Connecting Science to Practice
Patients with cancer are at increased risk for venous thromboembolism,
and those with gastrointestinal cancers may have a
higher risk for bleeding with direct oral anticoagulants
(DOACs) compared with low-molecular-weight heparin
(LMWH), according to recent landmark trials. NCCN guidelines
recommend using LMWH rather than DOACs in this
population. However, several physicians from our institution
preferentially prescribe DOACs over LMWH. Our single-center, retrospective study evaluated real-world data from
our institution to assess the bleeding risks associated with
apixaban and rivaroxaban. Our study showed a numerically
lower bleeding risk with apixaban. These findings may support
the use of apixaban as a viable alternative to rivaroxaban
or LMWH in select patients with gastrointestinal cancers.
Drug selection would involve careful consideration of patient
preference, adherence, and financial burden.
Patients with cancer have a higher risk for venous thromboembolism (VTE), which can lead to further complications and death.1 VTE can be caused by a number of factors, including the primary cancer site, the chemotherapy regimen, and the stage of the cancer.2 The risk for VTE increases further in the presence of metastatic disease.3,4 Although cancer itself can lead to a higher risk for VTE, certain types of cancers are associated with increased risk, such as gastric, lung, and pancreatic cancers.2
There is a 4- to 8-fold increased mortality rate for patients with cancer who had a VTE event compared with patients without cancer who had a VTE event, demonstrating the need for optimal treatment in these patients.2 Current NCCN guidelines recommend the use of low-molecular-weight heparin (LMWH) or direct oral anticoagulants (DOACs) for the treatment of VTE in patients with cancer.3
The SELECT-D trial compared rivaroxaban with dalteparin, and the CARAVAGGIO trial compared apixaban with dalteparin for the treatment of VTE in patients with cancer.5,6 In the SELECT-D trial, the primary outcome of VTE recurrence was 4% in the rivaroxaban group versus 11% in the dalteparin group.5 In the CARAVAGGIO trial, VTE recurrence was 5.6% in the apixaban group versus 7.9% in the dalteparin group.6 The 2 trials also examined major bleeding and clinically relevant nonmajor bleeding as safety outcomes. The SELECT-D trial showed a higher trend for major bleeding, of 6% for the rivaroxaban group versus 4% for the dalteparin group, and clinically relevant nonmajor bleeding was 13% for the rivaroxaban group versus 4% for the dalteparin group.5 Patients with gastrointestinal (GI) cancer, specifically esophageal or gastroesophageal cancer, had a higher risk for bleeding. The CARAVAGGIO trial’s results showed no increased risk for major bleeding (3.8% in the apixaban group vs 4% in the dalteparin group). The rates of clinically relevant nonmajor bleeding were numerically higher with apixaban than dalteparin (9% in the apixaban group vs 6% in the dalteparin group).6
As a result of the risk for major bleeding in these trials, the NCCN’s guidelines recommend treatment with LMWH for patients with gastric or gastroesophageal cancer.3 In real-world practice at the Winship Cancer Institute, GI physicians prescribe DOACs and LMWH for the treatment of cancer-associated VTE. The purpose of this study is to assess the real-world outcomes of patients with GI cancer who received apixaban or rivaroxaban for VTE, including for bleeding, and to determine the risk for VTE recurrence.
Methods
This single-center, retrospective chart review was approved by the institutional review board at Emory University and the Winship Cancer Institute. Winship Cancer Institute, a National Cancer Institute–designated Comprehensive Cancer Center, is part of Emory Healthcare, which spans across several sites in the greater Atlanta, GA, area. This study included patients aged ≥18 years who were diagnosed with a primary GI cancer between January 1, 2017, and December 31, 2021, and were prescribed apixaban or rivaroxaban for the treatment of VTE by an Emory Healthcare provider. Primary GI cancer included esophageal, gastric, appendiceal, liver, pancreatic, duodenal, colon, rectal, anal, and gallbladder cancers. Patients were excluded from the study if they were prescribed apixaban or rivaroxaban by anyone other than an Emory Healthcare provider, were prescribed an initial dose that deviated from the standard starting dose of 10 mg twice daily for 7 days followed by 5 mg twice daily for apixaban or 15 mg twice daily for 21 days followed by 20 mg daily for rivaroxaban, had a VTE other than deep vein thrombosis (DVT) or pulmonary embolism (PE), such as port-induced clots, or were prescribed the medication for an indication other than VTE.
