Connecting Science to Practice
This study aimed to evaluate the incidence of bleeding
events in adult patients receiving ibrutinib monotherapy
compared with those on ibrutinib with concomitant anticoagulant
and/or platelet-inhibiting medications. Using a retrospective,
observational design, all patients treated for ≥30
days at a single center were included, with bleeding events
graded per Common Terminology Criteria for Adverse
Events Version 4.0. Incidence rates were calculated per person-
year to account for varying lengths of follow-up contributed
by each patient. The findings demonstrate that concomitant
anticoagulant use significantly increases the risk of
any bleeding, whereas antiplatelet agents do not. It is important
to note that major bleeding rates were not significantly
elevated with either class. These results underscore the need
for cautious anticoagulant use in ibrutinib-treated patients
and support close pharmacist-led monitoring to mitigate
bleeding risk in everyday practice.
Bruton tyrosine kinase (BTK) is an important signaling molecule required for the proliferation and survival of normal and malignant B cells.1,2 Ibrutinib is an oral covalent binding BTK inhibitor that has been used for the treatment of B-cell malignancies, including chronic lymphocytic leukemia (CLL), mantle-cell lymphoma (MCL), and lymphoplasmacytic lymphoma.1,2 Initial ibrutinib clinical trials identified bleeding as an important adverse event (AE) of ibrutinib, with overall bleeding rates of up to 44%3 and major bleeding in 1% to 4% of patients.3-5 Ibrutinib-induced bleeding is attributed to decreased platelet function because of targeted BTK inhibition and off-target inhibition of TEC family kinases.2
The concomitant use of anticoagulants or platelet-inhibiting medications could increase the risk for bleeding in patients who are receiving ibrutinib therapy.6 In addition, ibrutinib increases the risk for atrial fibrillation, with a rate of 4% to 16%,7 which is often an indication for anticoagulation.2 In one clinical trial, approximately 55% of patients who were receiving ibrutinib were also receiving an anticoagulant, platelet-inhibiting medications, or both, suggesting that these medications could increase the risk for bleeding.8 In a multicenter, phase 2 study of patients with MCL who are receiving ibrutinib and an anticoagulant and/or platelet-inhibiting medications had an overall bleeding rate of 69% (grade 3 or 4, 8%) versus 28% (grade 3 or 4, 4%) with ibrutinib alone.8 However, most bleeding events in this study were low grade, and only 2 patients who were receiving concomitant anticoagulant medication discontinued ibrutinib treatment because of bleeding events.8
We examined the incidence of bleeding events in a real-world population of patients receiving ibrutinib therapy alone versus ibrutinib in combination with anticoagulants, platelet-inhibiting medications, or both. Our patients were followed closely by hematology/oncology clinical pharmacists, providing extensive and detailed long-term follow-up. We tested the hypothesis that adding anticoagulants and/or platelet-inhibiting medications to ibrutinib therapy would be associated with a greater risk for bleeding.
