Connecting Science to Practice
Hyperglycemia is common in patients with non–small cell
lung cancer receiving immune checkpoint inhibitors (ICIs),
particularly those with preexisting diabetes or receiving
chemotherapy. Hyperglycemia does not negatively impact
disease progression and may lead to longer time to disease
progression in those with hyperglycemia and/or immune-
related adverse events. Routine glucose monitoring
before and during ICI treatment is critical, especially in
patients with risk factors such as type 2 diabetes or baseline
hyperglycemia. Multidisciplinary management of hyperglycemia
using dietary interventions, glucose-lowering medications,
and potentially insulin may mitigate complications
and optimize patient outcomes. Prospective studies are
needed to confirm these findings and explore the mechanisms
linking hyperglycemia to the efficacy of ICIs.
Immune checkpoint inhibitors (ICIs) have positively impacted patients’ outcomes, including increasing survival, when used in the treatment of multiple malignancies, including metastatic non–small cell lung cancer (NSCLC).1 Currently, the National Comprehensive Cancer Network recommends ICIs, alone or in combination with cytotoxic chemotherapy, as a first-line treatment option for advanced or metastatic NSCLC in those without targetable driver mutations.1 ICIs for the treatment of NSCLC exert antitumor activity by blocking the programmed cell death protein 1/programmed death-ligand 1 (PD-L1) or cytotoxic T-lymphocyte antigen-4 pathways that cancerous cells use to evade the immune system.2 The use of ICIs to interrupt these pathways allows the body’s targeted immune response to recognize and attack malignant cells.2 Although ICIs have proven efficacy as therapeutic agents, they are associated with a myriad of immune-related adverse events (irAEs). These irAEs are inflammatory in nature, given that they are an extension of the therapeutic response produced by ICIs.2
The incidence and management of irAEs associated with ICIs have been well documented.2-4 In particular, endocrine irAEs frequently occur in patients receiving ICIs.5 Although hypophysitis and thyroiditis are the most documented endocrine irAEs, ICI-induced adrenal insufficiency and type 1 diabetes have also been noted.5 The proposed mechanism of ICI-induced type 1 diabetes and subsequent hyperglycemia during ICI treatment is via immune-mediated damage of pancreatic beta cells.6 However, hyperglycemia may also arise outside of ICI-induced type 1 diabetes and is often multifactorial (ie, steroid induced, type 2 diabetes/insulin resistance).1
Multiple studies have also examined the impact of irAEs on patients’ outcomes. Specific to endocrine effects, thyroid dysfunction secondary to ICI treatment has been associated with improved response and survival in multiple malignancies.7-9 A similar effect was demonstrated in patients with ICI-induced type 1 diabetes.10 Among 11 patients with cutaneous melanoma, researchers found a 73% partial or complete response rate for patients continuing ICI treatment after ICI-induced type 1 diabetes.10 This response rate among patients with melanoma and ICI-induced type 1 diabetes compares favorably with the response rates in clinical trials of ICI use in melanoma (overall response rate, 41%-58%).11,12
Although these results are reassuring for patients with melanoma who had ICI-induced type 1 diabetes, few studies have examined if the presence of ICI-induced type 1 diabetes or hyperglycemia during ICI treatment (ICI-related hyperglycemia) impacts the outcomes of patients with NSCLC. A recent Canadian study examined ICI-induced type 1 diabetes in 34 patients with heterogeneous cancer diagnoses, of whom 4 (12%) had a diagnosis of NSCLC.13 The researchers noted that patients with ICI-induced type 1 diabetes (although irreversible and requiring long-term dependence on insulin) had a high tumor response rate and may have prolonged survival (based on historic durations of survival).13 ICI-induced type 1 diabetes occurs in an estimated 0.2% to 1.4% of those receiving ICIs for the treatment of solid tumors.14 Alternatively, ICI-related hyperglycemia, without the presence of ICI-induced type 1 diabetes, occurs in up to 27% of patients, regardless of tumor type, and is multifactorial in its geneses.15 This suggests that the incidence of ICI-related hyperglycemia, which is much more common than ICI-induced type 1 diabetes, may wield a greater influence on patient outcomes.
Historically, in patients with NSCLC, preexisting diabetes and/or hyperglycemia have resulted in poorer patient outcomes when receiving traditional cytotoxic chemotherapy.16-20 An analysis of the impact of baseline hyperglycemia on outcomes in patients with NSCLC receiving ICI therapy demonstrated that those in the highest quartile (median glucose, 161 mg/dL) had a significantly shorter overall survival than patients with a baseline glucose in the lowest quartile (median glucose, 91 mg/dL).21 Whether ICI-related hyperglycemia during ICI therapy portends improved or worsened patient outcomes in this particular population is unknown. Therefore, the purpose of this study was to examine the frequency of ICI-related hyperglycemia and its impact on patient outcomes among those receiving ICI treatment for metastatic or unresectable NSCLC. The findings from this study provide valuable insights, informing the clinical practice of oncology providers who manage patients with NSCLC using ICI.
Methods
This study was a retrospective review of electronic health records (EHRs) from Franciscan Health Indianapolis, Indianapolis, IN, a community teaching hospital. The cohort consisted of patients who received first-line ICI treatment (pembrolizumab, atezolizumab, nivolumab with or without ipilimumab) with or without cytotoxic chemotherapy for the treatment of metastatic or unresectable NSCLC in the outpatient setting. The study’s eligibility criteria included age ≥18 years, a diagnosis of metastatic or unresectable NSCLC, and the initiation of first-line treatment with an ICI in the unresectable or metastatic NSCLC setting between January 1, 2019, and December 1, 2021. Patients were eligible for study inclusion if they received an ICI as consolidation treatment after chemoradiation or as definitive therapy for an earlier malignancy. Patients with preexisting type 1 diabetes were excluded from the study. Data were collected from the time of ICI treatment initiation until 6 weeks after drug discontinuation or for at least 6 months if the drug was not discontinued before the data collection cutoff of June 1, 2022. The local institutional review board approved this study.
