Connecting Science to Practice
This retrospective study evaluated whether patients with
multiple myeloma (MM) who were receiving immunomodulatory
drugs (IMiDs) were on appropriate anticoagulation
based on the SAVED score calculations. Patients were stratified
into those filling prescriptions at a health-system specialty
pharmacy and those filling prescriptions at a non–health-system specialty pharmacy. Secondary outcomes
included the number of emergency department visits related
to anticoagulation and the number of pharmacist interventions
related to anticoagulation. In calculating SAVED
scores for patients with MM who are receiving IMiDs and
evaluating the appropriateness of anticoagulation therapy,
pharmacists apply scientific risk assessment tools directly to
patient care in everyday practice, which unites evidence-based
approaches with individualized clinical decision-making to
improve patient outcomes.
Multiple myeloma (MM) is a malignant cancer forming in plasma cells, which then accumulate in the bone marrow and lead to bone destruction and marrow failure. The median age of patients at diagnosis is approximately 66 to 70 years.1 Patients presenting with active (symptomatic) MM are initially treated with primary therapy, followed by high-dose chemotherapy with autologous hematopoietic cell transplant (in transplant-eligible patients), and subsequently by maintenance therapy.2 Patients with newly diagnosed MM are typically responsive to a variety of classes of drugs, including immunomodulatory drugs (IMiDs), proteasome inhibitors, and monoclonal antibodies.
Patients with MM have a 9-fold increase in developing venous thromboembolism (VTE) when compared with the general population.3 The development of VTE is typically seen within the first 6 months of diagnosis,2 regardless of the treatment regimen. VTE is the second highest medical complication and the second highest cause of extended length of stay in hospitals.4 A meta-analysis showed that the cost per pulmonary embolism hospitalization increased from $5198 to $6928 in 2000 to $8764 in 2010.4 In addition, yearly healthcare plan expenditures for VTE-related services also increased from $10,804 to $16,644 during the 1998 to 2004 period to an average cost of $15,123 for a VTE event from 2008 to 2011.4
IMiDs are frequently prescribed as part of the induction and maintenance therapy regimen. This class of medications includes oral drugs such as thalidomide, lenalidomide, and pomalidomide. Because of the increased risk for teratogenic effects, these medications are administered through Risk Evaluation and Mitigation Strategy (REMS) programs and are only available through a select network of specialty pharmacies.
A specialty pharmacy model focuses on the overall care of patients with complex disease states. Only specialty pharmacies that are contracted and certified with the REMS program are able to dispense IMiD prescriptions. Clinical pharmacists working in specialty pharmacies assess for appropriateness of therapy, make clinical recommendations, educate patients on their specialty medications, monitor patients over the course of therapy, and complete REMS requirements, such as counseling patients.
The NCCN published updated recommendations detailing anticoagulation recommendations based on calculating IMPEDE and SAVED scores.2 These guidelines recommend pharmacologic thromboprophylaxis in patients with MM who are receiving IMiD therapy. According to Sanfilippo and colleagues, the IMPEDE VTE score outperformed previous International Myeloma Working Group/NCCN VTE prophylaxis guidelines because it also considers comorbid conditions, medical history, and concurrent therapies.3 In contrast, Li and colleagues concluded that the SAVED score outperformed previous NCCN guidelines in risk stratification of patients with MM receiving IMiD therapy.5 Use of either the IMPEDE or SAVED scores is currently considered an NCCN guideline 2A level strength recommendation.2 Given that either one of these scores can be used to assess appropriate anticoagulation therapy in patients with MM before the start of therapy, the SAVED score offers a more user-friendly approach to calculations in a specialty pharmacy setting.
The literature is limited regarding studies that assess the role of clinical specialty pharmacists in the prevention of thromboembolism in patients with MM receiving IMiD therapy. Therefore, the objective of this study was to compare utilization of NCCN guidelines and the SAVED score in the management of VTE in patients with MM receiving IMiDs from a health-system specialty pharmacy (HSSP) versus non-HSSPs. One of the key differences between an HSSP and a non-HSSP is that patient information and communication with providers is less accessible for a non-HSSP. This retrospective evaluation assessed appropriate anticoagulation use based on SAVED scores in patients diagnosed with MM. Inappropriate anticoagulation is defined as no anticoagulation or a discrepancy between the SAVED score and the anticoagulation therapy. Additional outcomes included the number of relevant emergency department visits related to thromboembolic events and the number of pharmacist interventions associated with the prevention of VTE.
