Connecting Science to Practice
Palbociclib, which is used in the treatment of advanced or metastatic breast cancer in adults, has a risk for neutropenia. This retrospective cohort study looked at the incidence of grade 3 or 4 neutropenia and febrile neutropenia (FN) in patients aged ≥18 years who received palbociclib; it also considered outcomes of patients who received pharmacist-managed medication therapy disease management (MTDM) for the neutropenia versus those who did not receive this service. Although the relationship between the incidence of FN in the setting of grade 3 or 4 neutropenia in the patients who received palbociclib was nonsignificant, this study showed that pharmacist involvement in MTDM services can positively contribute to improved patient outcomes in patients with grade 3 or 4 FN.
In the year 2020, female breast cancer was the most diagnosed cancer worldwide, with an estimated 2.3 million new cases; furthermore, it was the fifth leading cause of cancer mortality worldwide.1 The course of treatment is determined by review of diagnostic bilateral mammogram, pathology results, and tumor estrogen receptor status and can be assessed using the NCCN treatment guidelines.2 Neutropenia is a common hematologic consequence of chemotherapy.3 Neutropenia is classified into 4 categories by Common Terminology Criteria for Adverse Events version 5, including grade 1 (absolute neutrophil count [ANC] >1500 cells/mm3), grade 2 (ANC 1000-1500 cells/mm3), grade 3 (ANC 500-1000 cells/mm3), and grade 4 (ANC <500 cells/mm3).3,4 The presence of neutropenia increases the risk for bacterial and fungal infections resulting from decreased immunologic response.3
Palbociclib is indicated for the treatment of adults with hormone receptor–positive, HER2-negative, advanced or metastatic breast cancer, in combination with an aromatase inhibitor, as initial endocrine-based therapy in postmenopausal women or with fulvestrant in patients with disease progression after endocrine therapy.5 Palbociclib is an inhibitor of cyclin-dependent kinases 4 and 6 and thus inhibits cellular proliferation.5 The safety and efficacy of palbociclib were evaluated in the PALOMA-1, PALOMA-2, and PALOMA-3 studies.5-8
The phase 2 PALOMA-2 study, which was a continuation of the PALOMA-1 trial, evaluated palbociclib 125 mg daily plus letrozole 2.5 mg daily versus placebo plus letrozole.5-7 PALOMA-2 was a phase 2, double-blind, randomized study in a 2:1 ratio of 666 postmenopausal women with estrogen receptor–positive, HER2-negative breast cancer who did not previously receive treatment for advanced disease.7 The most common adverse event (AE) in the treatment group was neutropenia. In all, 249 (56.1%) patients had grade 3 neutropenia and 46 (10.4%) patients had grade 4 neutropenia. In the treatment group, grade 3 or 4 febrile neutropenia (FN) occurred in 8 (1.8%) patients and in none of the patients in the placebo group.7
PALOMA-3 was a phase 3, double-blind study of 521 patients who were randomized 2:1 to receive oral palbociclib (125 mg daily for 3 weeks, followed by 1 week off) and intramuscular fulvestrant (500 mg every 14 days for the first 3 injections and then every 28 days) or matching placebo and fulvestrant.8 As in PALOMA-2, the most common AE in the treatment group in PALOMA-3 was neutropenia. A total of 184 (53.3%) patients had grade 3 neutropenia and 30 (8.7%) patients had grade 4 neutropenia. FN occurred in 2 (0.6%) patients in the treatment group and in 1 (0.6%) patient in the placebo group.8
The PALOMA-1, PALOMA-2, and PALOMA-3 studies did not distinguish the incidence of FN between patients with grade 3 neutropenia and those with grade 4 neutropenia.6-8 Thus, palbociclib’s prescribing information is reflective of these studies and endorses the interruption of therapy when a patient has grade 3 neutropenia.5 Ultimately, there is no clear distinction in the incidence of FN between patients with grade 3 neutropenia and those with grade 4 neutropenia while receiving palbociclib therapy.
When managing treatment with palbociclib, there are 2 different practices that may be followed, including following the prescribing information and therapy continuation through grade 3 neutropenia. Per the prescribing information for palbociclib, therapy should be held if a patient has grade 3 neutropenia until symptoms resolve to grade ≤1 neutropenia or to grade ≤2 neutropenia if doing so is not considered a safety risk for the patient.5 When the patient’s symptoms recover, the dose of palbociclib is reduced to 100 mg daily if it is the patient’s first occurrence of grade 3 neutropenia or to 75 mg daily if this is the second occurrence of grade 3 neutropenia.5
Deviating from the prescribing information, the practice of continuing therapy through grade 3 neutropenia is derived from the mechanism of action of palbociclib, which slows down the cell cycle for white blood cells and is not cytotoxic.9 These data have been supported by Hu and colleagues, whose study results confirm the dose-dependent ANC suppression effects of palbociclib as well as the reversibility of the drug.9 In addition, a study by Kim and colleagues evaluated the efficacy of limited dosage modifications for palbociclib-related grade 3 neutropenia in hormone receptor–positive metastatic breast cancer and concluded that limited dosage modifications while the patient has grade 3 neutropenia may lead to progression-free survival without increasing AEs.10
With respect to our institution’s practices, patients continue receiving their current dosage of palbociclib through grade 3 neutropenia unless the patient has adverse drug reactions or FN. If a patient has adverse drug reactions or grade 3 FN, the dose of palbociclib is reduced to 100 mg daily if this is the patient’s first occurrence of grade 3 neutropenia or to 75 mg daily if this is the second occurrence of grade 3 neutropenia. In regard to our institution’s protocols, all patients presenting with grade 3 or 4 FN are admitted to the health system’s medical oncology service at one of our academic institutions for culture collection and antimicrobial therapy.