Methods
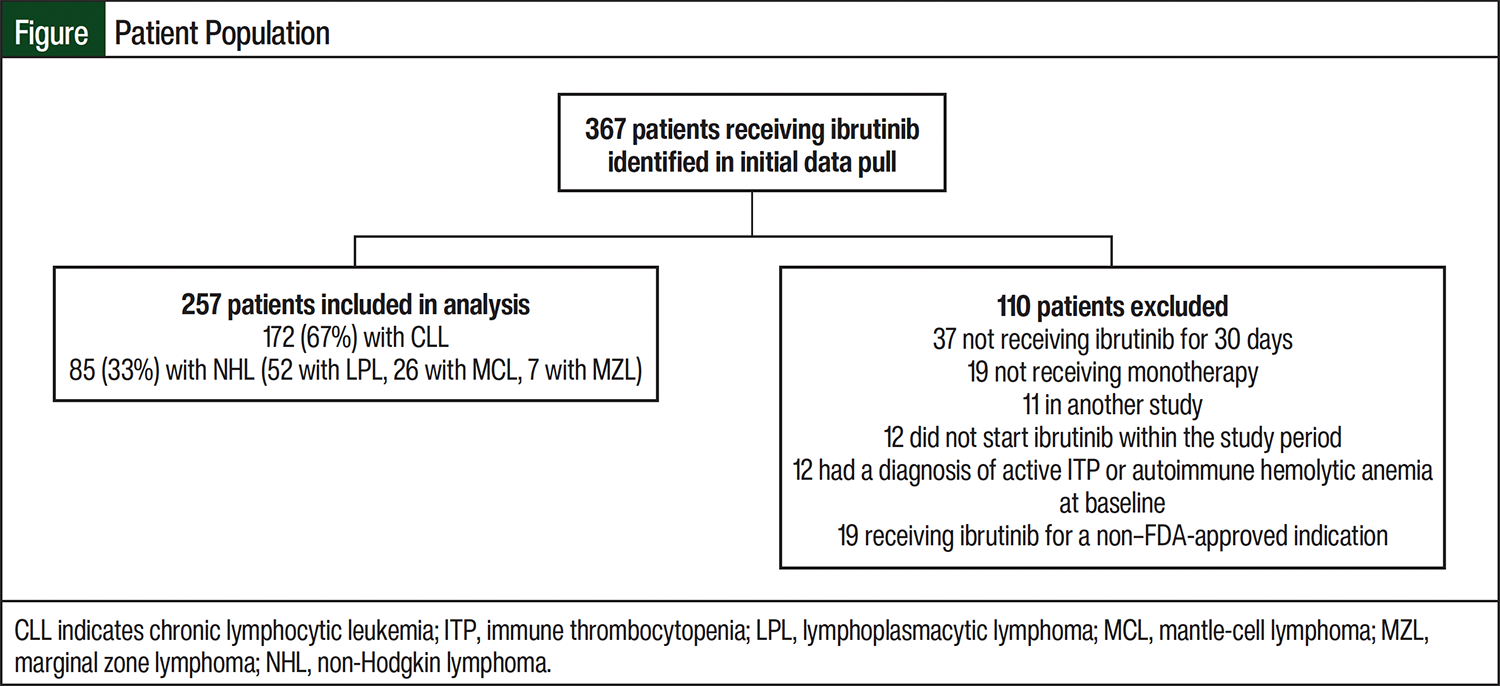
This was a retrospective, observational study of patients receiving ibrutinib between January 1, 2014, and July 1, 2018, at the University of Rochester Medical Center. The study was approved by the institution's research subjects review board. The patients were identified by an electronic report based on ibrutinib treatment and were screened for study inclusion. Patients were included in the study if they were aged ≥18 years, were receiving ibrutinib monotherapy for cancer, and received ≥30 days of treatment with ibrutinib during the study period. Patients were excluded from the study if they were enrolled in a clinical trial, receiving ibrutinib for a non–FDA-approved indication during the study period, or had a diagnosis of active autoimmune hemolytic anemia or immune thrombocytopenia at baseline.
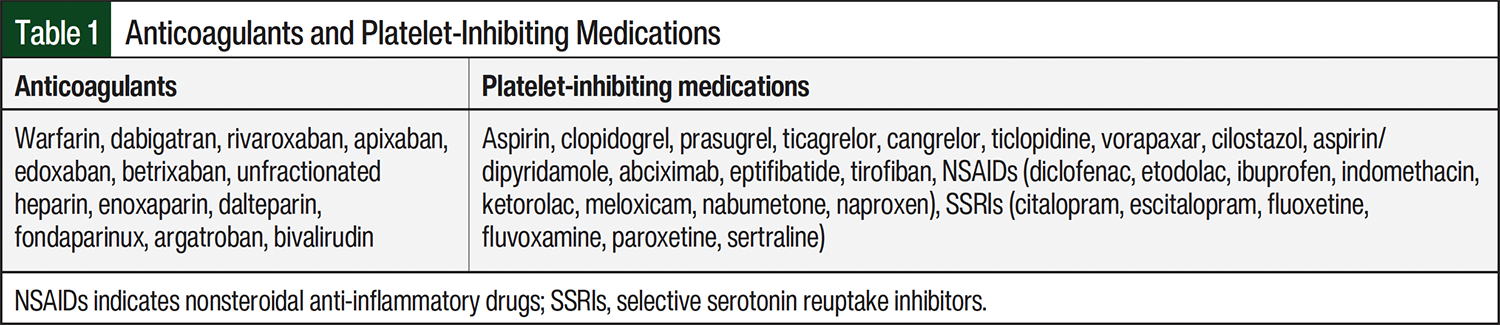
The primary objective was to evaluate the incidence of major bleeding events in patients who received ibrutinib alone or in combination with anticoagulant agents, medications with platelet-inhibiting properties, or both. Nonsteroidal anti-inflammatory drugs and selective serotonin reuptake inhibitors were included because of their platelet-inhibiting effects. A complete list of anticoagulant and platelet-inhibiting agents is shown in Table 1. The secondary objectives included the incidence of Common Terminology Criteria for Adverse Events (CTCAE) Version 4.0 grade 1 through grade 5 bleeding events,9 the incidence of bleeding (any bleeding event) among patients receiving ibrutinib for CLL versus other non-Hodgkin lymphomas (NHLs), and the incidence of bleeding rates in patients with CLL who received ibrutinib as first-line therapy versus therapy for relapsed or refractory disease (ie, previous treatment).
