Allogeneic hematopoietic stem-cell transplantation (HSCT) is a potentially curative therapy for children and adults with hematologic malignancies, immune deficiencies, and congenital disorders, such as hemoglobinopathies.1-3 More than 7000 allogeneic HSCTs were performed in children aged 0 to 17 years in the United States between 2016 and 2020.4
Cytomegalovirus (CMV), a herpes virus, is a potentially devastating complication after allogeneic HSCT.1 CMV has the potential to reactivate during periods of stress and severe immunosuppression. CMV infection can replicate within blood, tissue, and bodily fluids without causing symptoms; however, CMV end-organ disease should be considered with symptoms of life-threatening organ involvement (eg, hypoxia or abdominal pain), as in cases of pneumonia or gastrointestinal disease.1 The severity of the disease depends on several factors, including donor and recipient CMV status before transplant, the type of conditioning therapy, and allogeneic versus autologous transplant.1
Preventive measures to limit CMV reactivation include the administration of antiviral agents in the early posttransplant period when patients are at highest risk.5,6 Letermovir is an antiviral medication that has been approved by the FDA for CMV prophylaxis in CMV-seropositive adults who have had an allogeneic HSCT.7 Letermovir works by inhibiting the packaging of the CMV genome into preformed virus capsids.8 However, data on pediatric patients and adolescents and young adults (AYAs) are limited, and the use of letermovir in patients aged <18 years is considered off label.6,8,9 The currently available formulations of letermovir, which include intravenous (IV) injection and oral tablets,7 also limit the ability to administer letermovir to pediatric patients. The oral tablet form of letermovir should be swallowed whole and not crushed,7 which can be difficult for younger children who are unable to swallow tablets.
Currently, most pediatric HSCT programs adopt preemptive strategies to help prevent CMV disease.1 The patients who are at risk for reactivation are monitored with serial CMV quantitative polymerase chain reaction (PCR) testing until adequate immune recovery after HSCT is achieved.10 CMV antiviral therapy is initiated once viral CMV PCR levels rise above a particular threshold.1 The decision to initiate treatment is weighed in conjunction with the patient’s degree of immune suppression and the rate of rise in viral load. Currently, there is no standard universal viral load threshold to initiate treatment.1 Some centers have adopted universal prophylaxis as an alternative method. This method involves all individuals who have a high risk for CMV disease (eg, CMV-seropositive patients, those receiving a T-cell–depleted graft, or human leukocyte antigen [HLA]-mismatched, haploidentical, or umbilical cord blood donors) receiving posttransplant anti-CMV agents, such as letermovir, valganciclovir, ganciclovir, or foscarnet.1 Agents such as ganciclovir and foscarnet have high incidences of neutropenia and renal adverse events (AEs), respectively, making letermovir a more appealing agent for treatment in this population.1
Although data on treatment with letermovir in pediatric patients and AYAs are limited, Strenger and colleagues conducted a study on treatment with letermovir in 2 pediatric patients aged 6 years and 11.5 years.8 Both patients initially started treatment with ganciclovir prophylaxis and were switched to 240 mg of letermovir orally once daily, with no coadministration of cyclosporine, because they had CMV in the blood for several weeks. Both patients’ CMV viremia resolved within 1 to 3 weeks of starting treatment with letermovir, and they remained negative for CMV. One patient’s CMV reactivated 3 months after the withdrawal of treatment with letermovir. The treatment-related AEs with letermovir included hepatic dysfunction in both patients; however, both received additional hepatotoxic agents, such as chemotherapy, immunosuppressants, and azoles, and 1 patient had confirmed liver graft-versus-host disease (GVHD).8
In a retrospective, single-center evaluation, Daukshus and colleagues evaluated adolescent (aged <18 years) HSCT recipients and studied the effects of letermovir administration until their CD4+ T-cell count was >200 cells/µL.9 All patients received letermovir at the recommended adult dose of 480 mg or 240 mg daily in combination with cyclosporine.9 The median duration of letermovir treatment was 221 days (range, 3-501 days); there were no breakthrough CMV infections while receiving letermovir in any of the 9 pediatric patients enrolled in the study.9
Last, Kuhn and colleagues followed 9 pediatric patients (median age, 14 years) who were undergoing allogeneic HSCT.5 Letermovir prophylaxis was initiated peritransplant or as secondary prophylaxis. The patients received letermovir 480 mg or 240 mg daily, with the smallest and youngest patients receiving the lower dose; none of the patients received concomitant cyclosporine. CMV reactivation was observed in 1 of the 9 patients in the study. Brief transaminitis occurred in 2 patients, which resolved spontaneously.5
In this study, we evaluated the efficacy and safety of letermovir for CMV primary prophylaxis in pediatric and AYA populations.