Pharmacist-managed medication therapy disease management (MTDM) services are offered for all patients who receive oral chemotherapy. Providers may refer patients to receive pharmacist-managed MTDM services through a collaborative practice agreement; however, this is not a requirement. Patients are referred to pharmacist-managed MTDM services based on the provider’s preference of pharmacist involvement and patient acceptability of the additional service. Through our collaborative practice agreement, pharmacists order laboratory testing and monitor laboratory values, as well as check for side effects and drug-related AEs, throughout the course of the patient’s oral chemotherapy treatment. When a pharmacist identifies a medication-related concern or issue, the patient’s oncologist is alerted and appropriate recommendations are relayed, such as therapy interruption or dosage adjustments, as outlined above. At our institution, when a patient has grade 4 neutropenia, therapy is typically held for 7 days and laboratory tests are repeated before therapy is continued.
In this study, we evaluated the incidence of FN in patients with grade 3 or 4 neutropenia who received palbociclib. The secondary outcomes included pharmacist-managed MTDM involvement versus no involvement with respect to grade 3 and 4 neutropenia, the occurrences of grade 3 and 4 neutropenia per patient, and pharmacist-managed MTDM involvement in occurrences of grade 3 FN.
Methods
This retrospective cohort study evaluated the incidence of FN in patients with grade 3 or 4 neutropenia who received palbociclib. This study included patients from Geisinger Health System from January 1, 2018, to December 31, 2020. This health system is comprised of 10 academic institutions and 11 hematology and oncology clinics serving several regions in northeastern, central, and western Pennsylvania. The patient data of the study population were retrieved with information from hospital admissions as well as from outpatient clinic appointments, with all patients receiving their primary oncology services through our health system. The patients’ data were retrieved by a data scientist from Geisinger’s Center for Pharmacy Innovation and Outcomes and were further evaluated by the study team. This study was approved by the Geisinger Institutional Review Board.
This study included patients aged ≥18 years who received treatment with palbociclib and had grade 3 or 4 neutropenia. This population was further evaluated for the incidence of FN in patients with grade 3 or grade 4 neutropenia. FN was defined as a single body temperature of >38.3°C (101°F) or a temperature of >38°C (100.4°F) sustained for >1 hour within 7 days of having grade 3 or 4 neutropenia. A diagnosis of FN was identified by International Classification of Diseases, Tenth Revision codes, as well as by chart review, to identify the administration of antibiotics while the patient had grade 3 or 4 neutropenia. There were no exclusion criteria for this study.
The primary study outcome evaluated the incidence of FN in patients with grade 3 or 4 neutropenia who received palbociclib. The occurrence of neutropenia was assessed using laboratory values extracted from the electronic health record (EHR). Laboratory values that were recorded within 7 days of one another were included as 1 occurrence, and the grade of neutropenia corresponded to the first recorded laboratory value.
The secondary outcomes evaluated the effect of pharmacist-managed MTDM involvement, including pharmacist-managed MTDM involvement versus no involvement with respect to grade 3 or 4 neutropenia, the occurrence of grade 3 or 4 neutropenia per patient, and pharmacist-managed MTDM involvement in patients with grade 3 FN. Patients who received pharmacist-managed MTDM services were identified by manual chart review.
Statistical Analysis
The primary and secondary outcomes were assessed using descriptive statistics and the Fisher’s exact test. Descriptive statistics were used to present the patients’ baseline characteristics and study results. Data that were presented as descriptive statistics use average (percent), standard deviation (SD), range, median, and interquartile ranges to show the study findings, as appropriate. Statistical testing was performed using Real Statistics Resource Pack for Excel 2022 (XRealStats; 2022).
