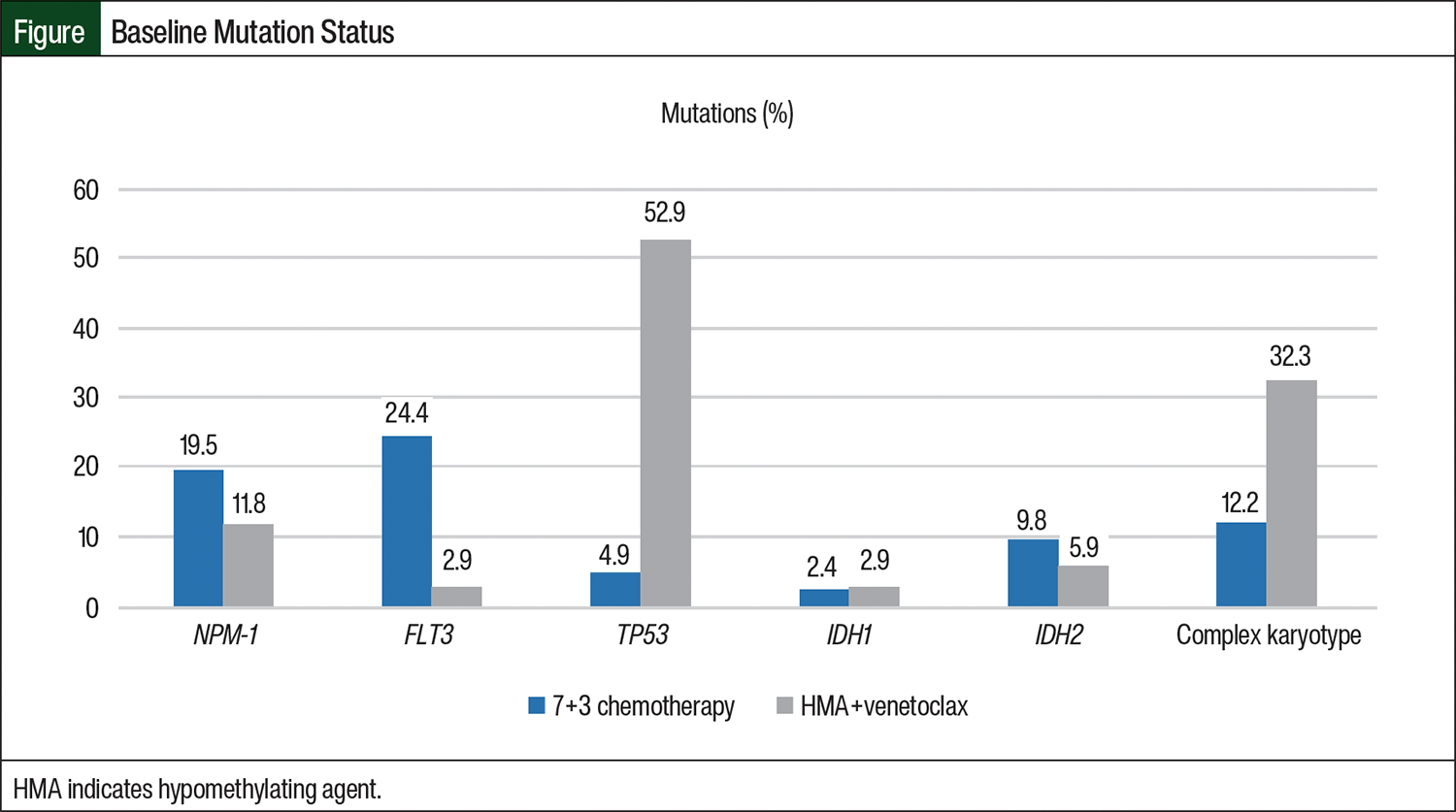
We identified 75 patients with newly diagnosed AML who received 7+3 chemotherapy or HMA+venetoclax induction therapy at our institution and met the study’s inclusion criteria. Of these patients, 41 received 7+3 chemotherapy and 34 received HMA+venetoclax. The baseline patient characteristics are shown in Table 1. There was a significant difference in median age between the 2 groups with the patients who received HMA+venetoclax being older than the patients who received 7+3 chemotherapy (61 years vs 57 years, respectively; P<.001). The patients in the HMA+venetoclax group had significantly more comorbidities, including chronic and severe cardiac history, pulmonary disease, renal disease, and diabetes. The common mutations in patients with AML were evaluated between the groups, as is shown in the Figure. There was significant difference between the groups, with more patients in the 7+3 chemotherapy group having FLT3 mutations and more patients in the HMA+venetoclax group having TP53 mutations or complex karyotype. The odds of receiving HMA+venetoclax and having TP53 mutation were 22 times higher than for patients who did not have this mutation. Other baseline characteristics were not significantly different between the groups.
