Imatinib mesylate is a tyrosine kinase inhibitor (TKI) of BCR-ABL, a fusion gene resulting from a translocation of genetic material between chromosome 9 and chromosome 22.1 Imatinib was first approved for the treatment of chronic myelogenous leukemia (CML), which transformed the treatment landscape of CML and drastically improved the survival of patients.1 In the phase 3 IRIS trial and subsequent follow-up studies, some of the common adverse events (AEs) of treatment with imatinib included edema, diarrhea, nausea, myalgias, and rash, with grade 3 or 4 events mostly comprised of hematologic AEs.1-4
We present a case of a patient with chronic-phase CML and multiple comorbidities, including gastrointestinal (GI) and cardiovascular diseases, who had AEs after receiving standard-dose imatinib at 400 mg once daily, but who had improved tolerance and a sustained molecular response to treatment with imatinib at split and reduced doses of 200 mg once in the morning and 100 mg once in the evening.
Case Report
A Black man aged 57 years was referred to The Lifespan Cancer Institute in Providence, RI, as a result of persistent leukocytosis, unintentional weight loss, and fatigue. He weighed 76 kg and had a BMI of 26.1 kg/m2 at the time of diagnosis. He had an extensive history of insulin-dependent diabetes, peripheral vascular disease, hyperlipidemia, coronary artery disease necessitating stenting on 2 separate occasions, hypertension, left ventricular hypertrophy, congestive heart failure with preserved ejection fraction, gastroesophageal reflux disease (GERD), and a renal transplant in 2010 that was maintained with tacrolimus treatment. His serum creatinine was 0.88 mg/dL at diagnosis, with creatinine clearance of 100 mL/min according to the Cockroft-Gault equation, and his hepatic function panel levels were all within normal limits. The patient had BCR-ABL1 fusion by polymerase chain reaction (PCR) in the peripheral blood. His Sokal score was 0.7, which is categorized as low risk.
The patient was initiated on treatment with imatinib 400 mg once daily. Other pertinent medications that the patient was receiving included tacrolimus to prevent the rejection after his renal transplant, pantoprazole for GERD, insulin aspart and insulin glargine for diabetes, as well as amlodipine, aspirin, atorvastatin, clopidogrel, losartan, and metoprolol as a result of his cardiac history. The possible drug interactions with imatinib included increasing concentrations of atorvastatin and tacrolimus via CYP3A4 inhibition. Neither of these were classified as major interactions, so no dosage modifications were made to either medication, and the patient was monitored.
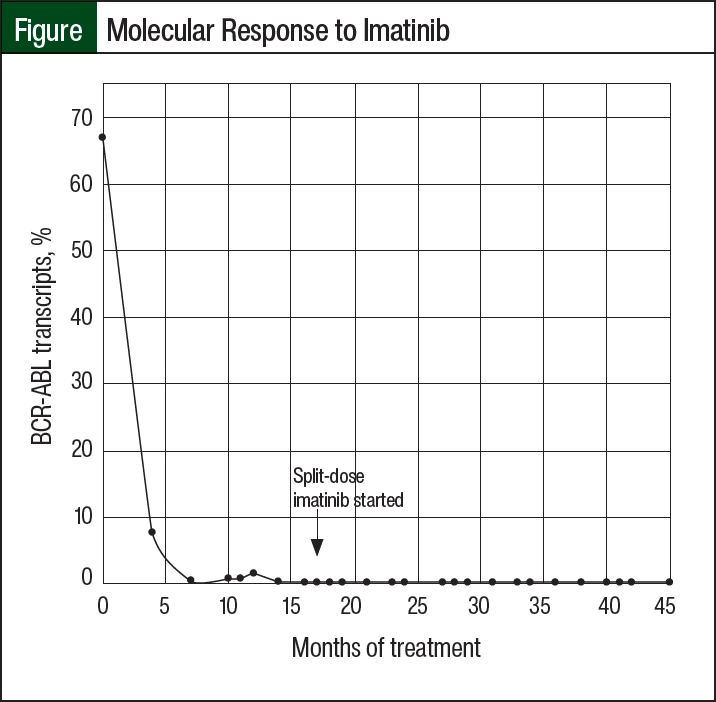
Within 6 months, the patient achieved a major molecular response (MMR; 0.3% BCR-ABL transcripts on PCR). He was admitted 2 months later with pancytopenia, grade 2 neutropenia and thrombocytopenia, and anasarca with a new pericardial effusion without tamponade, which was thought to result from treatment with imatinib; this was unusual timing for the onset of these AEs, because the majority of imatinib-related AEs occur during the initial 3 months of treatment.5,6 The patient did not receive any new medications in the preceding 4 weeks, did not receive any supratherapeutic levels of tacrolimus (target range, 6-8 ng/mL), and did not have other notable medical changes to otherwise account for these new symptoms. Treatment with imatinib was resumed as an outpatient at a lower dose of 200 mg once daily with improved tolerance. Unfortunately, BCR-ABL transcripts rose to 0.75% and then to 1.48% in subsequent PCRs, indicating relapse. The dose of imatinib was increased to 300 mg daily, however, his transcripts continued to climb.