Clinical information was collected through a data warehouse system and via retrospective chart review through Emory’s electronic medical record system for patients meeting all inclusion criteria within the specified date range.
The primary outcome was the rates of major bleeding with apixaban and rivaroxaban at any point throughout therapy. Major bleeding was defined as a decrease in hemoglobin of ≥2 g/dL over 24 hours, a transfusion of ≥2 units of red blood cells, bleeding that occurs in critical sites (intracranial, intraspinal, intraocular, pericardial), or bleeding that necessitates surgical intervention. The secondary outcomes included clinically relevant nonmajor bleeding at any point during therapy, the recurrence of VTE within 6 months, or death from bleeding. Clinically relevant nonmajor bleeding was defined as any bleeding that compromises hemodynamics, such as hypotension or irregular heart rate, epistaxis or gingival bleeding requiring tamponade or other medical intervention, hemoptysis, hematemesis, spontaneous rectal bleeding, spontaneous macroscopic hematuria, or hematoma >25 cm2. If a patient had a primary outcome of major bleeding, they were not included in the secondary outcome of having clinically relevant nonmajor bleeding.
Statistical Analysis
SAS version 9.4 (SAS Institute, Inc; Cary, NC) was used for all data analyses. The significance level (α) was set at .05. Baseline characteristics between the 2 groups (apixaban vs rivaroxaban) were compared using chisquare or Fisher’s exact tests for categorical covariates and the Analysis of Variance or Kruskal-Wallis tests for numerical covariates. Univariate logistic regression was used to compare the proportions of patients with the incidence of major bleeding, clinically relevant nonmajor bleeding, and VTE recurrence between the 2 groups. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated to evaluate the strengths of observed associations. Chi-square or Fisher’s exact tests were used to compare rectal bleeding and hematuria between the treatment groups.
