Connecting Science to Practice
This case report details a 68-year-old man with follicular lymphoma who had serum sickness, a type III hypersensitivity, in response to receiving the rituximab biosimilar Riabni. This patient was able to tolerate subsequent therapy with obinutuzumab, despite its similar mechanism to Riabni, likely because obinutuzumab is a fully humanized monoclonal antibody with less complement-dependent cytotoxicity. Fully humanized monoclonal antibodies are less immunogenic than mouse/human CD20 monoclonal antibodies, and therefore have a lower risk for immune complex formation than the chimeric rituximab-based monoclonals. When he received an obinutuzumab-containing regimen, this patient had significant improvement on staging scans at months 3 and 6. This case report provides evidence that may allow patients with type III hypersensitivity reactions to rituximab-based therapies to continue treatment with the alternate anti-CD20 monoclonal antibody obinutuzumab.
Serum sickness is an immune complex–mediated type III hypersensitivity caused by the presence of a heterologous or chimeric protein that leads to antibody formation and subsequent immune complexes.1 One to 2 weeks after initial exposure to the causative agent, immunoglobulin (Ig)M antibodies develop, followed shortly by IgG antibodies.2 Immune complexes are formed when IgM or IgG bind to circulating chimeric proteins. Activation of the complement system occurs after immune complexes deposit in tissues and, more frequently, in synovial fluid.3 C3a, a complement fragment, is responsible for histamine release, vasodilation, and urticarial lesions.4 The presentation of serum sickness is delayed after the first exposure to the offending agent, with symptoms typically present around the time of IgM and IgG formation 1 to 2 weeks later.2
A hallmark feature of serum sickness is arthralgia. Patients report pain in the knees, wrists, ankles, shoulders, and spine, as well as in the jaw and temporomandibular joints. This pain may be accompanied by tenderness to palpitation, swelling, and erythema in the extremities, often in the hands, feet, ankles, knees, and shoulders. Patients may also have fever, fatigue, and pruritic rash without the involvement of mucous membranes.1
Laboratory results in patients with serum sickness can be variable, and there is no one laboratory test or result that can diagnose serum sickness. A complete blood count can show leukopenia or mild leukocytosis with or without eosinophilia, and mild thrombocytopenia may occur.5 Acute-phase reactants, such as erythrocyte sedimentation rate and C-reactive protein, may be elevated.5 Some patients with serum sickness have abnormalities in serum chemistries, such as serum creatinine, blood urea nitrogen, and liver function tests.5 Diagnosis is made through clinical judgment in patients who have a characteristic pattern of subacute onset of a rash, fever, and severe arthralgias and myalgias occurring 1 to 2 weeks after the first exposure to a potentially causative agent.6
Similarly, serum sickness-like reaction (SSLR) can occur with similar symptoms days to weeks after the administration of nonchimeric protein therapeutics or other drugs, or in association with infections.1 SSLR is also known as drug-induced urticaria with arthritis, because this disease state, unlike true serum sickness, is not dependent on circulating immune complexes.1,7 Serum sickness is diagnosed in patients who received a heterologous therapy, whereas SSLR can be diagnosed in patients with other drug administrations.8 Serum sickness and SSLR are self-limiting and typically resolve 1 to 2 weeks after discontinuation of the causative agent.8 Patients with serum sickness related to an agent should be switched to a different therapy for the remainder of their treatment if such an option is feasible. Mild-to-moderate symptoms can be managed with nonsteroidal anti-inflammatory agents. More severe symptoms may require the use of oral or intravenous glucocorticoids.9