Methods
This is a retrospective evaluation that was conducted at a large HSSP. Patients who received IMiD therapies were identified using reports from the pharmacy’s case management program and the electronic health record. A medication use evaluation was performed during the study period (January 1, 2021, to July 31, 2022). Data collected included demographic data, previous VTE history, surgical history, emergency department visits, emergency department costs, and pharmacist interventions. Patients were then classified into 2 groups according to where they filled their IMiD prescription (HSSP or non-HSSP). Patients were included in the study if the following inclusion criteria were met: aged ≥18 years, diagnosed with MM, and prescribed continuous IMiD therapy (thalidomide, lenalidomide, pomalidomide) for ≥1 months during the study period. Patients with contraindications to anticoagulants as outlined in the NCCN guidelines were excluded from the study.
SAVED score was calculated for all patients following a complete clinical review as follows2: add 2 points for surgery within 90 days, add 1 point for age ≥80 years, add 3 points for VTE history, add 1 point for standard dexamethasone dose (120-160 mg/cycle), add 2 points for high dexamethasone dose (≥160 mg/cycle), and subtract 3 points for Asian race. Appropriate anticoagulation for patients with a score <2 is 81 to 325 mg of aspirin daily.2 Appropriate anticoagulation for patients with a SAVED score ≥2 is one of the following: low-molecular-weight heparins (equivalent to enoxaparin 40 mg daily), rivaroxaban 10 mg daily, apixaban 2.5 mg twice daily, fondaparinux 2.5 mg daily, or warfarin (target international normalized ratio, 2.0-3.0).2 SAVED score calculation was also based on consideration of general principles listed in the NCCN guidelines that account for other patient-specific factors (eg, atrial fibrillation, cytopenias, renal function, concurrent medications).2
