Connecting Science to Practice
Irinotecan remains an important backbone chemotherapeutic agent in the treatment of various types of malignancies. Although many common adverse events are manageable, irinotecan-induced muscle cramping and hypertonia are rare but serious complications. This case report describes a patient who had irinotecan-induced muscle cramping and hypertonia, likely exacerbated by underlying hypokalemia and cholinergic overstimulation, ultimately resulting in treatment discontinuation. Given the severity of these symptoms, clinicians should closely monitor and correct electrolyte imbalances throughout treatment and provide anticholinergic therapies when appropriate. Early recognition and proactive management of irinotecan-induced muscle cramping and hypertonia can help prevent treatment interruption and improve patient outcomes.
Irinotecan, a topoisomerase I inhibitor, is extensively prescribed for the treatment of various tumors. Irinotecan is hepatically metabolized to its active metabolite, SN-38, which enhances the stability of the topoisomerase I-DNA complex, thus preventing the relegation of strands and leading to DNA breaks and cell apoptosis.1-5 SN-38 is further metabolized in the liver through conjugation by UDP-glucuronosyltransferase 1A1 and is eliminated via biliary excretion.1-5
The adverse events (AEs) that are frequently associated with irinotecan include myelosuppression, diarrhea, nausea, vomiting, alopecia, weakness, and neutropenia. Irinotecan-induced cholinergic syndrome is another well-known AE.1,3,6 Acute symptoms may occur within the first 24 hours after the infusion of irinotecan and can include diarrhea, flushing, diaphoresis, lacrimation, bradycardia, rhinorrhea, and peristaltic abdominal pain.1,6 Delayed diarrhea can occur more than 24 hours after the administration of irinotecan and is often induced by the reactivation of SN-38 within the intestinal lumen, leading to direct mucosal damage and AEs.6,7 To manage cholinergic symptoms, treatment with anticholinergic medications, such as atropine, and antidiarrheal medications, such as loperamide or diphenoxylate and atropine, can be used.1,5,7 For severe, delayed diarrhea, treatment with a somatostatin analog, such as octreotide, may be necessary.5-7 Some rare, but serious AEs, including muscle twitching, dysarthria, and generalized weakness, may occur during or after the infusion of irinotecan and may be related to other underlying causes.5-13
Case Report
We present the case of a 58-year-old woman who had upper thoracic back pain with radiation into the interscapular region, associated chest pain, and abdominal discomfort for several months. A thoracic spine magnetic resonance imaging scan revealed vertebral T3-T4 neoplastic process with epidural extension that was causing spinal cord or nerve root compression. Subsequently, the patient underwent multiple computed tomography scans and an endoscopic ultrasound, which revealed adenocarcinoma of the pancreas with metastatic lesions in the skull, femur, and liver. In addition, she had a notable history of ovarian cyst, severe endometriosis, back pain, depression, anxiety, and hypertension.
A month later, the patient presented to our infusion center and was initiated on systemic chemotherapy with 5-fluorouracil 400 mg/m2 intravenous bolus, irinotecan 180 mg/m2, and oxaliplatin 85 mg/m2 (FOLFIRINOX), followed by 2.4 g/m2 of continuous infusion (leucovorin was omitted) and concomitant radiation therapy. She tolerated the first 3 chemotherapy cycles without any issues. During the fourth cycle, however, she had mild cramping in one of her upper extremities and had progressive neuropathy in her lower and upper extremities after the infusion. The primary oncologist was notified and intravenous dexamethasone was administered to resolve the patient’s symptoms.
Electrolytes were drawn before the treatment fourth cycle, and the patient had mild hypokalemia with a potassium level of 3.3 mmol/L (normal range, 3.5-5.2 mmol/L); her corrected calcium (9.48 mg/dL; normal range, 8.4-10.3 mg/dL) and magnesium (1.9 mg/dL; normal range, 1.6-2.6 mg/dL) levels were within the normal limit. It is unknown whether the patient’s symptoms were alleviated after receiving dexamethasone or if the symptoms self-resolved after discharge.
Two weeks later, the patient returned for her fifth chemotherapy cycle, and the oxaliplatin infusion was successfully completed. Before treatment, she continued to have mild hypokalemia, with a potassium level of 3.2 mmol/L, a normal corrected calcium level (9.04 mg/dL), and a slightly elevated magnesium level (2.7 mg/dL). After 42 minutes of starting the irinotecan infusion, the patient complained of rhinorrhea, cramping in both hands, and weakness in her bilateral upper extremities. Consequently, the infusion rate was reduced from 195 mL/hr to 150 mL/hr, after which her rhinorrhea subsided without further intervention. However, the patient continued to have persistent cramping in the bilateral upper extremities. The irinotecan infusion was then held, and her primary oncologist was notified.