Data collection included patient demographics, such as age, sex, race, diagnosis (CLL or NHL and type), if previous treatment was received in the patients with a CLL diagnosis, if ibrutinib was received as first-line therapy or for relapsed or refractory disease in patients with CLL, the ibrutinib dose at initiation, and if an anticoagulant and/or a platelet-inhibiting medication was administered concomitantly (Table 1). Throughout the study period, patients could have been included in multiple groups depending on which concomitant medications they were receiving at the time of the bleeding.
Bleeding event occurrence throughout the ibrutinib treatment period was determined through documentation by the clinician, hematology/oncology clinical pharmacist, or nurse in the electronic medical record. A clinical pharmacist followed most of the patients receiving ibrutinib at planned intervals via telephone and at clinic visits, and their documentation of clinical information in the patients’ chart provided a collection of comprehensive data on AEs, adherence, and drug interactions that were used as a source for data collection. Additional information collected related to the bleeding event included the site where the bleeding occurred, the severity based on CTCAE criteria, and the action taken as a result of the bleeding event (ie, transfusion, hospitalization, discontinuation of offending agent[s]). If multiple bleeding events occurred in 1 patient, all bleeding events were captured if the patient was still receiving ibrutinib. Major bleeding was defined as any treatment-emergent hemorrhagic AEs of grade ≥3. Specific CTCAE definitions9 can be found for several AEs (bleeding site/location), and if a specific CTCAE definition does not exist, then the AE was graded according to the generic CTCAE criteria and was confirmed with a physician investigator.
Investigators collected data using a data dictionary and standardized electronic data collection tool. All data abstractors were trained in data collection procedures by one investigator using example patients to demonstrate the retrospective medical record review process and database input. This same investigator oversaw this process and assisted with any questions or clarifications regarding the data collection process or variables.
Statistical Analysis
The baseline characteristics are reported descriptively. To account for the varying lengths of follow-up contributed by each patient, the incidence rate of bleeding while receiving ibrutinib therapy was calculated as the number of observed bleeding events over the number of person-years at risk. Person-years were defined as the time from a patient’s initiation of ibrutinib until treatment discontinuation or the end of the study period, whichever occurred first. A mixed Poisson model was used to assess the change in the incidence of bleeding events in patients receiving ibrutinib alone versus patients receiving ibrutinib in combination with anticoagulants, platelet-inhibiting medications, or both. This allowed us to account for correlations between the data coming from the same patient and the varying amounts of person-time contributed within each risk period. The incidence rate ratios are also presented.