Results
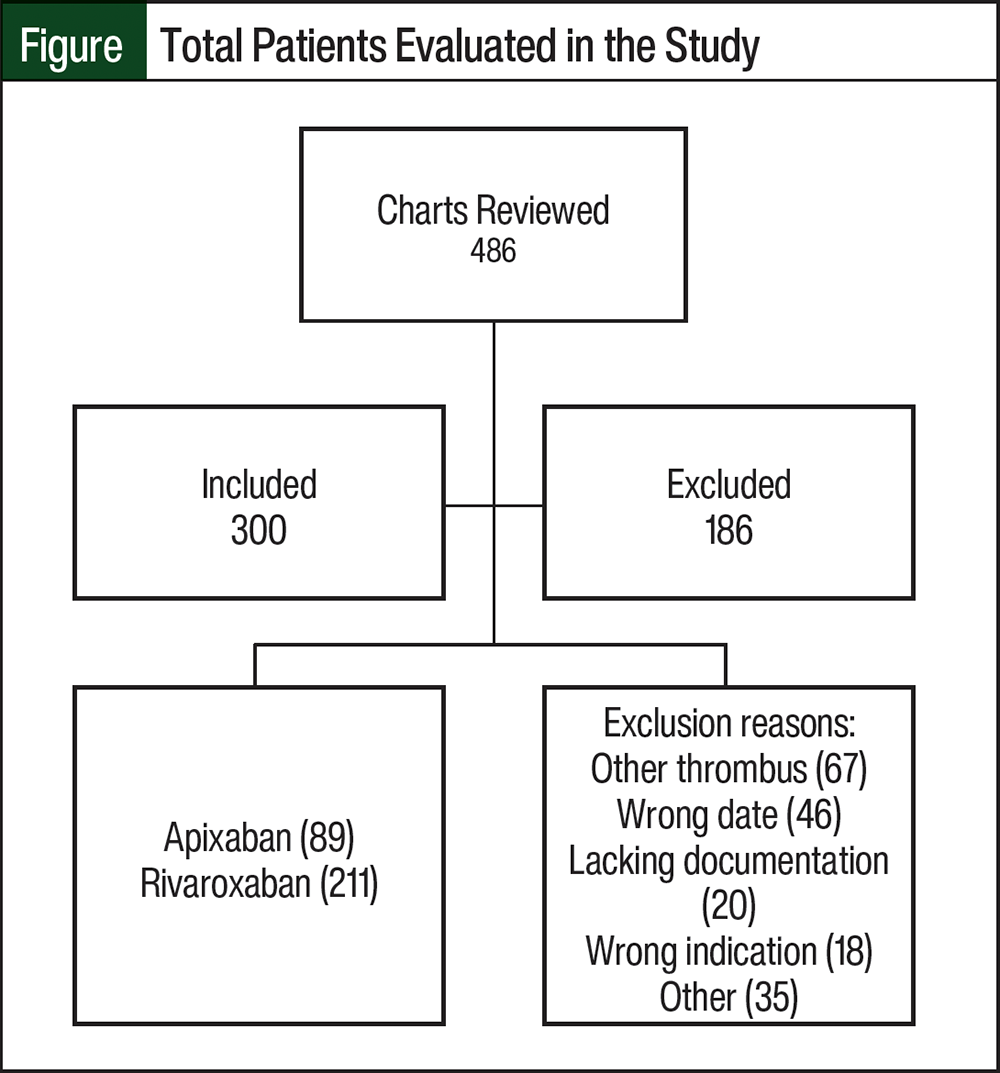
A total of 486 patients were evaluated to receive apixaban or rivaroxaban between 2017 and 2021 (Figure). Of these patients, 186 were excluded mainly because they had VTEs other than DVT or PE. Thus, the study included 300 patients, with 89 (30%) patients in the apixaban group and 211 (70%) in the rivaroxaban group.
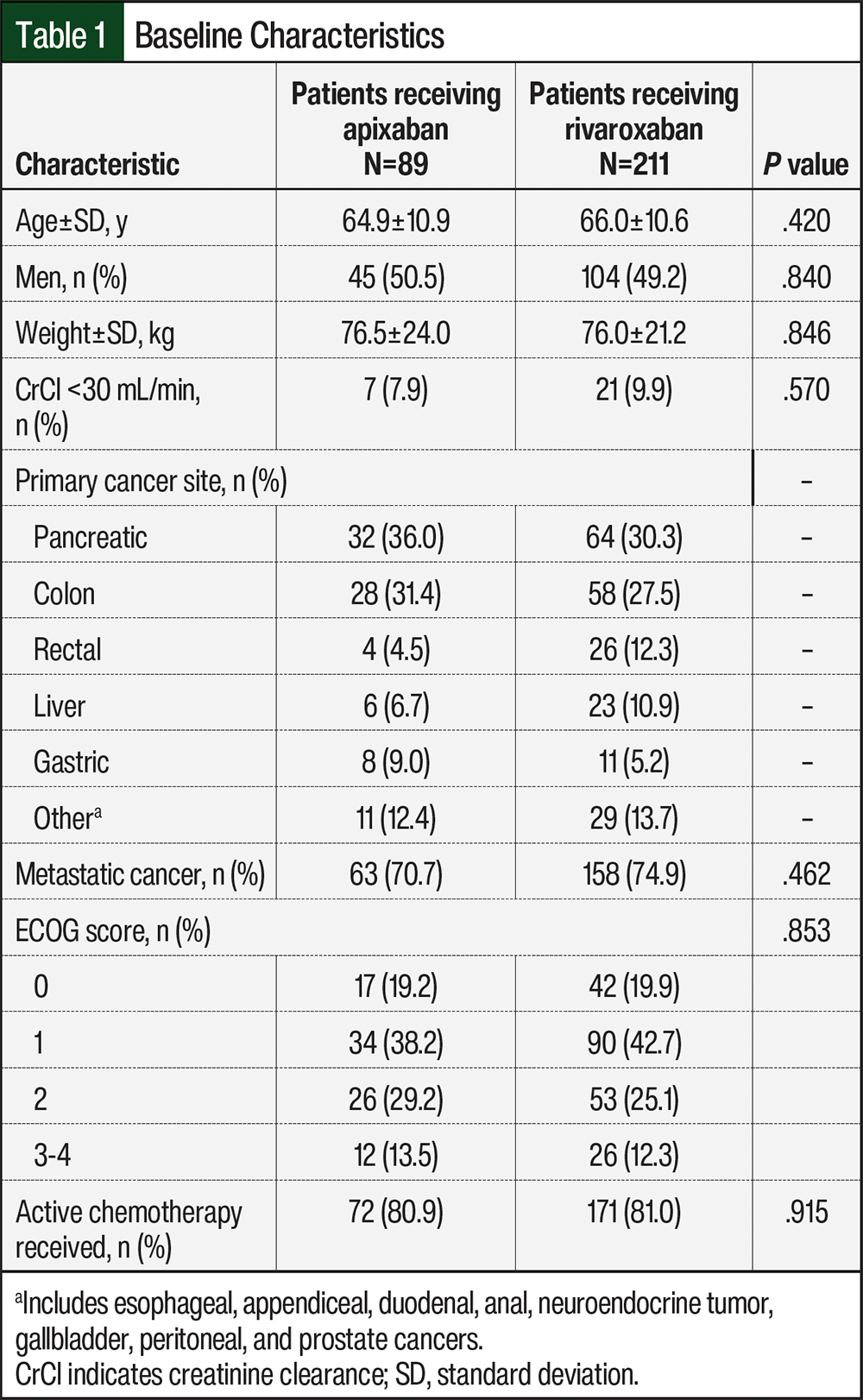
The patients’ baseline characteristics are shown in Table 1. Of these 300 patients, 70.7% and 74.9% had metastatic cancer in the apixaban and rivaroxaban groups, respectively. Most patients from each group were receiving active chemotherapy (80.9% and 81%, respectively). The most common cancers were pancreatic (36%) and colon (31.4%) in the apixaban group and pancreatic (30.3%), colon (27.5%), and rectal (12.3%) in the rivaroxaban group. The median patient age was 65.5 years, which was similar in both groups.
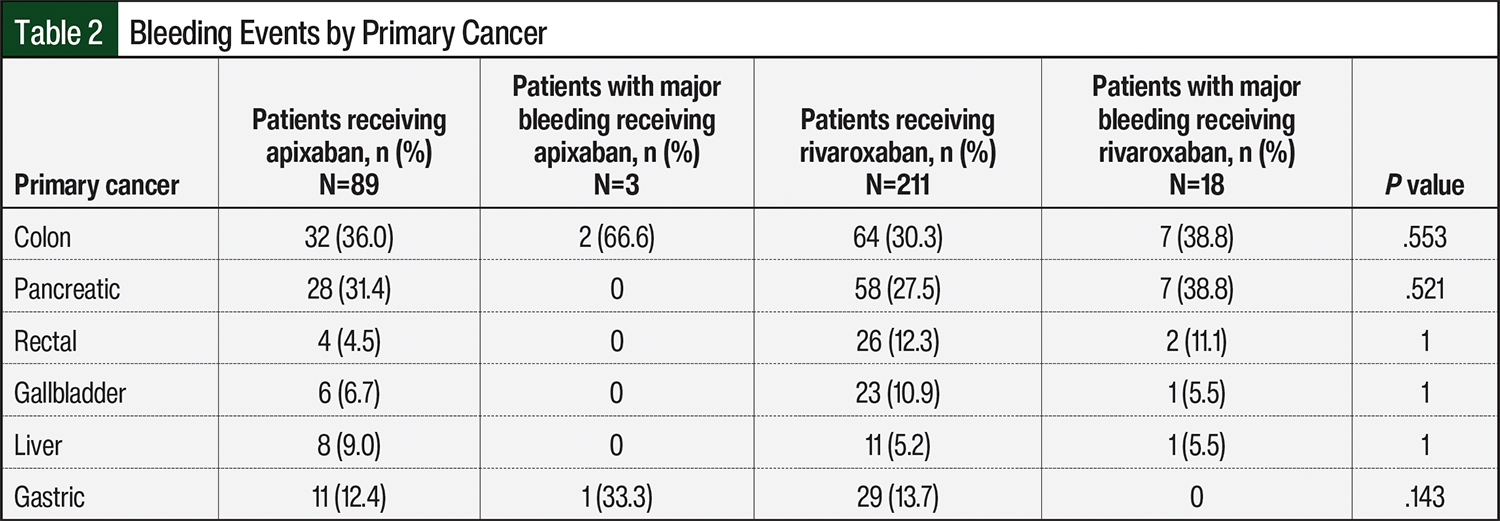
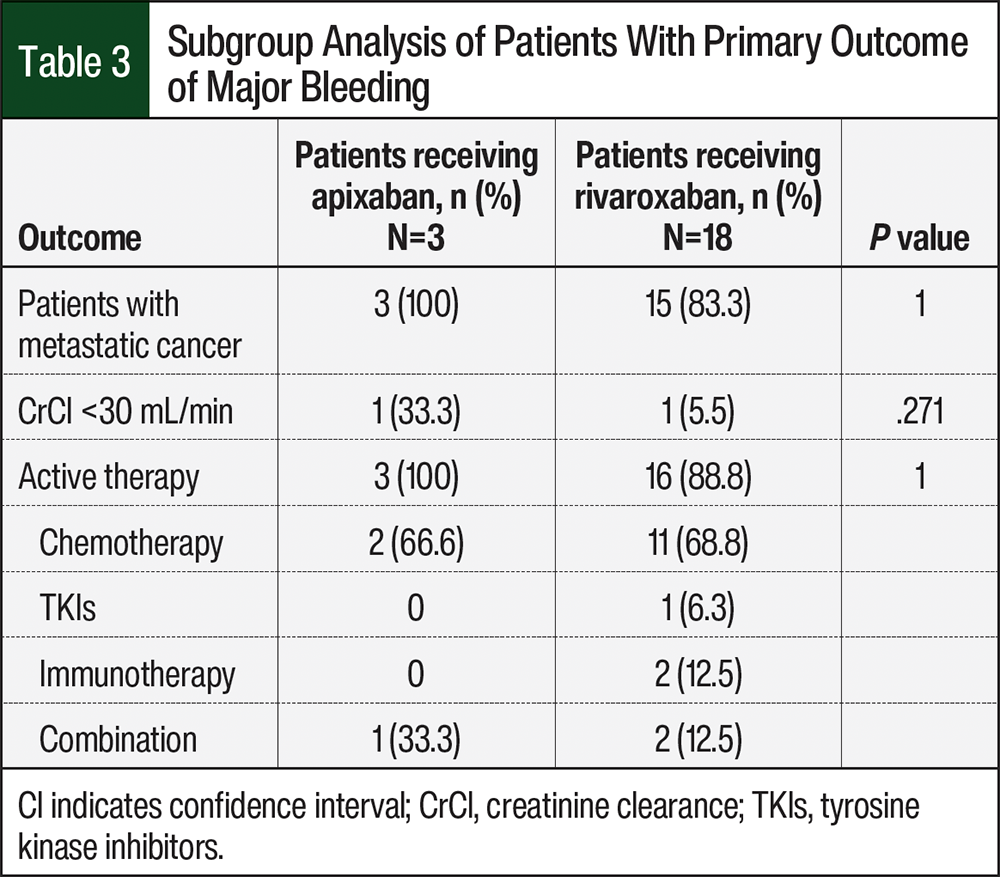
The primary outcome of major bleeding occurred in 3 of 89 (3.4%) patients in the apixaban group and in 18 of 211 (8.5%) patients in the rivaroxaban group (OR, 0.42; 95% CI, 0.13-1.37; P=.152). The median follow-up time was 23.8 months (range, 1.5-60.3 months). A subgroup analysis of major bleeding showed that patients with colon cancer had more major bleeding with apixaban than with rivaroxaban (66.6% vs 38.8%, respectively; P=.553; Table 2). Further analysis showed that patients with pancreatic cancer had more major bleeding events with rivaroxaban than with apixaban (38.8% vs 0%, respectively; P=.521; Table 2). All 3 patients who had major bleeding in the apixaban group had metastatic cancer and were receiving active treatment compared with the rivaroxaban group, which had 15 (83.3%) patients with metastatic cancer and 16 (88.8%) patients receiving active treatment who had a bleeding event (Table 3). There was no statistical difference in any of our primary outcomes between the groups.