Rituximab is a chimeric anti-CD20 monoclonal antibody that is used for the treatment of many hematologic malignancies, including follicular lymphoma (FL).9 Rituximab is often associated with infusion reactions, during or immediately after the first dose,9 as well as type IV allergic hypersensitivity reactions.10 Type IV hypersensitivities differ from type III reactions in that they are T-cell–mediated and do not require the formation of immune complexes.11 Symptoms can be mitigated with the concurrent administration of corticosteroids and antihistamines, as well as a slowed infusion rate if symptoms present during administration.10
Because of its chimeric makeup (an antibody that contains murine-derived variable regions), patients receiving rituximab are also at risk for rituximab-induced serum sickness.9 Obinutuzumab is a glycoengineered anti-CD20 monoclonal antibody.12 It has less complement-dependent cytotoxicity than rituximab, but greater antibody-dependent cellular cytotoxicity and phagocytosis and greater direct B-cell–depleting effects.12 Fully humanized monoclonal antibodies, such as obinutuzumab, are less immunogenic than mouse/human CD20 monoclonal antibodies, and therefore cause less type III and IV hypersensitivity reactions.13 However, as with rituximab, obinutuzumab is also associated with a high rate of severe infusion-related reactions with the first dose (65% vs 77% obinutuzumab vs rituximab).14,15 This higher rate of reactions results from the more rapid B-cell depletion and associated cytokine release with obinutuzumab, which are unrelated to its humanized makeup. Most non-Hodgkin lymphomas are derived from B cells, and anti-CD20 therapy and chemotherapy has shown improved outcomes in these patients.16 The successful use of obinutuzumab in patients with multiple types of hypersensitivity to rituximab have been reported in patients with hematologic disorders, but these rarely included type III hypersensitivities.16 This case report details treatment with obinutuzumab in a patient with FL who had serum sickness after receiving rituximab-arrx.
Case Report
A 68-year-old man was diagnosed with grade 1, stage IIE follicular non-Hodgkin lymphoma after discovering a palpable mass in his left inguinal region 8 weeks earlier. At treatment initiation, the patient underwent a left groin incisional lymph node biopsy. He denied having any fevers, night sweats, or weight loss. Because the patient was asymptomatic at this point, he was offered the option of observation. He wanted to initiate treatment and received 1 treatment cycle of 90-mg/m2 bendamustine on days 1 and 2 and 375 mg/m2 of the biosimilar rituximab-arrx on day 1. The patient received premedication with 20 mg of intravenous dexamethasone, 1 mg of granisetron, 25 mg of diphenhydramine, and 20 mg of famotidine 30 minutes before the start of the first infusion. Bendamustine was then infused for more than 10 minutes, followed by the initial titration of rituximab-arrx.
The patient tolerated treatment well on both days and he received sulfamethoxazole plus trimethoprim for neutropenia prophylaxis to start 1 week from treatment cycle 1 day 1. Eleven days after his first day of treatment, the patient had a sore throat, joint aches, and full-body hives, which began after starting treatment with sulfamethoxazole plus trimethoprim 4 days earlier. The patient alerted the oncology clinic to these symptoms. Although he did not have a documented allergy to sulfamethoxazole plus trimethoprim, it was thought by his oncologists that this could be an allergic reaction. The patient received a methylprednisolone dose pack and the antibiotic was discontinued.
The next day, the patient stated that his symptoms were worsening, and he had a low-grade fever and swollen legs, and was unable to eat, drink, or ingest the prescribed steroids. He was sent to the emergency department, received intravenous fluids, and returned home. A total of 14 days after receiving treatment, the patient returned for a follow-up appointment at our clinic. He again received intravenous fluids and 20 mg of intravenous dexamethasone in the office, but his symptoms did not improve. At this time, it was determined that the patient needed to be admitted to the hospital.
