Connecting Science to Practice
Serum sickness is a rare complication associated with the
use of certain medications, such as antithymocyte globulin.
Although the syndrome has been well described, standardized
management strategies have not been clearly established
in the literature. This report presents a unique case
of a patient with serum sickness who received individualized
supportive care to manage symptoms, including the
use of tocilizumab to address serum sickness symptoms,
particularly in the context of concomitant manifestations
similar to cytokine release syndrome.
Aplastic anemia is a life-threatening hematologic disorder resulting in pancytopenia caused by bone marrow failure.1,2 Although rare, severe aplastic anemia is a medical emergency and warrants prompt initiation of triple immunosuppression in medically fit patients. This regimen consists of cyclosporine, equine antithymocyte globulin (eATG), and eltrombopag.2 Since the introduction of this regimen as the standard of care, response rates have significantly improved.2 The use of eATG, however, continues to predispose patients to increased risks for myelosuppression, hypersensitivity reactions, and serum sickness.3 These symptoms may be prevented and treated, but there is limited literature available regarding optimal prophylaxis and management strategies for patients who had hypersensitivity reactions and serum sickness resulting from the treatment of their underlying aplastic anemia. Understanding optimal treatment strategies and the management of these reactions can improve the tolerability and safety of this regimen. This case report aims to highlight the management strategies that we used for this patient.
Case Report
A 50-year-old woman presented with vaginal bleeding and progressive pancytopenia. Before initiation of therapy, a bone marrow examination showed a hypocellular aspirate with 1% blasts. Erythroid precursors were markedly decreased. Cytogenetic studies showed +15 in 3 of 20 metaphases. Molecular analysis showed a DNMT3A mutation with a variable allelic frequency of 3%. These findings were consistent with aplastic anemia. The patient’s pertinent medical history is significant for antinuclear and antithyroglobulin antibody elevations.
The patient was admitted to start treatment with eATG, cyclosporine, and eltrombopag. On admission, the patient was hemodynamically stable with a blood pressure (BP) reading of 102/70 mm Hg, heart rate of 86 beats per minute (bpm), temperature of 36.7°C, respiratory rate of 18 breaths per minute, and O2 saturation of 99% on room air. A complete blood count with differential showed white blood cells of 2.5 × 103/μL, hemoglobin of 8 g/dL, hematocrit of 23%, a platelet count of 18 × 103/μL, and an absolute neutrophil count of 360 cells/μL. Her baseline weight was 70 kg.
Day 1
We started therapy on day 1 of admission, with the patient tolerating a 0.005-mg intradermal test dose of eATG, 150 mg once daily of eltrombopag, and 200 mg twice daily of cyclosporine. Subsequently, 650 mg of acetaminophen, 50 mg of diphenhydramine, and 1 mg/kg of methylprednisolone were administered as premedications before the eATG infusion, beginning at 60 mL/hr for a planned 6-hour infusion. Approximately 7 hours into the infusion, the patient had flushing, headache, and hives along her left arm and right hip. eATG was held and her vital signs were as follows: BP, 95/59 mm Hg; heart rate, 103 bpm; temperature, 37.3°C; respiratory rate, 18 breaths per minute; and O2 saturation, 96% on room air. The patient received additional doses of acetaminophen (650 mg) and diphenhydramine (50 mg) for her headache and hives, respectively, and later the patient received a subsequent dose of 50 mg of hydrocortisone for persistent hives. Once the hives resolved, eATG was restarted at half the initial rate (30 mL/hr). Despite the interventions, 2 hours later, the patient again had diffuse hives. Her vital signs were BP, 93/58 mm Hg; heart rate, 81 bpm; temperature, 37°C; respiratory rate, 18 breaths per minute; and O2 saturation, 96% on room air. The patient received 20 mg of famotidine, 50 mg of hydrocortisone, and 50 mg of diphenhydramine, and the remainder of the day-1 eATG infusion was held.
Day 2
The patient remained hemodynamically stable, so day 2 of the eATG infusion was started. Premedications were altered, and the patient received 650 mg of acetaminophen, 50 mg of diphenhydramine, 20 mg of famotidine, and 1 mg/kg of methylprednisolone, with a plan to repeat the methylprednisolone dose 4 hours into the infusion. We started her infusion at 30 mL/hr, and it was completed after 12 hours. However, 2 hours after the completion of the infusion, the patient had recurrent diffuse hives necessitating additional doses of 50 mg of diphenhydramine, 20 mg of famotidine, and 100 mg of hydrocortisone.