The primary end point of the study was the time to NSCLC progression by hyperglycemia status during ICI treatment. Additional end points included a description of the incidence, timing of, and potential risk factors for hyperglycemia during ICI treatment, the incidence and timing of additional irAEs by hyperglycemia status during ICI treatment, and differences in the time to disease progression between patients with NSCLC who have neither hyperglycemia nor irAE, hyperglycemia only, irAE only, or hyperglycemia and irAE during ICI treatment. Based on earlier studies, the American Diabetes Association (ADA) guidelines for the diagnosis and classification of type 2 diabetes, and the lack of control over when glucose levels were measured, hyperglycemia was defined as at least 1 random blood glucose reading of ≥200 mg/dL during ICI treatment.15,22,23
The baseline data were collected from the EHRs. The baseline random blood glucose of ≥200 mg/dL before ICI treatment initiation was based on laboratory values immediately before the first dose of ICI. Corticosteroid use was collected and considered baseline corticosteroid use if it was initiated before starting an ICI (ie, for the treatment of brain metastases or chronic obstructive pulmonary disease) or as corticosteroid use during treatment if it was started or continued after receiving an ICI (including maintenance corticosteroids, short corticosteroid course/corticosteroid taper, and corticosteroids used as supportive care for cytotoxic chemotherapy).24,25
The data for the study outcomes were collected from the patients’ EHRs. The patients had hyperglycemia during ICI if they had at least 1 random blood glucose reading of ≥200 mg/dL after the administration of the first ICI treatment. The time-to-event data were calculated from the start of the ICI treatment, and the time until NSCLC progression was defined as the time from the start of ICI treatment to the documented progression of malignancy by the treating oncologist. Last, irAE occurrence and timing were determined according to the documentation of the irAE by the treating oncologist.
Statistical Analysis
