Connecting Science to Practice
Biosimilars are helping lower healthcare costs and improve access
to medicines by increasing competition. However, their adoption
faces challenges, including limited uptake in certain drug categories.
This study showed that strategies such as step therapy, prior
authorization, and leveraging electronic health records can boost
biosimilar use and reduced health plan spending by 21% over 2
years at an academic medical center. Overall, effective management
and collaboration can promote biosimilar adoption and cut
healthcare costs.
Biosimilars offer a significant opportunity for cost-savings while presenting logistical hurdles for optimal implementation.1-4 Biosimilar savings nationwide are estimated to be $54 billion from 2017 to 2026, but these savings hinge on industry, regulatory, and policy changes to strengthen competition and sustainability in the biosimilar marketplace.5 The barriers to biosimilar adoption include provider acceptance, payer preferences, lack of an interchangeability designation for most biosimilars, and that biosimilars are assigned separate Healthcare Common Procedure Coding System codes requiring distinct build records within the electronic health record (EHR).1
At a multistate academic medical center, a biosimilar-first strategy was adopted starting in September 2020.1 The biosimilar-first strategy leveraged tools within the EHR to drive conversion from reference drugs to biosimilars—because guidance in the EHR is the standard approach when managing new drug coverage strategies—and to support pharmacist-driven therapeutic interchange.1,4 The preferred biosimilars were listed as the default drug or were listed first within ordering pathways and were noted as being preferred.1 Nonpreferred reference biologics and biosimilars were demoted in the drug list with a notation of being restricted to assist prescribers in appropriate drug selection.1 A pharmacist-driven therapeutic interchange authorized the pharmacy to change to a drug that is covered by the patient’s insurance if they did not cover the biosimilar that was ordered and to optimize biosimilars in the inpatient setting.1
Traditional utilization management techniques in healthcare include prospective, concurrent, and retrospective reviews.6 Step therapy and prior authorization or precertification before rendering a service or medication are in the prospective review category.6 For the employee health plan population, 85% of total medical benefit drug use occurs at employer-owned facilities. In September 2020, a substantial proportion of employee-plan patients received therapy with a reference drug, and member disruption from switching to a biosimilar was a significant concern of prescribers. Although the direct cost of biosimilars was lower than the direct cost of reference drugs, traditional utilization management techniques, such as step therapy and prior authorization, were considered less optimal from a provider and care team perspective because of the increased time and administrative burden to facilitate prior authorization before starting therapy.7 The uptake of rituximab, filgrastim, epoetin alfa, and infliximab biosimilars was driven through EHR optimization for proper drug selection. Bevacizumab and trastuzumab are often prescribed for longer durations of therapy and require a mix of traditional utilization management using step-therapy prior authorization and nontraditional case-management approaches for patient conversion from a reference drug to a biosimilar.1,6 Notably, for bevacizumab and trastuzumab only, the reference drugs required prior authorization, whereas biosimilars did not require prior authorization in an effort to successfully drive patients toward lower-cost biosimilars and reduce prior authorization burden.
For the purposes of this article, we consider nontraditional approaches to encompass driving patients from reference drugs to biosimilars within the EHR, the use of custom EHR reports to identify patients who continue receiving reference drugs, and having a lack of prior authorization requirement for biosimilars. The objective of this approach was to transition the employee health plan population from reference drugs to biosimilars more quickly and to realize cost-savings while minimizing the risk for therapy interruption, prior authorization burden, and the costs of conversion. Biosimilars to professionally administered medications with longer than 1 year of market availability were included in the initiative.
Methods
The biosimilar-first strategy was implemented beginning in September 2020 for professionally administered biosimilars to bevacizumab, filgrastim, epoetin alfa, infliximab, rituximab, and trastuzumab. Quarterly reports on utilization and health plan spending were generated from medical claims data for the reference and biosimilar drugs, absent of rebates. The number of unique users and corresponding health plan spending on reference and biosimilar drugs were evaluated by supply chain management to calculate the shifts to biosimilars and the spending reduction. The data reporting was provided by clinical colleagues at the medical carrier for the health plan population beginning in January 2021 and continues to date.
In general, the oncology indications for filgrastim, epoetin alfa, and rituximab do not require long-term treatment.8-10 Moreover, because of the lower cost associated with reference filgrastim and epoetin alfa versus biosimilars, the use of prior authorization to drive patients from reference drugs to biosimilars was not ideal because it was presumed to result in high prior authorization administrative burden to care teams.6 Therefore, for filgrastim, epoetin alfa, and rituximab, defaulting the preferred drugs within the EHR was the primary utilization management tool that was employed.