Results
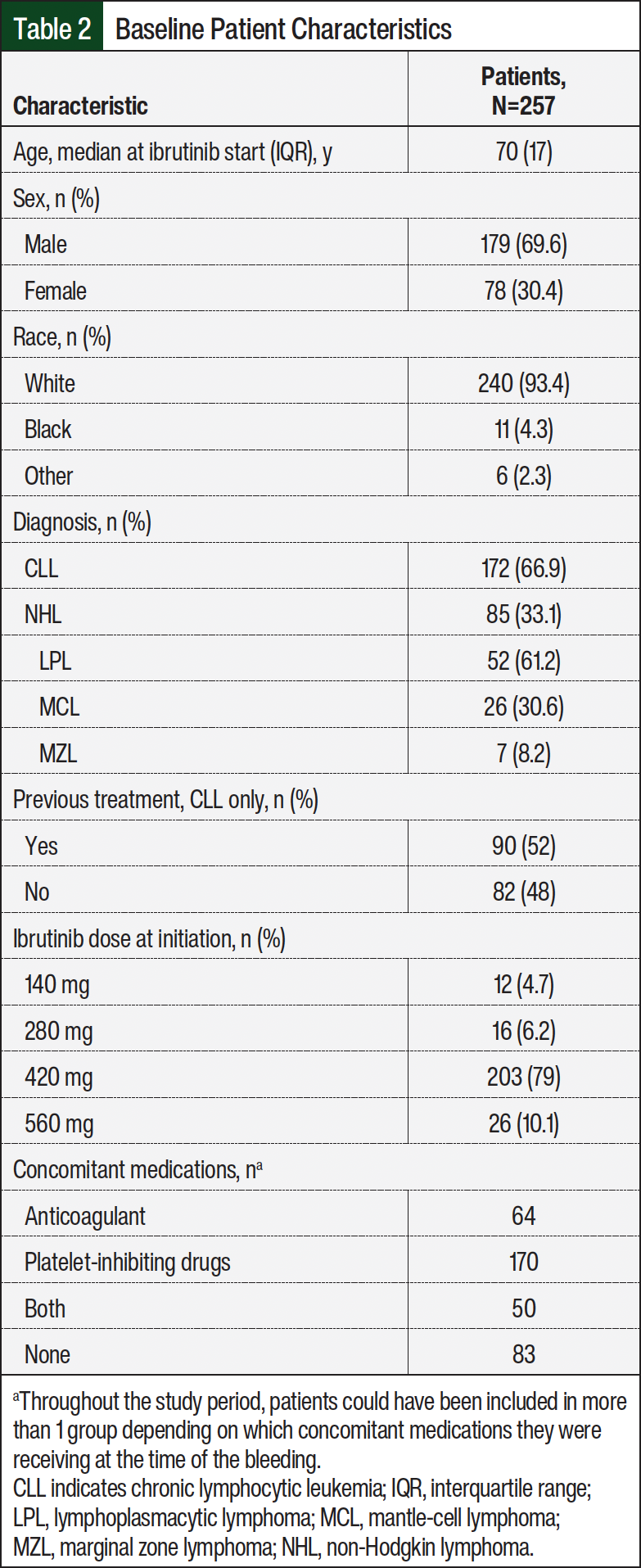
A total of 367 patients who were receiving ibrutinib were screened (Figure). Of those patients, 257 were included in the final analysis, contributing to a total of 386 person-years of follow-up. The most common reasons for exclusion from the study were not receiving ibrutinib monotherapy or not receiving treatment for at least 30 days (Figure). Most (67%) patients were receiving ibrutinib for CLL, and 33% were receiving treatment for other NHL diagnoses. Of the patients with other NHL, most (61.2%) had a diagnosis of lymphoplasmacytic lymphoma. The baseline characteristics of the patients in our study were similar to previous studies3,4 and are representative of the general population receiving ibrutinib (Table 2). A vast majority (95%) of the patients filled their ibrutinib prescriptions with the healthcare system’s internal specialty pharmacy and were followed closely by a hematology/oncology clinical pharmacist.
Throughout the study period, there were 64 patients receiving concomitant anticoagulants, 170 patients receiving platelet-inhibiting medications, 50 patients receiving both, and 83 patients who did not receive concomitant therapy (Table 2). With regard to anticoagulant medications, 41 patients received a heparin product (26 low–molecular-weight heparin and 15 unfractionated heparin). A total of 28 patients were receiving a direct oral anticoagulant (17 apixaban, 10 rivaroxaban, and 1 dabigatran) and 4 patients received warfarin. The most frequently received platelet-inhibiting medication was aspirin (111 patients). A total of 17 patients received clopidogrel, 2 received aspirin plus dipyridamole, and only 1 received ticagrelor. In addition, 41 patients were receiving selective serotonin reuptake inhibitors, and 56 patients were receiving nonsteroidal anti-inflammatory drugs during their treatment.