Most other TKIs were contraindicated given the patient’s extensive cardiovascular and GI comorbidities, and the initiation of dasatinib resulted in an inpatient admission for increasing pericardial effusion size, as well as pulmonary edema, which improved after he received diuretics. After he was discharged, the patient began receiving a trial of imatinib 200 mg twice daily, which achieved a second MMR. As a result of GI AEs, including abdominal discomfort and persistent grade 2 nausea affecting adherence, his treatment was reduced to doses of 200 mg once in the morning and 100 mg once in the evening, with no detrimental effect on BCR-ABL transcripts. At the time of this writing, the patient had achieved and maintained an MMR for 24 months with this dosing strategy, and he had minimal AEs related to imatinib treatment (Figure).
Discussion
Currently, 800-mg imatinib is approved for twice-daily dosing for the treatment of accelerated- and blast crisis–phase CML but not for the treatment of chronic-phase CML.7,8 Based on the existing literature, it is unknown whether imatinib 200 mg once in the morning and 100 mg once in the evening would yield a therapeutic response in patients with chronic-phase CML. Clinical data initially established that imatinib steady-state plasma concentrations of >500 ng/mL in vitro were needed to achieve tyrosine kinase inhibition of the BCR-ABL fusion protein.9 Since then, clinical data have established that plasma trough concentrations of >1000 ng/mL are associated with a better clinical response, particularly in high-risk patients.10
A phase 1 trial investigated the pharmacokinetics of once- or twice-daily administration of imatinib and mean plasma trough concentrations of ≥570 ng/mL at 24 hours after the administration of imatinib once-daily doses of ≥350 mg.9 The lowest dose for the twice-daily regimen was 300 mg twice daily, which had a trough concentration of 972 ng/mL at steady state. Interestingly, 600 mg of imatinib daily yielded a higher trough level of 1213.9 ng/mL compared with twice-daily dosing of 300 mg, but generalization on trough levels achieved by once-daily versus twice-daily dosing is limited by the small patient sample size in this study.9
Plasma concentrations have been correlated with hematologic response, as well as cytogenetic response and MMR; a subanalysis of the IRIS study showed higher imatinib plasma trough levels correlated with significantly higher rates of complete cytogenetic response and MMR.11 Our patient had additional reasons for lowered imatinib concentrations, including being a man and being of African-American descent; lower levels of imatinib have been documented in men compared with women and in patients of African descent compared with European and Asian ethnicities.10-12 This, taken together with the pharmacokinetic data not fully supporting the split and reduced doses that our patient has been maintained on, make for an interesting case report to document his continued response at 24 months.
This patient was unable to tolerate imatinib 400 mg once daily because of GI AEs and fluid retention, which was likely compounded by his history of heart failure with preserved ejection fraction and peripheral vascular disease. Despite receiving tacrolimus after having a kidney transplant, the patient’s serum creatinine remained within normal range (0.77-1.14 mg/dL during the 24- month reporting period). In addition, all levels of tacrolimus were within range or were lower than the target of 6 to 8 ng/mL, despite imatinib, which is a CYP3A4 inhibitor, theoretically increasing the concentrations of tacrolimus. The patient also had an interaction with atorvastatin, but this was not felt to have an impact on any of our patient’s AEs.
Regarding therapeutic alternatives, the patient received a trial of dasatinib treatment, but within a week he presented with increasing pericardial effusion size and pulmonary edema. Treatment with nilotinib and ponatinib were avoided because of their greater cardiotoxic profiles compared with imatinib.13-15 Bosutinib was approved based on the BFORE trial, in which patients who received bosutinib had an increase in diarrhea, vomiting, and abdominal pain—all of which our patient was more prone to have given his history of GERD and diabetic gastroparesis.16 Therefore, he did not receive bosutinib.