For trastuzumab and bevacizumab, EHR reports were generated by the formulary manager project lead on a quarterly basis to identify and address biosimilar conversion opportunities within the managed care population. Local care teams of clinical pharmacists practicing within the specialties were asked to change the order to a preferred biosimilar, leveraging the pharmacist-driven therapeutic interchange. For trastuzumab and bevacizumab, because patients may remain on therapy for a year, step-therapy prior authorization was used, which required patients who were treatment-naïve to have a contraindication or intolerance to at least 1 treatment with a biosimilar before allowing coverage of the reference drug.
Initially, established patients who were receiving trastuzumab and bevacizumab were grandfathered in and were allowed to finish their treatment course with the reference drug. At 1 year postimplementation, pharmacy staff members who were embedded in the practice were asked to intervene on health plan members who had not yet transitioned from a reference drug to a biosimilar. The only patients who were allowed to continue receiving reference drugs were those who were receiving treatment as part of a clinical trial in which the reference drug was mandated or when a patient was receiving copay assistance from a drug manufacturer. However, it was emphasized that many biosimilars have manufacturer patient-assistance programs to ensure that this was not a primary driver for members to continue receiving a higher-cost reference drug.
For infliximab, biosimilar conversion employed a longer-term, 2-phase approach because of sensitivity to the challenges faced by a patient population receiving long-term biologic therapy. Hesitancy by providers and patients to adopt the use of biosimilars needed a conversion approach that differed from that used for the previously mentioned therapies. Biosimilars were first preferred for all treatment-naïve patients using EHR tools. Then, clinician-led teams began the slower process of manually converting established patients, which was supported via reporting touchpoints, consistent workflows, and patient education tools. For all market categories, there was open dialogue with our managed care partners to ensure an understanding of the strategy and alignment with the overall goal of reducing spending to the plan and its members.