Methods
This single-center, retrospective chart review was conducted at Monroe Carell Jr Children’s Hospital at Vanderbilt in Nashville, TN, and was approved by the Vanderbilt University Medical Center Institutional Review Board. Patients were included in the study if they received treatment with letermovir for CMV prophylaxis after having an allogeneic HSCT between January 1, 2017, and December 31, 2021. Based on the providers’ agreed-on standards, the patients were eligible for letermovir prophylaxis if the donor or the recipient was CMV seropositive, if the patient was aged ≥12 years, and if the patient weighed >35 kg. Outpatient insurance approval for treatment with letermovir was also required for therapy initiation at our institution. The patients who did not meet these criteria were excluded from the study.
Letermovir dosing in our population was based on the FDA-approved dose for adults of 480 mg once daily.7 The study patients were required to weigh >35 kg for consideration of treatment with letermovir. Based on previously published literature, we felt comfortable administering the approved adult dose to our study’s patient populations.5,6,8,9,11,12 As a result of known drug–drug interaction that causes increased drug serum concentration, in the event of concomitant cyclosporine administration, the letermovir dose was reduced to 240 mg once daily per the prescribing information for letermovir.7 All patients were started on treatment with oral letermovir; however, 2 patients temporarily transitioned to the IV administration of letermovir during periods of mucositis. Letermovir therapy was started between day 0 and day 5 after stem-cell infusion. Quantitative CMV PCR levels were monitored once weekly starting at day 14, and the systemic treatment of CMV was started if the PCR level increased to >1000 IU/mL.