The categorical data were analyzed using a chi-square or Fisher’s exact test, as appropriate. The continuous data were compared using the Mann–Whitney U test (nonparametric) or Student’s t-test (parametric), and the ordinal data were compared using the Kruskal-Wallis test. A binomial logistic regression was used to determine the factors that may increase a patient’s risk for hyperglycemia during ICI treatment in the entire population and the population without a history of type 2 diabetes. The variables included in the binomial logistic regression included age, body mass index (BMI), sex, race/ethnicity, type of treatment (ICI/chemotherapy plus ICI maintenance therapy or ICI/chemotherapy or ICI monotherapy), a medical history of type 2 diabetes, baseline blood glucose of ≥200 mg/dL, and corticosteroid use during treatment.15
A Kaplan–Meier analysis (unadjusted for covariates) was used to examine the differences in the time to disease progression. Analysis of variance was used for continuous variable comparisons between more than 2 groups, and Bonferroni correction was used, as necessary, to account for multiple comparisons associated with pairwise analyses. The data were censored as of June 1, 2022, if the patients continued treatment past this date. All statistical tests were conducted with IBM SPSS Statistics, version 25 (IBM Corporation; Armonk, NY).
Results
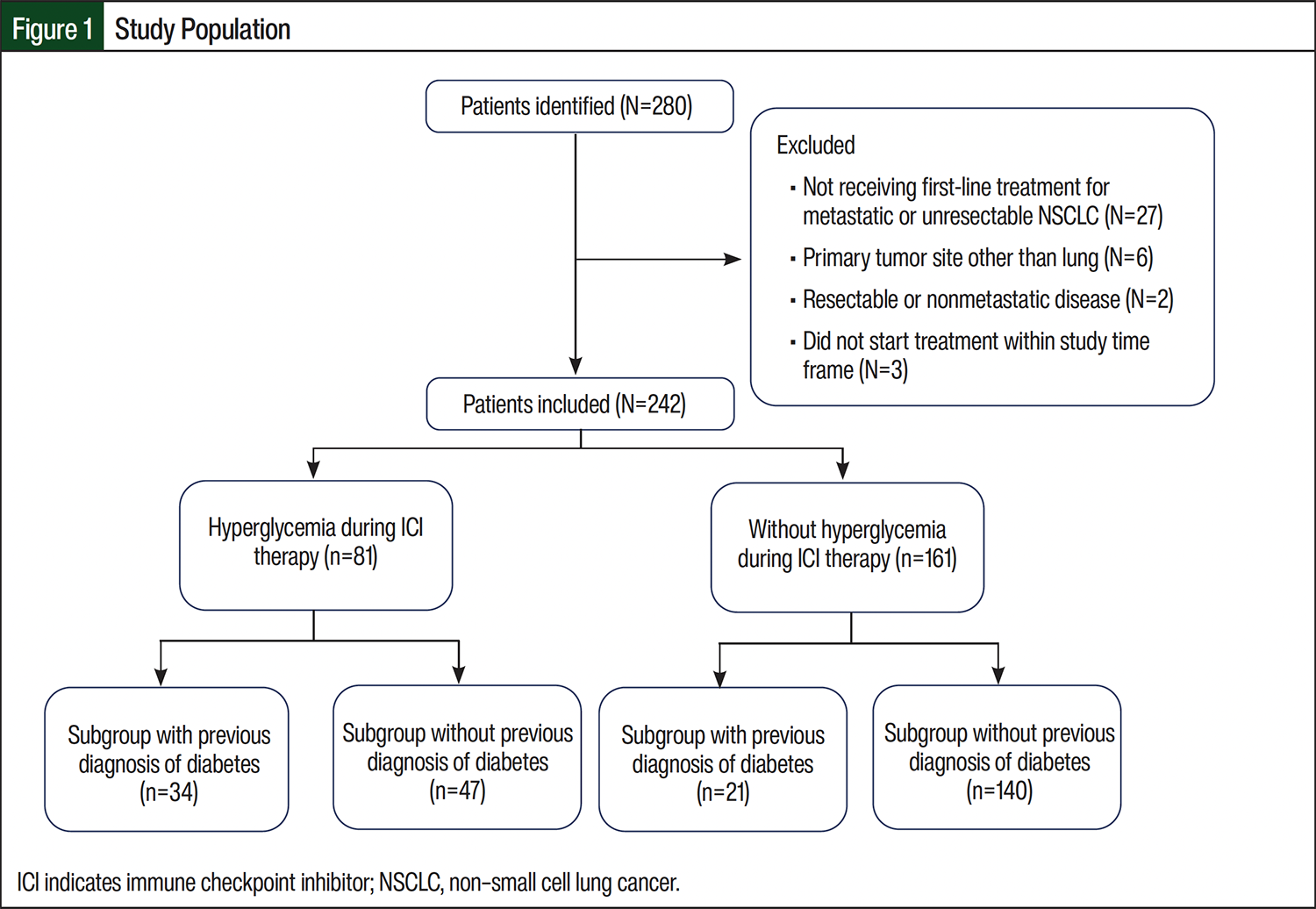
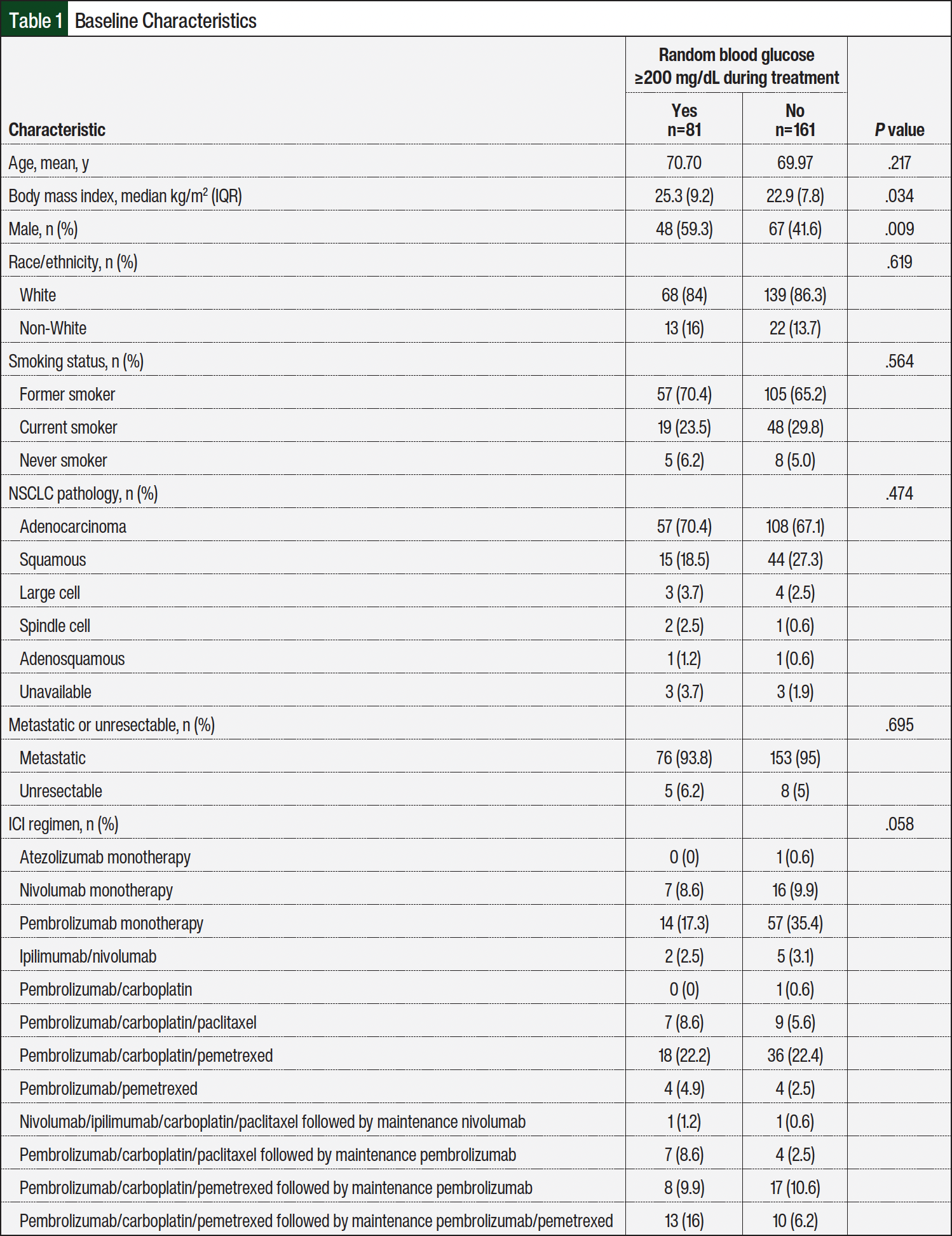
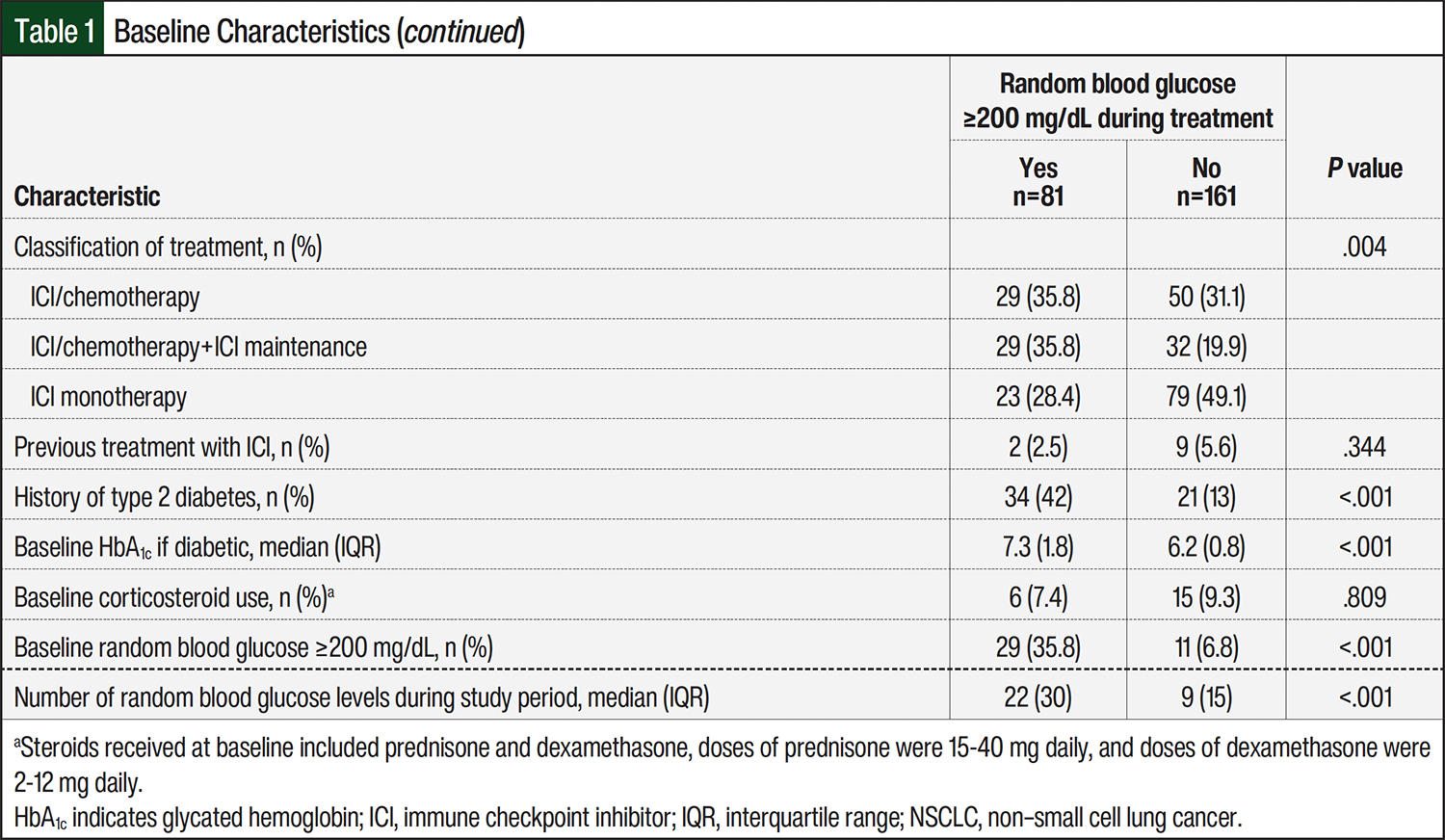
A total of 280 patients were identified, and after excluding patients who did not meet the inclusion criteria (n=38), the total sample size for analysis was 242 patients (Figure 1). A total of 12 distinct ICI regimens with or without cytotoxic chemotherapy were administered to patients (Table 1). In all, 81 patients (approximately 33%) had hyperglycemia during ICI treatment. The patients who had hyperglycemia during ICI treatment had a higher BMI, were more often male, and were more likely to receive cytotoxic chemotherapy with their ICI. Likewise, the patients with NSCLC and a history of type 2 diabetes, with a higher baseline glycated hemoglobin (HbA1c) or baseline random blood glucose of ≥200 mg/dL, and who received corticosteroids during ICI treatment were more likely to have hyperglycemia. There was no difference between the groups in the steroid regimen received during ICI treatment; among