Connecting Science to Practice
This study explored whether administering pembrolizumab
every 6 weeks at a higher dose (400 mg) causes more serious
immune-related adverse events (AE) than the standard dose,
which is administered every 3 weeks at a lower dose (200
mg). Researchers reviewed records from adult patients treated
across 16 hospitals and compared how often patients
had serious AEs leading to treatment delay or discontinuation.
The results showed no major difference in serious AEs
between the 2 dosing schedules. In clinical practice, this
longer dosing interval may offer more convenience for patients
and reduce clinic visits without increasing the risk for
serious AEs.
Pembrolizumab is a monoclonal antibody that acts as a programmed cell death protein 1 (PD-1) inhibitor. Binding to the PD-1 receptor blocks interaction with programmed cell death-ligand 1 (PD-L1) and PD-L2, releasing PD-1 pathway-mediated inhibition of the immune response, and secretes cytokines to restore antitumor immune responses. Blocking the PD-1/PD-L1 signaling pathway allows activated tumor-specific T cells to kill tumor cells. Pembrolizumab has been FDA approved for a wide variety of indications, including melanoma, non–small cell lung cancer (NSCLC), microsatellite instability-high or mismatch repair colorectal cancer, gastric cancer, esophageal cancer, cervical cancer, hepatocellular carcinoma, and renal cell carcinoma. It can be administered as a first line or subsequent line of therapy, in combination with chemotherapy or as a monotherapy.1
Pembrolizumab is associated with immune-related adverse events (irAEs) that can affect every organ system, such as pneumonitis, colitis, hepatitis, endocrinopathies, nephritis and renal dysfunction, and severe skin reactions.1 These can be severe or fatal in patients and may warrant additional treatment and/or withholding or discontinuing pembrolizumab based on severity. The mainstay for the treatment of irAEs is glucocorticoids (topical, oral, or intravenous).2 Additional immunosuppressant medications can be used if the patient’s response to steroids is suboptimal or for more severe irAEs. Other medications may be warranted depending on the organ system affected, such as insulin and thyroid medications.2
The conventional dose of pembrolizumab is 200 mg intravenously (IV) every 3 weeks. As of April 2020, the FDA approved a new dosage regimen consisting of 400 mg IV of pembrolizumab every 6 weeks across all approved adult indications.3 The new dosage was approved based on the results of the KEYNOTE-555 cohort B study.4 This was an open-label clinical study in patients with metastatic melanoma designed to evaluate a dosing regimen consisting of 400 mg of pembrolizumab every 6 weeks. The observed concentration-time profiles from cycle 1 were compared with predictions from a previously established pharmacokinetic model, which was based on data from 5 clinical trials across tumor types.5 The KEYNOTE-555 study validated previous findings from the pharmacokinetic model and provided a consistent benefit-risk profile for both dosing regimens.4,5
A retrospective study that included 45 patients with advanced NSCLC evaluated the incidence of irAEs when patients were switched from 200 mg IV every 3 weeks of pembrolizumab to 400 mg IV every 6 weeks.6 This study showed that switching regimens resulted in the occurrence of new or worsening irAEs in 37.8% of the patients, and 47.1% of these irAEs were grade ≥3.6 In contrast, another retrospective study including 80 patients with advanced NSCLC compared overall survival, progression-free survival, and occurrence and severity of irAEs in 2 groups: 200 mg IV of pembrolizumab every 3 weeks and 400 mg IV every 6 weeks. This study showed a comparable incidence and severity of irAEs, where grade ≥3 events occurred in 18% of patients in the group receiving 400 mg of pembrolizumab every 6 weeks and 19% of patients receiving 200 mg of pembrolizumab every 3 weeks.7
The 400-mg regimen given every 6 weeks would provide potential advantages to patients and healthcare systems, such as convenience, reduced infusion center visits for patients, and cost reduction. However, the data are limited regarding whether this regimen poses unacceptable risk for irAEs, which may diminish the potential benefits. Therefore, the purpose of this study was to evaluate the incidence of clinically significant irAEs with 200 mg IV of pembrolizumab every 3 weeks compared with 400 mg IV every 6 weeks.
Methods
This was a multicenter retrospective and prospective cohort study including patients receiving care at all BayCare Health System hospitals and outpatient infusion centers, including Winter Haven Hospital, St. Joseph’s Hospitals (Main, North, South), St. Anthony’s Hospital, Morton Plant Hospital, Morton Plant North Bay Hospital, Mease Countryside Hospital, Mease Dunedin Hospital, Bartow Regional Medical Center, BayCare Alliant Hospital, BayCare Hospital Wesley Chapel, and South Florida Baptist Hospital. Eligible patients included adults (aged ≥18 years) who had received fixed-dose pembrolizumab between January 1, 2020, and December 31, 2023, at any BayCare Health System hospital and outpatient infusion center. There were 2 study arms: a group consisting of patients receiving 200 mg of pembrolizumab every 3 weeks and a group consisting of patients receiving 400 mg every 6 weeks. Patients were enrolled in reverse chronological order within the study period until the desired sample size was reached.