Results
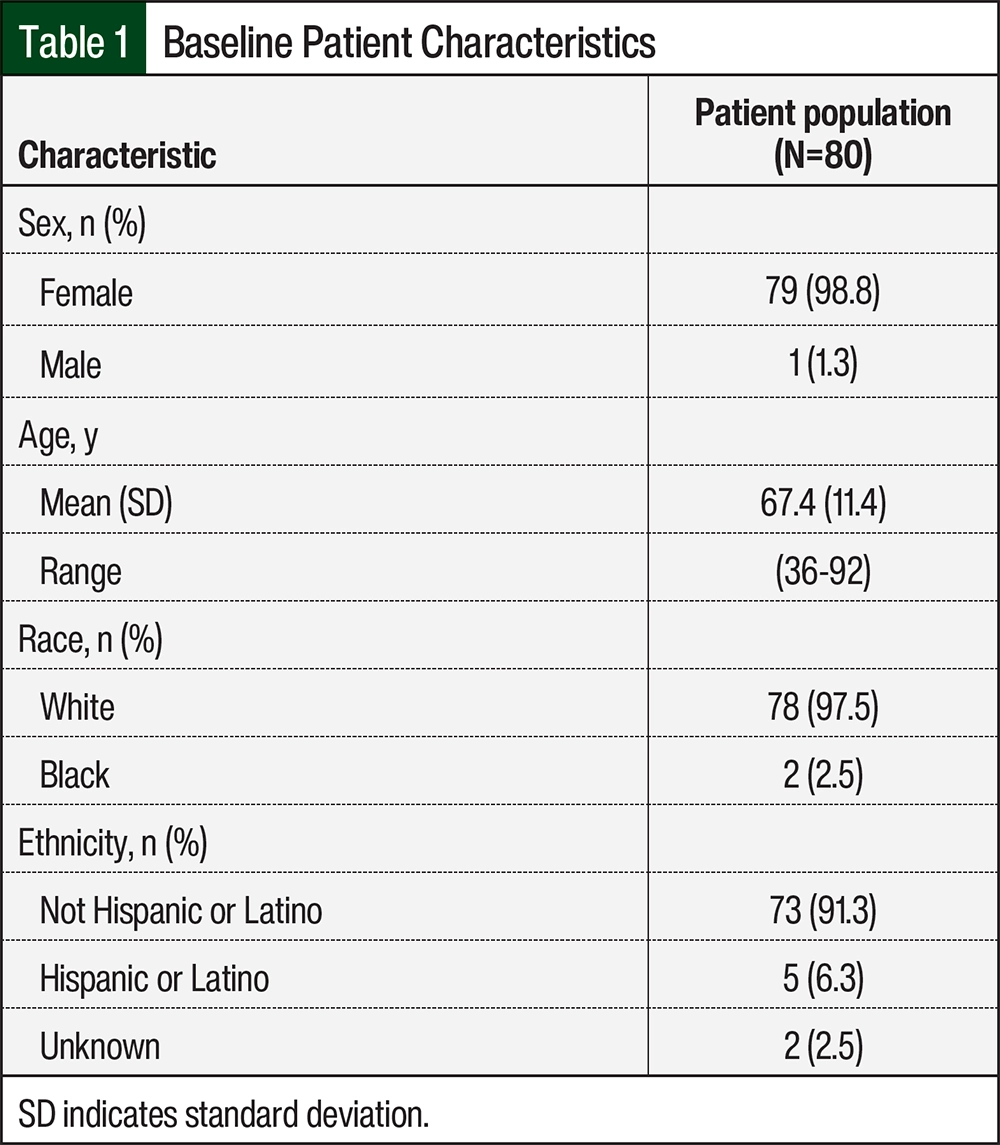
Between January 1, 2018, and December 31, 2020, 200 patients were prescribed palbociclib. Of these 200 patients, 80 had grade 3 or 4 neutropenia. The patients’ baseline characteristics included sex, age, race, and ethnicity (Table 1). The study population included 79 women diagnosed with breast cancer and 1 male patient diagnosed with dedifferentiated liposarcoma. The average patient age was 67.4 years (SD, 11.4), with the youngest patient aged 36 years and the oldest patient aged 92 years. Of the 80 patients, 78 (97.5%) were White and 73 (91.3%) patients identified as non-Hispanic or non-Latino (Table 