Day 3
In this patient with persistent hives, we postponed dose 3 of the eATG infusion by 1 day. She had progressive symptomatic improvement with doses of 50 mg of diphenhydramine, 20 mg of famotidine, 10 mg of loratadine, and 2 doses of 1-mg/kg methylprednisolone.
Day 4
The patient remained hemodynamically stable, and her hives resolved; therefore, we continued therapy with the third dose of eATG. Again, premedications were altered to 650 mg of acetaminophen, 50 mg of diphenhydramine, 20 mg of famotidine, 10 mg of loratadine, 1 mg/kg of methylprednisolone (with a plan to repeat the methylprednisolone dose 4 hours into the infusion), and 100 mg of hydrocortisone 8 hours into the infusion. The infusion was started at 30 mL/hr and completed after 12 hours. The patient completed the entire dose without having hives.
Days 5 to 9
The patient remained hemodynamically stable, so she received the fourth dose of eATG. Premedications and infusion rate were administered in the same manner as day 4. Again, the patient completed the entire dose without having hives. On days 6 to 9 after eATG completion, she was maintained on 1 mg/kg of methylprednisolone daily, 10 mg of loratadine once daily, 20 mg of famotidine twice daily, and 50 mg of diphenhydramine as needed.
Day 10
On day 10, the patient had a new rash on her arms, legs, trunk, and back, as well as periorbital edema. Her dose of methylprednisolone (1 mg/kg) was increased to twice daily for 3 days, 10 mg of loratadine was increased to twice daily, 20 mg of famotidine was continued twice daily, and 50 mg of diphenhydramine was increased to every 6 hours.
Day 11
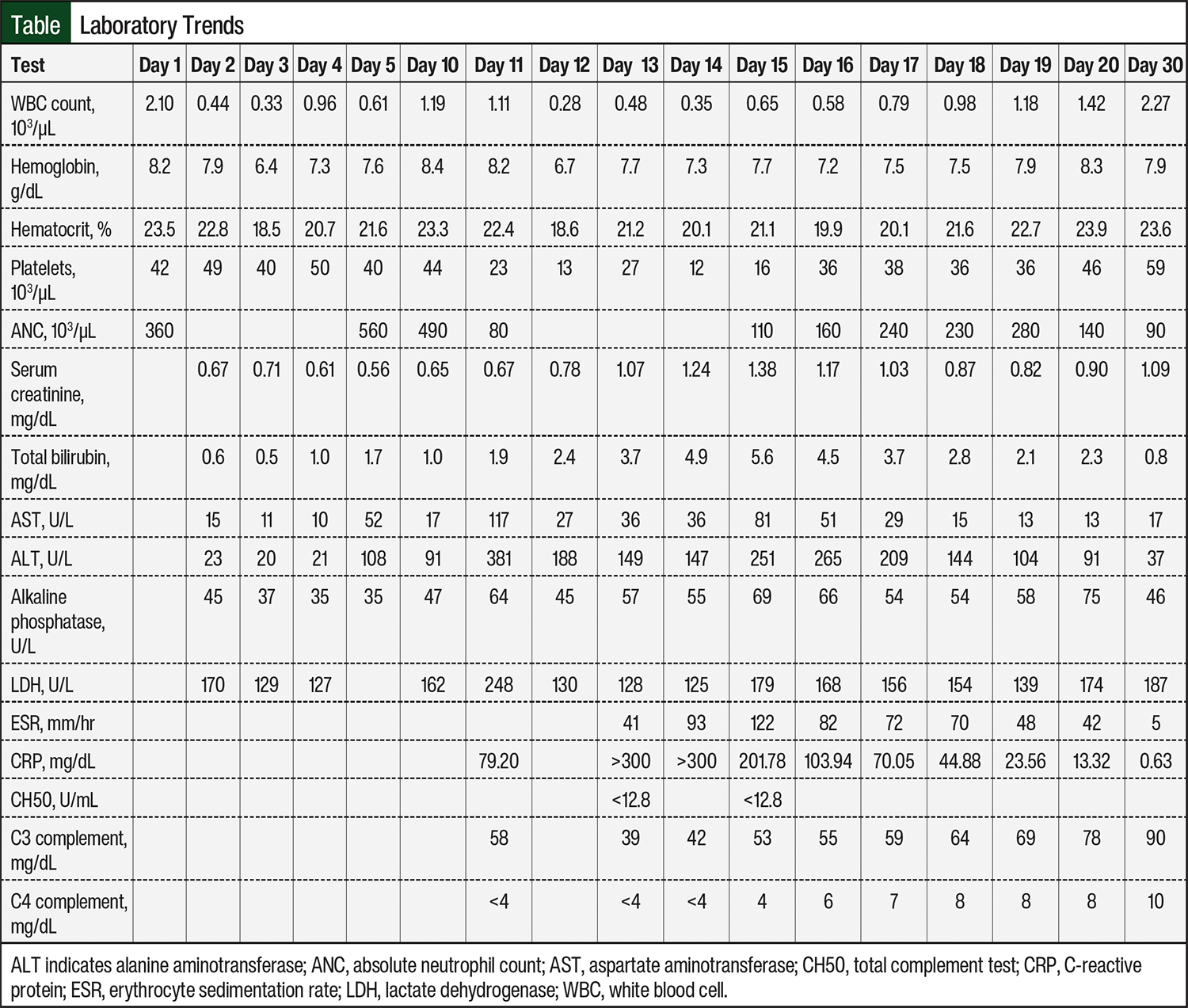
The patient’s symptoms continued to worsen, and she had odynophagia, left wrist pain, pleuritic chest pain, and epigastric pain with a fever. Laboratory tests, including C-reactive protein (CRP) and complement levels, suggested the presence of serum sickness (Table). Methylprednisolone 1 mg/kg was continued twice daily, 10 mg of loratadine was increased to 4 times daily, 20 mg of famotidine was continued twice daily, and 50 mg of diphenhydramine was increased to every 4 hours as needed. Intensive care unit triage was consulted overnight for an event of tachycardia and hypotension, which improved with fluids.