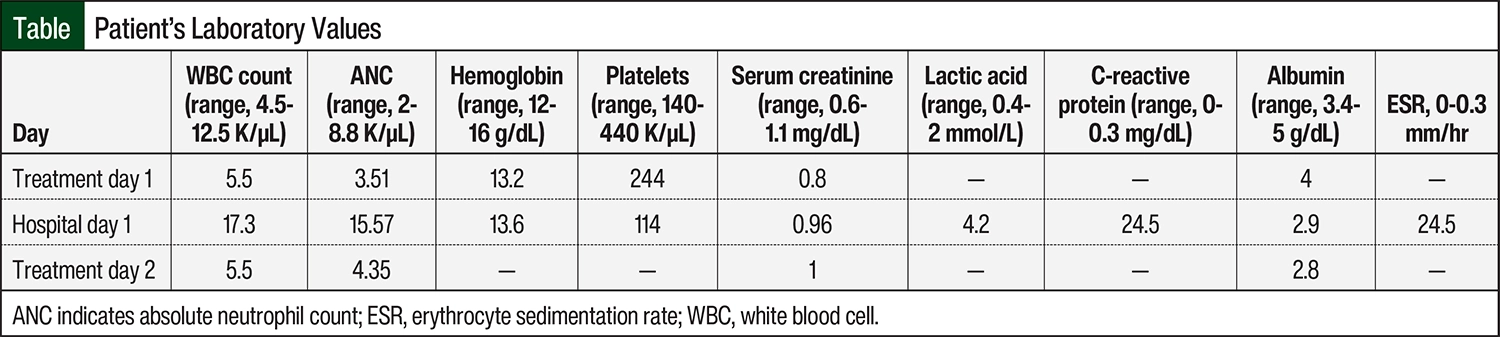
Inpatient laboratory testing results showed that the patient had leukocytosis, normal hemoglobin, and baseline serum creatinine (Table). He was admitted to the hospital with an elevated white blood cell count, muscle aches, severe soreness in his mouth, congestion, and difficulty breathing, which are consistent with serum sickness, as a delayed reaction to treatment with rituximab-arrx. The patient received 3 separate doses of methylprednisolone 125 mg over the span of his inpatient stay. The following day, the patient was discharged from the hospital when he clinically improved after receiving fluids and intravenous steroids.
The patient returned to the clinic for treatment cycle 2, but he was no longer deemed a candidate for further therapy with rituximab-arrx. For the treatment of noncontiguous stage II FL, the National Comprehensive Cancer Network’s guidelines for B-cell lymphoma recommend an anti-CD20+ monoclonal antibody.17 As a result of findings from the GALLIUM study, which showed superiority of obinutuzumab and bendamustine over rituximab and bendamustine for the treatment of FL,18 substituting obinutuzumab for rituximab was discussed with the patient. The risk for possible serum sickness as a result of treatment with obinutuzumab was discussed with the patient because of the redundant mechanism of action of obinutuzumab. The patient agreed to the formulation change, and in treatment cycle 2 he began receiving treatment with bendamustine 90 mg/m2 on days 1 and 2 and with obinutuzumab 1000 mg on days 1, 8, and 15 for cycle 1 and then every 28 days.
The patient tolerated the infusion well and was prescribed prednisone 10 mg to be received as a taper if he began to have symptoms of serum sickness. When the patient returned for day 8 of treatment cycle 2, he remained symptom free. On day 15, he stated that he did not receive oral steroids and that he did not have symptoms similar to his previous reaction. The patient continued to receive treatment with obinutuzumab and bendamustine for a total of 5 cycles (6 chemotherapy cycles in total) without further issue.
Staging scans at months 3 and 6 showed significant improvements in the patient’s previously identified areas of malignancy. The lymph node that was initially detected in the iliac fossa, which measured 9.9 cm in its greatest dimension and reported a standard uptake value of 20.3, showed normal measurements in month 3 with a standard uptake value of 2.6. A repeat positron emission tomography scan was performed 6 months after therapy initiation, which showed a further decrease in hypermetabolic activity to a standard uptake value of 1.8. At the 1-year follow-up, his physical examination was unremarkable for peripheral lymphadenopathy or hepatosplenomegaly and the surveillance computed tomography did not show any convincing evidence of disease progression or recurrence.