The primary study outcome was the efficacy of letermovir use for CMV prophylaxis in our pediatric and AYA cohorts based on CMV reactivation rates, which were defined by a rise in CMV levels to ≥1000 IU/mL. The secondary study outcomes included AEs associated with letermovir use. The data collected from our institutional electronic medical records (EMRs) included the baseline patient demographics; date of HSCT; CMV serostatus for the donors and recipients, donor source, and HLA match; letermovir dose, duration, and frequency of therapy; quantitative CMV PCR results while receiving letermovir and after the discontinuation of therapy; information relating to GVHD prophylaxis, occurrence, and treatment; renal function; and the relevant dates. The AEs that were possibly attributed to treatment with letermovir, including nausea, vomiting, cough, headache, myalgia, tachycardia, edema, and dyspnea, were included in the data collection because of their previous evaluation in similar research.7 The occurrence of AEs was determined by documentation in the patients’ EMRs during the time frame that they received letermovir treatment. All data collected were evaluated using descriptive statistics.
Results
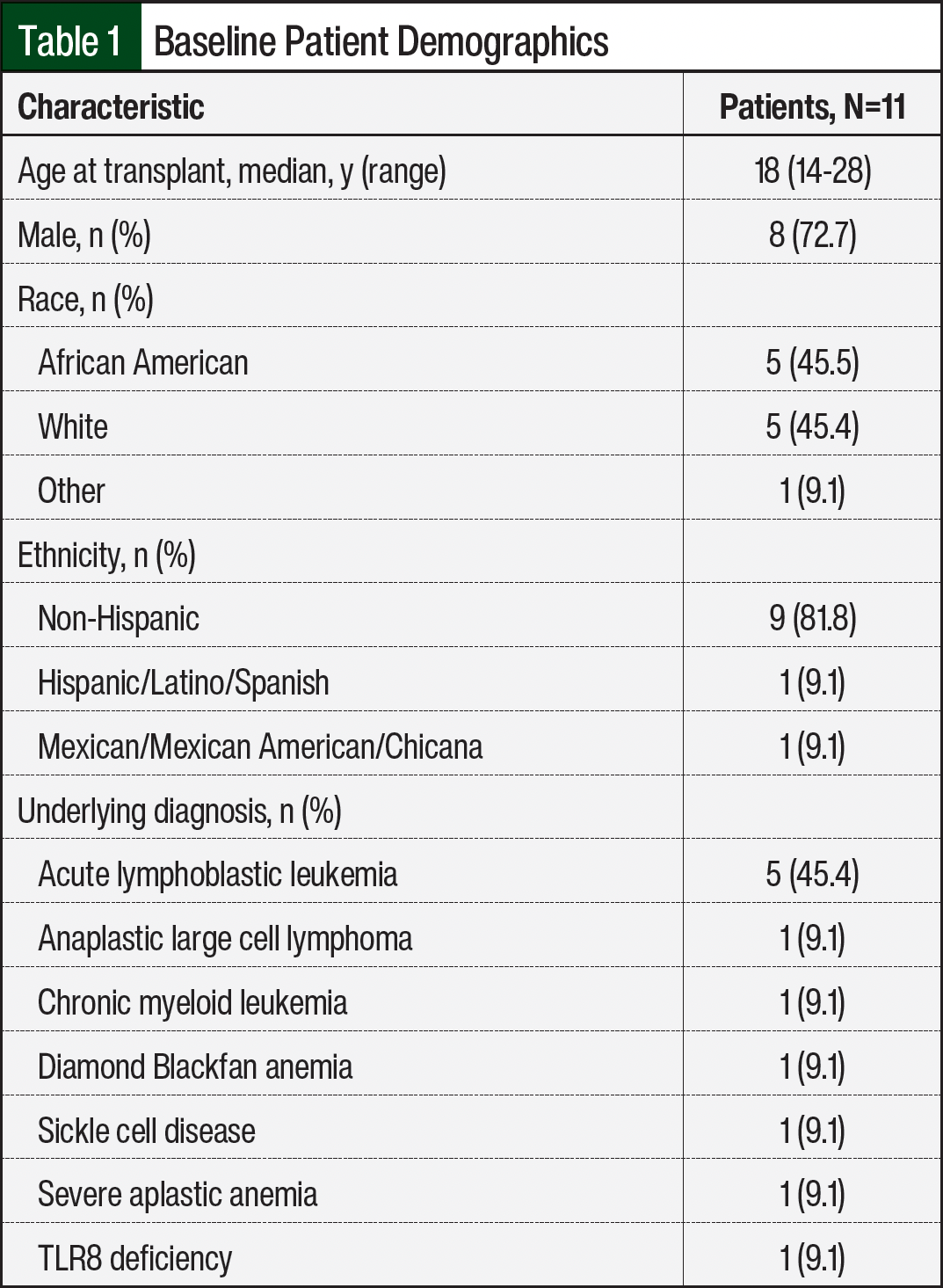
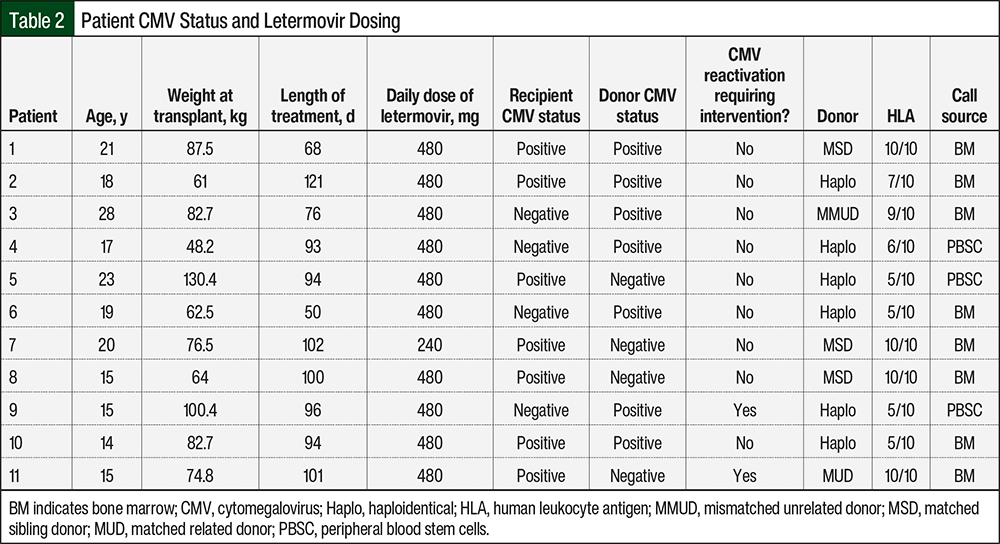
A total of 11 patients were evaluated based on the inclusion criteria during the study time frame. The patients’ baseline demographics, including age, sex, race, ethnicity, and underlying diagnosis, as defined in the EMRs, were collected and are shown in Table 1. In all, 10 patients were undergoing their first HSCT, and no patients received T-cell–depleted grafts. Letermovir dose and duration of treatment, donor and recipient CMV status, and recipient CMV reactivation were also evaluated and are shown in Table 2. Of the 11 patients, 10 received a dose of 480 mg daily of letermovir and 1 patient received a dose of letermovir of 240 mg daily as a result of concomitant cyclosporine administration. The median duration of letermovir treatment was 94 days (range, 50-121 days), with all patients stopping therapy approximately 100 days after transplant.