On physical examination, the patient presented with hypertonia of bilateral hands and fingers, and she received an intravenous dose of atropine 0.25 mg. In addition, she received an intravenous dose of diphenhydramine 25 mg in case she had drug-induced hypersensitivity reaction; however, the patient denied having any such symptoms, and her vitals were stable. Her symptoms persisted for the next half hour with slowed improvement, and she continued to have mild cramping in both feet that lasted for another 50 minutes. Once the patient was stable, the rest of the chemotherapy regimen was administered, and the patient was stable and was discharged home. Her oncologist discontinued subsequent treatment with irinotecan to prevent further AEs. The patient did not have any additional muscle cramping, weakness, or hypertonia with subsequent treatment cycles.
Discussion
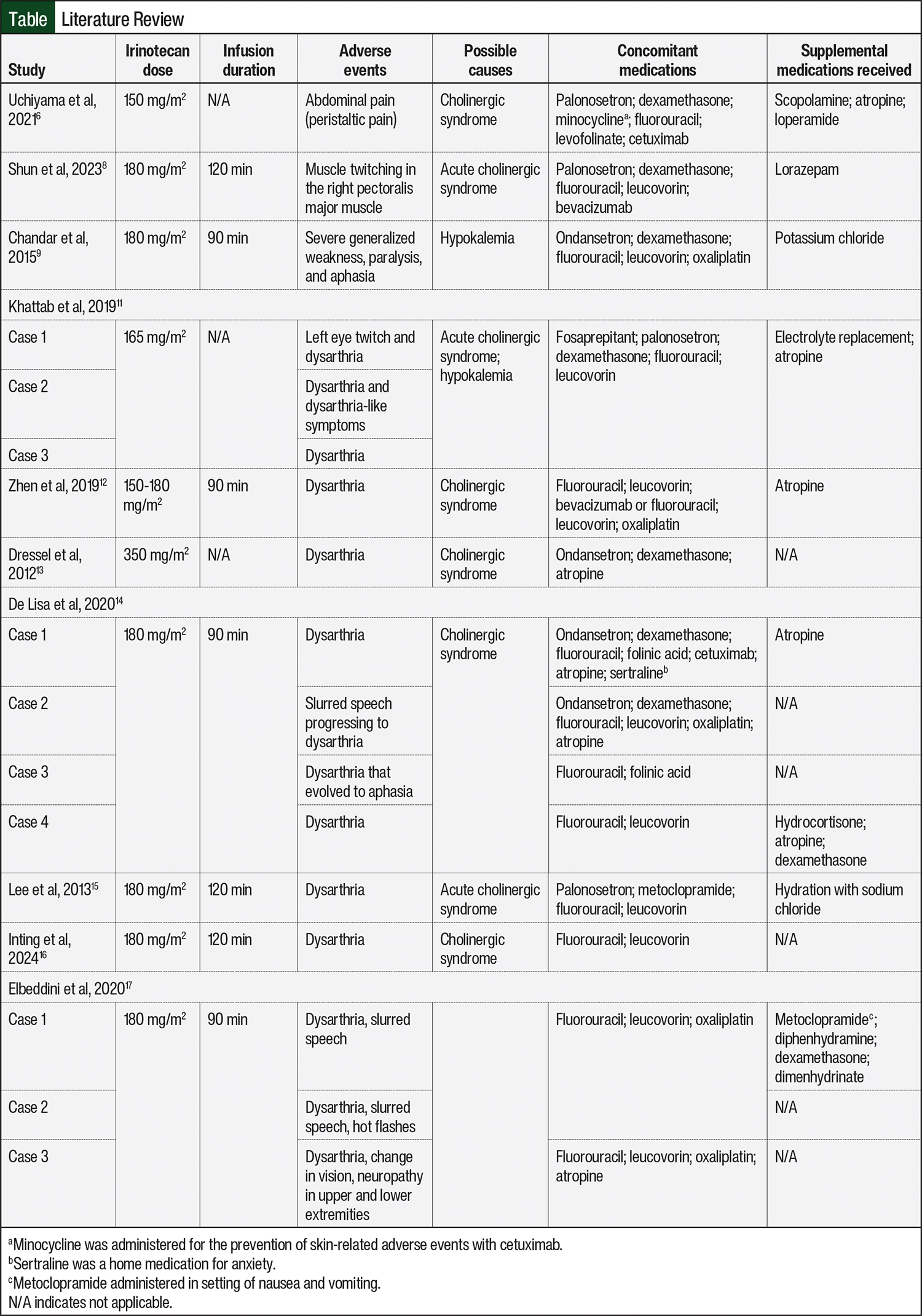
To date, the most frequent infusion reactions with irinotecan treatment are rhinorrhea, dysarthria, and generalized weakness,5,10-13 with dysarthria being the most common AE in the literature that we reviewed (Table6,8,9,11-17). To our knowledge, there are currently no other cases of irinotecan-induced muscle cramping and hypertonia. The underlying causes that can lead to these symptoms include electrolyte disturbances, particularly hypokalemia, or an excess of the neurotransmitter acetylcholine.5,10,13
In a study by Chandar and de Wilton Marsh, a patient with mild hypokalemia had generalized weakness and paralysis of extremities during an infusion of irinotecan.9 The researchers concluded that the coadministration of irinotecan and oxaliplatin may have resulted in severe symptoms in the setting of an electrolyte imbalance.9 In another study, Giampieri and colleagues proposed that in patients receiving oxaliplatin-containing regimens, an electrolyte imbalance can lead to motor impairment, particularly hypokalemia versus hypocalcemia or hypomagnesemia.10 Our patient had mild hypokalemia during the fourth and fifth treatment cycles, where she received oxaliplatin followed by irinotecan. Therefore, it is possible that the patient’s symptoms were exacerbated as a result of hypokalemia at baseline and the coadministration of oxaliplatin and irinotecan.
