Connecting Science to Practice
This research explores relevant concerns in the biosimilar
space, including the differences in adverse events (AEs)
that may be encountered when using different biosimilars
to injectable drugs. We specifically focus on bevacizumab
and its biosimilar, bevacizumab-awwb, which are approved
for the treatment of a variety of cancers. Medication-related
AEs affect patients’ quality of life and the decision-making
processes of providers and pharmacists.
Bevacizumab, a vascular endothelial growth factor inhibitor, is a frequently used monoclonal antibody in the treatment of a variety of cancer types. Bevacizumab was approved by the FDA under the brand name Avastin in 2004 as the first antiangiogenic targeted therapy.1 Bevacizumab is most notable for its use in cervical, colorectal, nonsquamous non–small cell lung, and ovarian cancers, as well as recurrent glioblastoma.
In 2017, the FDA approved Mvasi (bevacizumab-awwb), a biosimilar to Avastin. The biosimilar mirrored bevacizumab in approved indications, with the exception of an indication for use as adjuvant therapy in colon cancer.2,3 Bevacizumab-awwb has shown promising evidence as a clinical equivalent to bevacizumab, most notably in the MAPLE trial in which the 2 biologics were compared head-to-head.4 Within the study, patients receiving therapy for advanced nonsquamous non–small cell lung cancer were randomized to either bevacizumab or bevacizumab-awwb. The objective response rate of patients receiving bevacizumab-awwb was 39% versus 41.7% in those receiving bevacizumab, and the objective response rate risk ratio was 0.93 (90% confidence interval [CI], 0.80-1.09). Notably, the incidence of at least 1 adverse event (AE) of any grade was 95.1% in patients administered bevacizumab-awwb versus 93.5% in those who received bevacizumab.4
The production of biosimilars, specifically costly monoclonal antibodies, has become increasingly prevalent. The FDA deems that a drug can be labeled a biosimilar to a reference drug if “the biological product is highly similar to the reference product not withstanding minor differences in clinically inactive components” and “there are no clinically meaningful differences between the biological product and the reference product in terms of the safety, purity, and potency of the product.”5 Bevacizumab-awwb and other biosimilars have benefits such as the mitigation of drug shortage issues and affordability. With that comes a concern for the lack of sound clinical data in support of similarity and ability to extrapolate results to different oncologic disease states.6 The affordability and cost-savings of utilizing bevacizumab-awwb may drive institutions to list it as their preferred formulary agent over bevacizumab.
Although arthralgia is not a listed AE in either of the medications’ prescribing information, a noteworthy number of patients report arthralgia as a bothersome AE. Few postmarketing studies have addressed the incidence of arthralgia in patients receiving bevacizumab or biosimilars. A single-center study including 76 patients with ovarian cancer who received bevacizumab showed that the incidence rate of arthralgia was 30.3% (n=23) patients. Three of these patients received corticosteroids or methotrexate, whereas the other patients received analgesics alone.7 Other studies report 30% to 50% incidence rates for arthralgia in patients receiving bevacizumab, but they do not specify whether the patients were receiving bevacizumab or bevacizumab-awwb.8,9
Further investigation of the incidence of arthralgia specific to bevacizumab-awwb is warranted, as well as an evaluation of proper management when arthralgia does occur. The purpose of this study is to quantify and subsequently compare the incidence of arthralgia in bevacizumab versus bevacizumab-awwb, and to describe the current clinical management of arthralgia.
Methods
This study was a single-center, retrospective chart review that evaluated the incidence of arthralgia among patients receiving bevacizumab or bevacizumab-awwb at Froedtert and the Medical College of Wisconsin, Milwaukee, WI. The study further assessed grading, location, and management of arthralgia in patients who had arthralgia.
Patients were eligible for study inclusion if they started and received at least 1 dose of bevacizumab or bevacizumab-awwb within the tertiary academic medical center infusion clinic between July 1, 2017, and March 31, 2023. Patients required close follow-up with a provider at the study institution, which was defined as follow-up within 30 days of medication administration. Patients also had to be aged ≥18 years and could not be receiving bevacizumab or bevacizumab-awwb via participation in a clinical trial.