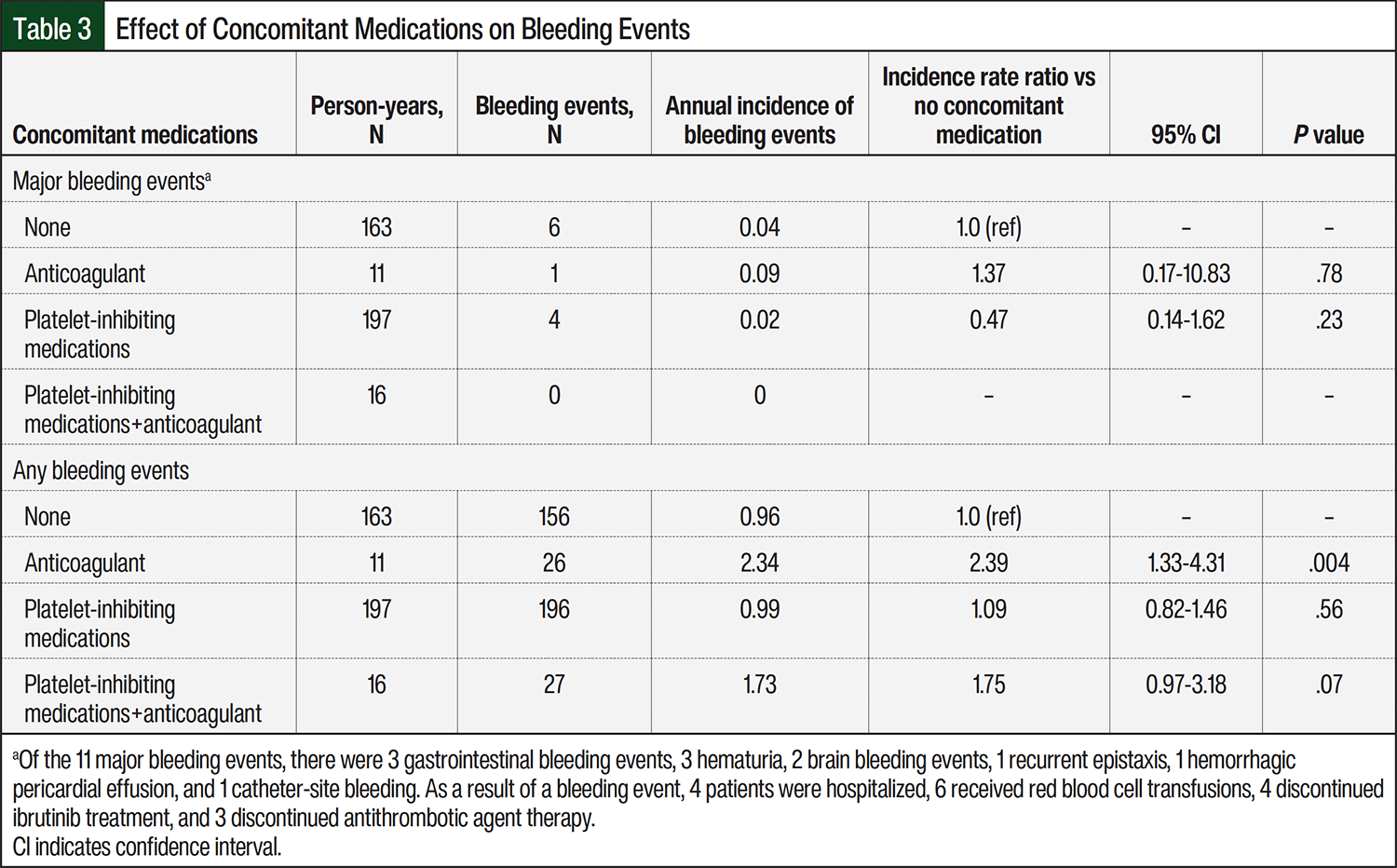
A total of 405 events of any grade were identified (1.05 bleeding events per person-year; 95% confidence interval [CI], 0.95-1.16; Table 3). Most (n=358, 88.4%) of the bleeding events were grade 1 (0.93 bleeding events per person-year; 95% CI, 0.83-1.03). There was no significant difference in the incidence of all bleeding events between the patients receiving either platelet-inhibiting medications only (197 person-years of follow-up; incidence rate ratio, 1.09; 95% CI, 0.82-1.46; P=.56) or platelet-inhibiting medications plus anticoagulant medications (16 person-years of follow-up; incidence rate ratio, 1.75; 95% CI, 0.97-3.18; P=.07) versus no concomitant therapy. Patients receiving concomitant anticoagulant medications without platelet-inhibiting medications had a significantly higher incidence of any bleeding compared with patients receiving ibrutinib alone (11 person-years of follow-up; incidence rate ratio, 2.39; 95% CI, 1.33-4.31; P=.004; Table 3).