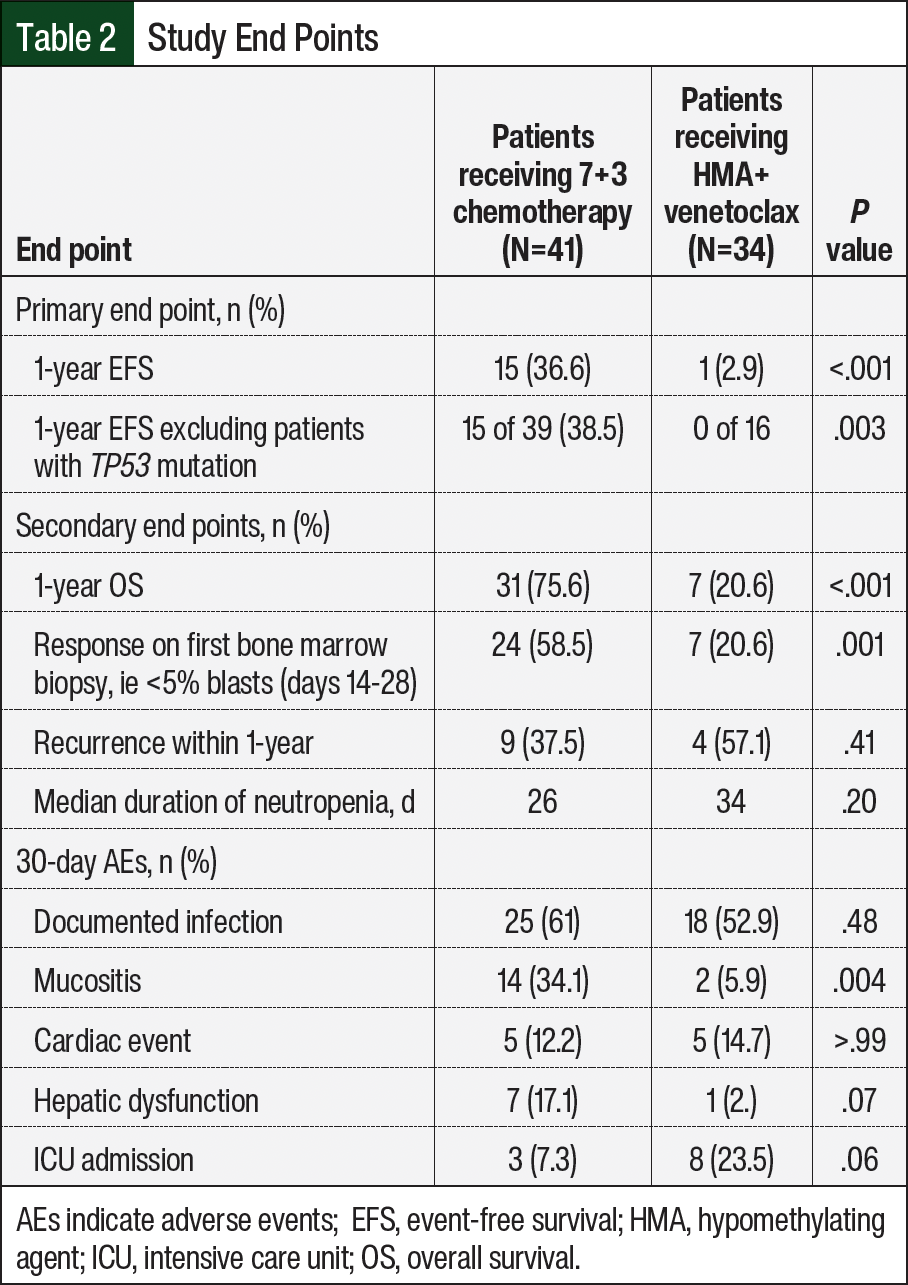
The study’s results are summarized in Table 2. The primary end point of the 1-year EFS rate was significantly different between the groups and favored the 7+3 chemotherapy group over the HMA+venetoclax group (36.6% vs 2.9%, respectively; P<.001). Because of the significantly higher proportion of patients with TP53 mutations in the HMA+venetoclax cohort, a sensitivity analysis was performed with these patients excluded. After the removal of patients with the TP53 mutation, the improved EFS rate in the patients who received 7+3 chemotherapy versus the patients who received HMA+venetoclax remained (38.5% vs 0%, respectively; P=.003).
Each of the 3 components making up the 1-year EFS rate were evaluated individually. The patients receiving 7+3 chemotherapy were more likely to be alive at 1 year compared with those receiving HMA+venetoclax (75.6% vs 20.6%; P<.001) and to have <5% blasts on nadir bone marrow biopsy (58.5% vs 20.6%; P=.001). In those who achieved a morphologic leukemia-free state on the nadir biopsy, the recurrence of disease within 1 year was not significantly different (37.5% vs 57.1%; P=.41). The median duration of neutropenia was slightly longer with HMA+venetoclax (26 days vs 34 days), but this was not statistically significant. Of the 30-day AEs evaluated, mucositis was more prevalent with 7+3 chemotherapy (34.1% vs 5.9%; P=.004). Although a higher proportion of patients receiving HMA+venetoclax had an ICU admission within 30 days of treatment initiation, this difference was not significant (7.3% vs 23.5%; P=.06). Other AEs were not significantly different between the groups (Table 2).
Discussion
When designing this study, a primary goal was to identify practice patterns within our institution to determine what patient characteristics drive treatment selection in this population. Although treatment for younger patients or patients aged >75 years is more well defined, patients aged between 50 years and 70 years can reflect a wide range of fitness for chemotherapy with significant interpatient variability.
This study showed a distinct difference in outcomes for patients aged 50 to 70 years who received each regimen. The primary outcome of 1-year EFS was achieved by 15 (36.6%) patients in the 7+3 chemotherapy group and by only 1 (2.9%) patient in the HMA+venetoclax group. The results with HMA+venetoclax in our real-world analysis of younger, although overall less-fit, patients showed poorer health than expected based on previous results,10,11 with only 20.6% of patients in our study alive at 1 year. The phase 3 VIALE-A study demonstrated an OS of 14.7 months in patients aged >75 years who received azacitidine and venetoclax, with 36.7% of patients achieving complete remission.11 When including patients with complete remission and only partial hematologic recovery (ie, incomplete neutrophil or platelet recovery), 64.7% of patients achieved the end point, and 39.9% reached this end point before the beginning of cycle 2.10