Discussion
Serum sickness is a clinical diagnosis made after observing characteristic patterns that cannot be definitively confirmed. Our patient had subacute onset of severe arthralgias, rash, and fever that began 11 days after receiving his initial dose of rituximab-arrx. The results of the JASMINE trial, which compared rituximab-arrx with reference rituximab, showed similar rates of type I infusion-related reactions (43% vs 42.9%, respectively).19 The timing and severity of adverse events, as well as the presence of antidrug antibodies, were similar between the 2 treatment groups. There were no episodes of type III reactions in either treatment arm; however, this remains a risk for all rituximab-based therapies.19 A recent study in France supports the conclusion that there is no additional risk for hypersensitivity reactions, including serum sickness, with rituximab biosimilars compared with originator rituximab.20
When our patient first called the clinic to inform the staff of his symptoms, he stated that he was having an allergic reaction to sulfamethoxazole plus trimethoprim, which is the antibiotic that was prescribed for neutropenia prophylaxis after his initial treatment cycle. He had received 3 doses of sulfamethoxazole plus trimethoprim at symptom presentation. Type I hypersensitivity reactions or IgE-mediated reactions are responsible for most classic drug allergies. This presentation can be similar to serum sickness with urticarial rash, but the timing of the symptoms differs. IgE-mediated reactions present minutes to hours after administration of a drug, depending on the time it takes to mount a significant IgE response.16 For this reason, it is unlikely that our patient had a true IgE-mediated allergic reaction to sulfamethoxazole plus trimethoprim.
When urticaria presents with fever and arthralgias, differential diagnosis leans more toward a type III serum sickness hypersensitivity. Sulfamethoxazole plus trimethoprim has been cited to cause SSLR,21 which would present in the same fashion as serum sickness, 1 to 2 weeks after initiation of treatment. It is unlikely that our patient would have an SSLR to sulfamethoxazole plus trimethoprim because symptom onset was too rapid relative to the initiation of therapy. In addition, types I and III hypersensitivities resolve with the withdrawal of the causative agent.
Our patient discontinued treatment with sulfamethoxazole plus trimethoprim at the onset of his symptoms, yet his symptoms still progressed. His serum sickness after receiving rituximab-arrx scored 6 on the Naranjo Adverse Drug Reaction Probability Scale, which indicates a probable relation between the reaction and the drug. To ameliorate infusion hypersensitivity reactions, the prescribing information for rituximab recommends premedication with acetaminophen and an antihistamine, in combination with 100 mg of intravenous methylprednisolone if the patient has rheumatoid arthritis.21 In addition to rituximab-arrx, our patient also received bendamustine, a moderately emetogenic chemotherapy agent, which is typically premedicated with dexamethasone and a 5-HT3 antagonist.22 The premedication regimen prescribed for our patient varied slightly from this recommendation, but the lack of acetaminophen did not have a role in his serum sickness syndrome. Similarly, because of the typical delayed symptom onset of serum sickness syndrome, the dose and type of steroid did not contribute to the serum sickness.
Despite this adverse reaction, our patient wanted to continue with treatment. Rituximab desensitization is a strategy used typically for type I IgE-mediated infusions.18 However, there are limited reports of desensitization for type III allergic reactions to rituximab drugs. A patient with serum sickness reaction had successful desensitization of a type III allergic reaction to rituximab through the use of a nonstandardized slow 2-day protocol.23 The treatment team hypothesized that changing the treatment plan to obinutuzumab would be a safer and more time-sensitive alternative, compared with a rituximab desensitization.
Although obinutuzumab is associated with a high risk for infusion reactions, delayed reactions, such as serum sickness, are not often reported. However, one case report detailed a patient with nonresponse to rituximab treatment who had serum sickness twice with obinutuzumab after the first and fifth infusions.24 The patient continued receiving treatment as part of salvage therapy because these reactions were uncomfortable, but ultimately self-limiting.24 The results of a retrospective cohort study of 17 patients receiving obinutuzumab after having drug hypersensitivity reactions of any type subsequent to treatment with rituximab showed that 94% of patients tolerated this newer anti-CD20 without having a severe reaction.13 Although this cohort had only 6 patients with serum sickness as a hypersensitivity reaction,13 it supported our hypothesis that the risk for such a reaction would be lower for our patient than re-exposure to rituximab-arrx.