all 242 patients enrolled, 43% received steroids as supportive care for chemotherapy, 16% received a short/tapered course, 9% received as supportive care and as a short/tapered course, and 7% received long-term/maintenance steroids. Other baseline characteristics were similar between the groups (Table 1). The median number of glucose readings per patient was significantly higher in those who had hyperglycemia than in those without hyperglycemia during ICI treatment (22 vs 9, respectively; P<.001).
Among the patients with NSCLC who had hyperglycemia during ICI treatment, the median time from treatment initiation to a random blood glucose of ≥200 mg/dL was 25 days and the median proportion of blood glucose readings of ≥200 mg/dL during treatment was 18%. Corticosteroid use during ICI treatment was common and occurred in 70 (86%) patients who had hyperglycemia compared with 112 (69%) patients without hyperglycemia (P=.004). Of the 81 patients with hyperglycemia during ICI therapy, 11 (14%) were not receiving corticosteroids and had potentially otherwise unexplained hyperglycemia. Among the patients with NSCLC who did not have a history of type 2 diabetes or hyperglycemia at baseline (n=174), 39 (22%) had hyperglycemia while receiving ICI treatment and 5 (13%) did not receive corticosteroids.
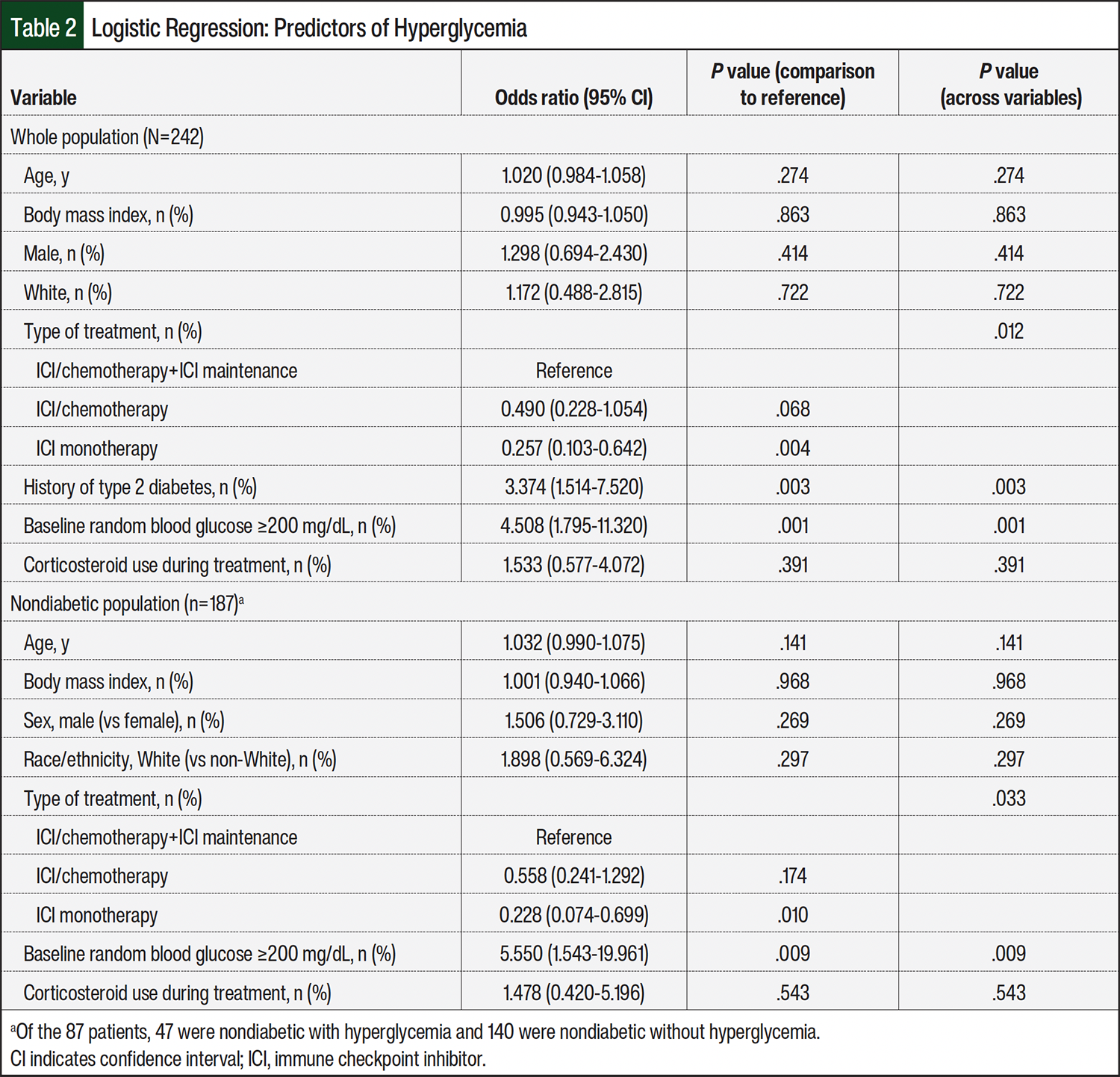
A logistic regression was completed to examine the risk factors for hyperglycemia during ICI treatment. The model was statistically significant compared with the null model; X2=56.98 (P<.001) explained 29% of the variation in outcomes (Nagelkerke R2) and correctly predicted 75.2% of cases (Table 2). The results of the logistic regression demonstrated that patients with NSCLC who received ICI monotherapy had lower odds of hyperglycemia (odds ratio [OR], 0.257; 95% confidence interval [CI], 0.10-0.64). Patients with NSCLC and a medical history of type 2 diabetes had higher odds of hyperglycemia than those without a diagnosis of type 2 diabetes (OR, 3.3; 95% CI, 1.51-7.52). Patients with NSCLC and a baseline random blood glucose of ≥200 mg/dL before ICI treatment initiation had higher odds of hyperglycemia during treatment (OR, 4.5; 95% CI, 1.79-11.3) than those with a baseline random blood glucose of <200 mg/dL.