In VIALE-A, the median event-free survival, which was defined as the number of days to disease progression, treatment nonresponse (the inability to achieve complete remission or <5% bone marrow blasts after at least 6 cycles of treatment), confirmed relapse, or death, was 9.8 months.10 In a real-world analysis of intensive chemotherapy for AML by the University of Pennsylvania, patients aged 60 to 75 years receiving 7+3 chemotherapy had a significantly longer OS than the group that received HMA+venetoclax (22 months vs 10 months, respectively; P<.0005), although the researchers noted imbalances between the groups in age, comorbidities, and disease biology that were similar to that in our study.12
A 2021 case series reported the use of HMA+venetoclax in 7 relatively young, but unfit, patients with newly diagnosed AML at a single center in India.13 The patients were deemed unfit for high-intensity chemotherapy because they had neutropenic sepsis at presentation, as well as comorbidities such as additional infectious complications, chronic kidney disease, or heart disease. Although the median age in this series was 45 years, 3 of these 7 patients were aged between 50 years and 60 years. At a median follow-up of 8 months (range, 2-10 months), 4 of the 7 total patients were alive and in remission and 3 patients had died (2 patients’ disease relapsed and they died, and 1 patient died as a result of sudden cardiac death). The researchers noted that although HMA+venetoclax is a feasible treatment option for younger patients who are unfit for traditional 7+3 chemotherapy, the outcomes remain poor and curative intent consolidation therapy should follow in suitable patients.13
Additional challenges in the real-world setting could contribute to worse outcomes compared with clinical trials. For example, although the 7+3 chemotherapy regimen is received entirely in the inpatient setting in a more condensed fashion over 1 week, the administration of HMA+venetoclax may have more opportunities for treatment delays, problems with adherence, and suboptimal management of supportive-care issues after hospital discharge. Patients with secondary AML could also have previously received treatment with HMAs for myelodysplastic syndrome, which could affect their response to HMA+venetoclax. It is also important to consider that in the real-world setting, the treatment decision is rarely as straightforward as clinical trial criteria. Although treatment selection is truly between more-intensive treatment and less-intensive treatment for some patients, treatment selection is often a choice between HMA+venetoclax and supportive care alone for the least-fit patients. Although all patients who received 7+3 chemotherapy in this review were in the former group, the patients in the HMA+venetoclax cohort were likely in the latter group. Although unavoidable, these additional challenges faced in the nontrial setting could potentially contribute to the differences in outcomes demonstrated with HMA+venetoclax versus with the outcomes in earlier trials.