Muscle cramping or weakness can also be exacerbated by an increase in the production of acetylcholine.13,14 Acetylcholine can bind to cholinergic (muscarinic and nicotinic) receptors and lead to symptoms such as rhinorrhea and muscle and abdominal cramping.5,6,8-10,13 Irinotecan inhibits the acetylcholinesterase enzyme, thus preventing metabolism and promoting the accumulation of acetylcholine within the system, leading to muscle weakness or cramping.6 Harel and colleagues proposed that the accumulation of acetylcholine at the hypoglossal nerve can lead to the overstimulation of the hypoglossal nerve, leading to dysarthria.18 Increased accumulation of and overstimulation from acetylcholine at various nerve endings can explain the hypertonia and weakness in our patient. Our patient received a single dose of atropine, an anticholinergic medication, to help alleviate her symptoms; however, this had minimal effect and resulted in slow improvement of her AEs, which could be indicative of a need for additional increased doses of atropine or of other potential etiologies that can contribute to the AEs.
Another rare AE, irinotecan-induced involuntary muscle twitching, has also been reported in a few patients who received treatment with irinotecan; however, the mechanism of such a reaction has not been fully explained.8,19,20 Shun and colleagues effectively managed and prevented irinotecan-induced muscle twitching in a patient by administering intravenous lorazepam before the initiation of irinotecan treatment.8 This approach was based on the known mechanism of benzodiazepines in the treatment of myoclonus, by facilitating gamma-aminobutyric acid receptors and thereby mitigating symptoms. By administering lorazepam before the chemotherapy infusion, they were able to improve the patient’s overall tolerance to the treatment and reduce the incidence of this particular AE.8
Certain nonpharmacologic strategies may be used to mitigate irinotecan-induced AEs. Patients with a heterozygous or homozygous UGT1A*28 allele have an increased risk for irinotecan-induced AEs, because the presence of this allele can lead to reduced elimination of the active metabolite.4 Pharmacogenomics testing for the UGT1A*28 allele can help identify patients who have an increased risk for such reactions.4 In addition, Lee and colleagues and Ramirez and colleagues have proposed that extending the duration of irinotecan infusions may help reduce the incidence of dysarthria.15,21 Their research suggests that shorter infusion times may lead to a rapid accumulation of SN-38, which may contribute to dysarthria.15,21
Conclusion
We present a rare case of irinotecan-induced muscle cramping and hypertonia in a patient with stage IV metastatic pancreatic adenocarcinoma who was receiving FOLFIRINOX. The patient started having muscle cramping, bilateral extremity weakness, and hypertonia during her irinotecan infusions. This patient’s symptoms persisted despite the infusion rate of irinotecan treatment being lowered and receiving pharmacologic treatment with steroids and anticholinergic medications. This case highlights that underlying conditions, such as electrolyte imbalances, at baseline or that result from the concomitant administration of other medications, can lead to muscle weakness and motor impairment. Therefore, electrolytes should be monitored and repleted before and during each irinotecan infusion.