Type III hypersensitivity reactions are initially the result of antigen-antibody complexes, which are also referred to as immune complexes.9 In type III hypersensitivity reactions, these immune complexes attach to endothelial tissues, glomerular tissues, the epithelial lining of alveoli, as well as synovial tissues, as was apparent in our patient.3 Soluble proteins—in this case, rituximab-arrx—are identified by IgG- or IgM-adaptive antibodies, which then stimulate the beginning of the classical complement cascade.25 In the classical cascade, C1 binds to the immune complex, resulting in activation and the subsequent production of the anaphylatoxins C3a and C3b. Through a series of reactions, C3b combines with C3 convertase to form C5 convertase, which converts C5 to C5a and C5b. C5a is an inflammatory peptide that helps recruit innate immune cells to the sites of immune complexes.25 The recruited neutrophils then bind to the tissue-bound immune complexes, which results in the release of cytotoxic granules and subsequently leads to tissue damage and necrosis.26 In our case, the patient’s joint pain was most likely associated with synovial damage from the complement cascade and synovial-bound immune complexes.
Obinutuzumab therapy results in lower complement-dependent cytotoxicity than reference rituximab and rituximab biosimilars,18 which may explain why patients who had serum sickness with rituximab tolerate treatment with obinutuzumab. In addition, obinutuzumab is a humanized monoclonal antibody whereas rituximab drugs are chimeric. Humanized monoclonal drugs are primarily human, with the complementarity determining regions of the variable domains being the only foreign portion. Conversely, the entire variable region of chimeric monoclonals is foreign, which may increase the immunogenicity of, and thereby the presence of immune complexes with, these agents.27 Although the mechanisms of action are the same for obinutuzumab and rituximab (CD20-directed, antibody-dependent cellular cytotoxicity), the structural differences of the 2 molecules are potentially distinct enough to prevent cross-reactivity.27 As in our case, the immune system did not recognize the soluble obinutuzumab protein, despite forming previous immune complexes with rituximab-arrx. To our knowledge, this is the first published case in which treatment with obinutuzumab was successful after serum sickness with a rituximab biosimilar.
Conclusion
Serum sickness has been reported in patients receiving rituximab and other biosimilars. The rates of similar reactions have not yet been documented for newer anti-CD20 drugs such as obinutuzumab. This case report describes a patient who had serum sickness after receiving rituximab-arrx and tolerated subsequent treatment with obinutuzumab, which is likely a result of the structural and pharmacologic differences between the 2 drugs.
The primary hypothesis for the toleration of obinutuzumab is that obinutuzumab induces lower complement-dependent cytotoxicity than rituximab-based drugs. In addition, obinutuzumab is structurally unique, possibly preventing a similar immune complex, which is one of the required components of serum sickness syndrome. This case report provides evidence that may allow patients with type III hypersensitivity reactions to rituximab-based therapies to continue treatment with the alternate anti-CD20 monoclonal antibody obinutuzumab.
Author Disclosure Statement
Dr Padgett, Dr Shely, and Dr Pierce have no conflicts of interest to report.
References
- Güngörer V, Ertan K, Arslan Ş. Diagnosis that clinicians don’t think of, serum sickness-like reaction. Pediatr Int. 2022;64:e14740.
- Joint Task Force on Practice Parameters; American Academy of Allergy, Asthma and Immunology; American College of Allergy, Asthma and Immunology; Joint Council of Allergy, Asthma and Immunology. Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105:259-273.
- Shiel WC, Andrews BS, Friou GJ, et al. Immune complexes in synovial fluid and serum from patients with disseminated gonococcal infection: evidence for local immune complex formation within the joint. Ann Rheum Dis. 1986;45:816-820.
- Giang J, Seelen MAJ, van Doorn MAB, et al. Complement activation in inflammatory skin diseases. Front Immunol. 2018;9:639.
- Patterson-Fortin J, Harris CM, Niranjan-Azadi A, Melia M, M. Serum sickness-like reaction after the treatment of cellulitis with amoxicillin/clavulanate. BMJ Case Reports. 2016;bcr2016217608.
- Yorulmaz A, Akın F, Sert A, et al. Demographic and clinical characteristics of patients with serum sickness-like reaction. Clin Rheumatol. 2018;37:1389-1394.
- Bakshi D, Tang X, Waserman S. A case of pediatric serum sickness like reaction (SSLR) after a 2-month re-exposure to amoxicillin. Allergy Asthma Clin Immunol. 2024;20:29.