1). Overall, the baseline characteristics revealed that most of this study’s patients were women, White, and identified as non-Hispanic or non-Latino.
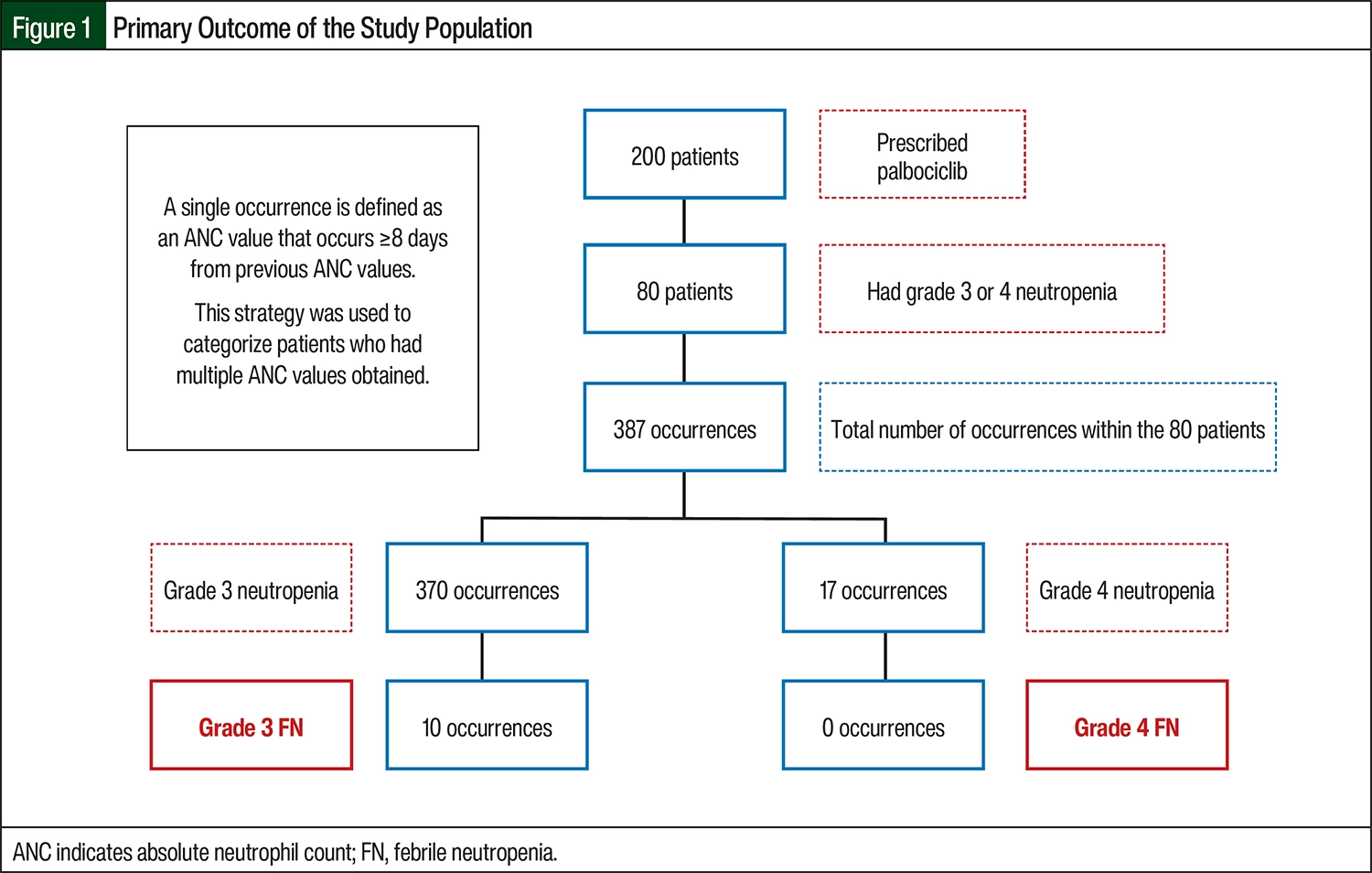
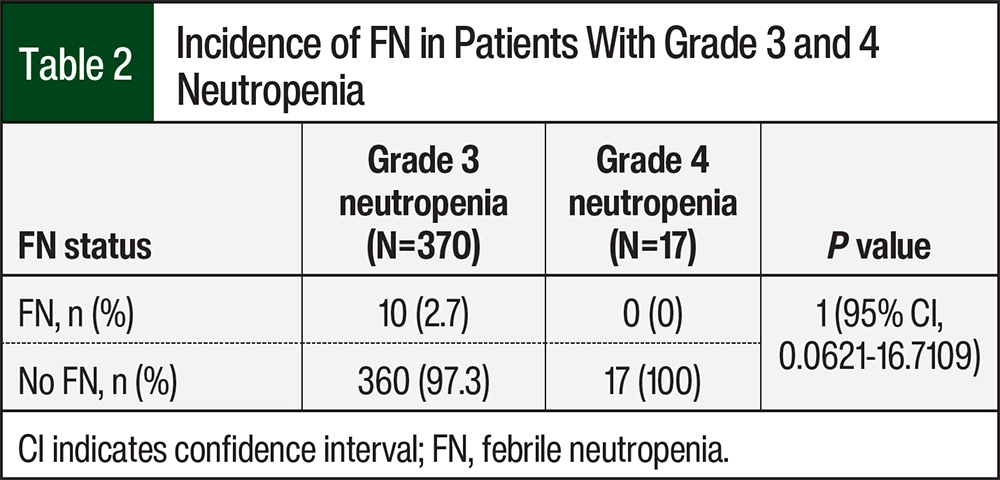
According to laboratory results, the 80 patients had a total of 387 occurrences of grade 3 or 4 neutropenia (Figure 1). Of the 387 occurrences of neutropenia, 370 were grade 3 and 17 were grade 4. There were 10 (2.7%) occurrences of FN in patients with grade 3 neutropenia and no occurrences of FN in those with grade 4 neutropenia (relative risk [RR], 1.0189; 95% confidence interval [CI], 0.0621-16.7109; P=1; Figure 1, Table 2).
