Connecting Science to Practice
Automated dose rounding to the nearest 10% was piloted
on 3 biologic chemotherapy agents, bevacizumab, bevacizumab-
awwb, and paclitaxel protein-bound, which were selected
based on high cost and high usage. Cost-savings was calculated
using drug acquisition cost and number of vials
saved by dose rounding down to the nearest vial. Automated
dose rounding was found to contribute to significant costsavings
and waste avoidance while improving pharmacist
verification efficiency and technician compounding accuracy.
These improvements contribute to more consistent practices
and alignment with Hematology/Oncology Pharmacy
Association recommendations.
Spending on cancer medications has had a significant effect on direct patient costs, hospital expenditures, and healthcare as a whole. In 2022, global expenditures on cancer medicine increased by 4% to $196 billion and are projected to climb to $375 billion by 2027.1 A large contribution to this increase is the introduction of 100 new treatments with the shift from traditional chemotherapy agents to new biologic and targeted agents.2 The addition of the biologic agents bevacizumab and panitumumab to the traditional mFOLFOX treatment regimen for breast cancer has led to 213% and 357% increases in cost, respectively.3 These growths in cost and spending are propelled by ongoing innovation and are counterbalanced by the increasing adoption of biosimilars in key markets.
Biosimilars for 3 oncology drugs—bevacizumab, rituximab, and trastuzumab—were introduced in 2019 in the United States.1,4 The adoption of oncology biosimilars has resulted in savings of $5.5 billion in 2022 and a total savings of $12 billion over the past 5 years.1 More than half of the savings in the United States contributes to cost reductions for patients and healthcare systems, while also broadening access to medications.2 In 2022, the median annual cost for new oncology medications reached $260,000, marking a significant increase from $63,534 a decade ago.1 This surge is largely attributed to novel modalities tailored for smaller patient populations, which have become predominant in recent launches.1 Although the expense of specific drugs may be substantial, the overall cost of a patient’s treatment remains subject to variations that are determined by the comprehensive regimens administered, nondrug aspects of their care, and, notably, insurance coverage.1
In an effort to promote the reduction of healthcare expenditures, minimize drug waste, and promote accuracy during drug preparation, the Hematology/Oncology Pharmacy Association (HOPA) issued a position statement recommending that chemotherapy doses be rounded to within 10% of the prescribed dose.5 This statement has been reviewed and endorsed by the International Society of Oncology Pharmacy Practitioners and the National Comprehensive Cancer Network. The rationale for recommending dose rounding within 10% of the prescribed dose is based on the principle that this practice will not negatively affect the safety or effectiveness of the drug therapy. In congruence with this 2018 HOPA recommendation,5 our organization updated our chemotherapy policy to permit pharmacists to manually round non-institutional review board–supplied chemotherapy doses within 10% of the ordered dose for adults and pediatric patients to avoid drug waste.
In recent years, many organizations have successfully implemented automated dose-rounding algorithms within their electronic health record (EHR). The Mayo Clinic shared its process for developing and implementing dose-rounding rules and the cost-savings afforded 6 months after implementation.6 Dose rounding was automated to round within 10% or to a convenient measurable volume. The Mayo Clinic’s initial implementation included 91 oncology drugs that were categorized as biologics, oncolytics, or miscellaneous. From January 2019 to June 2019, there were 9814 vials saved, for a total cost-savings of $7.3 million. Rituximab and paclitaxel protein-bound had the highest cost-savings at $1.32 million and $340,541, respectively.6
After the publication of its original data, the Mayo Clinic published a 3-year analysis of cost-savings that resulted from automated dose rounding and drug waste prevention.7 These new data showed that from 2019 to 2021, 36.1% of doses were rounded down, 35.8% were rounded up, and 28.1% remained unrounded. The automated dose rounding resulted in the saving of more than 62,000 vials of medication. The cost-savings totaled $39.75 million, with $31.92 million attributed to biologic agents. Among the oncolytic agents, paclitaxel protein-bound attributed to the highest cost-savings, which totaled $2.05 million. The cost associated with waste avoidance, which was rounded up to use the entire vial, totaled $9.95 million.7
Dela-Pena and colleagues at Michigan Medicine implemented a similar dose-rounding initiative, for which they acquired approximately $3.6 million using wholesale acquisition cost (WAC) pricing.8 They separated their results by medication as well as by inpatient and outpatient cost-savings.8 Fahey and colleagues at UW Health demonstrated the benefit of automated dose rounding in terms of cost-savings compared with manual dose rounding, as well as medication safety improvements, in an approximately 3-month period.9 The initial implementation included the 3 medications bevacizumab, rituximab, and trastuzumab, in which they configured dose-rounding rules to round doses to 5% or 10%. Before the implementation of automated dose rounding, the results showed that only 36.2% of doses were manually rounded during pharmacist verification. Postimplementation of automated dose rounding, 100% of administrations were automatically dose rounded, which saved pharmacists at least 9297 additional clicks and 11,363 additional keystrokes. Using WAC pricing, manual dose rounding and automated dose rounding translated to cost-savings of $8420 and $224,746, respectively. Dose rounding led to cost-savings of $56,631 for bevacizumab, $126,027 for rituximab, and $42,088 for trastuzumab.9
The purpose of this study was to demonstrate the effect of a piloted automated chemotherapy dose-rounding program on reducing drug waste and achieving cost-savings at an academic medical center while providing an in-depth discussion of difficulties faced and addressed during the piloting phase. The chemotherapy infusion pharmacy within the academic medical center prepares chemotherapy medication for 57 infusion bays. Within the past year, the chemotherapy infusion pharmacy compounded more than 14,000 medication orders per month, 2000 of which were chemotherapy medications.