Morphologic leukemia-free state on first bone marrow biopsy was also significantly different between the groups, although notably the patients who received HMA+venetoclax often require more than 1 cycle to achieve complete remission, which was not fully assessed in this study. We evaluated the first bone marrow biopsy in this study in an effort to have a more even comparison of one treatment versus another treatment, because 7+3 chemotherapy reinductions were not included. These findings suggest that patients who are less fit but can potentially tolerate more intensive therapy with additional supportive care may benefit from more aggressive therapy selection.
The rates of recurrence within 1 year were numerically higher in the HMA+venetoclax group than in the 7+3 chemotherapy group at 57.1% versus 37.5%, respectively; however, this was not statistically significant. The patients in this study, particularly in the HMA+venetoclax group, had multiple baseline comorbidities and mutations that are associated with worse outcomes, which likely played a large role in this difference in outcomes. In addition, as a large academic medical center and tertiary referral center, a number of patients were referred to our facility as a result of a high level of acuity and medical complexity, which could have created a population that is predisposed to poorer outcomes compared with historical trials. Ultimately, when broken down by each component making up the primary end point, the significant difference in OS and response on bone marrow biopsy combined with no difference in the rates of recurrence suggest that a lack of remission achieved rather than an inability to stay in remission was the driver of the primary end point.
Perhaps the most notable factor contributing to the difference between the cohorts was the proportion of patients with TP53 mutations in the HMA+venetoclax group compared with the 7+3 chemotherapy group. We postulate that this difference reflects the practice patterns that were in place at the time that HMA+venetoclax was approved. Patients with TP53 mutations have worse overall outcomes and extremely poor prognosis, with disease that is less responsive to chemotherapy.14-16 Intensive traditional treatment regimens such as 7+3 chemotherapy have inadequate results in this patient population; therefore, physicians at our institution favored treatment with HMA+venetoclax in an effort to leverage the potential advantage of venetoclax and its different mechanism of action. As more data became available that showed no real improvement in outcomes, this prescribing preference gradually changed over time.