Because of the confounding influence of a type 2 diabetes diagnosis on the outcomes, a second logistic regression was completed in a subgroup of patients without a diagnosis of type 2 diabetes at baseline (Table 2). Again, the model was statistically significant compared with the null model; X2=25.73 (P=.001) explained 19% of the variation in outcomes (Nagelkerke R2) and correctly predicted 78.6% of hyperglycemia cases. Similarly, patients with NSCLC without a diagnosis of type 2 diabetes receiving ICI monotherapy remained at reduced odds of hyperglycemia (OR, 0.22; 95% CI, 0.07-0.69; Table 2). Likewise, patients with NSCLC without type 2 diabetes but with a random blood glucose of ≥200 mg/dL before ICI treatment initiation had increased odds of hyperglycemia during treatment (OR, 5.5; 95% CI, 1.54-19.9).
The occurrence of additional irAEs was similar between those who had hyperglycemia during ICI treatment and those who did not (42% vs 37.9%, respectively; P=.539), as was the median time until additional irAEs (3.3 months vs 3.2 months, respectively; P=.507; Table 3). The median time to NSCLC progression was longer in the group that had hyperglycemia during ICI treatment than the group that did not have hyperglycemia; however, this difference did not reach statistical significance (14.5 months vs 11.3 months, respectively, P=.138; Table 3).
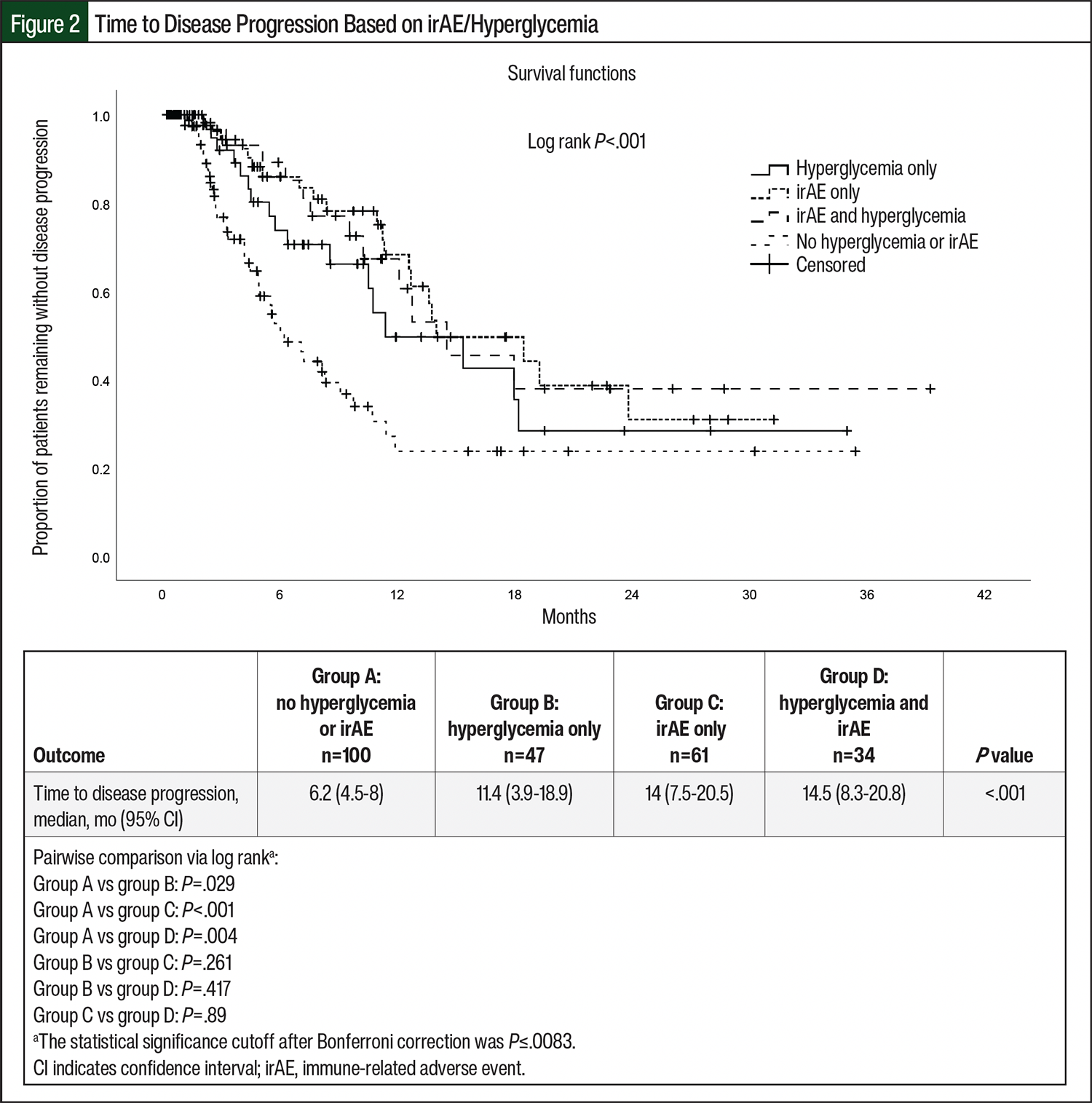
An additional Kaplan–Meier analysis was completed to determine if there were any differences in the time to disease progression between patients with NSCLC who had neither hyperglycemia nor irAEs, hyperglycemia only, irAEs only, or hyperglycemia and irAEs. There was a significant difference in the median time until disease progression across the 4 groups of patients with NSCLC (P<.001; Figure 2). Specifically, patients with NSCLC who had neither hyperglycemia nor irAEs had a shorter time to disease progression than those who had hyperglycemia only, irAEs only, or hyperglycemia and irAEs during ICI treatment (6.2 months vs 11.4 months, 14 months, and 14.5 months, respectively; P<.001; Figure 2). When the groups were analyzed via pairwise comparisons, a significant difference remained after Bonferroni correction (corrected statistical significance cutoff: P≤.0083) between those with neither hyperglycemia nor irAEs and those with irAEs only (P<.001) and between those with neither hyperglycemia nor irAEs and those with hyperglycemia and irAEs (P=.004). There was a trend toward significance when comparing those with neither hyperglycemia nor irAEs and those with hyperglycemia only (P=.029). There was no difference between the hyperglycemia-only and irAEs-only groups or compared with the group with hyperglycemia and irAEs.