The primary outcome of this study was to determine the number of patients undergoing IMiD therapy who were on appropriate anticoagulation based on NCCN guidelines and the SAVED score. Secondary outcomes included the number of emergency department visits for anything related to anticoagulation and the number of pharmacist interventions involving anticoagulation therapy. The cost of the emergency department visit was provided by the health system’s finance department.
In terms of statistical analyses, goal sample size was set to 100 patients so that the study was powered to 80%. The P value was set to <.05% to determine the statistical significance, and various statistical tests were performed for baseline demographic data. The Kruskal-Wallis test was used to compare patient age between the 2 groups, and the chi-square test was used to compare race and sex. For the primary and secondary outcomes of interest, chi-square statistical tests were conducted.
Results
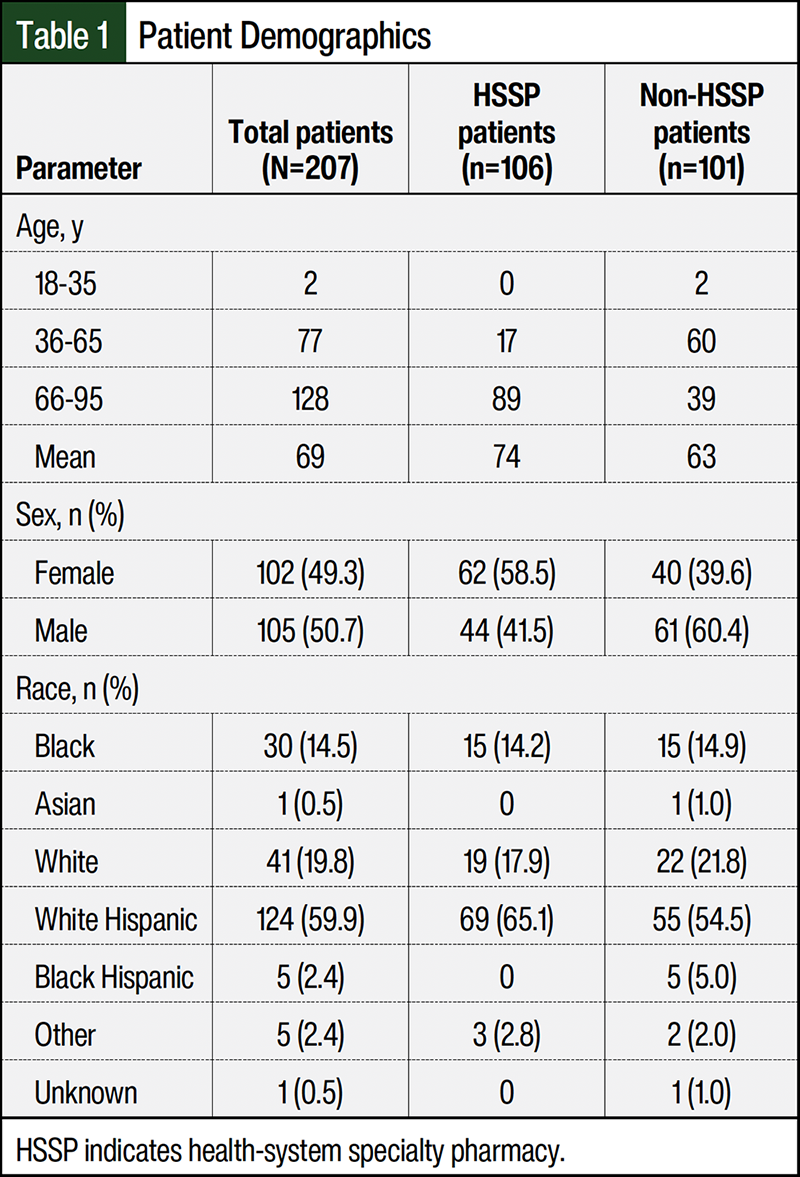
A total of 207 patients met the inclusion criteria. Of these, 106 patients received IMiD therapy from an HSSP, and 101 patients received IMiD therapy from non-HSSPs. Baseline characteristics across study groups are outlined in Table 1, and characteristics were similarly distributed across the 2 groups except age. The mean age for the overall study population was 69 years. A significant difference was seen in median age among the HSSP and the non-HSSPs groups: 74 (range, 68-78) in the HSSP group versus 63 (range, 57-69) in the non-HSSP group (P<.001). In total, 62 (58.5%) patients in the HSSP group were women compared with 40 (39.6%) women in the non-HSSP group.
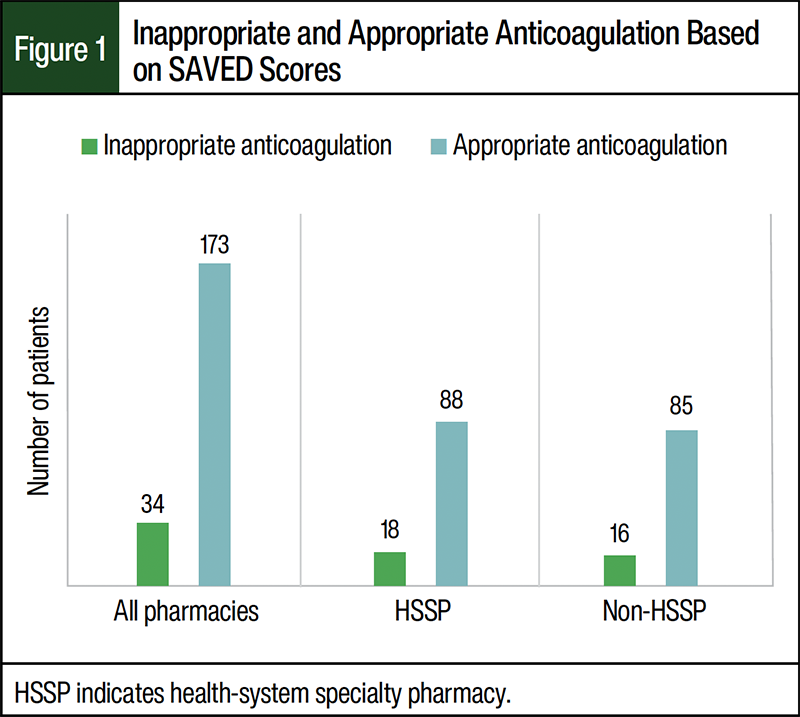
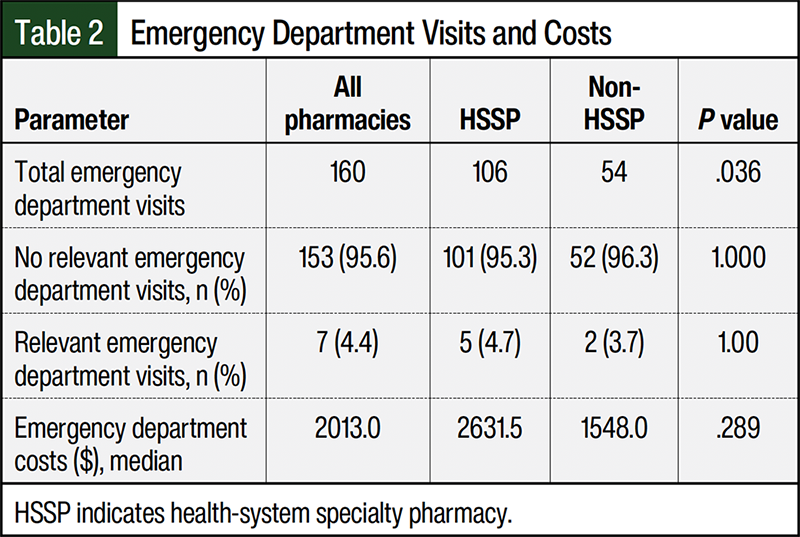
Of the 207 patients who met the inclusion criteria to participate in this study, 173 patients received appropriate anticoagulation: 83% of the patients who filled their prescriptions through the HSSP and 84% of the patients who filled through a non-HSSP received appropriate anticoagulation (Figure 1) (P=.973). No significant difference was seen in the average number of relevant emergency department visits among the HSSP and non-HSSP groups. We found a total of 5 relevant emergency department visits in the HSSP group and 2 relevant emergency department visits in the non-HSSP group.