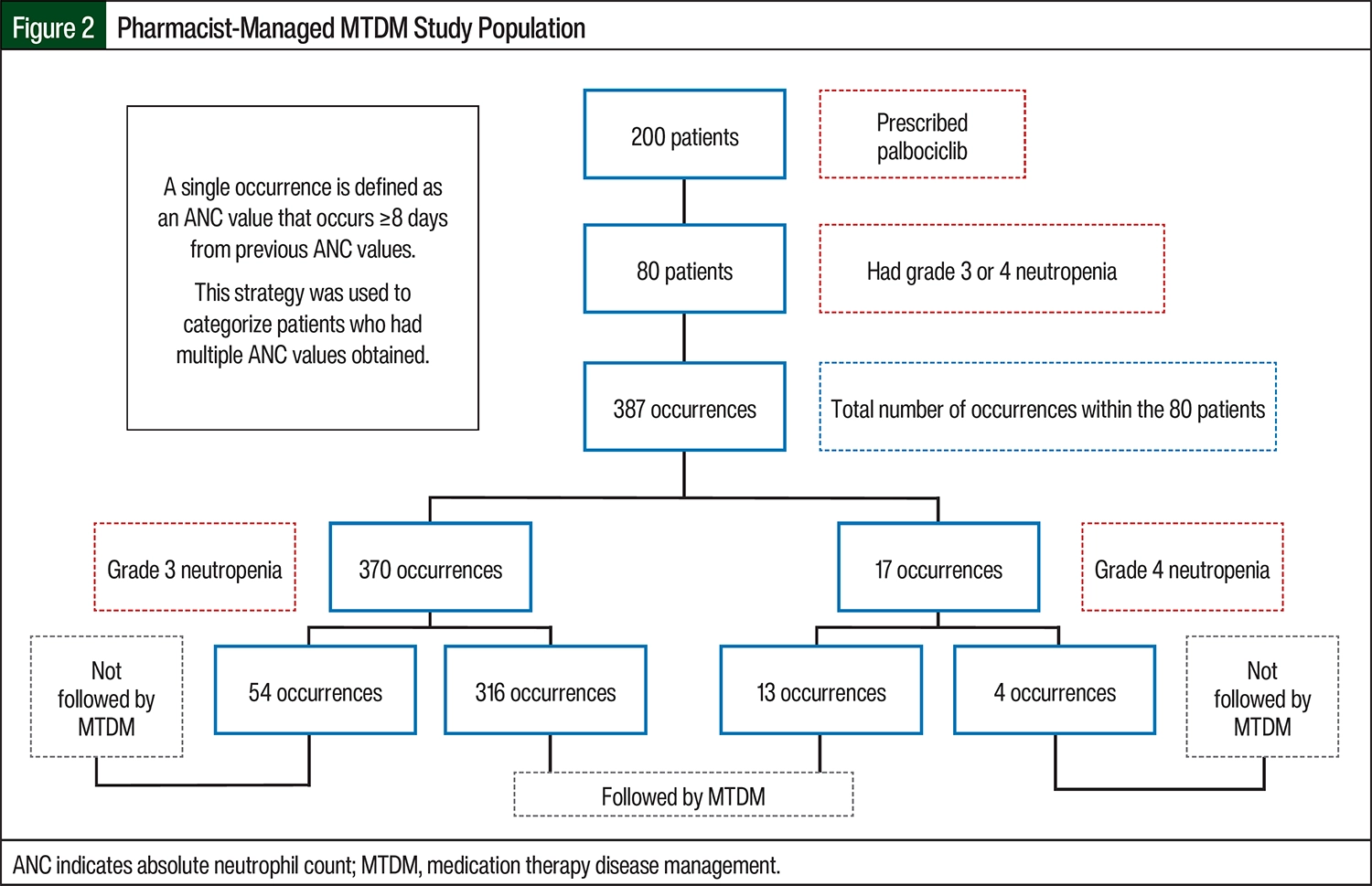
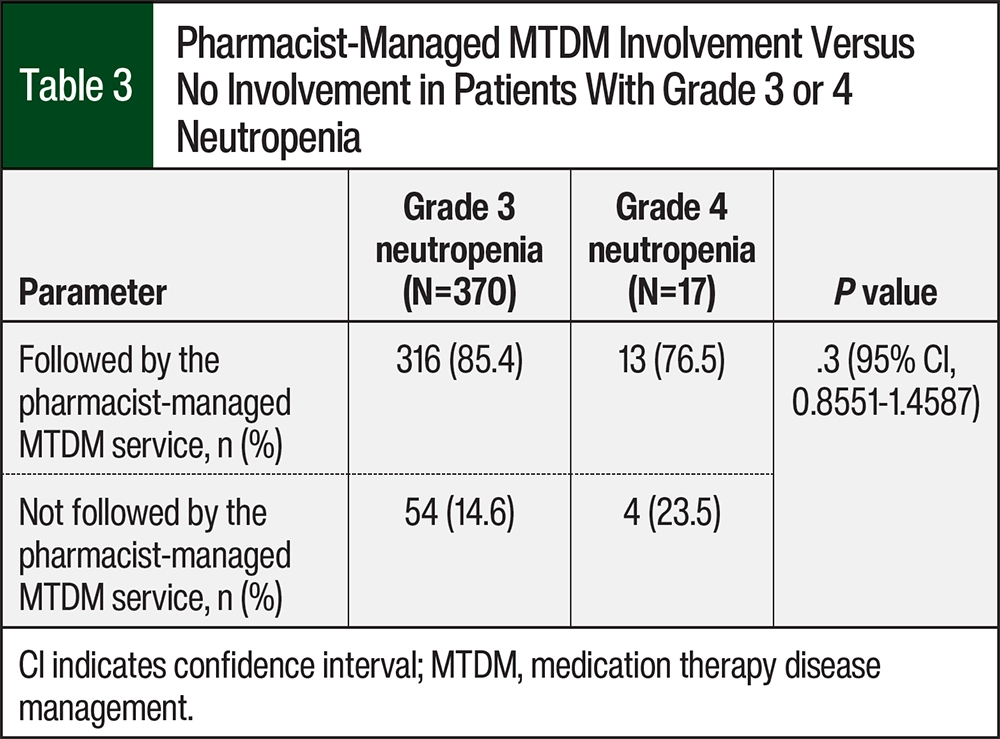
A total of 72 (90%) patients received pharmacist-managed MTDM services. Of the 370 occurrences of grade 3 neutropenia, 54 were in patients who were not followed by the pharmacist-managed MTDM service and 316 were in patients who were followed by the pharmacist-managed MTDM service (Figure 2, Table 3). Of the 17 occurrences of grade 4 neutropenia, 4 were among patients who were not followed by the pharmacist-managed MTDM service and 13 were in patients who were followed by the pharmacist-managed MTDM service (RR, 1.1168; odds ratio, 1.8; 95% CI, 0.8551-1.4587; P=.3; Figure 2, Table 3).