After 1 year, case management was used for all therapeutic categories to transition patients who continued to receive reference drugs to treatment with biosimilars. This was achieved through medical center EHR reports, provider education, and clinical pharmacy support for direct patient conversion to biosimilars. For rituximab, trastuzumab, bevacizumab, filgrastim, and epoetin alfa, the conversion was streamlined, in which patients were converted to biosimilars and providers were informed of the change.
Results
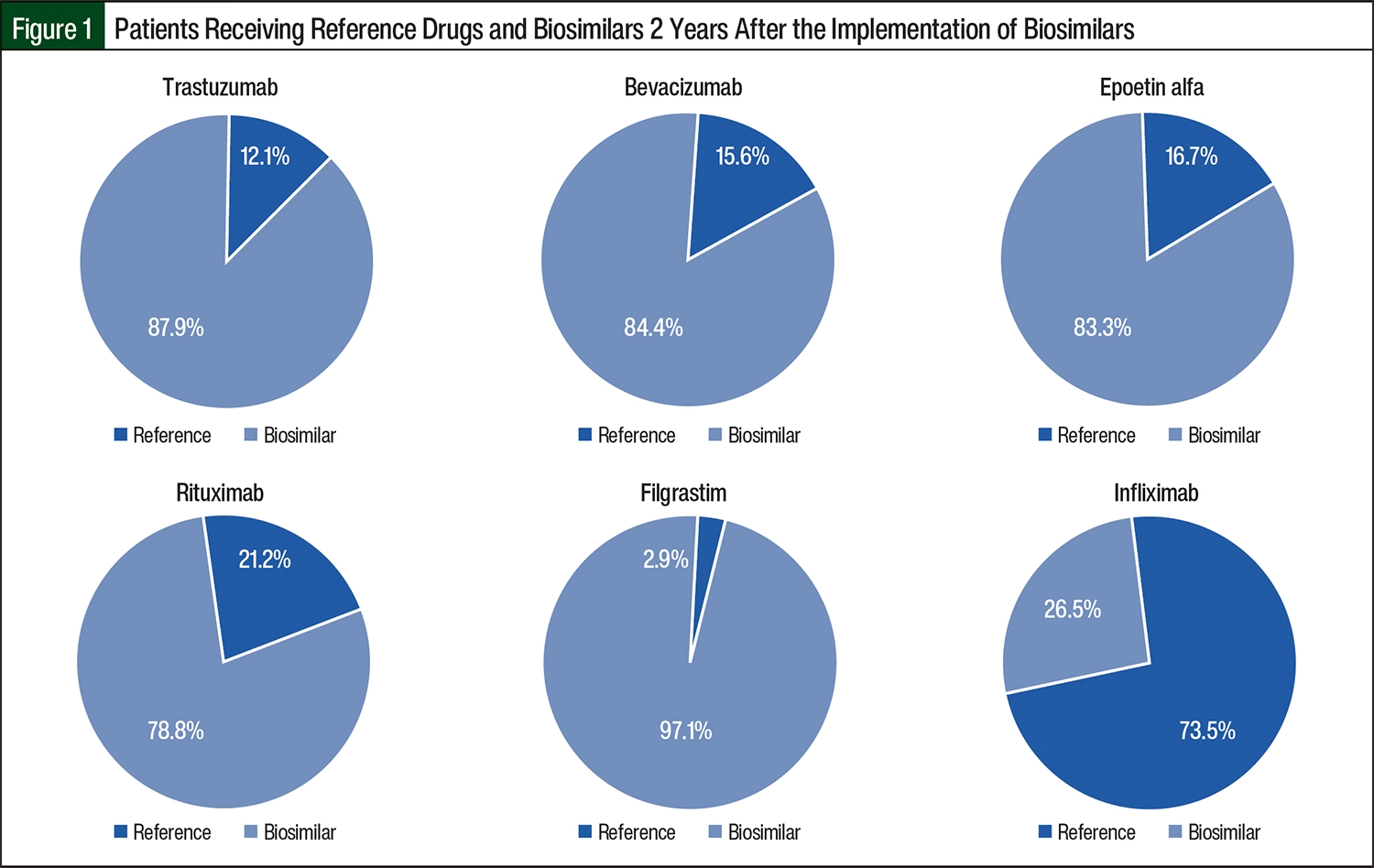
From September 2019 to August 2020 was considered the baseline period before the implementation of biosimilars. At baseline, a total of 434 patients received biosimilars or reference drugs; biosimilar utilization by members was <3% across all drug groups, except for filgrastim, which had a 6.1% biosimilar utilization rate in the baseline time frame. Two years after the implementation of biosimilars, a total of 463 patients received biosimilars or reference drugs, and the biosimilar utilization rate for members was 78.8% for rituximab; >80% each for the trastuzumab, bevacizumab, and epoetin alfa drug groups; and 97.1% for filgrastim (Figure 1). For infliximab, where implementation followed a different timeline and process based on formulary adoption, 26.5% of members were receiving biosimilars at year 2 (Figure 1).
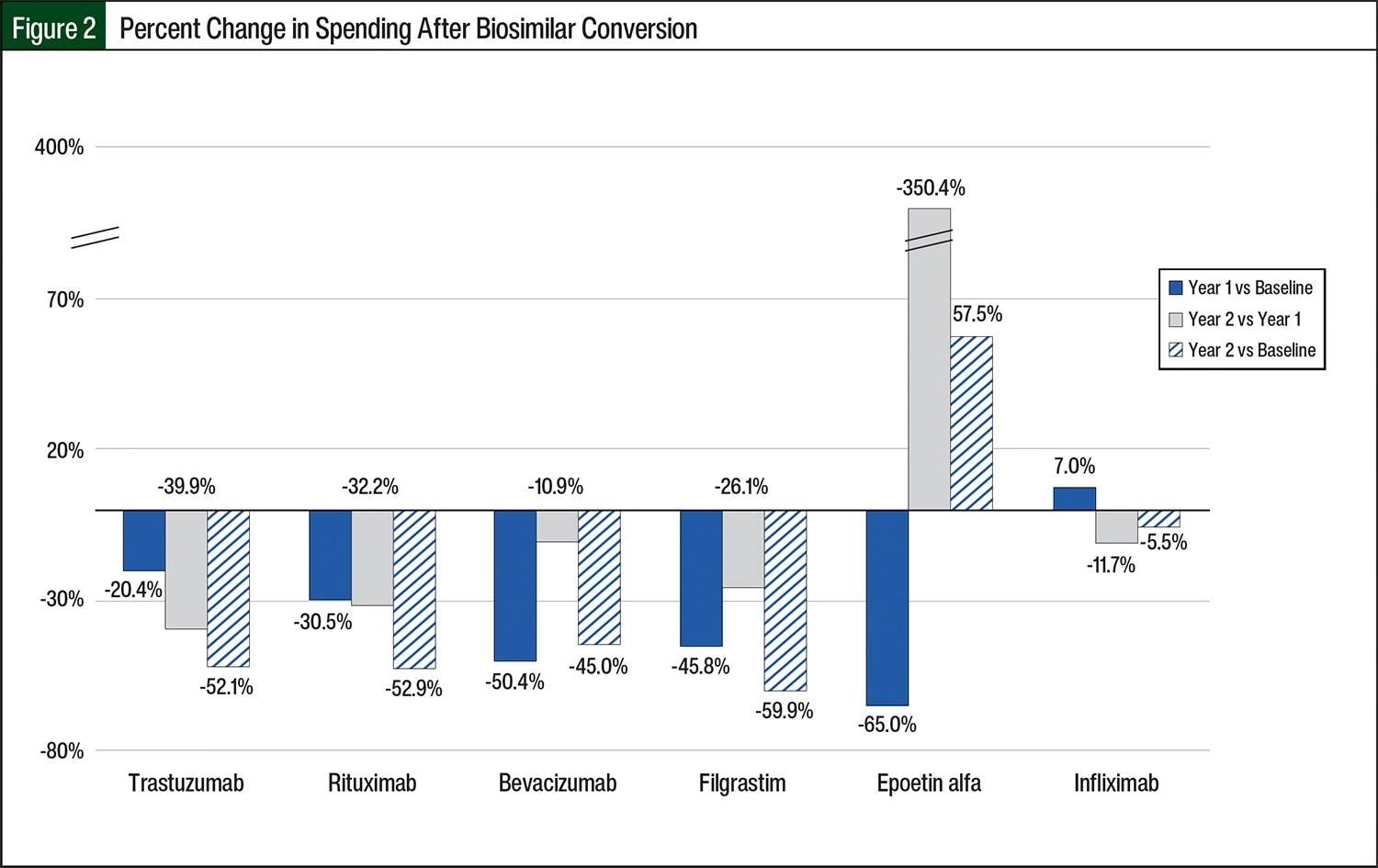
Increased health plan spending was observed in 2 of the 6 drug categories (Figure 2). This included infliximab at year 1 compared with baseline and epoetin alfa at year 2 compared with year 1 and with baseline. The infliximab market category accounted for 65.6% of the total baseline spending. Significant savings remained across all categories, with a total spending reduction of 21% at year 2 (Figure 2). Comparing baseline with year 2 after the implementation of the biosimilars, the savings amounted to $5.878 million for the health plan.