Methods
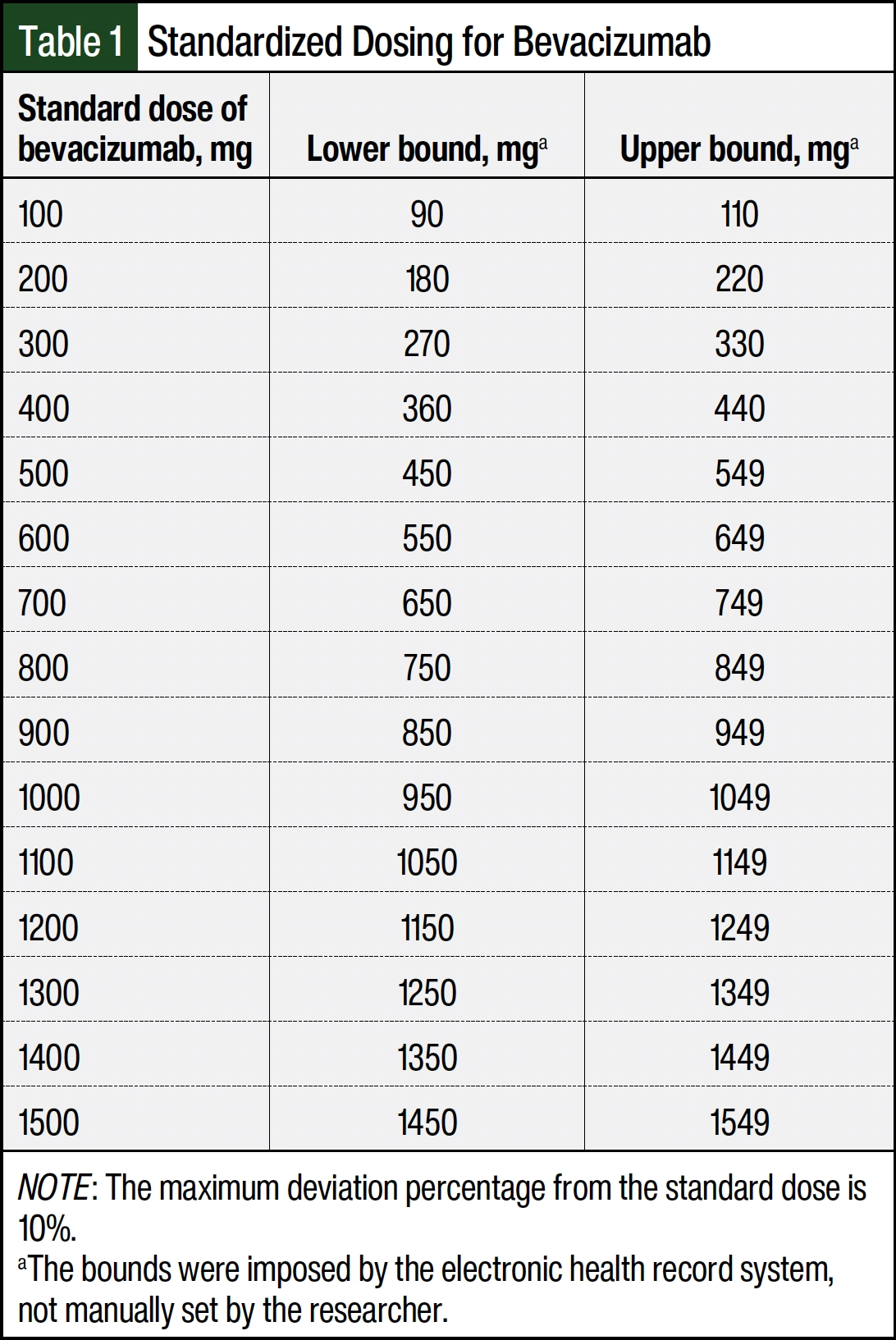
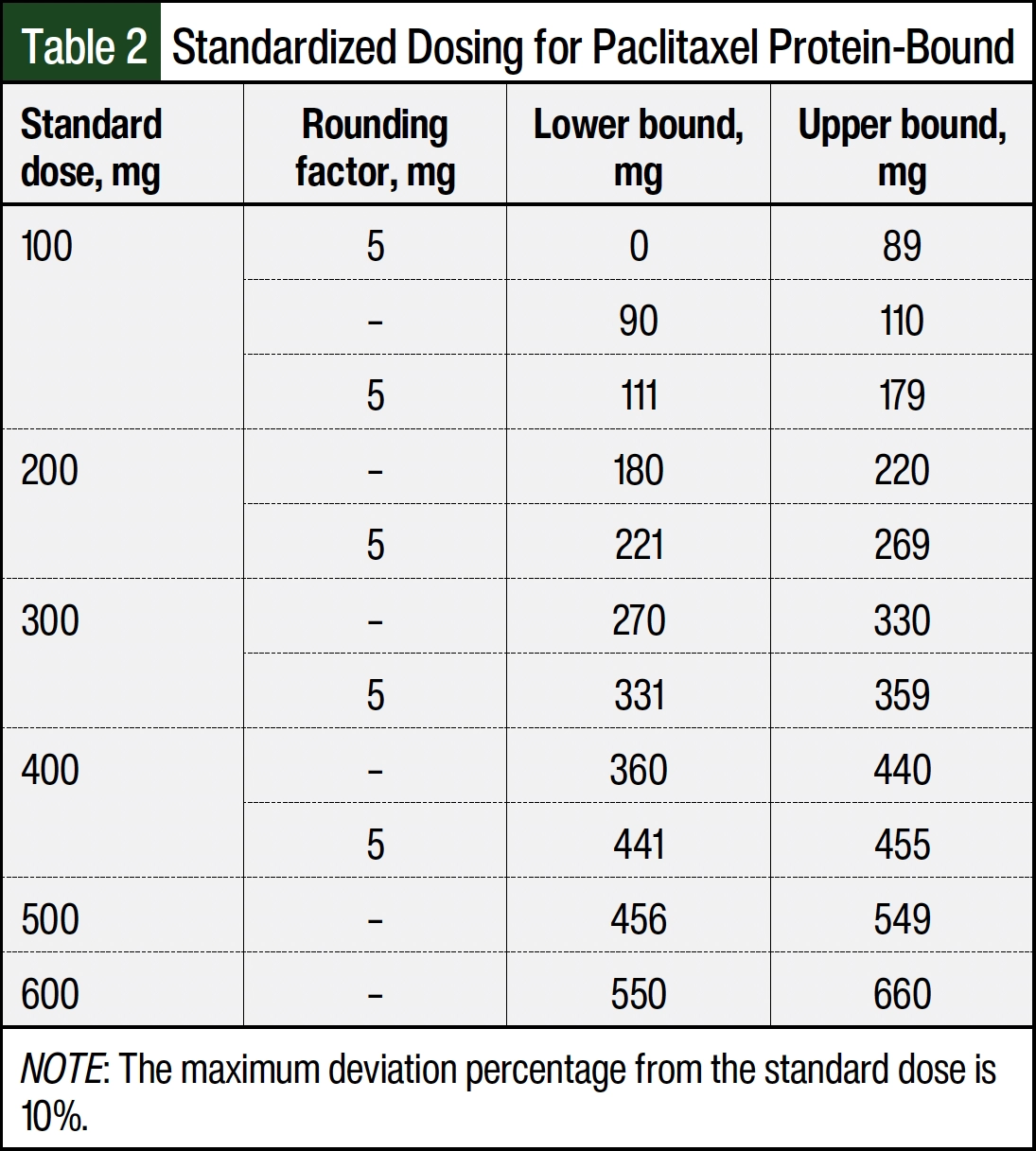
This is a single-center, retrospective chart review, quality improvement study performed within a chemotherapy infusion pharmacy located in an academic medical center from July 5, 2023, to October 4, 2023. Before the implementation of automated dose rounding, an analysis was performed to determine the high-cost medications that would most benefit from automated dose rounding. The pilot medications that were selected for inclusion in this study included paclitaxel protein-bound, bevacizumab, and bevacizumab-awwb based on their high cost and frequent use. Within the chemotherapy infusion pharmacy, the average number of doses compounded monthly were 29 for paclitaxel protein-bound, 3 for bevacizumab, and 42 for bevacizumab-awwb. Bevacizumab is available as 100-mg/4-mL and 400-mg/16-mL single-dose vials. Paclitaxel protein-bound is available as a 100-mg single-dose vial. Medication-specific dose-rounding algorithms (Table 1 and Table 2) were created and were agreed on by the interdisciplinary Oncology Drug Safety Committee.