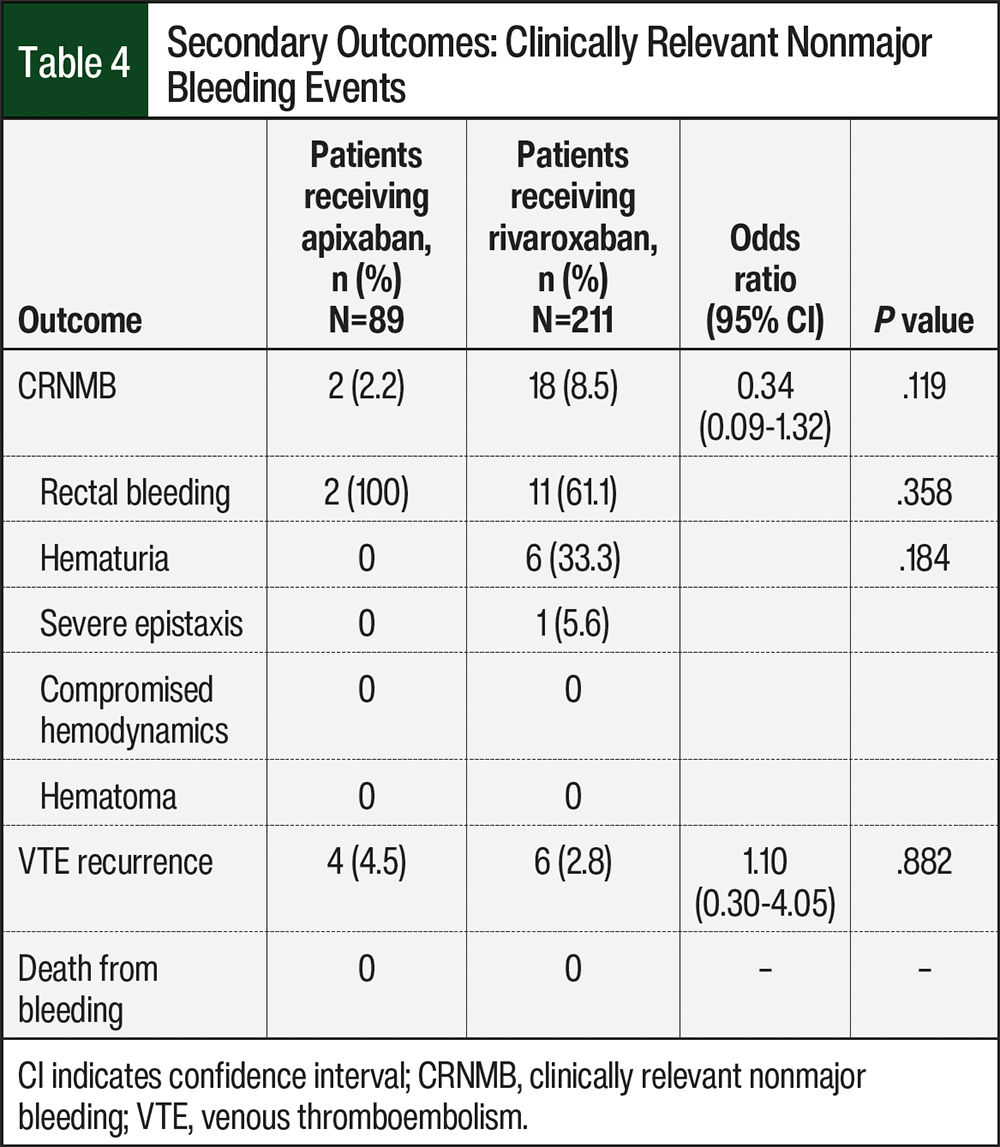
The secondary outcome of clinically relevant nonmajor bleeding occurred in 2 of 89 (2.2%) patients in the apixaban group and in 18 of 211 (8.5%) patients in the rivaroxaban group (OR, 0.34; 95% CI, 0.09-1.32; P=.119; Table 4). More patients receiving apixaban had rectal bleeding than those receiving rivaroxaban (100% vs 61.1%; P=.358), but more patients who received rivaroxaban had hematuria than those who received apixaban (33.3% vs 0%; P=.184; Table 4). VTE recurrence occurred in 4 of 89 (4.5%) patients in the apixaban group and in 6 of 211 (2.8%) patients in the rivaroxaban group (OR, 1.10; 95% CI, 0.30-4.05; P=.882; Table 4). There were no significant differences between the groups in the secondary outcomes. There were also no deaths from any type of bleeding.