Day 13
On day 13, the patient had acute kidney injury, and her serum creatinine increased to 1.07 mg/dL. In addition, her total bilirubin was 3.7 mg/dL, indicating hepatic injury (Table). Several medications were then discontinued, including cyclosporine and pantoprazole as a result of her worsening renal function (despite cyclosporine remaining in therapeutic range on daily monitoring), progesterone because of hyperbilirubinemia, eltrombopag because of transaminases, and piperacillin plus tazobactam, vancomycin, and sulfamethoxazole plus trimethoprim as a result of possible drug-related rash. We also switched the patient to 60 mg of fexofenadine 4 times daily from loratadine based on her preference.
Day 14
The patient’s organ function continued to worsen, with increases in serum creatinine to 1.24 mg/dL and total bilirubin to 4.9 mg/dL. Her inflammatory markers were also on the rise; her erythrocyte sedimentation rate (ESR) was 93 mm/hr, and her CRP was >300 mg/dL. Simultaneously, her vital signs were BP, 109/70 mm Hg; heart rate, 82 bpm; temperature, 36.6°C; respiratory rate, 18 breaths per minute; and O2 saturation, 95% on a 3L nasal canula. Given the need for O2 supplementation and rising inflammatory markers, we determined the patient had a grade 2 cytokine release syndrome (CRS)-like appearance and administered 1 dose of 8 mg/kg of tocilizumab.
Day 16
The patient had symptomatic improvement in her vital signs, with a BP of 132/88 mmHg, heart rate of 74 bpm, respiratory rate of 18 breaths per minute, and O2 saturation of 98% on a 3L nasal canula. Laboratory work also showed improved organ function, indicated by serum creatinine decrease to 1.17 mg/dL, total bilirubin decrease to 4.5 mg/dL, and a decrease in inflammatory markers, most notably CRP decreased to 103.94 mg/dL (Table). Subsequently, given the downward trend in inflammatory markers and improvement in arthralgias, we began a methylprednisolone twice-daily taper, decreasing the dose by approximately 15% every 3 days.
Day 19
We noted continued improvement, leading to further tapering of methylprednisolone to a once-daily oral prednisone regimen and continued with approximately 15% reductions every 3 days. Cyclosporine 200 mg twice daily was also resumed because of stable renal function.