In all, 11 major bleeding episodes (all grade 3) were identified in 11 unique patients (0.03 major bleeding events per person-year; 95% exact Poisson CI, 0.01-0.05). Of these patients, 1 was receiving an anticoagulant, 4 were receiving platelet-inhibiting medications, and 6 were not receiving concomitant therapy. All major bleeding events were grade 3, and there were no grade 4 or 5 events. Although most (n=10) of these patients were receiving 420 mg of ibrutinib, 1 patient was receiving 140 mg of ibrutinib. A total of 4 patients discontinued ibrutinib treatment secondary to their bleeding event. There were no significant differences between the groups receiving concomitant anticoagulant medications, platelet-inhibiting medications, or both in the occurrence of major bleeding events (Table 3).
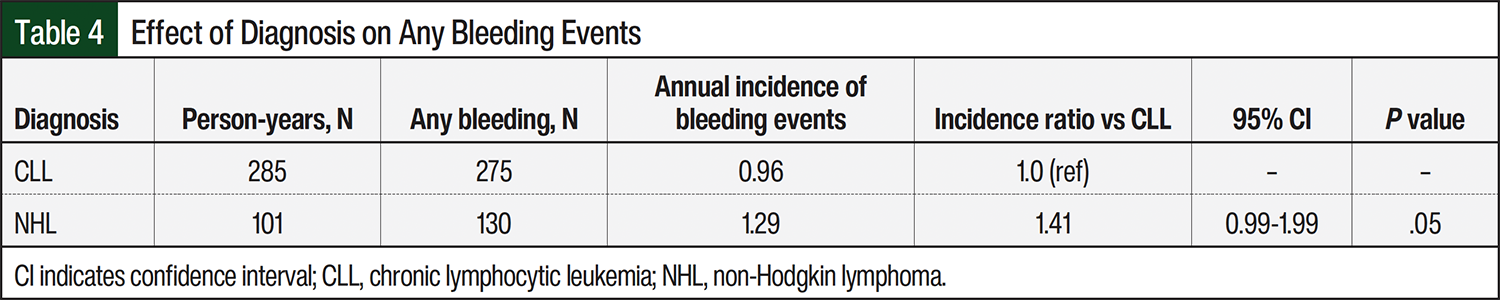
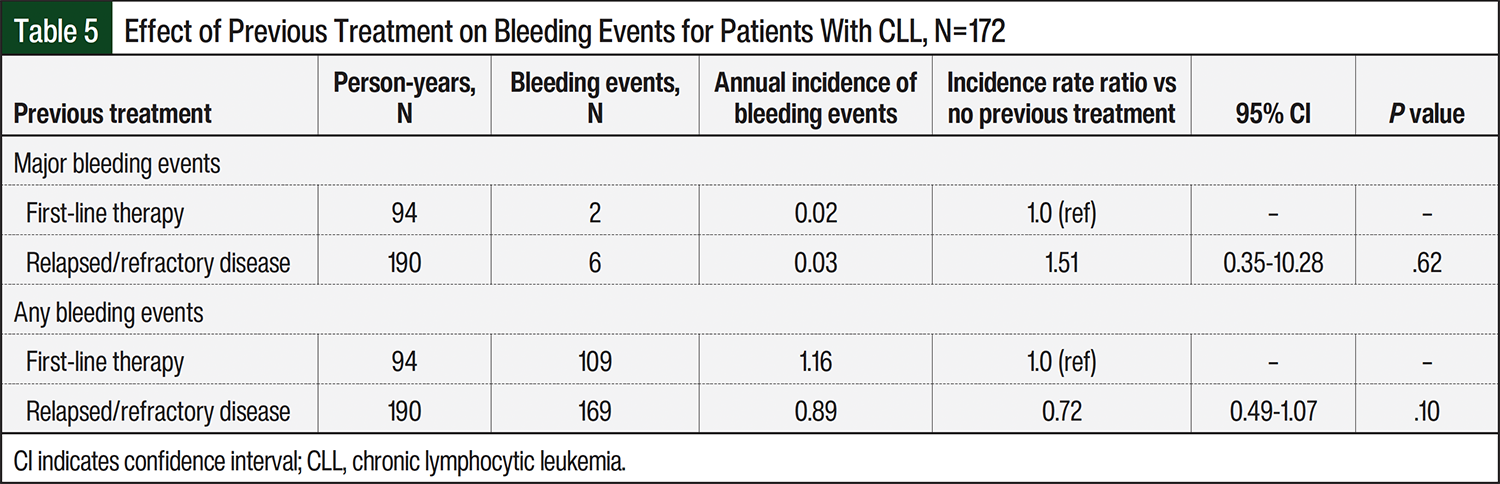
There were no significant differences between all patient diagnoses and the risks for all bleeding events or major bleeding events (Table 4). Of the 172 patients who were receiving ibrutinib treatment for CLL, those with relapsed or refractory disease did not have any significant increase in the risk for bleeding compared with those receiving first-line treatment (Table 5).
Discussion
Our observational study of a real-world population of patients with B-cell lymphoid malignancies who were receiving ibrutinib monotherapy showed that the addition of anticoagulant therapy but not platelet-inhibiting medications increased their risk for bleeding. Receiving treatment with anticoagulants did not increase the risk for major bleeding.
The rate of bleeding events in patients receiving ibrutinib is generally consistent with previous literature, which reported bleeding events in up to 44% of patients and major bleeding in 1% to 4% of patients during the duration of the study.3-5 In contrast, we reported the incidence of bleeding as bleeding events per person-year, which allowed us to account for correlations between data coming from the same patient and the varying amounts of person-time contributed within each risk period.