Discussion
To date, limited data are available regarding the impact of ICI-related hyperglycemia on patient outcomes in metastatic or unresectable NSCLC, and to our knowledge, this is the first study to describe outcomes with ICI-related hyperglycemia during ICI treatment. In our study, 33% of patients with metastatic or unresectable NSCLC receiving ICI treatment had hyperglycemia, and none of the patients had ICI-induced type 1 diabetes or became dependent on insulin therapy. In addition, patients with hyperglycemia during ICI treatment had 18% of their random blood glucose readings at ≥200 mg/dL, highlighting the importance of glycemic monitoring before initiation of and throughout ICI treatment.
Our results are consistent with previous reports.15,22 In a retrospective study of 959 patients with multiple malignancies receiving ICIs, 228 (24%) had a lung cancer diagnosis, of which 13.5% had hyperglycemia (defined as a random blood glucose ≥200 mg/dL).22 In another retrospective chart review by Leiter and colleagues, 411 patients receiving ICIs for multiple malignancies of all stages, 105 (27%) had hyperglycemia.15 In that study, 29 (28%) of the 105 patients with hyperglycemia during ICI treatment had new-onset hyperglycemia. Treatment with corticosteroids often results in hyperglycemia, and patients receiving ICI treatment frequently receive corticosteroids to manage irAEs or as supportive care if cytotoxic chemotherapy is used in combination with ICIs. Leiter and colleagues purported that some of the ICI-related hyperglycemia could be attributed to glucocorticoids; however, a subset of the patients (N=10) had otherwise unexplained hyperglycemia, suggesting that ICIs may have a role in promoting elevations in blood glucose.15 Our results likewise suggest that corticosteroids may not account for all patients who had hyperglycemia while receiving ICI therapy. In all, 14% of the patients with NSCLC in our study did not receive corticosteroids, and their otherwise unexplained hyperglycemia could be attributed to the effects of ICI treatment alone.
Additional risk factors for ICI-related hyperglycemia beyond corticosteroid use were also assessed in the study by Leiter and colleagues, and identifying as Hispanic or Asian, having a BMI of ≥30 kg/m2, and a history of hyperglycemia were significantly associated with hyperglycemia after starting ICI treatment.15 Race/ethnicity and obesity did not increase the risk for ICI-related hyperglycemia in our study; however, there was a limited number of non-White patients in our cohort. More studies are needed to explore the outcomes of patients with NSCLC of diverse race/ethnicity. We did find that having a history of type 2 diabetes and the occurrence of hyperglycemia before ICI treatment increased a patient’s risk for hyperglycemia while receiving ICI treatment. Screening patients for hyperglycemia and diabetes before systemic cancer treatments is recommended, especially in those with risk factors.26 Assessing patients with NSCLC for a history of type 2 diabetes and/or hyperglycemia at baseline before ICI treatment initiation can guide clinical practice and help oncology practitioners predict patients who may be at the greatest risk for ICI-related hyperglycemia and proactively include glucose monitoring during therapy to ensure this potential irAE is adequately managed. Given the median onset of ICI-related hyperglycemia of 25 days in our study, oncology practitioners should be attentive to glucose monitoring, particularly during the first few cycles of therapy.
Interestingly, 22.4% of the patients with NSCLC in our study population had type 2 diabetes. This was higher than that of other research, which showed the prevalence rate of type 2 diabetes to be 10% to 16% among patients diagnosed with lung cancer.27-30 Our study’s rate of type 2 diabetes is also significantly higher than the defined prevalence rates in the general US population (11.6%) and in the state of Indiana (11.7%).31,32 Of note, in the study by Leiter and colleagues assessing risk factors for hyperglycemia in 411 patients receiving ICI treatment for multiple malignancies (24% with NSCLC), 22% had preexisting diabetes.15 This potentially higher frequency of diabetes diagnoses among those with advanced NSCLC demonstrates the importance of monitoring for hyperglycemia in this population, especially when receiving several medications that can increase the risk for diabetes, such as ICIs and corticosteroids.
In previous research, preexisting diabetes and/or hyperglycemia were associated with poorer outcomes in patients with NSCLC who received traditional cytotoxic chemotherapy.16-20 In a multicenter study of 438 patients with NSCLC receiving ICI therapy, the quartile of patients with the highest baseline glucose levels had significantly shorter survival compared with the quartile of patients with the lowest baseline glucose.21 Conversely, ICI-induced type 1 diabetes and insulin dependence were associated with improved outcomes in patients.13 Interestingly, in the patients with NSCLC who had hyperglycemia during ICI treatment in our study, the time to disease progression was approximately 3 months longer than in the patients without hyperglycemia. These findings were further supported by the results of the 4-group Kaplan–Meier analysis, which demonstrated that patients who did not have hyperglycemia or irAEs during ICI treatment had a shorter time to disease progression than those who had hyperglycemia, an irAE, or both.