- Zhang Z, Xiang Y, Wang B, et al. Intestinal mucosal permeability of children with cefaclor-associated serum sickness-like reactions. Eur J Pediatr. 2013;172:537-543.
- Blase JR, Frame D, Michniacki TF, Walkovich K. Case report: use of obinutuzumab as an alternative monoclonal anti-CD20 antibody in a patient with refractory immune thrombocytopenia complicated by rituximab-induced serum sickness and anti-rituximab antibodies. Front Immunol. 2022;13:863177.
- Fouda GE, Bavbek S. Rituximab hypersensitivity: from clinical presentation to management. Front Pharmacol. 2020;11:572863.
- Posadas SJ, Pichler WJ. Delayed drug hypersensitivity reactions - new concepts. Clin Exp Allergy. 2007;37:989-999.
- Marcus R, Davies A, Ando K, et al. Obinutuzumab for the first-line treatment of follicular lymphoma. N Engl J Med. 2017;377:1331-1344.
- Schieber T, Li A, Lundberg J, et al. Successful obinutuzumab use after rituximab discontinuation due to intolerance in patients with hematologic disorders. Blood Adv. 2023;7:3431-3434.
- Gazyva (obinutuzumab) injection, for intravenous use [prescribing information]. Genentech; July 2022. Accessed August 28, 2024. www.gene.com/download/pdf/gazyva_prescribing.pdf
- Rituxan (rituximab) injection, for intravenous use [prescribing information]. Biogen Idec and Genentech; December 2021. Accessed August 28, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2021/103705s5467lbl.pdf
- Prica A, Crump M. Improving CD20 antibody therapy: obinutuzumab in lymphoproliferative disorders. Leuk Lymphoma. 2019;60:573-582.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines) B-Cell Lymphomas. Version 3.2024; August 26, 2024. www.nccn.org/professionals/physician_gls/pdf/b-cell.pdf
- Younes A, Burke JM, Diefenbach C, et al. Safety and efficacy of atezolizumab with obinutuzumab and bendamustine in previously untreated follicular lymphoma. Blood Adv. 2022;6:5659-5667.
- Niederwieser D, Hamm C, Cobb P, et al. Efficacy and safety of ABP 798: results from the JASMINE trial in patients with follicular lymphoma in comparison with rituximab reference product. Target Oncol. 2020;15:599-611. Erratum in: Target Oncol. 2020;15:807.
- Jourdain H, Hoisnard L, Sbidian E, Zureik M. Severe hypersensitivity reactions at biosimilar versus originator rituximab treatment initiation, switch and over time: a cohort study on the French National Health Data System. BioDrugs. 2023;37:397-407.
- Surdacki M, Fernandez E, Phillips EJ, Stone CA. Drugs associated with serum sickness and serum sickness–like reactions: a review of FDA Adverse Event Reporting System (FAERS) data. J Allergy Clin Immunol Pract (Cambridge, MA). 2024;12:1351-1354.
- Tombleson RL, Ho V, Sokol L, et al. Optimizing premedications in the prevention of bendamustine infusion-related reactions. Cancer Control. 2012;19:245-247.
- Wong JT, Long A. Rituximab hypersensitivity: evaluation, desensitization, and potential mechanisms. J Allergy Clin Immunol Pract. 2017;5:1564-1571.
- Saba J, Logan AC. Obinutuzumab-induced serum sickness following salvage therapy for chronic lymphocytic leukemia. Clin Case Rep. 2017;5:891-893.
- Janeway CA Jr, Travers P, Walport M, et al. The complement system and innate immunity. Immunobiology: The Immune System in Health and Disease. 5th ed. Garland Science; 2001. Accessed July 11, 2023. www.ncbi.nlm.nih.gov/books/NBK27100/
- Mayadas TN, Tsokos GC, Tsuboi N. Mechanisms of immune complex-mediated neutrophil recruitment and tissue injury. Circulation. 2009;120:2012-2024.
- Boyer-Suavet S, Andreani M, Lateb M, et al. Neutralizing anti-rituximab antibodies and relapse in membranous nephropathy treated with rituximab. Front Immunol. 2020;10:3069.