Discussion
This study captures the real-world utilization of apixaban and rivaroxaban and its impact on rates of major bleeding and clinically relevant nonmajor bleeding in patients with GI malignancies. Within the GI cancer subset, patients in the SELECT-D trial had an increased rate of major bleeding and clinically relevant nonmajor bleeding in the rivaroxaban group compared with the group that received dalteparin,5 whereas in the CARAVAGGIO trial, there were similar rates of major bleeding and clinically relevant nonmajor bleeding between the apixaban and dalteparin cohorts.6 The results from the CARAVAGGIO trial were inconsistent with previous studies that demonstrated a higher incidence of bleeding when comparing other DOACs with dalteparin.5,6
GI cancer pathophysiology can affect drug absorption by damaging the GI physiology. Gastric cancers can increase the pH in the stomach to ≤6 to 77 and can cause delayed gastric emptying.8 Pancreatic and colon cancer can cause motility disorders, and motility disorders are associated with increased risk for pancreatic and colon cancers.9-11 All of these changes can affect drug absorption.9 Apixaban is absorbed throughout the GI tract, with 55% of the absorption occurring in the distal small bowel and ascending colon.12 Rivaroxaban is absorbed primarily in the stomach, and there can be decreases in AUC and Cmax when the medication reaches the small intestines and colon.13 The pharmacokinetic properties of apixaban and rivaroxaban further demonstrate the need to perform our study to determine their utility and safety in patients with different GI cancers.
Our study provides clinically relevant results to better understand the impact of DOACs in the upfront treatment setting of VTE in patients with GI cancer. The demographics between each group in our study were similar. However, there were significantly more patients in the rivaroxaban group than in the apixaban group. One insight as to why this may have happened may be the 2018 publication date of the SELECT-D trial that showed reduced VTE recurrence in patients receiving rivaroxaban, whereas the CARAVAGGIO trial was published 2 years later in 2020. The time difference in publishing would have led more physicians to practice evidence-based medicine by prescribing rivaroxaban versus apixaban.
The definition for the primary outcome of major bleeding aligned with the criteria used in the SELECT-D and CARAVAGGIO trials.5,6 In our study, the occurrence of major bleeding was numerically lower in the apixaban group than in the rivaroxaban cohort, although this was not statistically significant (3.4% vs 8.5%, respectively; P=.152). These rates were similar to the rates in the SELECT-D and CARAVAGGIO trials.5,6 The apixaban bleeding rate in our study was 3.4% versus 3.8% in the CARAVAGGIO trial,6 and the rivaroxaban bleeding rate in our study was 8.5% versus 6% in the SELECT-D trial,5 showing possible clinical significance.
The secondary outcome of clinically relevant nonmajor bleeding showed a numerical difference favoring apixaban over rivaroxaban, although this was not statistically significant (2.2% vs 8.5%, respectively; P=.119). Compared with the earlier trials, our study showed apixaban-related clinically relevant nonmajor bleeding was 2.2% versus 9% in the CARAVAGGIO trial.6 Rivaroxaban-related clinically relevant nonmajor bleeding was 8.5% in our study versus 13% in the SELECT-D trial.5
The lower rate of clinically relevant nonmajor bleeding with apixaban in our study may have been influenced by several factors. The first factor is the size of the groups: our study evaluated 89 patients compared with 576 patients in the CARAVAGGIO study, of which more than 150 patients had GI cancer.6 Another factor is the retrospective nature of this study, which limited the ability to identify all instances of clinically relevant nonmajor bleeding because of a lack of documentation. Further analysis of our data showed a greater rate of rectal bleeding in the patients receiving apixaban versus the patients who received rivaroxaban, and a greater rate of hematuria in the patients who received rivaroxaban. As a result of the small numbers of patients in each group, we were unable to find any correlation between this trend. However, more patients with colon or anal cancer had rectal bleeding (35% of clinically relevant nonmajor bleeding) than patients with the other cancer types.
The VTE recurrence rates were similar between the apixaban and rivaroxaban cohorts, without being significant (4.5% vs 2.8%, respectively; P=.882). We found similar rates of VTE recurrence in our study compared with other trials.5,6 The VTE recurrence rates in our study were 4.5% in the apixaban group compared with 5.6% in the CARAVAGGIO trial and 2.8% in our study in the rivaroxaban group compared with 4% in the SELECT-D trial.5,6 Our results demonstrate that there is no significant difference in VTE recurrence between the 2 groups within our study.