Having an irAE is believed to be a marker of a robust immune response in tumors and healthy tissue; however, there is much heterogeneity in the data assessing irAE impact on outcome.33 In addition, mechanistic data explaining the association between hyperglycemia and outcomes are limited, and it is plausible that the proinflammatory state induced by hyperglycemia in patients with NSCLC receiving ICI treatment facilitates beneficial effects on the time to disease progression versus deleterious effects in the same patient population receiving traditional cytotoxic chemotherapies.
Further complicating any association between hyperglycemia and outcomes to ICI treatment in NSCLC is the established obesity paradox, in which patients with NSCLC and an increased BMI (≥25 kg/m2) have a better prognosis and respond better to treatment, including to ICIs.34 Given the competing data regarding the outcomes of patients with hyperglycemia and NSCLC and the known deleterious effects of uncontrolled hyperglycemia in general, those patients who have hyperglycemia while receiving an ICI should be monitored and managed using a multidisciplinary approach (ie, education, dietary interventions, glucose-lowering medications).26 Because of the lack of a hypoglycemia risk, metformin has been suggested as the treatment of choice in patients with cancer and hyperglycemia; however, treatment with metformin may be limited by gastrointestinal adverse events (AEs) in this population.26 Patients with disease that is uncontrolled with or who are unable to tolerate metformin may receive additional oral antidiabetes medications; however, one must consider the potential hypoglycemic effects in this population who may have alterations in their nutritional intake. Likewise, newer agents, such as glucagon-like peptide 1 agonists, could be considered; however, these agents may be difficult to tolerate because of gastrointestinal AEs and the potential to exacerbate weight loss. If hyperglycemia is uncontrollable with oral treatment options, insulin may be necessary. More studies are warranted to explain the biologic and pharmacologic mechanisms of hyperglycemia in patients with NSCLC who are receiving ICI treatment.
To our knowledge, this study is the first investigation of ICI-related hyperglycemia during ICI treatment in patients with NSCLC. Our findings are among the first to describe understudied AEs that frequently occur with ICI treatment in this vulnerable population. This study has several strengths, including a large homogeneous sample size of patients with NSCLC and the use of EHRs, which facilitated comprehensive and accurate data collection. In addition, the findings of this study describe outcomes that patients with NSCLC had during ICI treatment in a real-world setting.
Limitations
The findings of this study should be considered in the context of its limitations. The descriptive, retrospective study design precludes the ability to interpret causality. Most of the patients with NSCLC in this study were White and were from 1 institution, thereby limiting the generalizability of the findings. Larger multisite, prospective studies are needed to validate the findings of this study. The high number of patients with NSCLC and preexisting type 2 diabetes may have influenced the prevalence of ICI-related hyperglycemia and the study’s outcomes. However, to control for the impact of type 2 diabetes on hyperglycemia, we completed logistic regressions to identify the risk factors for ICI-related hyperglycemia in the entire population and the subpopulation without type 2 diabetes.
HbA1c is not routinely monitored in patients with cancer nor are glucose levels monitored outside of routine pretreatment laboratory testing; therefore, it is plausible that some patients with NSCLC may have had prediabetes or undiagnosed type 2 diabetes that was not documented in the EHR, which may have impacted the study’s findings, highlighting the importance of baseline and interval screening and the assessment of type 2 diabetes and hyperglycemia before the initiation of and throughout treatment with an ICI. Because of the retrospective design and nature of glucose monitoring in patients with cancer, a random blood glucose result of ≥200 mg/dL was used instead of fasting glucose, a 2-hour postprandial blood glucose, or home-monitored results, which limited the ability to identify additional cases of hyperglycemia. The use of a cutoff of 200 mg/dL for a random blood glucose reading is supported by previous studies and the ADA guidelines for the diagnosis of type 2 diabetes.15,22,23 The patients in our study with hyperglycemia had a longer time to documented treatment progression; therefore, it is possible that those patients receiving treatment for an extended period of time have more opportunities to have hyperglycemia. However, it must be noted that hyperglycemia tended to occur relatively quickly after starting ICI treatment.
Last, although we grouped patients by whether they received chemotherapy in combination with ICI treatment, we did not assess the impact of individual chemotherapy agents on ICI-related hyperglycemia rates. Likewise, we relied on the documentation of progression by the treating oncologist and did not assess the impact of tumor PD-L1 status on the time to NSCLC progression. Future studies should examine the impact of ICI monotherapy compared with combinations with chemotherapy on ICI-related hyperglycemia, irAEs, and subsequent patient outcomes in those who receive treatment for NSCLC.
Conclusion
Based on our results, hyperglycemia occurs often among patients with NSCLC receiving ICI treatment for metastatic or unresectable disease. Hyperglycemia may be more likely to occur in patients receiving concomitant chemotherapy and those with type 2 diabetes or hyperglycemia before starting ICI therapy. Despite many patients receiving corticosteroids, more than 10% of patients without exposure to corticosteroids had hyperglycemia. Furthermore, based on our results, hyperglycemia does not negatively impact the time to NSCLC progression or increase the risk for other irAEs. Our study shows that having hyperglycemia, an irAE, or both during ICI treatment is associated with a longer time to NSCLC progression. Prospective studies are needed to confirm these findings and explore the biologic processes that contribute to them, especially with the emergence of new immunotherapies and the increasing prevalence of diabetes.
Funding Source
This study was funded in part by the Indiana University Simon Comprehensive Cancer Center Cancer Prevention and Control pilot grant (Dr Storey).
Author Disclosure Statement
Dr Thurman, Dr Storey, and Dr Reeves have no conflicts of interest to report.
References
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): non-small cell lung cancer. Version 8.2025. Accessed November 24, 2025. www.nccn.org/professionals/physician_gls/pdf/nscl.pdf
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): management of immunotherapy-related toxicities. Version 1.2025. December 20, 2024. Accessed January 9, 2025. www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf
- Brahmer JR, Lacchetti C, Schneider BJ, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2018;36:1714-1768.