For infliximab, there was a 10.2% increase in users and an 8.8% increase in the number of treatments administered in year 1 compared with baseline. Most treatment-naïve patients were initiated on biosimilar therapy, resulting in significant cost avoidance. Comparing year 2 with the baseline for infliximab, there was a 32% increase in unique users, an 18.7% increase in the number of treatments, and a 5.5% reduction in expenses and $1.1 million in reduced spending over 2 years (Figure 2).
For epoetin alfa, the number of unique users remained steady throughout the period studied, with 10, 7, and 6 patients at baseline, year 1, and year 2, respectively, but the number of treatments significantly increased in year 2. For epoetin alfa, increased spending in year 2 was driven by an overall increased use of end-stage renal disease therapy for patients on dialysis, with 404% and 127% increases in utilization compared with year 1 and at baseline, respectively. Comparing baseline with year 2 in increased utilization, there was increased spending of $22,000 for the health plan.
Rituximab represented the second largest spending category at baseline after infliximab, resulting in an impressive 30.5% savings in year 1, 32.2% savings in year 2, and 52.9% overall savings comparing year 2 with baseline and $2.5 million in savings to the health plan (Figure 2).
For the trastuzumab category, the total spending decreased by 20.4% from baseline to year 1, and another 39.9% from year 1 to year 2 (Figure 2). Overall, from baseline to year 2, the total spending decreased by 52.1% and $1.2 million in savings to the health plan (Figure 2).
For the bevacizumab category, the total spending decreased by 50.4% from baseline to year 1, 10.9% savings in year 2, and 45% in overall savings comparing year 2 with baseline, which amounted to $1.1 million in savings to the health plan (Figure 2).
For the filgrastim category, the total spending decreased by 45.8% from baseline to year 1, 26.1% from year 1 to year 2, and 59.9% overall from baseline to year 2, resulting in $100,000 of savings to the health plan (Figure 2).