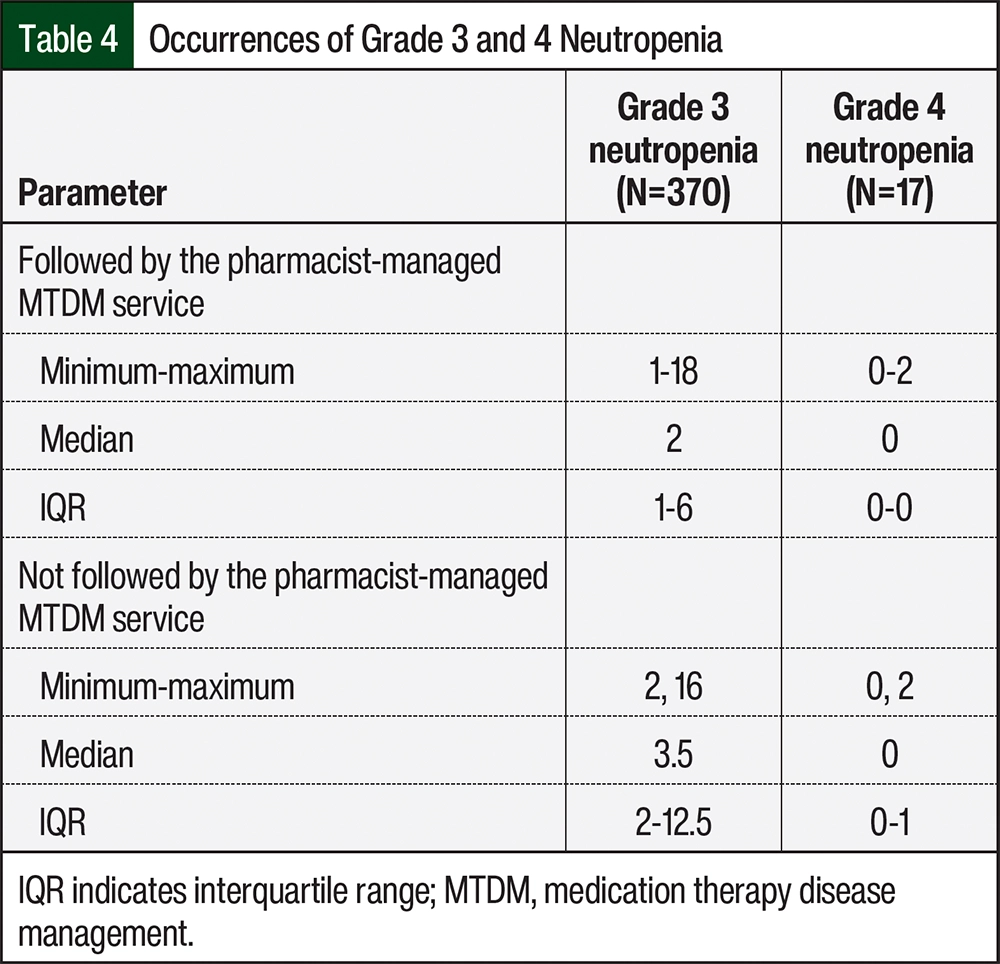
To establish an understanding of the relationship between the number of occurrences of grade 3 and 4 neutropenia between patients who were followed by the pharmacist-managed MTDM service and those who did not receive this service, we evaluated the median, minimum value, maximum value, and interquartile range (IQR) of each group. With respect to grade 3 neutropenia, the median number of occurrences per patient who was followed by the pharmacist-managed MTDM service was 2 (minimum, 1-maximum, 18; IQR, 1-6) and the median number of occurrences per patient who was not followed by the pharmacist-managed MTDM service was 3.5 (minimum, 2-maximum, 16; IQR, 2-12.5; Table 4). The occurrence of grade 4 neutropenia was similar in the 2 groups.