In general, dose-rounding algorithms round doses that fall within 10% of the nearest vial size based on the recommendation from the 2018 HOPA Position Statement.5 Bevacizumab-awwb doses were rounded to the nearest 100 mg within 10% of the ordered dose. All doses in which rounding was not within 10% of the ordered dose retained their original ordered dose. Similarly, paclitaxel protein-bound doses were rounded to the nearest 100 mg.
Additional dose-rounding strategies were applied to round doses that were not within 10% to a convenient measurable dose, 5 mg, based on input from the hematology/oncology pharmacists and the infusion pharmacy team with approval from the Oncology Drug Safety Committee. The dose-rounding algorithms were incorporated into the Rx Standardized Dosing table within the Epic medication master file. For bevacizumab and bevacizumab-awwb, the dose-rounding algorithm was applied to the bevacizumab orderable parent record. At order entry and order verification, the rounded dose and the original calculated dose are displayed, allowing the provider or pharmacist to adjust the dose if necessary.
Automated dose rounding requires a newly placed medication order after implementation that has complex dosing (mg/kg, mg/m2). The dose will be calculated based on the dosing weight within the chemotherapy treatment plan. Although a new weight is obtained at each treatment and cycle, the dose will continue to be based on the initial dosing weight on the treatment plan entry. The go-live date of the dose-rounding algorithms was July 5, 2023, which we consider the start of our postimplementation period for this study.
The EHR data were extracted for a 3-month postimplementation period of July 5, 2023, to October 4, 2023. The medication order data that were extracted included the order number, order name, administration date, administration time, rounded dose, and ordered dose. The patients’ demographics that were extracted included age, sex, race, ethnicity, and insurance. To assess if a dose was automatically rounded, the original ordered dose was subtracted from the rounded dose. The orders in which this calculation did not equal zero were labeled as automatically rounded. All orders for bevacizumab, bevacizumab-awwb, and paclitaxel protein-bound that were compounded by the chemotherapy infusion pharmacy and underwent dose rounding via the applied dose-rounding algorithm were included in this study. All other medication orders and orders that were not automatically rounded via the dose-rounding algorithm within the EHR were excluded from this study. Based on institutional policy, sponsor-supplied medications were excluded from dose rounding.
The drug acquisition cost of the nearest commercial vial was used to perform the cost analysis in this study. The cost analysis was only performed on orders for which the dose was automatically rounded down to the nearest vial, thereby saving at least 1 full vial. The number of vials saved was calculated by subtracting the original calculated dose from the rounded dose and then dividing this by the nearest vial size. For example, if there is an ordered dose of 320 mg, the automated rounding will round the dose down to 300 mg, thereby preventing the use of 1 extra 100-mg vial. The cost-savings was calculated by multiplying the number of vials saved by the acquisition cost of that vial. Doses that were not rounded to the nearest vial were excluded from the cost-savings analysis.
During the postimplementation phase, we received multiple reports of dose rounding of >10% based on the patient’s weight collected on the day of treatment. We learned that within the organization’s EHR settings, the calculated chemotherapy dose is based on the initial treatment plan dosing weight, and the system then automatically rounds the dose within 10% of that ordered dose. As such, the same height and weight is carried forth each cycle as well as the same rounded dose. To address this situation, we dispersed a Situation, Background, Assessment, and Recommendation (SBAR) document to our pharmacy and nursing teams. The recommendation was for the nurse to notify the pharmacist if a rounded dose exceeded the 10% rule based on the new weight and body surface area (BSA) but the patient’s actual weight and BSA are within 10% of the treatment plan weight and BSA. The pharmacist would then assess and change the dose on verification, as appropriate, to meet the 10% rule.