Discussion
Building and strengthening relationships across a highly matrixed organization was crucial to the overall success of the utilization management strategy that was implemented. Educating multiple practice groups, explaining the primary goals of reducing drug expenditures, reducing inventory burden, and using reporting were key contributors to gaining buy-in throughout the conversion process.11 Moreover, communicating savings and recognizing team members’ hard work was integral to maintaining momentum in reaching these goals. This multidisciplinary collaborative effort was successful at significantly increasing biosimilar use for members by year 2.
When compared with commercial industry benchmarks, our approach yielded improved or equivalent conversion to biosimilars across all market categories.12 We had greater success compared with benchmarks as a result of driving medication selection from reference drugs to biosimilars within the EHR, leveraging step-therapy prior authorization for reference drugs for bevacizumab and trastuzumab, having a multimodal approach for patients who remained on reference drugs 1 year after the implementation of biosimilars, and case-management approaches.12 Our strategies achieved ≥80% biosimilar utilization for bevacizumab, epoetin alfa, filgrastim, rituximab, and trastuzumab. For infliximab, our strategy yielded a nearly 30% shift from reference drugs to biosimilars compared with only 5% in the industry.12 The use of infliximab biosimilars is hindered by reasons such as patient or provider hesitancy and financial disincentives.13
The 2021 industry benchmark provided by Magellan Rx Management was before several market changes that impacted the space, including increased biosimilar competition, the launch of unbranded drugs, reduced list prices, and the resultant impact on average sales prices (ASPs).12,14,15 Samsung Bioepis’s biosimilar market report further illustrates challenges in this drug category, because infliximab’s biosimilar market share had only reached 50% as of quarter 4 of 2022, whereas most oncology biosimilars had previously achieved much greater conversion.15
As expected, biosimilars are increasing competition and reducing healthcare expenditures while increasing access and affordability.1,16 Biosimilar competition for drugs billed to the medical benefit has resulted in reduced list prices and ASPs for reference drugs and biosimilars alike.14 Moreover, the removal of prior authorization requirements from biosimilar drugs expedites access to care and generates reduced healthcare costs from prior authorization itself, which was estimated to cost $10.92 per authorization and approximately 21 minutes per manual transaction in 2019.17
After the COVID-19 pandemic, the remote workforce has continued to grow, and an increasing number of plan members now receive treatment outside of the academic medical center. To support biosimilar use moving forward, or if an increase in reference drug prescribing from a nonowned facility occurs, traditional utilization management techniques, such as step therapy and prior authorization, will have to be considered. For medical centers that have a similar biosimilar strategy and use the EHR to drive utilization from reference drugs to biosimilars, there likely is a direct benefit for employees who receive treatment at their facilities. For medical centers that do not leverage biosimilars within the EHR, reporting capabilities and partnering with the medical plan carrier may be suitable strategies for patient conversion from reference drugs to biosimilars.
Last, for payers without a designated medical facility, preferring biosimilars and implementing step-therapy prior authorization or formulary exclusion for reference biologics would be effective utilization management strategies to reduce health plan costs.
Biosimilars to adalimumab, as well as the near-term launch of a biosimilar to ustekinumab in the self-administered space, offer new opportunities that can build on previous experience with the adoption of professionally administered biosimilars. Pricing dynamics for pharmacy benefit medications will create added complexity. It is our goal that experience with conversion from reference drugs to biosimilars covered under the medical benefit will offer critical learnings. Continued collaboration and communication with multiple stakeholders will be vital to address these complexities.
Limitations
Our study had limitations. Almost 85% of the employee health plan utilization occurs at employer-owned facilities, which creates a unique opportunity for internal management strategies within this population. The impact may not be as pronounced for plans where members receive care primarily from non–employer-owned facilities.
The overall health plan spending data were based on claims reimbursement and did not consider medical center expenses or administrative time spent on reporting and clinical pharmacy interventions with providers. The rebates to the medical plan were considered negligible for professionally administered biologics and were not included in the analysis.
Conclusion
Leveraging traditional and nontraditional utilization management techniques across a multidisciplinary team with shared goals and clear communication was an effective strategy to drive biosimilar adoption and reduce plan spending by 21% over 2 years at an academic medical center with a self-insured employee health plan population. For medical plan carriers or employee health plan populations, a combination of these utilization management techniques can be leveraged to promote biosimilar use and reduce healthcare costs.