Our most clinically relevant finding is that the addition of platelet-inhibiting medications to ibrutinib therapy did not increase the risk for bleeding. The validity of this conclusion is supported by data from 197 person-years of follow-up. In contrast, we had less data for analysis on the risk for the addition of anticoagulants alone (11 person-years) or anticoagulants plus platelet-inhibiting medications (16 person-years). The low amount of person-years of follow-up could potentially explain the lack of a statistically significant increase in bleeding events in these groups.
Previous data on bleeding events in patients receiving ibrutinib in combination with antithrombotic medications are limited and inconclusive.3-8,10 Our study was consistent with a retrospective study of patients receiving ibrutinib for CLL, MCL, or Waldenström’s macroglobulinemia that reported 14 (3.2%) major bleeding events in 437 patients, half of which occurred in the absence of antithrombotic medications.10 Conversely, the results of another retrospective study of patients with CLL or MCL showed a major bleeding rate of 18% in a cohort of 71 patients.5 Of the 9 patients receiving an anticoagulant and a platelet-inhibiting medication, 78% had a major bleeding event, indicating a much higher risk with concomitant therapy than in our study.5
Our relatively low rate of bleeding events could be a result of the integration of specialty pharmacists into the care team at our institution. These hematology/oncology clinical pharmacists work closely with patients in various oncology clinics and provide counseling services to patients, closely monitor for AEs with frequent phone check-ins, and provide medication delivery programs. Pharmacists also perform medication reviews to avoid drug–drug interactions and drug AEs. Patients are counseled to hold BTK inhibitors around surgical procedures for 3 days before and after low or moderate bleeding risk procedures and for 7 days before and after high bleeding risk procedures. The close follow-up by hematology/oncology clinical pharmacists could be reducing patient risk by identifying and resolving at-risk issues and thereby reducing the risk for major bleeding events.