The primary end point of this study was the estimated cost-savings based on drug vials saved as a result of automated dose rounding down to the nearest vial size. The secondary end point was drug waste prevented from rounding up to the nearest whole vial. This study received a formal Determination of Quality Improvement status according to the organization’s institutional policy. As such, this initiative was deemed as not being research on human subjects and was therefore not reviewed by the institutional review board.
Results
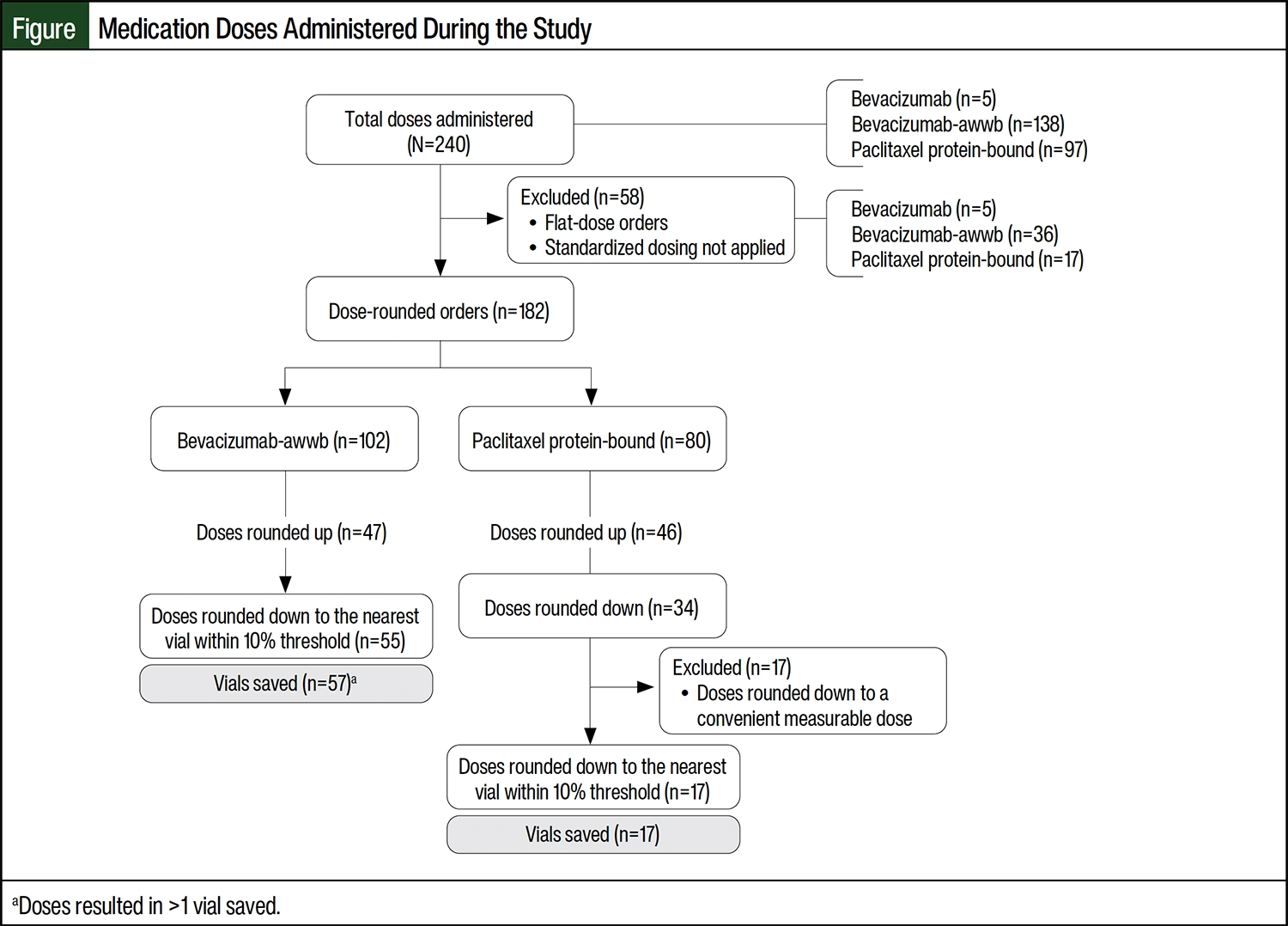
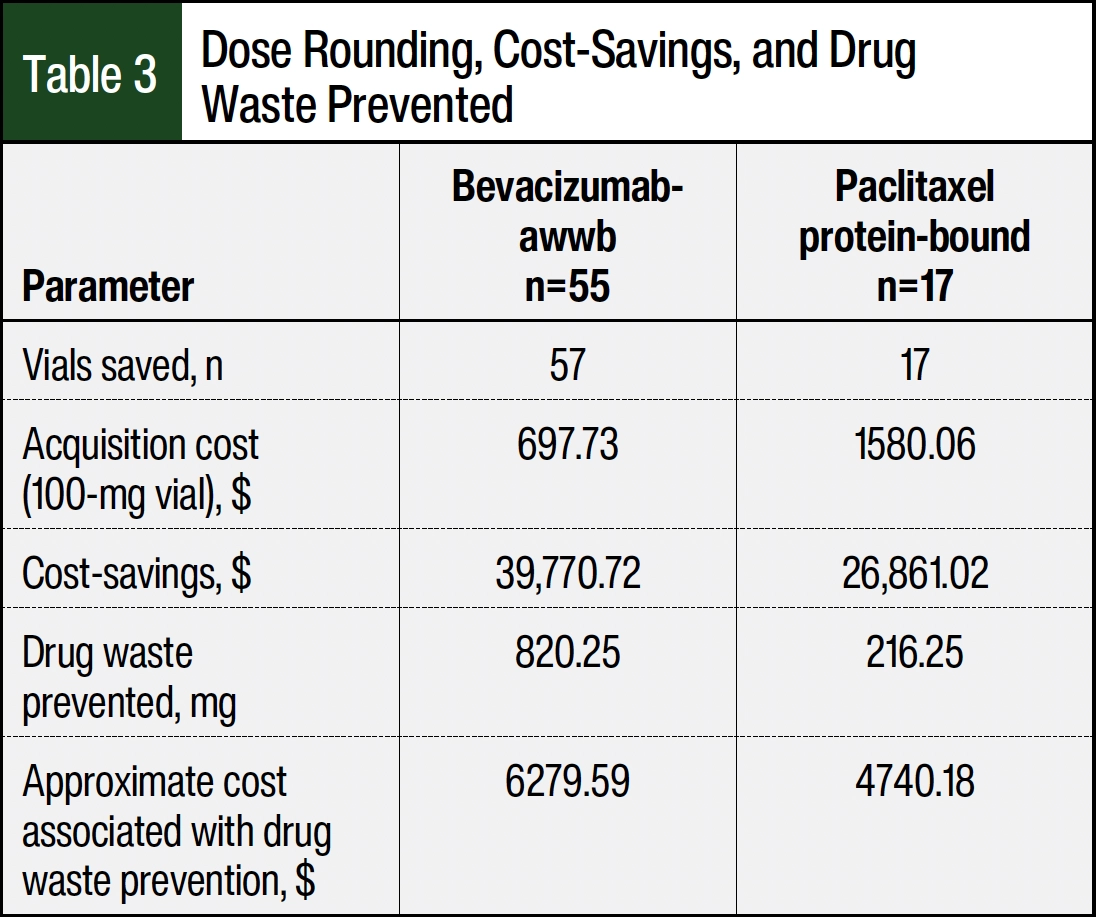
From July 5, 2023, to October 4, 2023, there were 240 doses of the study medications compounded in a chemotherapy infusion pharmacy. All medications in this study were compounded within the time of analysis. Of the 240 orders compounded, 58 were excluded from the study because they were entered as flat-dose orders or doses that did not undergo automated dose rounding (Figure). The 58 orders that were excluded included all 5 of the bevacizumab orders because they did not undergo automated dose rounding. The remaining 182 (75.8%) doses were automatically dose rounded, with a percent change ranging from 0.2% to 8.2%. Bevacizumab-awwb accounted for 102 (56%) administrations, 55 (53.9%) of which the dose was automatically rounded down to the nearest vial size, accounting for 57 vials saved. Paclitaxel protein-bound accounted for 80 (44%) administrations, 34 (42.5%) of which were rounded down to the nearest vial size, each accounting for 1 vial saved. Of the doses rounded down, 17 were rounded to a convenient measurable dose, which was set as the nearest 5 mg, rather than rounded to the nearest whole vial; therefore, these were excluded from the cost analysis. Using acquisition cost, the total cost-savings as a result of automatic rounding down of doses to the nearest vial size was $66,631.74. Table 3 presents the amount saved per drug. The extrapolated estimated annual cost-savings is $266,526.98.