- Puzanov I, Diab A, Abdallah K, et al. Managing toxicities associated with immune checkpoint inhibitors: consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J Immunother Cancer. 2017;5:95.
- Elia G, Ferrari SM, Galdiero MR, et al. New insight on endocrine-related adverse events associated to immune checkpoint blockade. Best Pract Res Clin Endocrinol Metab. 2020;34:101370.
- Yim C, Mansell K, Hussein N, Arnason T. Current cancer therapies and their influence on glucose control. World J Diabetes. 2021;12:1010-1025.
- Phillips AL, Reeves DJ. Outcomes and management of immune checkpoint inhibitor-induced hypothyroidism: a retrospective analysis. Ann Pharmacother. 2022;56:1100-1105.
- Thuillier P, Joly C, Alavi Z, et al. Thyroid dysfunction induced by immune checkpoint inhibitors is associated with a better progression-free survival and overall survival in non-small cell lung cancer: an original cohort study. Cancer Immunol Immunother. 2021;70:2023-2033.
- Lima Ferreira J, Costa C, Marques B, et al. Improved survival in patients with thyroid function test abnormalities secondary to immune-checkpoint inhibitors. Cancer Immunol Immunother. 2021;70:299-309.
- Stamatouli AM, Quandt Z, Perdigoto AL, et al. Collateral damage: insulin-dependent diabetes induced with checkpoint inhibitors. Diabetes. 2018;67:1471-1480.
- Hamid O, Robert C, Daud A, et al. Five-year survival outcomes for patients with advanced melanoma treated with pembrolizumab in KEYNOTE-001. Ann Oncol. 2019;30:582-588.
- Wolchok JD, Chiarion-Sileni V, Gonzalez R, et al. Long-term outcomes with nivolumab plus ipilimumab or nivolumab alone versus ipilimumab in patients with advanced melanoma. J Clin Oncol. 2022;40:127-137.
- Muniz TP, Araujo DV, Savage KJ, et al. CANDIED: a Pan-Canadian cohort of immune checkpoint inhibitor-induced insulin-dependent diabetes mellitus. Cancers. 2022;14:89.
- Quandt Z, Young A, Anderson M. Immune checkpoint inhibitor diabetes mellitus: a novel form of autoimmune diabetes. Clin Exp Immunol. 2020;200:131-140.
- Leiter A, Carroll E, Brooks D, et al. Characterization of hyperglycemia in patients receiving immune checkpoint inhibitors: beyond autoimmune insulin-dependent diabetes. Diabetes Res Clin Pract. 2021;172:108633.
- Bergamino M, Rullan AJ, Saigi M, et al. Fasting plasma glucose is an independent predictor of survival in patients with locally advanced non-small cell lung cancer treated with concurrent chemoradiotherapy. BMC Cancer. 2019;19:165.
- Zeng X, Xu C, Cheng J, et al. Poor glycemic control might compromise the efficacy of chemotherapy in non-small cell lung cancer patients with diabetes mellitus. Cancer Med. 2020;9:902-911.
- Qian J, Wang W, Wang L, et al. The survival benefit for optimal glycemic control in advanced non-small cell lung cancer patients with preexisting diabetes mellitus. Front Oncol. 2021;11:745150.
- Zhang S, Pease DF, Kulkarni AA, et al. Real-world outcomes and clinical predictors of immune checkpoint inhibitor monotherapy in advanced lung cancer. Clin Med Insights Oncol. 2021;15:1-8.
- Wu WY, Luke B, Wu XC, et al. Glycemic control in diabetic patients improved overall cancer survival across diverse populations. JNCI Cancer Spectr. 2024;8:1-13.
- Osataphan S, Awidi M, Jan YJ, et al. Association between higher glucose levels and reduced survival in patients with non-small cell lung cancer treated with immune checkpoint inhibitors. Lung Cancer. 2024;198:108023.
- Mulla K, Farag S, Moore B, et al. Hyperglycaemia following immune checkpoint inhibitor therapy–incidence, aetiology, and assessment. Diabet Med. 2023;40:e15053.
- American Diabetes Association Professional Practice Committee. 2. Diagnosis and classification of diabetes: standards of care in diabetes–2024. Diabetes Care. 2024;47(suppl 1):S30-S42.
- Hammer M, Storey S, Hershey DS, et al. Hyperglycemia and cancer: a state-of-the-science review. Oncol Nurs Forum. 2019;46:459-472.
- Brady VJ, Grimes D, Armstrong T, LoBiondo-Wood G. Management of steroid-induced hyperglycemia in hospitalized patients with cancer: a review. Oncol Nurs Forum. 2014;41:E355-E365.
- Shahid RK, Ahmed S, Le D, Yadav S. Diabetes and cancer: risk, challenges, management, and outcomes. Cancers. 2021;13:5735.
- Edwards BK, Noone AM, Mariotto AB, et al. Annual report to the nation on the status of cancer 1975-2010, featuring prevalence of comorbidity and impact on survival among persons with lung, colorectal, breast, or prostate cancer. Cancer. 2014;120:1290-1314.
- Islam KMM, Jiang X, Anggondowati T, et al. Comorbidity and survival in lung cancer patients. Cancer Epidemiol Biomarkers Prev. 2015;24:1079-1085.
- Lee K, Kang S, Hwang J. Lung cancer patients’ characteristics and comorbidities using the Korean National Hospital discharge in-depth injury survey data. J Epidemiol Glob Health. 2022;12:258-266.
- Fowler H, Belot A, Ellis L, et al. Comorbidity prevalence among cancer patients: a population-based cohort study of four cancers. BMC Cancer. 2020;20:2.
- Centers for Disease Control and Prevention (CDC). National Diabetes Statistics Report: Estimates of Diabetes and Its Burden in the United States. Accessed November 24, 2025. https://stacks.cdc.gov/view/cdc/148231.
- American Diabetes Association. The Burden of Diabetes in Indiana. Accessed November 24, 2025. https://diabetes.org/sites/default/files/2023-09/ADV_2023_State_Fact_sheets_all_rev_Indiana.pdf.
- Zhou X, Yao Z, Yang H, et al. Are immune-related adverse events associated with the efficacy of immune checkpoint inhibitors in patients with cancer? A systematic review and meta-analysis. BMC Med. 2020;18:87.
- Vedire Y, Kalvapudi S, Yendamuri S. Obesity and lung cancer–a narrative review. J Thorac Dis. 2023;15:2806-2823.