Patients were included in the group receiving 200 mg every 3 weeks if they only received 200 mg of pembrolizumab during their treatment course. Patients were included in the group receiving 400 mg every 6 weeks if they received ≥1 dose of 400 mg of pembrolizumab during their treatment course. Key exclusion criteria included receiving weight-based dosing, receiving a nonconventional dosing regimen (something other than 200 mg every 3 weeks or 400 mg every 6 weeks) before the data collection period, receiving a PD-1/PD-L1 inhibitor in the past, receiving long-term glucocorticoid treatment, and having >1 primary cancer site.
Patients were identified by examining drug utilization through Discern Analytics 2.0 to generate reports of patients who received 200 mg or 400 mg of pembrolizumab between January 1, 2020, and December 31, 2023. Patients were screened for inclusion in the study through individual patient chart review in the electronic health record. Data were collected through individual patient chart review, including medication administration record, medication history, laboratory results, AE reports, and clinical notes.
The primary outcome was the composite incidence of clinically significant irAEs, defined as >1 of the following: discontinuing pembrolizumab because of irAEs; holding or delaying pembrolizumab treatment because of irAEs; documented irAEs grade ≥3 or irAEs meeting grade ≥3 based on the NCCN guidelines2; switching from 400 mg of pembrolizumab IV every 6 weeks to 200 mg IV every 3 weeks because of irAEs or to improve medication tolerance; altering conventional dosing frequency because of irAEs or to improve medication tolerance (ie, extending the frequency of 200 mg of pembrolizumab to every 3-4 weeks or every 6 weeks); and initiating new medications/interventions to treat irAEs, such as oral/intravenous glucocorticoids (>10 mg daily of prednisone or an equivalent), high- to very high–potency topical steroids, abatacept, mycophenolate, intravenous immunoglobulin, rituximab, cyclosporine, tacrolimus, alemtuzumab, antithymocyte globulin, plasmapheresis, tacrolimus 0.1% ointment, long-acting insulin, levothyroxine, methimazole, propylthiouracil, vedolizumab, conventional synthetic disease-modifying antirheumatic drugs (methotrexate, sulfasalazine, leflunomide, or hydroxychloroquine), tumor necrosis factor inhibitors (etanercept, adalimumab, infliximab, golimumab, or certolizumab), tocilizumab, and azathioprine.
Secondary outcomes were defined as incidence of individual components of the composite primary outcome and incidence of documented irAEs by type (cardiovascular: myocarditis, pericarditis, large vessel vasculitis; dermatologic: maculopapular rash, pruritus, bullous dermatitis, Stevens-Johnson syndrome, toxic epidermal necrolysis, psoriasis; oral mucosa: inflammation, dry mouth, oral dysesthesia; endocrine dysregulation: hyperglycemia, hypothyroidism, thyrotoxicosis, hypophysitis, adrenal insufficiency; gastrointestinal: diarrhea, colitis; hepatobiliary: hepatitis, pancreatitis; musculoskeletal: inflammatory arthritis, myositis, polymyalgia rheumatic, giant cell arteritis; nervous system: myasthenia gravis, Guillain-Barré syndrome, peripheral neuropathy, aseptic meningitis, encephalitis, demyelinating disease; ocular: uveitis, episcleritis, scleritis; and pulmonary: pneumonitis).
Statistical analysis was conducted using Minitab, a statistics software package. To achieve 80% power, a minimum of 177 patients were required in each group, with an alpha level set at 0.05 to observe an estimated difference of 10% in the incidence of irAEs between study groups. Continuous, normally distributed data were analyzed using a Student’s t-test. Continuous data that are not normally distributed were analyzed with a Mann–Whitney U test. Discrete data were analyzed using the chi-square test.