Of the 102 doses of bevacizumab-awwb that were rounded, 47 (46.1%) doses were rounded up to the nearest whole vial, accounting for 820.25 mg of drug waste prevented, or approximately 9 vials (100-mg vials). Of the 80 doses of paclitaxel protein-bound that were rounded, 46 (57.5%) doses were rounded up to the nearest whole vial, accounting for 255.70 mg of drug waste prevented, or approximately 3 vials (100-mg vials). The total savings from drug waste prevented was approximately $11,019.77.
Discussion
The implementation of automated dose rounding for 2 chemotherapy medications over 3 months provided cost-savings of approximately $67,000 based on doses that were rounded down. Extrapolated over a 1-year period, the estimated cost-savings would be approximately $267,000. In the implemented dose rounding, we also obtained drug waste prevention as a result of the doses being rounded up to the nearest vial. This prevented drug waste led to an approximate $11,000 cost-savings. In our study, we only calculated the cost-savings for vials saved from doses that were rounded down a vial and thereby prevented an extra vial from being opened. This is important to note, because our paclitaxel protein-bound dose-rounding algorithm included a rounding factor of 5 mg for doses that were not 10% of the standard doses. The doses that were only rounded to the nearest 5 mg were not included in our cost analysis, although it has an effect on workflow efficiencies for our compounding technicians, including more safe and accurate compounding volumes.
Our findings align with those of previous studies that have investigated the cost-savings associated with implementing automated dose rounding on chemotherapy medications. Because of the high-cost nature of these medications, our pilot study aimed to target those with the greatest impact, namely paclitaxel protein-bound and bevacizumab. Within our 3-month period, we were able to observe similar cost-savings and the avoidance of drug waste.
The implementation of this study served as a pilot for the capabilities and financial benefits of automated dose rounding while also providing a better understanding of limitations and considerations as we expand the implementation of automated dose rounding to other high-cost medications in our organization. Through this study, we showed areas in our system that may hinder the safe implementation of automated dose rounding. We showed limitations of and considerations for how to implement automated dose rounding based on the type of medication record, such as simple versus orderable. To promote change and foster trust in automated dose rounding, we found it imperative to understand and address challenges as they arose.
Safety is our primary goal because we wish to ensure our efforts do not negate serving our patients. As previously mentioned, an SBAR instruction on how nursing and pharmacy teams were to manage changes in weight and BSA and dose rounding was put in place. This process entails the nurse releasing the order, being notified by the EHR system that the rounded dose exceeds the 10% threshold, then notifying the pharmacy staff for an assessment during order verification. At that time, the pharmacist will assess and update the dose manually if deemed appropriate. We understand that the process described here negates the workflow efficiencies gained by automated dose rounding, but the organization plans to discuss the implications of updating the EHR settings to recalculate doses each time a new weight or height is documented. Of note, any manual dose changes by the pharmacist did not affect the results of this study, because we only assessed the doses that underwent automated standardized dosing.
Another aspect of this study that was met with challenges was the difference between automated dose rounding and rounding decisions made by a human doing manual calculations. In institutional practice, a pharmacist may not round doses that are exactly 10%, or even 9%. In our study, we assessed the percent change of our doses rounded to assess for extremes made by the automated dose rounding. The results show a change in the range of 0.2% to 8.2%, thereby more likely aligning with decisions that a pharmacist may make for the same doses. The goal was to assess the risk and concern for increasing adverse events by rounding to a higher dose, while balancing the notion that patients may not be receiving an adequate dose if the dose is rounded down to the nearest vial size. It is important to note that as we reach higher doses of >500 mg, the 10% of the standard doses begin to overlap.
Table 1 depicts how the EHR system interpreted and set the dosing bounds. The standard dose column lists the doses in increments of 100 mg and sets the maximum deviation percentage from standard dose to 10%. For doses of >500 mg in which the standard dose bounds overlapped, the method in which the pharmacist would round did not always align with how the system rounded. When we encountered these situations, we re-engaged our pharmacy stakeholders to devise the agreed-on bounds for each standardized dose. This allowed the pharmacists to be more confident about the system’s automated dose rounding because it better aligned with their clinical judgment.