Currently, providers continue to favor HMA+venetoclax in patients with TP53 mutation to meet the patients’ goals of care and to maximize their quality of life, but this selection is no longer the default, which is a practice change supported by these data. Although outside the scope of this study, alternative treatment strategies such as investigational therapies or clinical trial enrollment may be appropriate in this population as well. TP53 mutation, with its poor survival outcomes and low response rates, continues to affect the population with AML. However, the difference in EFS after the removal of patients with TP53 mutation from this study makes it less likely that this was the sole driver of the differences between the groups.
As expected, the patients’ comorbidities, including chronic cardiac conditions, such as hypertension and congestive heart failure; a history of myocardial infarction or coronary bypass surgery; cerebrovascular disease; diabetes; chronic kidney disease; and pulmonary disorders, such as chronic obstructive pulmonary disease, were significantly more prevalent in the patients who received HMA+venetoclax. Most of these patients had multiple concurrent comorbidities, which presumably reflected a lower overall performance status. In patients with a history of multiple cardiac comorbidities, particularly baseline heart failure, anthracyclines may not be a suitable therapeutic option. In this study, 85% of patients had a baseline left ventricular ejection fraction of >55%; so, although this is a consideration when evaluating therapy options, it was not likely a primary driver in this study. In addition, a liposomal formulation of cytarabine and daunorubicin (CPX-351) was approved in 2017 for patients with therapy-related AML or AML with myelodysplastic changes, which are 2 high-risk subtypes of AML that are associated with poor prognosis.17,18 Our study excluded patients who received CPX-351 as induction therapy, although 19.5% of patients in the 7+3 chemotherapy cohort and 35.3% in the HMA+venetoclax group had secondary AML.
According to a post-hoc analysis of a phase 3 study comparing CPX-351 with 7+3 chemotherapy in patients aged 60 to 75 years with newly diagnosed secondary AML, the patients with adverse-risk AML without TP53 mutations had higher remission rates with CPX-351 than with 7+3 chemotherapy.18 In contrast, for patients who are not candidates for intensive chemotherapy, HMA+venetoclax remains the recommended treatment option for secondary AML. Thus, in this study, the patients with secondary AML who were candidates for more intensive induction therapy were likely excluded from the study, whereas those who were less fit were included in the HMA+venetoclax group.18
Predictive models and algorithms to assist with treatment selection have been proposed. The German AML cooperative group proposed a treatment decision-making algorithm for treatment-naïve, medically fit patients aged ≥60 years based on data from a study of 1406 patients with AML.19 Age, de novo leukemia versus secondary AML, hemoglobin, platelets, fibrinogen, body temperature, and lactate dehydrogenase were significantly associated with complete response or early death.19 Alternative predictive models for early death have suggested that age may reflect other covariates that can more accurately predict poor outcomes than age itself.5 These may include performance status, platelets, secondary AML, peripheral blood blast percentage, and serum creatinine.5 A retrospective study divided patients into 3 categories (fit for intensive chemotherapy, unfit for intensive chemotherapy, or unfit even for nonintensive chemotherapy) based on criteria such as age >75 years, medical comorbidities, active resistant infection, and performance status score of ≥3 not related to leukemia. These categories were independent predictors of survival.20 Although these models suggest more objective factors beyond age that may correlate with treatment outcomes and therefore assist with treatment selection, these models and algorithms are not often used directly in daily clinical practice.
The AEs between the groups were fairly similar, with the exception of mucositis, which was more common with 7+3 chemotherapy. HMA+venetoclax induces prolonged cytopenias even after achieving remission.1,3 In this study, the duration of neutropenia was numerically longer with HMA+venetoclax, but this was not statistically significant. The rates of documented infection, hepatic dysfunction, cardiac event, or admission to an ICU within 30 days of treatment initiation were similar between the groups. Although not significantly different, it is interesting that more patients in the HMA+venetoclax group were admitted to an ICU than in the 7+3 chemotherapy group. This number includes patients who were in the ICU at the time of treatment initiation, which is likely why it is higher than anticipated.