In our study, 4 patients had low-level reactivation of CMV while receiving letermovir prophylaxis; however, all of these individuals’ CMV subsequently became undetectable on repeated testing without any intervention. Of the 4 patients, 3 had single isolated CMV quantitative PCR levels of <137 IU/mL, whereas 1 patient had a peak level of 708 IU/mL. CMV reactivation requiring intervention occurred in 2 (18.2%) patients, who had CMV reactivation at least 30 days after the discontinuation of letermovir treatment, at 30 days and 113 days. The donor sources for these 2 patients included a haploidentical peripheral blood transplant with a 5/10 HLA–mismatched sibling donor and a matched unrelated donor bone marrow transplant with a 10/10 HLA–matched donor.
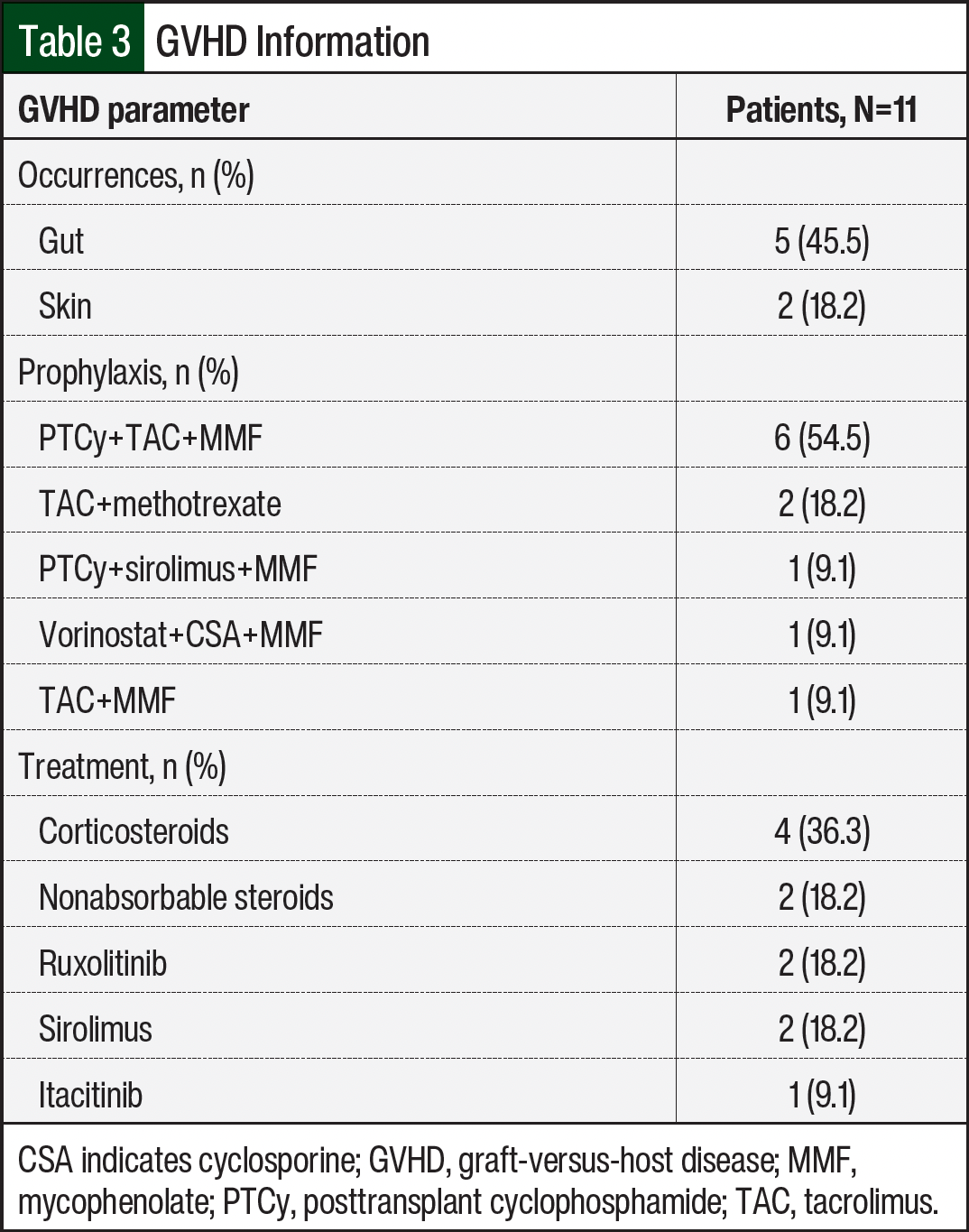
All patients received standard prophylaxis for GVHD per our institutional protocols, depending on the donor source. Grades 2 to 4 acute GVHD, as classified on presentation and reported in the patients’ charts, occurred in 5 (45.6%) of the 11 patients, who primarily received treatment with 1 to 2 mg/kg daily of systemic corticosteroids (Table 3). All 5 patients had gut GVHD, and 2 of the 5 patients also had skin GVHD. Of these 5 patients, 2 had CMV reactivation. Both patients who had CMV reactivation were not receiving letermovir therapy at the time of reactivation. Patient 9 was receiving prednisone, tacrolimus, budesonide, and ruxolitinib at the time of CMV reactivation. This patient had previously received sirolimus in place of tacrolimus; but because of an AE, treatment with sirolimus was transitioned back to tacrolimus.