We would be remiss if we did not discuss the lack of inclusion of our third agent in this study, bevacizumab. Midway through the study, we were notified by our pharmacy stakeholders that bevacizumab dose rounding was not functioning as expected. The main concern was the insurance restrictions for waste billing for bevacizumab and bevacizumab-awwb. When implementing automated dose rounding for bevacizumab drugs, we enacted standardized dosing on the orderable record to ensure that all mixtures it encompasses will incur automated dose rounding. On investigation, we learned that our orderable record did not have criteria to instruct the system when to select the second medication listed, which was bevacizumab in this case. Therefore, standardized dosing only applied to the first medication listed, bevacizumab-awwb. To resolve this issue, we added the standardized dosing to the main ingredient medication record, so any mixture records using this main ingredient will incur automated dose rounding. This update was made outside of the time frame of this pilot study; therefore, bevacizumab was not included in this study’s results.
The organization plans to continue the implementation of automated dose rounding to other high-cost biologics and chemotherapy medications, as listed in Appendix A. We will use lessons learned through this pilot toward the remaining implementations and continue to engage our frontline staff throughout the process.
As we consider generalizability, we recognize that not every organization encounters waste drug billing and those that do may not obtain the same reimbursement. We included automated rounding up to the nearest vial size to minimize drug waste and to simplify the cumbersome process of waste drug billing, which can be complicated by payer type and billing codes. Often, dose rounding is associated only with rounding down to the nearest vial size, so having automated dose rounding in both directions helps to ensure consistency within practices without compromising efficacy or safety. The aim of this study is to reduce drug waste and demonstrate cost-savings while sharing obstacles and lessons learned through this process.
Conclusion
Automated dose rounding has a significant effect on cost-savings and drug waste prevention when applied to high-cost chemotherapy medications. There is also a benefit to pharmacist workflow efficiencies because automated dose rounding removes the manual rounding process. By rounding to a convenient measurable dose, workflow efficiencies were gained for compounding technicians. This pilot study has allowed us to become more vigilant as we work to expand the implementation of automated dose rounding on additional chemotherapy medications and other high-cost biologics throughout our health system.
As we review the lessons learned through this pilot study, the major considerations we derived included the determination of which medication record in the EHR to apply automated rounding rules to; the mixture record versus the vial record, based on which will affect the appropriate orders; the EHR settings for treatment dosing weight, which may affect the appropriateness of the new automated dose; and the significance of stakeholder engagement throughout implementation, as well as postimplementation, because this ensures real-time feedback and problem resolution and fosters trust in the new process. We recommend devising standardized testing scenarios for each medication to which automated rounding rules will be applied. Testing should be completed by the pharmacy informatics team as well as oncology pharmacy specialists and oncology nurses to ensure that each step of the treatment process is considered. We continue to work with our pharmacy and nursing stakeholders to ensure that any EHR updates are appropriate and safe for patients and to encourage the frontline staff to report challenges within the system.
Author Disclosure Statement
Dr Davis, Dr Zhang, and Dr Moser have no conflicts of interest to report.
References
- Aitken M, Kleinrock M, Connelly N, Pritchett J. Global Oncology Trends 2023: Outlook to 2027. IQVIA Institute for Human Data Science; 2023.
- Aitken M, Kleinrock M, Pritchett J. Global Use of Medicines 2024: Outlook to 2028. IQVIA Institute for Human Data Science; 2024.
- Nadeem H, Jayakrishnan TT, Rajeev R, et al. Cost differential of chemotherapy for solid tumors. J Oncol Pract. 2016;12:251, e299-e307.
- Aitken M, Kleinrock M, Pritchett J. Biosimilars in the United States 2023-2027: Competition, Savings, and Sustainability. IQVIA Institute for Human Data Science; 2023.
- Fahrenbruch R, Kintzel P, Bott AM, et al. Dose rounding of biologic and cytotoxic anticancer agents: a position statement of the Hematology/Oncology Pharmacy Association. J Oncol Pract. 2018;14:159, e130-e136.
- Shah V, Spence A, Bartels T, et al. Decreasing drug waste, reducing drug costs, and improving workflow efficiency through the implementation of automated chemotherapy dose rounding rules in the electronic health record system. Am J Health Syst Pharm. 2022;79:676-682.
- Shah VS, Irvine C, McWilliams RR, et al. Reducing cancer drug cost: 3-year analysis of automated dose rounding in electronic health records. JCO Oncol Pract. 2024:OP2300688.
- Dela-Pena JC, Eschenburg KA, LaRocca VW, et al. Financial impact of an automated oncology dose-rounding initiative: one-year analysis. JCO Clin Cancer Inform. 2021;5:805-810.
- Fahey OG, Koth SM, Bergsbaken JJ, et al. Automated parenteral chemotherapy dose-banding to improve patient safety and decrease drug costs. J Oncol Pharm Pract. 2020;26:345-350.