Days 21 to 40
Steroids were tapered further, continuing to follow the approximately 15% reduction every 3 days plan, and sulfamethoxazole plus trimethoprim was also reinitiated without evidence of rash. The patient’s laboratory tests and symptoms continued to improve during her remaining time in the hospital (Table). She had intermittent complaints of joint discomfort/pain, which were treated on an as-needed basis with analgesic medications. On day 40, we restarted eltrombopag at 150 mg once daily, and the prednisone taper was completed.
Discussion
This case report details the management strategies that we used for a patient with severe infusion-related reactions and serum sickness secondary to treatment with eATG. Despite maximal dosing of supportive medications, the patient had ongoing signs of an uncontrolled inflammatory response, as evidenced by hypotension, hypoxia, end-organ damage, and a marked increase in CRP. Our patient’s presentation mimicked CRS, which is defined as a systemic inflammatory response causing the release of various cytokines. CRS is often caused by infection or certain medications. Because the patient had an O2 requirement, rising inflammatory markers, and evidence of end-organ damage, we used tocilizumab, which resulted in prompt improvement in her clinical symptoms and laboratory values. Tocilizumab is an interleukin (IL)-6 receptor antagonist and is frequently used for CRS secondary to cellular therapies. Increases in IL-6 have the potential to result in nonspecific inflammation, leading to complications such as capillary leak, hypotension, and activation of the complement pathway.4
Infusion-related reactions (eg, fevers, chills, rash) resulting from the use of eATG have been reported in 25% to 40% of patients and can range in severity.3 As a result of the acuity of aplastic anemia, the administration of eATG is often continued despite the presence of infusion-related reactions. Two recommended modalities to anticipate the development of and mitigate the risk for infusion-related reactions include the administration of an intradermal test dose of the medication and premedication with acetaminophen, diphenhydramine, and steroids.2 Our case report shows that we used a test dose before day 1 of the eATG infusion, which did not show a positive reaction. We also provided premedication with acetaminophen, diphenhydramine, and steroids, as well as additional doses as needed, and the addition of famotidine and loratadine as the infusion reactions worsened.
Serum sickness is a type-III immune-mediated hypersensitivity reaction. Patients often present with nonspecific symptoms, including fever, rash, and arthralgia.5 Although rare, serum sickness has an incidence of 0.6% to 10% in children and adults receiving eATG, respectively.3,6 In the landmark study of cyclosporine, eltrombopag, and eATG for the treatment of severe aplastic anemia, 7% of patients had serum sickness, all of which were grade ≥3 in severity.7 Symptoms typically manifest 8 to 13 days after exposure to the causative agent. The introduction of a foreign serum protein causes an excessive quantity of antigen–antibody complexes to form. The mononuclear phagocyte system is unable to clear these complexes, and subsequently they are deposited in the blood vessels and synovial fluid. On deposition, the complement pathway is activated, leading to lower levels of C3 and C4 complements.5 Furthermore, histamine production will increase, causing an inflammatory response of the tissue and associated joints.8 Other common laboratory findings of nonspecific inflammatory markers include elevations in ESR and CRP.8 The clearance of these antigen–antibody complexes and the resolution of symptoms are often self-limiting and typically resolve in 7 to 14 days.8
As seen in our case report, the patient had serum sickness symptoms at approximately day 10, and laboratory tests showed decreased complement levels and elevated ESR and CRP on days 11 to 13 (Table). Given the concern for IL-6’s potential to activate the complement pathway in this patient leading to serum sickness, our decision to use tocilizumab to mitigate the activation of this pathway was supported.