Results
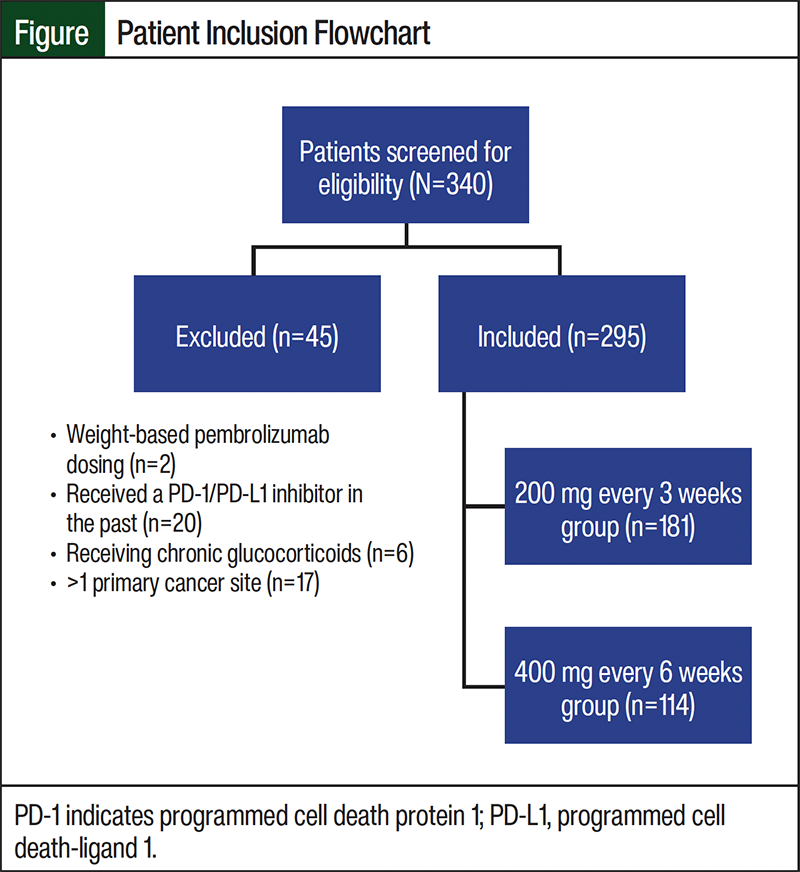
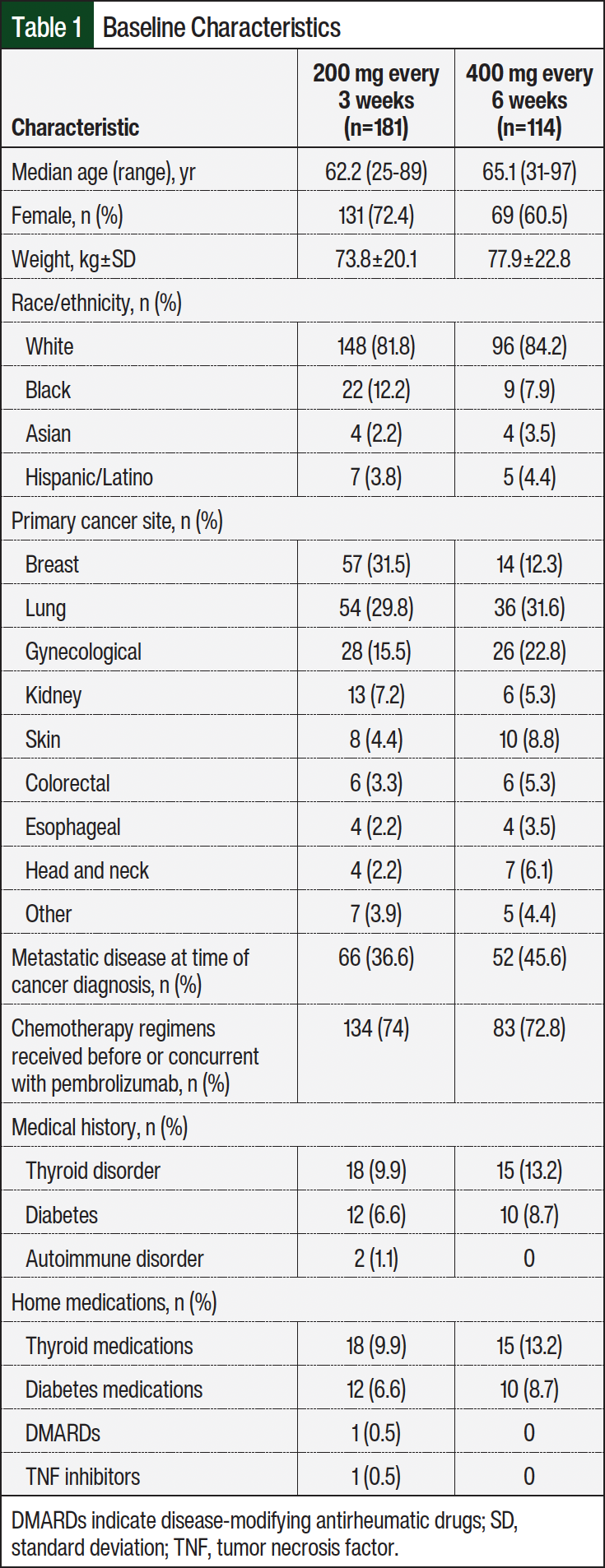
A total of 340 patients were screened for eligibility between January 1, 2020, and December 31, 2023, and 295 patients were included in the final analysis. In all, 181 patients were included in the 200 mg every 3 weeks group, and 114 patients were included in the 400 mg every 6 weeks group (Figure). Table 1 shows the demographics of patients enrolled in the study. Median age was 62.2 years (range, 25-89) in the group receiving 200 mg every 3 weeks and 65.1 years (range, 31-97) in the group receiving 400 mg every 6 weeks. Most patients were female, 72.4% and 60.5%, respectively. Other baseline characteristics were similar between both groups, except patients with breast cancer (31.5% and 12.3%, respectively) and those with metastatic disease at time of diagnosis (36.6% and 45.6%, respectively).