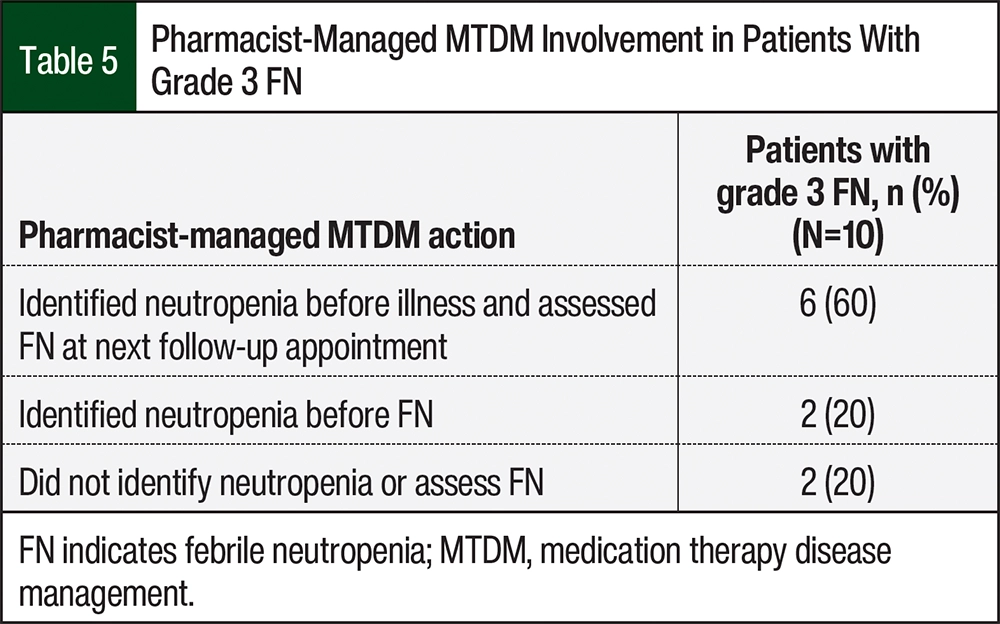
All 10 of the occurrences of grade 3 FN were in patients who were followed by the pharmacist-managed MTDM service. As stated previously, there were no occurrences of FN in the patients with grade 4 neutropenia. This study collected information to assess if the pharmacists identified the patients’ neutropenia before illness and assessed FN at the next follow-up appointment. The patients receive MTDM services via telephone calls; thus, follow-up appointments after FN consisted of subjective data relayed to the pharmacist from the patient. Of these 10 patients, 6 (60%) had a pharmacist identify neutropenia and assess FN at their next follow-up appointment (Table 5). There were also 2 (20%) patients who only had an identification of neutropenia by a pharmacist before their illness. Thus, 8 (80%) of the 10 patients had a pharmacist identify their neutropenia before they were diagnosed with FN (Table 5).
Discussion
In this study, there were more occurrences of grade 3 neutropenia than grade 4 neutropenia. This was comparable with the results of the PALOMA-1, PALOMA-2, and PALOMA-3 studies.6-8 Regarding grade 3 and 4 neutropenia, there were only cases of grade 3 FN in this study. At the time of this study, the incidences of grade 3 and 4 FN were not compared in patients who received palbociclib. Although the primary outcome of this study was nonsignificant, there are multiple additional considerations that should be discussed.
Palbociclib’s prescribing information recommends therapy interruption if the patient has grade 3 neutropenia on day 1 of the cycle until the ANC has recovered to grade 2 neutropenia.5 If the patient has grade 3 neutropenia on day 15 of therapy, the prescribing information recommends the continuation of therapy at the current dose to complete the cycle and that the complete blood count is repeated on day 22, with dosage modification if the patient has grade 4 neutropenia on day 22.5 Although the prescribing information advocates for a holding period in patients with grade 3 neutropenia,5 recent practices consist of therapy continuation with close monitoring parameters. Even with therapy continuation at our site in patients with grade 3 neutropenia, there were only 17 occurrences of grade 4 neutropenia during the 2-year study period.
Various studies have investigated the value of pharmacist-integrated services within the hematology and oncology healthcare team.11-13 A study by Vulaj and colleagues evaluated the effect an oncology pharmacist specialist may have on ASCO Quality Oncology Practice Initiative achievement metrics.11 Through this study, areas of actionable metrics that could be accomplished with pharmacist involvement were identified, which included areas such as counseling and education, participating in protocol-based care, managing symptoms, providing supportive care, and medication reconciliation.11
Our study shows that the overall incidence of grade 3 or 4 FN in patients who received palbociclib at our institution was low, with only 10 occurrences of grade 3 FN and no occurrences of grade 4 neutropenia. Thus, it is reasonable to assume that there was a positive effect on patient care with pharmacist involvement, as is demonstrated by a lower median of occurrences of grade 3 neutropenia per patient receiving pharmacist-managed MTDM services. Furthermore, McKee and colleagues used a survey to evaluate the role of the patient–pharmacist relationship in patients undergoing chemotherapy.12 The survey results showed that patients undergoing chemotherapy were interested in meeting with a pharmacist regularly.12 Likewise, Munro and colleagues completed a prospective mixed-methods study involving surveys and patient interviews that assessed patients’ perception of ambulatory oncology clinical pharmacists.13 The survey and interview results confirmed that patients felt the services offered by oncology clinical pharmacists were important to their care and that the patients were highly satisfied with the services they received.13 Thus, our study correlates with and supports the idea that pharmacists develop important relationships with patients undergoing chemotherapy.