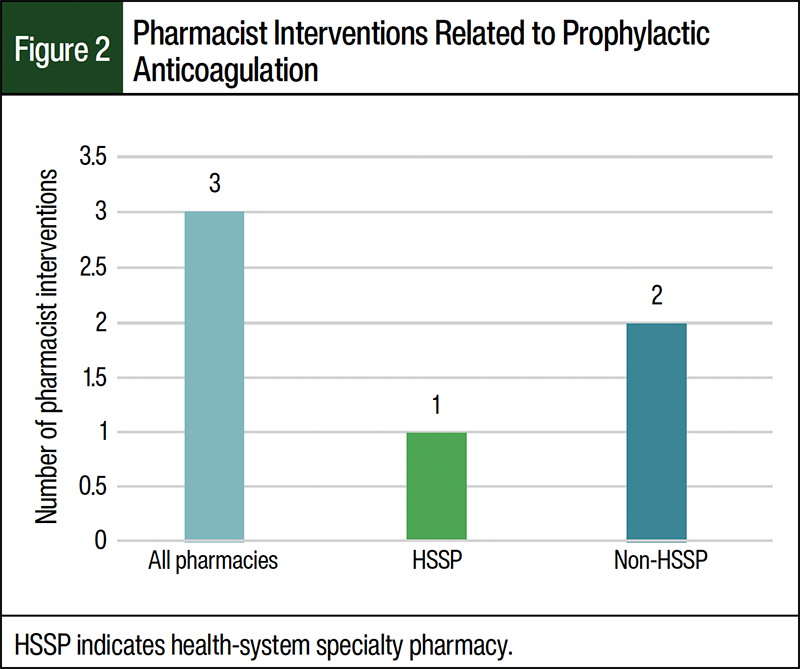
Table 2 shows total and relevant emergency department visits and costs for both groups. A total of 4.4% of all the emergency department visits were classified as relevant. Of these, 1 patient had 2 relevant emergency department visits and the remaining emergency department visits were for 1 patient each. The reasons for emergency department visits included 3 deep vein thromboses, 2 epistaxis, 1 cerebrovascular accident, and 1 nontraumatic subconjunctival hemorrhage. The average cost per emergency department visit was $2631.50 for the patients in the HSSP group and $1548.00 for patients in the non-HSSP group (Table 2). A total of 3 pharmacist interventions surrounding anticoagulation occurred among all of the patients: 1 intervention in the HSSP group and 2 interventions in the non-HSSP group (Figure 2). All 3 interventions were recommendations to prescribe an anticoagulant. Of these, 2 patients were not taking an anticoagulant and 1 patient stopped taking the anticoagulant because refills for the prescription ran out. The patients with interventions did not have relevant emergency department visits.
Discussion
This is a retrospective evaluation of the appropriateness of anticoagulation therapy in patients taking an IMiD based on the SAVED score. It has not been standard practice in specialty pharmacy to evaluate for appropriateness of anticoagulation during clinical review of new IMiD prescriptions based on NCCN guidelines.2 NCCN guidelines recommend management of VTE in MM based on calculated IMPEDE or SAVED score and this has been validated in several papers.3,5,6 Although data from studies that link VTE and inferior overall survival in patients with MM lack consensus, a clear association has been established between adverse events that may lead to treatment interruptions, increased morbidity, and increased economic burden in these patients.7,8 The primary objective of this study was to assess appropriate anticoagulation therapy rates based on NCCN guidelines for patients receiving their IMiD prescriptions and education from clinical pharmacists at a large HSSP compared with patients receiving their prescriptions at non-HSSPs. Li and colleagues showed that concurrent anticoagulation prophylaxis is prescribed in <20% of patients receiving IMiDs.6 During the time of our study, standard clinical practice included clinical pharmacists reviewing that patients were on an anticoagulant before the start of IMiD therapy, 2 weeks after therapy started, and every 6 months thereafter (or as per clinically appropriate based on pharmacists’ clinical judgment). Standard care did not include evaluating anticoagulation appropriateness based on SAVED or IMPEDE scores as stated in the NCCN guidelines. Our study results showed that 18 patients receiving their IMiD prescriptions at the HSSP had inappropriate anticoagulation therapy compared with 16 patients receiving their IMiD prescriptions from non-HSSPs (Figure 1). In addition, patients filling their IMiD prescriptions at the HSSP had 5 (4.7% of patients) emergency department visits related to anticoagulation during the study period, with a total cost of $10,555, compared with patients filling their IMiDs at non-HSSPs, who had 2 (3.7% of patients) relevant emergency department visits during the study period (Table 2) and a total cost of $4785.