Acknowledgments
The authors acknowledge Tina Seekamp, CPhT, for her assistance with the figures and graphs; Gary Effertz for the data extraction; and Ray Creighton-Lewis, PharmD, BCACP, BCPPS, AE-C, for coordination between the medical plan carrier and clinical teams and data analysis.
Author Disclosure Statement
Ms Wescott’s Mayo Clinic salary is funded by Alluma, a pharmacy benefit manager, and she has received honoraria from the Academy of Managed Care Pharmacy; and Dr Jensen was a consultant and received honoraria from Amgen and from Aptitude Health Cases. Dr Erickson, Dr Bartoo, and Dr Wallingford have no conflicts of interest to report.
References
- Jensen CJ, Tichy EM, Lempke MB, et al. Implementing and optimizing biosimilar use at Mayo Clinic. Mayo Clin Proc. 2022;97:1086-1093.
- Webster J, Smith RE, Wieland D, et al. Cost savings of biosimilar pegfilgrastim in a Medicare OCM population. J Clin Oncol. 2020;38(15 suppl):e19362.
- Aschermann LM, Forshay CM, Kennerly-Shah J, Pilz J. The formulary process for biosimilar additions at a comprehensive cancer center. J Oncol Pharm Pract. 2022;28:185-189.
- Kar I, Kronz M, Kolychev E, et al. Biosimilar strategic implementation at a large health system. Am J Health Syst Pharm. 2022;79:268-275.
- Mulcahy AW, Hlavka JP, Case SR. Biosimilar cost savings in the United States: initial experience and future potential. Rand Health Q. 2018;7:3.
- Weller J. An introductory guide to utilization management in healthcare. Smartsheet. January 9, 2020. Accessed October 25, 2024. www.smartsheet.com/content/utilization-management
- Turner A, Miller G, Clark S. Impacts of prior authorization on health care costs and quality: a review of the evidence. National Institute for Health Care Reform. November 2019. Accessed October 25, 2024. www.nihcr.org/wp-content/uploads/Altarum-Prior-Authorization-Review-November-2019.pdf
- Neupogen (filgrastim) injection, for subcutaneous or intravenous use [prescribing information]. Amgen; April 2023. Accessed October 25, 2024. http://pi.amgen.com/united_states/neupogen/neupogen_pi_hcp_english.pdf
- Epogen (epoetin alfa) injection, for intravenous or subcutaneous use [prescribing information]. Amgen; April 2024. Accessed October 25, 2024. www.pi.amgen.com/~/media/amgen/repositorysites/pi-amgen-com/epogen/epogen_pi_hcp_english.pdf
- Rituxan (rituximab) injection, for intravenous use [prescribing information]. Biogen and Genentech; December 2021. Accessed October 25, 2024. www.gene.com/download/pdf/rituxan_prescribing.pdf
- Skerik S. The key to leading change: employee buy-in. Engagement Multiplier. Accessed September 9, 2024. www.engagementmultiplier.com/resources/the-key-to-leading-change-employee-buy-in/
- Magellan Rx Management. Medical Pharmacy Trend Report, Twelfth Edition. 2022. Accessed August 14, 2023. www1.magellanrx.com/documents/2022/12/medical-pharmacy-trend-report-2022.pdf
- Chen AJ, Gascue L, Ribero R, Van Nuys K. Uptake of infliximab biosimilars among the Medicare population. JAMA Intern Med. 2020;180:1255-1256.
- Joszt L. Price discounts have driven higher biosimilar usage, especially in oncology. Am J Manag Care; July 13, 2023. Accessed July 13, 2023. www.ajmc.com/view/price-discounts-have-driven-higher-biosimilar-usage-especially-in-oncology
- Samsung Bioepis. Biosimilar Market Report: 2nd Edition, Q3 2023. 2023. Accessed September 10, 2024. www.samsungbioepis.com/upload/attach/SB+Biosimilar+Market+Report+Q3+2023.pdf
- Gascón P, Tesch H, Verpoort K, et al. Clinical experience with Zarzio in Europe: what have we learned? Support Care Cancer. 2013;21:2925-2932.
- Daly R. Prior-authorization cost and time burdens increase for providers, report finds. Healthcare Financial Management Association. January 22, 2020. Accessed September 1, 2024. www.hfma.org/payment-reimbursement-and-managed-care/health-plan-payment-and-reimbursement/prior-authorization-cost-and-time-burdens-increase-for-providers/