There were no occurrences of grade 4 FN within our study. The monitoring of patient laboratory values and clinical status by qualified oncologists in our institution, as well as MTDM pharmacist–managed services may have contributed to the lack of occurrences of grade 4 FN in our patient population. Mathur and colleagues studied the components of a hematology/oncology pharmacist telehealth visit at our institution.14 This study evaluated the multiple medication-related interventions made by pharmacists, which included increased medication monitoring, care coordination, nonpharmacologic interventions, clarification with a provider, patient education, modification of drug therapies, and other interventions. More important, because our oral chemotherapy pharmacist–managed MTDM service is telephone-based, these interventions can be performed while patients are at home. Transportation can be difficult for many patients, and these telephone encounters offer quality care without the need for an in-person clinic visit.14 Although our study did not result in any significant differences between the patients who received MTDM services and those who did not, it is imperative to acknowledge the positive clinical implications and value that pharmacists add to the healthcare team.
Limitations
This study does have several limitations. This was a single health system, retrospective cohort study. The data were collected using information documented in the EHR. Thus, the study team’s manual chart review relied on accurate documentation within appropriate locations of the EHR. Next, our study population consisted primarily of patients who were female, White, and did not identify as Hispanic or Latino, therefore applicability may be limited in other regions with a differing demographic.
A study by Lee and colleagues concluded that a low baseline ANC, white blood cell count, platelets, and body surface area were significant risk factors for early palbociclib-induced grade 3 or 4 neutropenia.15 These low baseline values were defined as an ANC <3700 cells/mm3, a white blood cell count <6.3×109/mm3, platelets of <230×109/mm3, and a body surface area of <1.58 m2.15 However, these risk factors were not evaluated in our study.
Subsequently, our data collection and analysis did not include the dose of palbociclib when patients had AEs or illness; thus, no conclusions can be assessed with respect to the dose of palbociclib that the patient received. Last, power was not calculated for this study, and the small patient population may have prohibited the identification of patients with grade 4 neutropenia.
Conclusion
In our study, the relationship between the incidence of FN in the setting of grade 3 or 4 neutropenia was nonsignificant in the patients who received palbociclib. This study’s results show that pharmacist-managed MTDM services may contribute to better outcomes in patients with grade 3 or 4 FN.
More robust research that incorporates multiple healthcare institutions and a larger study cohort is warranted to further investigate the relationship between FN in the setting of grade 3 or 4 neutropenia before definitive conclusions can be determined.
Author Disclosure Statement
Dr Ivey, Dr Maiers, and Dr Maley have no conflicts of interest to report.
References
- Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209-249.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): breast cancer. Version 4.2024. July 3, 2024. Accessed March 14, 2022. www.nccn.org/professionals/physician_gls/pdf/breast.pdf
- Dale DC. Merck Manual Professional Version. Neutropenia. Merck & Co., Inc. Revised April 2023. Accessed March 14, 2022. www.merckmanuals.com/professional/hematology-and-oncology/leukopenias/neutropenia
- National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0. November 27, 2017. Accessed May 19, 2022. https://academy.myeloma.org.uk/wp-content/uploads/2015/04/CTCAE_v5.pdf
- Ibrance (palbociclib) capsules, for oral use [prescribing information]. Pfizer; September 2023. Accessed August 14, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2023/207103s017s018lbl.pdf
- Finn RS, Crown JP, Lang I, et al. The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2-negative, advanced breast cancer (PALOMA-1/TRIO-18): a randomised phase 2 study. Lancet Oncol. 2015;16:25-35.
- Finn RS, Martin M, Rugo HS, et al. Palbociclib and letrozole in advanced breast cancer. N Engl J Med. 2016;375:1925-1936.
- Turner NC, Ro J, André F, et al. Palbociclib in hormone-receptor–positive advanced breast cancer. N Engl J Med. 2015;373:209-219.
- Hu W, Sung T, Jessen BA, et al. Mechanistic investigation of bone marrow suppression associated with palbociclib and its differentiation from cytotoxic chemotherapies. Clin Cancer Res. 2016;22:2000-2008.
- Kim SG, Kim MH, Park S, et al. Efficacy of limited dose modifications for palbociclib-related grade 3 neutropenia in hormone receptor–positive metastatic breast cancer. Cancer Res Treat. 2023;55:1198-1209.
- Vulaj V, Hough S, Bedard L, et al. Oncology pharmacist opportunities: closing the gap in quality care. J Oncol Pract. 2018;14:e403-e411.
- McKee M, Frei BL, Garcia A, et al. Impact of clinical pharmacy services on patients in an outpatient chemotherapy academic clinic. J Oncol Pharm Pract. 2011;17:387-394.
- Munro L, Myers G, Gould O, LeBlanc M. Clinical pharmacy services in an ambulatory oncology clinic: patient perception and satisfaction. J Oncol Pharm Pract. 2021;27:1086-1093.
- Mathur AD, Maiers TA, Andrick BJ. Impact of a pharmacist-led telehealth oral chemotherapy clinic. Am J Health Syst Pharm. 2022;79:896-903.
- Lee Y, Lee D, Seo I, et al. Risk factors for palbociclib-induced early developing neutropenia in patients with hormone receptor-positive metastatic breast cancer. Cancers (Basel). 2023;15:2810.