In patients with serum sickness, optimal management includes discontinuing the causative agent. Unfortunately, in patients with aplastic anemia, the causative agent, eATG, has already been completely administered, so the focus of management shifts to supportive care measures. Common supportive care measures include anti-inflammatory, antihistamine, and analgesic medications as needed.9 For mild symptoms, nonsteroidal anti-inflammatory drugs (NSAIDs) can be used, but their role in aplastic anemia is limited because of concomitant thrombocytopenia. Steroid courses used for the treatment of severe cases of serum sickness for 7 to 10 days at doses of 1 to 2 mg/kg per day have alleviated patients’ symptoms in case reports. 9,10 Antihistamine management includes nonselective first-generation H1 antihistamine (diphenhydramine), second-generation selective peripherally acting H1 antihistamines (loratadine and fexofenadine), and gastric H2 antihistamines (famotidine).11 The use of a first-generation H1 antihistamine allows for intravenous administration with more potent and quicker onset of action compared with second-generation antihistamines; however, second-generation antihistamines are associated with less sedation and reduced anticholinergic effects.11 The addition of an H2 antagonist can be considered if a patient has incomplete resolution of a rash with the use of H1 antihistamines alone.11
We avoided the use of NSAIDs in our patient because of persistent thrombocytopenia and worsening renal function; however, we used a similar steroid approach that included 1 mg/kg of methylprednisolone twice daily for 10 days starting at the conclusion of her infusion and continued this dose through the resolution of her serum sickness. We followed this dosing with a taper. At the height of the patient’s symptoms of serum sickness, we used the maximum dosing for all antihistamines (50 mg of diphenhydramine every 4 hours, 10 mg of loratadine 4 times daily, and 20 mg of famotidine twice daily). As her rash began to resolve, we switched the diphenhydramine dosing to as needed, and loratadine was switched to fexofenadine per the patient’s preference. Last, we tapered the fexofenadine and famotidine dosages back to once daily until they were no longer required. Given our experience with tocilizumab, we hypothesize that the inhibition of IL-6 in conjunction with steroids and antihistamines slows complement activation and leads to prompt resolution of serum sickness.
Conclusion
This case report illustrates the potential utility of tocilizumab in patients with serum sickness that is refractory to standard treatment with corticosteroids and antihistamines and who have ongoing evidence of a marked inflammatory response mimicking CRS. To date, little in the literature outlines real-world experiences in altering premedication and supportive care medications to ensure the safety of eATG and the management of severe serum sickness with end-organ damage with CRS-like features.
Author Disclosure Statement
Dr Jurcic has received research funding from Blueprint
Medicines, Forma Therapeutics, Seagen, and Sumitomo
Pharma, is a consultant to Syros Pharmaceuticals, and is on an ad hoc advisory board at Incyte. Dr Sabatino and Dr
Varabyeva have no conflicts of interest to report.
References
- Vaht K, Göransson M, Carlson K, et al. Incidence and outcome of acquired aplastic anemia: real-world data from patients diagnosed in Sweden from 2000-2011. Haematologica. 2017;102:1683-1690.
- Peffault de Latour R, Kulasekararaj A, Iacobelli S, et al. Eltrombopag added to immunosuppression in severe aplastic anemia. N Engl J Med. 2022;386:11-23.
- Murthy H, Iqbal M, Chavez JC, Kharfan-Dabaja MA. Cytokine release syndrome: current perspectives. Immunotargets Ther. 2019;8:43-52.
- Atgam (lymphocyte immune globulin, anti-thymocyte globulin [equine]), sterile solution, for intravenous use only [prescribing information]. Pfizer; September 2023. Accessed January 15, 2023. https://labeling.pfizer.com/ShowLabeling.aspx?format=PDF&id=525
- Lawley TJ, Bielory L, Gascon P, et al. A prospective clinical and immunologic analysis of patients with serum sickness. N Engl J Med. 1984;311:1407-1413.
- Rizkallah J, Cordova F, Malik A, Zieroth S. Serum sickness after antithymocyte globulin administration in a cardiac transplant patient. Transplantation. 2012;94(11):e68-e69.
- Townsley DM, Scheinberg P, Winkler T, et al. Eltrombopag added to standard immunosuppression for aplastic anemia. N Engl J Med. 2017;376:1540-1550.
- Joint Task Force on Practice Parameters; American Academy of Allergy, Asthma and Immunology; American College of Allergy, Asthma and Immunology; Joint Council of Allergy, Asthma and Immunology. Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105:259-273.
- Clark BM, Kotti GH, Shah AD, Conger NG. Severe serum sickness reaction to oral and intramuscular penicillin. Pharmacotherapy. 2006;26:705-708.
- Tanriover B, Chuang P, Fishbach B, et al. Polyclonal antibody-induced serum sickness in renal transplant recipients: treatment with therapeutic plasma exchange. Transplantation. 2005;80:279-281.
- Berstein JA, Lang DM, Khan DA, et al. The diagnosis and management of acute and chronic urticaria: 2014 update. J Allergy Clin Immunol. 2014;133:1270-1277.