Limitations
This study has several limitations. First, the difference in sample size was a limitation. This study was a single-center, retrospective chart review that relied exclusively on provider documentation for the primary and secondary outcomes. Many patients were excluded from the study because of a lack of documentation, which led to a diminished sample size in 1 group. This decreased our ability to determine proper adherence to the medication regimen.
Another limitation was the inability to control absorption rates between apixaban and rivaroxaban based on tumor types, given the variability in pathophysiology and pharmacokinetics.
Conclusion
This retrospective study provided safety and efficacy data of major bleeding, clinically relevant nonmajor bleeding, VTE recurrence, and death in a real-world setting. Our findings showed that patients who received apixaban had fewer major bleeding events and fewer clinically relevant nonmajor bleeding events than patients who received rivaroxaban. Therefore, apixaban may be safer to prescribe than rivaroxaban in patients who have primary GI cancer because of its more favorable safety profile of fewer major bleeding events and clinically relevant nonmajor bleeding. In addition, this study showed that the efficacy of apixaban and rivaroxaban in the prevention of VTE is similar.
Considering both of these findings together, DOACs can be a safe and effective treatment in patients with GI cancer. The results of this study warrant further investigation of the use of DOACs in patients with GI cancers.
Acknowledgments
Research in this publication was supported in part
by the Biostatistics Shared Resource of Winship Cancer
Institute of Emory University and National Institutes
of Health/National Cancer Institute under
award number P30CA138292. The content is solely
the responsibility of the authors and does not necessarily
represent the official views of the National Institutes
of Health. The authors would like to thank Marie
Diab, MD, for her contributions to this research.
Author Disclosure Statement
Dr Watson is on the Advisory Board of Bristol Myers
Squibb; Dr Abousaud is employed at Astellas; Dr Gbolahan
has received grant funding from Bayer and Bristol Myers
Squibb; Dr Gundersen, Dr Basilio, Dr Draper, Dr Tiao, and
Dr Goyal have no conflicts of interest to report.
References
- Blom JW, Doggen CJM, Osanto S, Rosendaal FR. Malignancies, prothrombotic mutations, and the risk of venous thrombosis. JAMA. 2005;293:715-722.
- Lee AYY, Levine MN. Venous thromboembolism and cancer: risks and outcomes. Circulation. 2003;107(suppl 1):I17-I21.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): cancer-associated venous thromboembolic disease. Version 2.2024. Accessed July 22, 2024. www.nccn.org/professionals/physician_gls/pdf/vte.pdf
- Chew HK, Wun T, Harvey D, et al. Incidence of venous thromboembolism and its effect on survival among patients with common cancers. Arch Intern Med. 2006;166:458-464
- Young AM, Marshall A, Thirlwall J, et al. Comparison of an oral factor Xa inhibitor with low molecular weight heparin in patients with cancer with venous thromboembolism: results of a randomized trial (SELECT-D). J Clin Oncol. 2018;36:2017-2023.
- Agnelli G, Becattini C, Meyer G, et al. Apixaban for the treatment of venous thromboembolism associated with cancer. N Engl J Med. 2020;382:1599-1607.
- Tatsuta M, Iishi H, Okuda S. Gastric emptying in patients with fundal gastritis and gastric cancer. Gut. 1990;31:767-769.
- Lu PJ, Hsu PI, Chen CH, et al. Gastric juice acidity in upper gastrointestinal diseases. World J Gastroenterol. 2010;16:5496-5501.
- Feig C, Gopinathan A, Neesse A, et al. The pancreas cancer microenvironment. Clin Cancer Res. 2012;1816:4266-4276.
- Park JY, Mitrou PN, Luben R, et al. Is bowel habit linked to colorectal cancer? Results from the EPIC-Norfolk study. Eur J Cancer. 2009;45:139-145.
- Shafi MA. Gastrointestinal motility issues in cancer patients. Curr Gastroenterol Rep. 2019;21:69.
- Cada DJ, Levien TL, Baker DE. Apixaban. Hosp Pharm. 2013;48:494-509.
- Martin K, Lee C, Farrell T, et al. Oral anticoagulant use after bariatric surgery: a literature review and clinical guidance. Am J Med. 2017;130:517-524.