In tenuous patients who already require ICU support (but who do not require intubation), the selection of HMA+venetoclax is a reasonable and potentially more flexible choice because of the variation in organ dysfunction. The similarities in AEs between the groups show that the assessment of patient tolerance for chemotherapy was likely appropriate. However, it may also suggest that select patients may have tolerated more intensive therapy in the setting of additional supportive care and adjustments for AEs. Alternatively, providers could consider if any factors that contribute to poor performance status could be improved via supportive measures before chemotherapy initiation to use a more intensive regimen.
Limitations
This study has several limitations. This study is a single-center, retrospective review relying on the documentation of what were likely nuanced discussions between families and providers. With its relatively small patient population, it is difficult to draw practice-changing conclusions. Significantly larger multicenter reviews would be required to overcome the significant differences in baseline characteristics, such as the prevalence of TP53 mutations in the HMA+venetoclax group, which clearly contributed to the difference in EFS in this study. A response on first bone marrow biopsy, which is typically performed at day 21 for patients receiving HMA+venetoclax, was included as part of our composite primary outcome for 1-year EFS. However, patients may require multiple cycles of HMA+venetoclax to achieve a complete response, which could have partially influenced the components of our composite primary outcome.
The results may have also been skewed by only including patients who received treatment in the inpatient setting, because many fit patients at our institution receive HMA+venetoclax in the outpatient setting. When assessing the fitness of patients, a lack of uniformity in evaluation and documentation made the retrospective collection of performance status difficult. The key comorbidities were collected in an attempt to capture a more complete picture of patient status; however, future prospective trials could use a more systematic method to evaluate the overall fitness of patients for intensive chemotherapy.
In addition, we did not collect information on which patients proceeded to transplant, although with the lack of response and significant comorbidities in the HMA+venetoclax group, it is unlikely that this was a significant portion of that group. Patients who received alternative agents such as mitoxantrone, which may be considered in place of daunorubicin in patients aged >60 years, were excluded from the study, as well as patients who received targeted agents such as midostaurin or quizartinib.1 Despite the exclusion of patients who received targeted agents, a number of patients remained in the study who had targetable mutations but did not receive targeted therapy. Although not entirely clear in retrospect, this may have been because of issues with medication access, as well a result of fewer targeted options being available at the beginning of our study period.
Conclusion
Ultimately, this study showed worse outcomes than expected in patients aged between 50 years and 70 years who received HMA+venetoclax based on earlier clinical trials. Although a multitude of compounding factors play into this result, including guideline-directed treatment algorithms that prefer HMA+venetoclax in patients who may be less fit for intensive therapy, the magnitude of difference between the patients who received HMA+venetoclax and the patients who received 7+3 chemotherapy lead us to question the shift in practice patterns in favor of HMA+venetoclax.
Multiple complex issues go into treatment selection beyond those evaluated in this study, including social factors, such as caregiver support; the length of expected hospital admission; the individual patients’ goals of care; and the ability of the institution to provide more intensive chemotherapy. Although these factors certainly continue to play a role in regimen selection for this difficult population, the results of this study support deep and thorough discussion between the interdisciplinary team and the patient to better identify those patients who could potentially tolerate more intensive chemotherapy with adequate support versus patients for whom the risk continues to outweigh the benefit. Additional large-scale studies are still needed to develop comprehensive predictive models and algorithms to guide treatment in this patient population.
Author Disclosure Statement
Dr Tumlinson, Dr Kennedy, Dr Parish, and Dr Erkanli have no conflicts of interest to report.
References
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Acute Myeloid Leukemia. Version 2.2025. January 27, 2025. Accessed March 25, 2025. www.nccn.org/professionals/physician_gls/pdf/aml.pdf
- US Food and Drug Administration. FDA approves venetoclax in combination for AML in adults. Accessed March 27, 2025. www.fda.gov/drugs/fda-approves-venetoclax-combination-aml-adults
- DiNardo CD, Pratz KW, Letai A, et al. Safety and preliminary efficacy of venetoclax with decitabine or azacitidine in elderly patients with previously untreated acute myeloid leukaemia: a non-randomised, open-label, phase 1b study. Lancet Oncol. 2018;19:216-228.