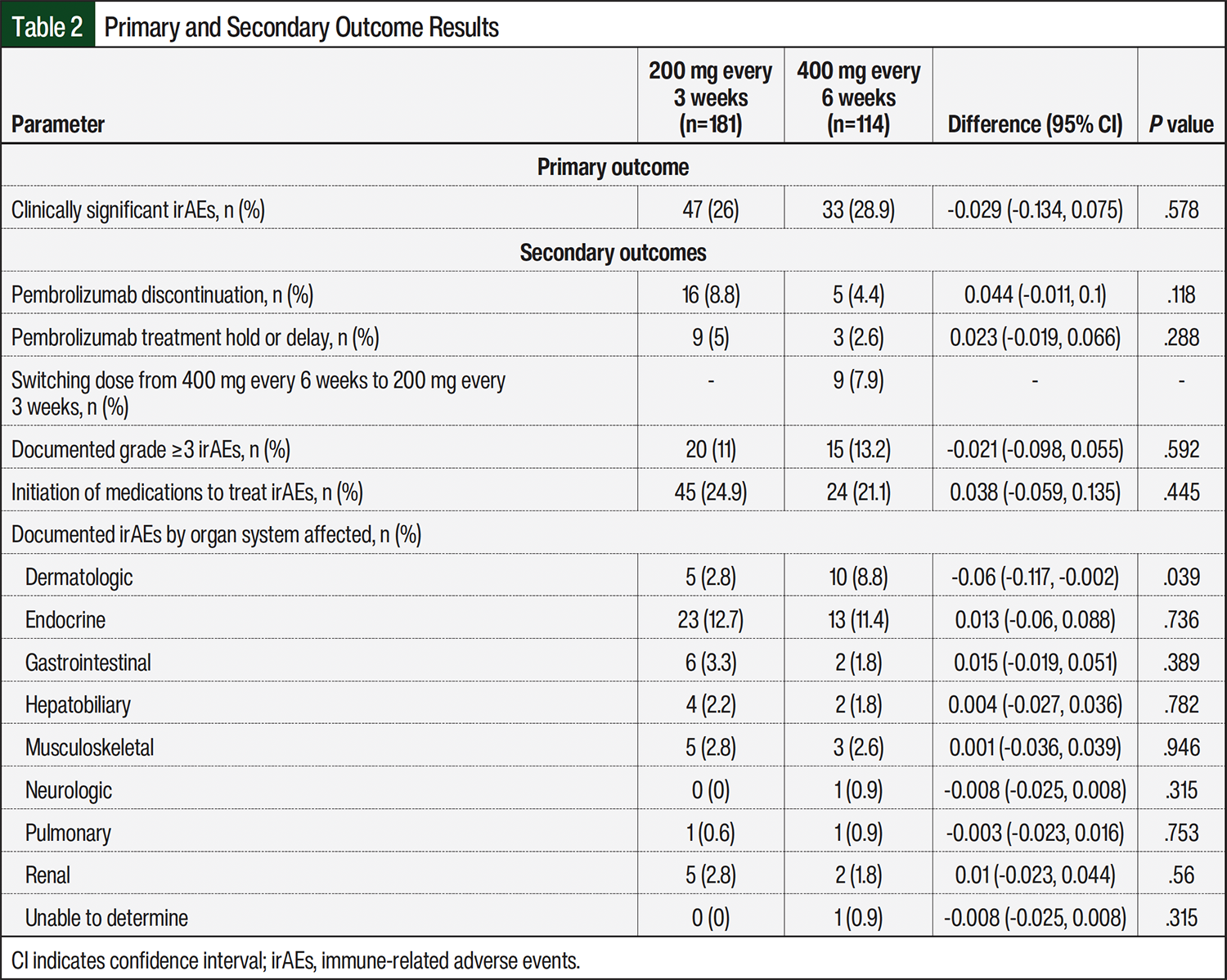
Results for the primary and secondary outcomes are listed in Table 2. Medications to treat irAEs were initiated in 45 (24.9%) patients in the group receiving 200 mg of pembrolizumab every 3 weeks and in 24 (21.1%) patients in the group receiving 400 mg every 6 weeks. The newly initiated medications included levothyroxine (33 patients), oral/intravenous glucocorticoids (26 patients), high- to very high–potency topical steroids (8 patients), and hydroxychloroquine (2 patients). There was a significant difference in the number of patients with dermatologic irAEs between the group receiving pembrolizumab every 3 weeks, 5 (2.8%) irAEs, and those receiving it every 6 weeks, 10 (8.8%) irAEs patients. Endocrine dysregulation, such as hypothyroidism, was the irAE with higher incidence: 23 (12.7%) patients versus 13 (11.4%) patients, respectively.