Conclusion
We report a case of splitting and reducing imatinib to doses of 200 mg once in the morning and 100 mg once in the evening to maintain a response in a patient who is intolerant to the standard once-daily dosing of imatinib 400 mg. This new dosing strategy was implemented because of the limited treatment alternatives for our patient. He had a loss of response to imatinib 200 mg once daily, was intolerant to 300 mg of imatinib once daily, and most recently, was also intolerant to 200 mg of imatinib twice daily.
To our knowledge, the existing pharmacokinetic data do not clearly indicate that the split and reduced dosing strategy for imatinib would achieve adequate tyrosine kinase inhibition. Our case shows that the therapeutic effect of this treatment strategy with imatinib is maintained, because this patient continues to respond 24 months after starting the split and reduced dose of imatinib.
Author Disclosure Statement
Dr Liauw, Dr Brunault, Dr Quesenberry, and Dr Bakow have no conflicts of interest to report.
References
- Druker BJ, Guilhot F, O’Brien SG, et al. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. N Engl J Med. 2006;355:2408-2417.
- Distler JHW, Distler O. Cardiotoxicity of imatinib mesylate: an extremely rare phenomenon or a major side effect? Ann Rheum Dis. 2007;66:836.
- Deininger M, O’Brien SG, Guilhot F, et al. International randomized study of interferon vs STI571 (IRIS) 8-year follow up: sustained survival and low risk for progression or events in patients with newly diagnosed chronic myeloid leukemia in chronic phase (CML-CP) treated with imatinib. Blood. 2009;114:1126.
- Hochhaus A, Larson R, Guilhot F, et al. Long-term outcomes of imatinib treatment for chronic myeloid leukemia. N Engl J Med. 2017;376:917-927.
- Thanopoulou E, Judson I. The safety profile of imatinib in CML and GIST: long-term considerations. Arch Toxicol. 2012;86:1-12.
- Francis J, Palaniappan M, Dubashi B, et al. Adverse drug reactions of imatinib in patients with chronic myeloid leukemia: a single-center surveillance study. J Pharmacol Pharmacother. 2015;6:30-33.
- Cortes JE, Kantarjian HM, Goldberg SL, et al. High-dose imatinib in newly diagnosed chronic-phase chronic myeloid leukemia: high rates of rapid cytogenetic and molecular responses. J Clin Oncol. 2009;27:4754-4759.
- Gleevec (imatinib mesylate) tablets, for oral use [prescribing information]. Novartis, March 2024. Accessed May 13, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2024/021588Orig1s063lbl.pdf
- Peng B, Hayes M, Resta D, et al. Pharmacokinetics and pharmacodynamics of imatinib in a phase I trial with chronic myeloid leukemia patients. J Clin Oncol. 2004;22:935-942.
- Guilhot F, Hughes TP, Cortes J, et al. Plasma exposure of imatinib and its correlation with clinical response in the Tyrosine Kinase Inhibitor Optimization and Selectivity Trial. Haematologica. 2012;97:731-738.
- Larson RA, Druker BJ, Guilhot F, et al. Imatinib pharmacokinetics and its correlation with response and safety in chronic-phase chronic myeloid leukemia: a subanalysis of the IRIS study. Blood. 2008;111:4022-4028.
- Adjiwidjaja J, Gross AS, Boddy AV, McLachlan AJ. Physiologically-based pharmacokinetic model predictions of inter-ethnic differences in imatinib pharmacokinetics and dosing regimens. Br J Clin Pharmacol. 2022;88:1735-1750.
- Saglio G, Kim DW, Issaragrisil S, et al. Nilotinib versus imatinib for newly diagnosed chronic myeloid leukemia. N Engl J Med. 2010;362:2251-2259.
- Kim TD, Rea D, Schwarz M, et al. Peripheral artery occlusive disease in chronic phase chronic myeloid leukemia patients treated with nilotinib or imatinib. Leukemia. 2013;27:1316-1321.
- Aghel N, Delgado DH, Lipton JH. Cardiovascular toxicities of BCR-ABL tyrosine kinase inhibitors in chronic myeloid leukemia: preventive strategies and cardiovascular surveillance. Vasc Health Risk Manag. 2017;13:293-303.
- Cortes JE, Gambacorti-Passerini C, Deininger MW, et al. Bosutinib versus imatinib for newly diagnosed chronic myeloid leukemia: results from the randomized BFORE trial. J Clin Oncol. 2018;36:231-237.