Irinotecan can also lead to the possible accumulation of acetylcholine and the overstimulation of nerve endings, which results in cholinergic syndrome and requires treatment with anticholinergic agents. Increased doses of anticholinergic medications during or before treatment may help alleviate such symptoms. Nonpharmacologic approaches, including pharmacogenomics testing for the UGT1A*28 allele and extending infusion durations, can be used to mitigate AEs. These strategies may help improve patient outcomes by reducing AEs and preventing the need for treatment interruption or discontinuation. Further research is warranted to fully understand the etiology and management of such severe reactions to prevent treatment discontinuation.
Author Disclosure Statement
Dr Mishra, Dr Gohil, Ms Gu, Ms Zhikharev, and Dr Jhawer have no conflicts of interest to report.
References
- Camptosar (irinotecan hydrochloride) injection, for intravenous use [prescribing information]. Pharmacia & Upjohn Company; June 2024. Accessed April 3, 2025. https://labeling.pfizer.com/ShowLabeling.aspx?id=533
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): pancreatic adenocarcinoma. Updated February 3, 2025. Accessed April 3, 2025. www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf
- Fujita K, Kubota Y, Ishida H, Sasaki Y. Irinotecan, a key chemotherapeutic drug for metastatic colorectal cancer. World J Gastroenterol. 2015;21:12234-12248.
- Karas S, Innocenti F. All you need to know about UGT1A1 genetic testing for patients treated with irinotecan: a practitioner-friendly guide. JCO Oncol Pract. 2022;18:270-277.
- Bailly C. Irinotecan: 25 years of cancer treatment. Pharmacol Res. 2019;148:104398.
- Uchiyama K, Saito Y, Takekuma Y, et al. Alleviation of abdominal pain due to irinotecan-induced cholinergic syndrome using loperamide: a case report. Case Rep Oncol. 2021;14:806-811.
- Stein A, Voigt W, Jordan K. Chemotherapy-induced diarrhea: pathophysiology, frequency and guideline-based management. Ther Adv Med Oncol. 2010;2:51-63.
- Shun YT, Lai HY, Chuang YT, Lin HF. Successful treatment of irinotecan-induced muscle twitching: a case report. Clin Med Insights Case Rep. 2023;16:11795476221150354.
- Chandar M, de Wilton Marsh R. Severe generalized weakness, paralysis, and aphasia following administration of irinotecan and oxaliplatin during FOLFIRINOX chemotherapy. Case Rep Oncol. 2015;8:138-141.
- Giampieri R, Maccaroni E, Sotte V, et al. Acute peripheral motor neuropathy induced by oxaliplatin-correlated hypokalaemia. Oncol Ther. 2020;8:161-169.
- Khattab A, Park SJ, Patruni S, et al. Case series of irinotecan-induced-dysarthria: a review of literature and proposition of a pre-medication regimen. Mathews J Gastroenterol Hepatol. 2019;4:13.
- Zhen DB, McDevitt RL, Zalupski MM, Sahai V. Irinotecan-associated dysarthria: a single institution case series with management implications in patients with gastrointestinal malignancies. J Oncol Pharm Pract. 2019;25:980-986.
- Dressel AJ, van der Mijn JC, Aalders IJ, et al. Irinotecan-induced dysarthria. Case Rep Oncol. 2012;5:47-51.
- De Lisa M, Ballatore Z, Marcantognini G, et al. Irinotecan-induced transient dysarthria: case series and updated literature review. Oncol Ther. 2020;w8:147-160.
- Lee KA, Kang HW, Ahn JH, et al. Dysarthria induced by irinotecan in a patient with colorectal cancer. Am J Health Syst Pharm. 2013;70:1140-1143.
- Inting KAE, Piano AN. Irinotecan-induced transient dysarthria in a patient with metastatic colorectal cancer: a case report. Cureus. 2024;16:e54416.
- Elbeddini A, Hooda N, Gazarin M, et al. Irinotecan-associated dysarthria in patients with pancreatic cancer: a single site experience. Am J Case Rep. 2020;21:e924058.
- Harel M, Hyatt JL, Brumshtein B, et al. The crystal structure of the complex of the anticancer prodrug 7-ethyl-10-[4-(1-piperidino)-1piperidino]-carbonyloxycamptothecin (CPT-11) with Torpedo californica acetylcholinesterase provides a molecular explanation for its cholinergic action. Mol Pharmacol. 2005;67:1874-1881.
- Naviglio S, Rabusin M. Irinotecan-induced muscular contractions. Pediatr Blood Cancer. 2018;65:e26948.
- Peters SS, Bettinger J, Philip J, et al. Irinotecan-induced muscle twitching from a possible drug interaction: a case report. J Oncol Pharm Pract. 2018;24:555-560.
- Ramirez KG, Koch MD, Edenfield WJ. Irinotecan-induced dysarthria: a case report and review of the literature. J Oncol Pharm Pract. 2017;23:226-230.