Patient 11 was receiving tacrolimus and ruxolitinib at the time of CMV reactivation. Before these therapies, this patient received itacitinib for 56 days as part of a clinical trial for the treatment of acute GVHD. While receiving itacitinib the patient continued to receive letermovir, but therapy was stopped at day 100, which was 3 days after the completion of treatment with itacitinib. In addition, the patient also received a trial treatment of sirolimus in place of tacrolimus, but treatment with sirolimus was stopped as a result of angioedema and tacrolimus was restarted. Both patients received successful treatment for CMV viremia with valganciclovir; their CMV levels became undetectable within 5 weeks of therapy, and they had no end-organ damage or clinical disease.
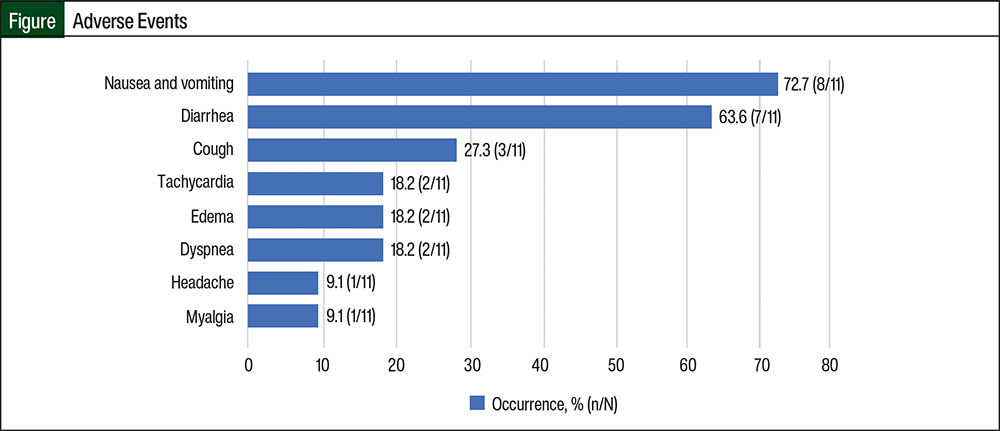
The secondary outcome of safety related to letermovir use was also evaluated in this study. All possible AEs related to letermovir therapy were well tolerated, and no patients discontinued treatment with letermovir because of AEs. Nausea and vomiting was the most common AE, occurring in 8 (72.7%) of the 11 patients, followed by diarrhea (63.6%), cough (27.3%), headache (18.2%), myalgia (18.2%), tachycardia (9.1%), edema (9.1%), and dyspnea (9.1%; Figure).