Discussion
This study showed a difference of approximately 3% in the clinically significant irAEs in the group receiving 400 mg of pembrolizumab every 6 weeks when compared with the group receiving 200 mg every 3 weeks. However, this difference was not significant. Secondary outcomes were also not significant, except the incidence of dermatologic irAEs, with a difference of approximately 6% in the group receiving pembrolizumab every 6 weeks compared with the group receiving it every 3 weeks. Previous studies comparing these regimens included <100 patients and were limited to patients with NSCLC. Our study included a larger sample size (295 patients) from multiple healthcare institutions and a variety of primary cancers. These were some of the strengths of the study, which provides generalizability of the results to a wider patient population.
Limitations
Our study had limitations as a result of the retrospective design. The majority of data collection was dependent on accurate documentation in the electronic health record, which may vary across institutions. Variability can also be seen in the documentation of irAEs across different BayCare institutions, because documentation practices are not standardized and the level of detail in the documentation varies by individual practitioners. Another limitation was that the study did not achieve the sample size needed to meet power. Also, patients may have presented to an institution outside of the BayCare hospitals with an irAE complaint, which may have been missed in the study. Finally, primary outcome definitions had to be limited because of study feasibility, which may have impacted internal validity.
Conclusion
In conclusion, this study adds to the available literature that both pembrolizumab regimens may be appropriate for patients. However, because of the limited sample size, future trials with larger study populations and prospective designs are needed to demonstrate whether receiving 400 mg every 6 weeks poses a higher risk for irAEs. Also, trials comparing solely patients receiving 400 mg or 200 mg of pembrolizumab would be beneficial to limit a possible confounding factor of patients transitioning from 200 mg every 3 weeks to 400 mg every 6 weeks.
Author Disclosure Statement
Dr Bacallao Molina, Dr Muirhead, Dr Rugay, Dr Thatcher, and Dr Hernandez have no conflicts of interest to report.
References
- Keytruda (pembrolizumab) injection, for intravenous use [prescribing information]. Merck Sharp & Dohme Corp, June 2025. Accessed July 7, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2025/125514s180lbl.pdf
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): management of immunotherapy-related toxicities. Version 1.2025. October 25, 2024. Accessed November 7, 2024. www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf
- US Food and Drug Administration. FDA approves new dosing regimen for pembrolizumab. April 29, 2020. Accessed July 16, 2024. www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-new-dosing-regimen-pembrolizumab
- Cohen G, Rapoport B, Chan SW, et al. Pembrolizumab 400 mg every 6 weeks as first-line therapy for advanced melanoma (KEYNOTE-555): results from cohort B of an open-label, phase 1 study. PLoS One. 2024;19(11):e0309778.
- Lala M, Li TR, de Alwis DP, et al. A six-weekly dosing schedule for pembrolizumab in patients with cancer based on evaluation using modelling and simulation. Eur J Cancer. 2020;131:68-75.
- Higashiyama RI, Yoshida T, Yagishita S, et al. Safety implications of switching pembrolizumab dosage from 200 mg every 3 weeks to 400 mg every 6 weeks in patients with advanced NSCLC. J Thorac Oncol. 2022;17:1227-1232.
- Dubé-Pelletier M, Labbé C, Côté J, Pelletier-St-Pierre AA. Pembrolizumab every 6 weeks versus every 3 weeks in advanced non-small cell lung cancer. Oncologist. 2023;28:969-977.