- DiNardo CD, Pratz K, Pullarkat V, et al. Venetoclax combined with decitabine or azacitidine in treatment-naive, elderly patients with acute myeloid leukemia. Blood. 2019;133:7-17.
- Walter RB, Othus M, Borthakur G, et al. Prediction of early death after induction therapy for newly diagnosed acute myeloid leukemia with pretreatment risk scores: a novel paradigm for treatment assignment. J Clin Oncol. 2011;29:4417-4423.
- Döhner H, Wei AH, Appelbaum FR, et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood. 2022;140:1345-1377.
- Jonas BA, Pollyea DA. How we use venetoclax with hypomethylating agents for the treatment of newly diagnosed patients with acute myeloid leukemia. Leukemia. 2019;33:2795-2804.
- Harris PA, Taylor R, Thielke R, et al. Research electronic data capture (REDCap)–a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377-381.
- Harris PA, Taylor R, Minor BL, et al. The REDCap consortium: building an international community of software partners. J Biomed Inform. 2019;95:103208.
- DiNardo CD, Jonas BA, Pullarkat V, et al. Azacitidine and venetoclax in previously untreated acute myeloid leukemia. N Engl J Med. 2020;383:617-629.
- Pratz KW, Jonas BA, Pullarkat V, et al. Long-term follow-up of VIALE-A: venetoclax and azacitidine in chemotherapy-ineligible untreated acute myeloid leukemia. Am J Hematol. 2024;99:615-624.
- Matthews AH, Perl AE, Luger SM, et al. Real-world effectiveness of intensive chemotherapy with 7&3 versus venetoclax and hypomethylating agent in acute myeloid leukemia. Am J Hematol. 2023;98:1254-1264.
- Senapati J, Dhawan R, Aggarwal M, et al. Venetoclax and azacitidine (VenAZA) combination therapy in young unfit patients with AML: a perspective from a developing country. Leuk Lymphoma. 2021;62:1514-1517.
- Papaemmanuil E, Gerstung M, Bullinger L, et al. Genomic classification and prognosis in acute myeloid leukemia. N Engl J Med. 2016;374:2209-2221.
- Rucker FG, Schlenk RF, Bullinger L, et al. TP53 alterations in acute myeloid leukemia with complex karyotype correlate with specific copy number alterations, monosomal karyotype, and dismal outcome. Blood. 2012;119:2114-2121.
- Stengel A, Kern W, Haferlach T, et al. The impact of TP53 mutations and TP53 deletions on survival varies between AML, ALL, MDS and CLL: an analysis of 3307 cases. Leukemia. 2017;31:705-711.
- Lancet JE, Cortes JE, Hogge DE, et al. Phase 2 trial of CPX-351, a fixed 5:1 molar ratio of cytarabine/daunorubicin, vs cytarabine/daunorubicin in older adults with untreated AML. Blood. 2014;123:3239-3246.
- Lancet JE, Uy GL, Cortes JE, et al. CPX-351 (cytarabine and daunorubicin) liposome for injection versus conventional cytarabine plus daunorubicin in older patients with newly diagnosed secondary acute myeloid leukemia. J Clin Oncol. 2018;36:2684-2692.
- Krug U, Röllig C, Koschmieder A, et al. Complete remission and early death after intensive chemotherapy in patients aged 60 years or older with acute myeloid leukaemia: a web-based application for prediction of outcomes. Lancet. 2010;376:2000-2008.
- Borlenghi E, Pagani C, Zappasodi P, et al. Validation of the “fitness criteria” for the treatment of older patients with acute myeloid leukemia: a multicenter study on a series of 699 patients by the Network Rete Ematologica Lombarda (REL). J Geriatr Oncol. 2021;12:550-556.