Discussion
CMV reactivation remains the most common clinically significant infection after allogeneic HSCT and is associated with high rates of morbidity and mortality. In recent years, the use of letermovir as prophylaxis for CMV significantly reduced the risk for CMV infection and all-cause mortality in adults who had a stem-cell transplant.11 However, data in pediatric patients and AYAs who have had allogeneic HSCT have been limited because of a lack of clinical studies in these patients.
The results of our retrospective chart review demonstrate that letermovir was an effective prophylactic medication against CMV infection in the adolescent population at our institution by preventing CMV reactivation in 9 (82%) patients. Our data are consistent with the results of other small, single-institution studies of pediatric patients that also show effective prevention of CMV reactivation with letermovir after HSCT.6,8,9 In all, 4 patients in our study had low-level CMV reactivation that resolved without intervention. In the study by Daukshus and colleagues, 1 patient in the pediatric cohort had low-level CMV reactivation while receiving letermovir prophylaxis, with a peak level of 382 IU/mL, which also self-resolved.9 In contrast, the studies by Richert-Przygonska and colleagues and Cheng and colleagues did not include any patients with detectable CMV reactivation while receiving treatment with letermovir.6,12 However, these studies included pediatric patients with younger age groups,6,12 which may have contributed to some of the differences in the findings. It is important to note that although some patients in our study may have had low-level viral detection while receiving letermovir, they were still protected against clinically significant disease.
Two patients in our study did have clinically significant CMV reactivation that required additional antiviral treatment after the discontinuation of treatment with letermovir. Both of the individuals who had CMV reactivation were diagnosed with acute GVHD and were receiving systemic corticosteroids at 1 mg/kg daily at the time of their viral reactivation. These CMV reactivations may have been prevented if letermovir treatment had been continued after day 100 or restarted when more intensive immunosuppression was required; however, we cannot definitively determine this result from our limited sample size, and further research is needed.
Similarly, in their single-center study of 9 patients aged 10 to 17 years, Daukshus and colleagues noted the reactivation of CMV after the discontinuation of treatment with letermovir in 2 patients.9 However, 1 patient discontinued treatment with letermovir before day 100 because of an inability to tolerate oral therapy as a result of severe nausea and vomiting, and CMV reactivation may have been prevented with continued prophylaxis.9 Taken together, these data suggest that some patients with high-risk features for CMV reactivation, such as the presence of GVHD and/or intensive immunosuppression, may benefit from continued letermovir treatment beyond 100 days after HSCT.
In general, the results of our analysis also demonstrated that treatment with letermovir resulted in well-tolerated AEs. The most common AE was nausea and vomiting, which occurred in 72.7% of patients. Other AEs such as cough; cardiac symptoms, including tachycardia, edema, and dyspnea; and headaches and myalgia were also noted in small percentages of patients. However, these AEs were relatively self-limiting, because none of our patients prematurely discontinued therapy as a result of these symptoms. It is important to note that we were unable to establish a true cause-and-effect relationship between AEs and treatment with letermovir because patients were receiving other medications concurrently that could have similar AEs, but the nonspecific nature of these AEs could also be related to continued recovery within 100 days of having an HSCT.
Our findings were similar to those of other studies of pediatric and adult patients.5,6,8,9 The symptoms of tachycardia, edema, and dyspnea have not been previously noted in pediatric cohorts. However, the results of a phase 3 study of adults (aged ≥18 years) who received letermovir for the treatment of CMV after having HSCT showed that 14.5% of patients had edema and 8% of patients had dyspnea.11 Similarly, the results of a phase 2 study of adults (aged ≥18 years) who received letermovir for the treatment of CMV after having HSCT showed that approximately 15% of patients had peripheral edema.13 The patients in our study span into adulthood, which may explain why our cohort had more of these AEs than patients in other dedicated pediatric studies. More dedicated studies in pediatric and AYA patients are needed to explore the incidence of these AEs in more detail.
Appropriate letermovir dosing has not been established in the pediatric population. Currently, dosing is based primarily on single-center studies in pediatric and adolescent patients, and we followed these data in our cohort.5,6,8,9 In our study, all patients received 480 mg of letermovir daily, except for 1 patient who received 240 mg of letermovir daily as a result of concurrent cyclosporine use. Although both patients who had reactivated CMV received 480 mg of letermovir daily, this study was not designed to assess optimal dosing in pediatric patients. Both patients responded appropriately to antiviral treatment and cleared CMV viremia, suggesting that prophylaxis with letermovir did not increase the risk for a resistant CMV strain. Additional studies in pediatric patients that optimize letermovir dosing for CMV prophylaxis are warranted, and more dedicated research would be beneficial in this area.