Limitations
Our study was strengthened by the statistical analysis that allowed us to account for multiple events from the same patient, as well as close pharmacist follow-up and documentation during the study period. Our study was limited to retrospective data collection, and therefore we only captured bleeding events recorded in the electronic medical record. This methodology is unlikely to have resulted in underestimating the incidence of major bleeding, because these events are of high acuity and likely require medical intervention and documentation, but could have decreased our capture of minor bleeding events.
Another potential limitation of the study is the fact that ibrutinib is currently the oldest BTK inhibitor available on the market, with newer and more specific agents now available. It is unknown how these data may translate to the other agents within this class.
Conclusion
Our data show that the use of BTK inhibitors may allow for the cautious addition of anticoagulant therapy without increasing the risk for major bleeding events. In addition, there was no increase in the risk for bleeding in patients receiving BTK inhibitors and platelet-inhibiting medications.
Acknowledgments
We would like to acknowledge Nicole M. Acquisto, PharmD, FASHP, FCCM, FCCP, BCCCP, for assistance with manuscript review and editing; Dana Cohan, PharmD; Kyle Farina, PharmD; Nathan Heitz, PharmD; Evan Horan, PharmD; and Zerina Zornic, PharmD, for assistance with data collection. We would also like to acknowledge the University of Rochester Pharmacy Research committee for its thoughtful review and suggestions.
Funding Source
This study was supported in part by the Cadregari Endowment Fund at the Wilmot Cancer Institute.
Author Disclosure Statement
Dr Moore is an Advisory Board participant at BeiGene; Dr Treptow is currently employed by GlaxoSmithKline, but was employed by the University of Rochester Medical Center at the time this research was conducted; Dr Delibert is currently employed by Astellas Pharma, but was employed by the University of Rochester Medical Center at the time this research was conducted; Dr Zent has received research funding from Acerta Pharma/AstraZeneca and Genmab; Dr Lattuca and Ms Baran have no conflicts of interest to report.
References
- Caron F, Leong DP, Hillis C, et al. Current understanding of bleeding with ibrutinib use: a systematic review and meta-analysis. Blood Adv. 2017;1:772-778.
- Aguilar C. Ibrutinib-related bleeding: pathogenesis, clinical implications and management. Blood Coagul Fibrinolysis. 2018;29:481-487.
- Byrd JC, Brown JR, O’Brien S, et al. Ibrutinib versus ofatumumab in previously treated chronic lymphoid leukemia. N Engl J Med. 2014;371:213-223.
- Burger JA, Tedeschi A, Barr PM, et al. Ibrutinib as initial therapy for patients with chronic lymphocytic leukemia. N Engl J Med. 2015;373:2425-2437.
- Kunk PR, Mock J, Devitt ME, et al. Major bleeding with ibrutinib: more than expected. Blood. 2016;128:3229.
- Shatzel JJ, Olson SR, Tao DL, et al. Ibrutinib-associated bleeding: pathogenesis, management and risk reduction strategies. J Thromb Haemost. 2017;15:835-847.
- Ganatra S, Sharma A, Shah S, et al. Ibrutinib-associated atrial fibrillation. JACC Clin Electrophysiol. 2018;4:1491-1500.
- Wang ML, Blum KA, Martin P, et al. Long-term follow-up of MCL patients treated with single-agent ibrutinib: updated safety and efficacy results. Blood. 2015;126:739-745.
- US Department of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE). Version 4. 2010. Accessed June 8, 2023. https://dctd.cancer.gov/research/ctep-trials/trial-development/ctcae-v4.03.xlsx
- Pavlik A, Barr H, Dotson E, et al. Major bleeding complications among patients treated with ibrutinib and concomitant antiplatelet, anticoagulant, or supplemental therapy. Blood. 2016;128:4387.