Study Limitations
This study has some limitations. Patients were not differentiated as newly diagnosed or previously diagnosed with MM. The highest risk for VTE is in the first 6 months after a new diagnosis of MM,2 therefore risk for VTE could have varied significantly between the 2 groups. Another limitation involves its retrospective design—there was no follow-up SAVED score calculation. This study served as a baseline for future implementation of assessing appropriate anticoagulation only. The cohort was limited in size because of only having 3 interventions and 7 emergency department visits. The interventions made and emergency department visits were not related and each corresponded to unique patients. All 3 interventions were recommendations to prescribe an anticoagulant: 2 of the interventions were to patients not taking anticoagulation therapy and 1 was to a patient who had ran out of refills and stopped taking the anticoagulant.
Other limitations included that health-system specialty pharmacists evaluated appropriateness of anticoagulation therapy for all patients who filled their IMiD prescriptions in-house and at non-HSSPs before the start of IMiD therapy. Pharmacists in clinic continuously monitored patients filling their IMiD prescriptions at non-HSSPs 2 weeks after therapy initiation and every 6 months thereafter. In addition, SAVED score variables such as surgery within 90 days and VTE history were dependent on a thorough history after patient encounter and follow-up progress notes. It is also uncertain if different IMiDs posed distinct thrombotic risks in patients with newly diagnosed MM.5
Future Direction
NCCN guidelines recommend calculating the SAVED scores for all new IMiD prescriptions received, assess for appropriateness of anticoagulation based on the calculated scores, and make recommendations as necessary. Calculations of SAVED scores should be completed during clinical review of new IMiD prescriptions to make appropriate recommendations before dispensing of an IMiD and then periodically during therapy. A prospective study is necessary to assess clinical outcomes, specifically the number of emergency department visits related to anticoagulation.
Conclusion
The difference in percentages of patients receiving appropriate anticoagulation at HSSPs versus non-HSSPs was not significant. There was also no statistical significance in the patients who had relevant emergency department visits or in the number of pharmacist interventions between patients filling prescriptions at HSSPs and non-HSSPs during the study period. This confirms that the majority of patients are on guideline-recommended anticoagulation.
Author Disclosure Statement
Dr Planas, Dr Alvarez, Dr Delgado, Dr Igwe, and Dr Pulido have no conflicts of interest to report.
References
- Palumbo A, Anderson K. Multiple myeloma. N Engl J Med. 2011;364:1046-1060.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): multiple myeloma. Version 4.2026. www.nccn.org/professionals/physician_gls/pdf/myeloma.pdf
- Sanfilippo KM, Luo S, Wang TF, et al. Predicting venous thromboembolism in multiple myeloma: development and validation of the IMPEDE VTE score. Am J Hematol. 2019;94:1176-1184.
- Fernandez MM, Hogue S, Preblick R, Kwong WJ. Review of the cost of venous thromboembolism. Clinicoecon Outcomes Res. 2015;7:451-462.
- Li A, Wu Q, Luo S, et al. Derivation and validation of a risk assessment model for immunomodulatory drug-associated thrombosis among patients with multiple myeloma. J Natl Compr Canc Netw. 2019;17:840-847.
- Li A, Wu Q, Warnick G, et al. The incidence of thromboembolism for lenalidomide versus thalidomide in older patients with newly diagnosed multiple myeloma. Ann Hematol. 2020;99:121-126.
- Khorana AA, Dalal MR, Lin J, Connolly GC. Health care costs associated with venous thromboembolism in selected high-risk ambulatory patients with solid tumors undergoing chemotherapy in the United States. Clinicoecon Outcomes Res. 2013;5:101-108.
- Lee AYY. Anticoagulation in the treatment of established venous thromboembolism in patients with cancer. J Clin Oncol. 2009;27:4895-4901.