Limitations
This study has limitations. Like other pediatric and AYA studies, our study is primarily limited by its single-center, retrospective nature; the patients’ ages; the lack of a control arm; and the study’s small sample size. Multicenter, prospective studies that enroll a larger pediatric cohort are an essential next step to validate our findings. Hospital formulary and insurance restrictions also limited the individuals who were able to receive letermovir, further restricting our sample size.
Given the efficacy and favorable side-effect profile of letermovir in the pediatric and AYA patients who had allogeneic HSCT in our cohort, we strongly recommend further prospective clinical studies in the pediatric population because it may help reduce a significant source of HSCT-associated morbidity and mortality.
Conclusion
Our findings add to the growing body of literature showing that letermovir is safe and effective in the prevention of CMV reactivation after HSCT in the pediatric and AYA populations. Although our data support the use of letermovir in pediatric and AYA patients, prospective, randomized controlled studies are needed to validate our findings.
Author Disclosure Statement
Dr Kitko is on the Advisory Board of Horizon Therapeutics, and is a paid speaker for i3Health, PER, and Sanofi. Dr Dietz, Dr Chakravarthy, Dr Rogozinski, Dr Ho, and Dr Gulley have no conflicts of interest to report.
References
- Hiskey L, Madigan T, Ristagno E, et al. Prevention and management of human cytomegalovirus in pediatric HSC recipients: a review. Front Pediatr. 2022;10:1039938.
- Fraint E, Holuba MJ, Wray L. Pediatric hematopoietic stem cell transplant. Pediatr Rev. 2020;41:609-611.
- Tan EY, Boelens JJ, Jones SA, Wynn RF. Hematopoietic stem cell transplantation in inborn errors of metabolism. Front Pediatr. 2019;7:433.
- US Health Resources and Services Administration. Transplant activity report: number of transplants per year by donor type and age. Updated April 14, 2022. Accessed June 7, 2024. https://bloodstemcell.hrsa.gov/sites/default/files/bloodstemcell/data/transplant-activity/transplants-year-donor-type-age.xlsx
- Kuhn A, Puttkammer J, Madigan T, et al. Letermovir as cytomegalovirus prophylaxis in a pediatric cohort: a retrospective analysis. Transplant Cell Ther. 2023;29:62.e1-62.e4.
- Richert-Przygonska M, Jaramek K, Debski R, et al. Letermovir prophylaxis for cytomegalovirus infection in children after hematopoietic cell transplantation. Anticancer Res. 2022;42:3607-3612.
- Prevymis (letermovir) tablets, for oral use, and injection, for intravenous use [prescribing information]. Merck; August 2023. Accessed June 18, 2024. www.merck.com/product/usa/pi_circulars/p/prevymis/prevymis_pi.pdf
- Strenger V, Sperl D, Kubesch K, et al. Letermovir in paediatric HSCT recipients. J Antimicrob Chemother. 2019;74:2820-2821.
- Daukshus NP, Cirincione A, Siver M, et al. Letermovir for cytomegalovirus prevention in adolescent patients following hematopoietic cell transplantation. J Pediatric Infect Dis Soc. 2022;11:337-340.
- Dadwal SS, Papanicolaou GA, Boeckh M. How I prevent viral reactivation in high-risk patients. Blood. 2023;141:2062-2074.
- Marty FM, Ljungman P, Chemaly RF, et al. Letermovir prophylaxis for cytomegalovirus in hematopoietic-cell transplantation. N Engl J Med. 2017;377:2433-2444.
- Cheng CN, Li SS, Yeh YH, et al. Letermovir prophylaxis for cytomegalovirus reactivation in children who underwent hematopoietic stem cell transplantation: a single-institute experience in Taiwan. J Microbiol Immunol Infect. 2022;55:323-327.
- Chemaly RF, Ullmann AJ, Stoelben S, et al. Letermovir for cytomegalovirus prophylaxis in hematopoietic-cell transplantation. N Engl J Med. 2014;370:1781-1789.