The primary end point was the incidence of arthralgia in patients receiving bevacizumab versus those receiving bevacizumab-awwb. The primary end point data were collected by searching the electronic health records of the included patients for keywords in the oncology provider notes, including arthralgia(s), joint pain, knee pain, shoulder pain, elbow pain, hip pain, ankle pain, bone pain, joint stiffness, and joint/body ache. On identifying patients that had provider notes that included any of the above keywords, the electronic health records were further assessed to determine if the keyword was associated with bevacizumab or bevacizumab-awwb or if the keyword finding was the result of an existing condition, metastases, or other acute illness. If a patient was positive for the primary end point (ie, had arthralgia), further evaluation was completed for the prespecified secondary end points. The secondary end points included the grade of arthralgia, location of arthralgia, pharmacologic and nonpharmacologic management prescribed for those reporting arthralgia, and the incidence of change in therapy from bevacizumab-awwb to bevacizumab. Arthralgias were graded based on the Common Terminology Criteria for Adverse Events version 5.0.
Statistical Analysis
Descriptive statistics were used to summarize the patient and treatment characteristics. Mean and standard deviation were used for the continuous variables, whereas counts and percentages were used for the categorical variables. Continuous variables were compared between the treatment groups using the Wilcoxon rank-sum test. Categorical variables were compared using a chi-square test. Kaplan-Meier curves were used to estimate the survival probability by treatment group. Patients were censored at the date of last follow-up. A log-rank test was used to compare the survival probabilities between the groups. The cumulative incidence of arthralgia (accounting for death as a competing risk) was calculated in each treatment group. Gray’s test was used to compare the cumulative incidence of arthralgia between the groups. All analyses were performed with R version 4.3.0 statistical software.
Results
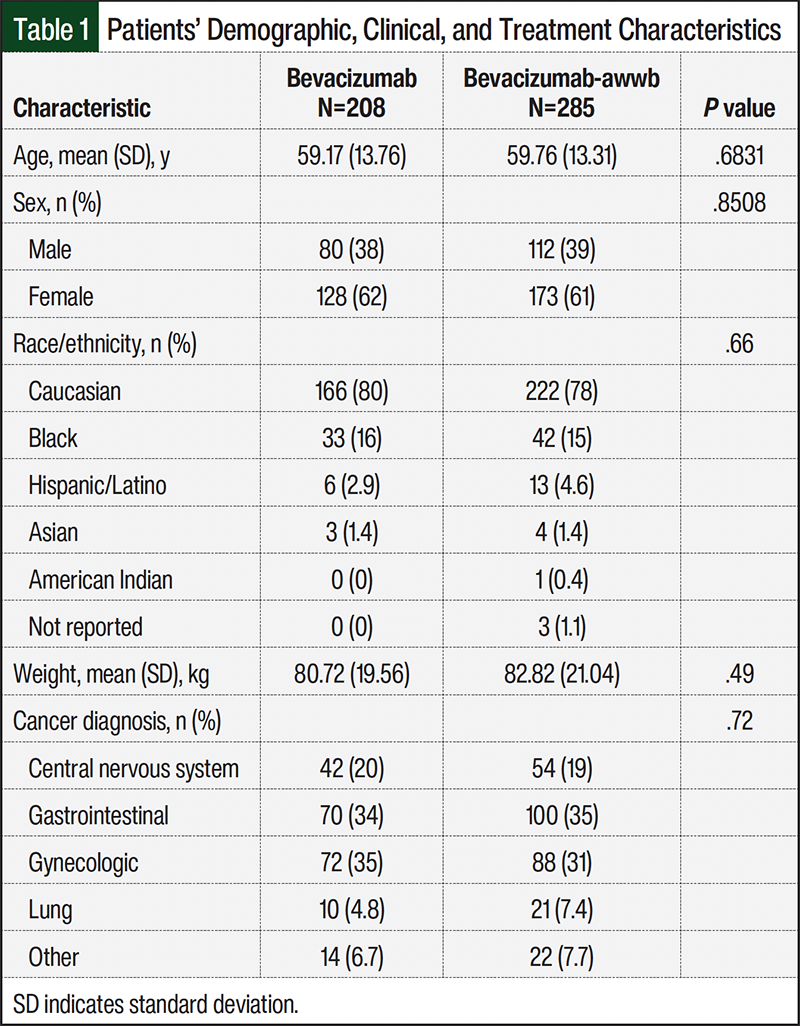
A total of 573 patients received bevacizumab or bevacizumab-awwb between July 1, 2017, and March 31, 2023, with close follow-up with an oncology provider. Of these patients, 493 patients were included in the study, with 208 in the bevacizumab arm and 285 in the bevacizumab-awwb arm (Figure 1). The baseline characteristics, including age, sex, race/ethnicity, weight, and cancer diagnosis, were similar between the groups (Table 1). Notably, most patients were receiving treatment for either a gastrointestinal or gynecologic malignancy. There was no significant difference between the groups regarding mortality (P=.8).
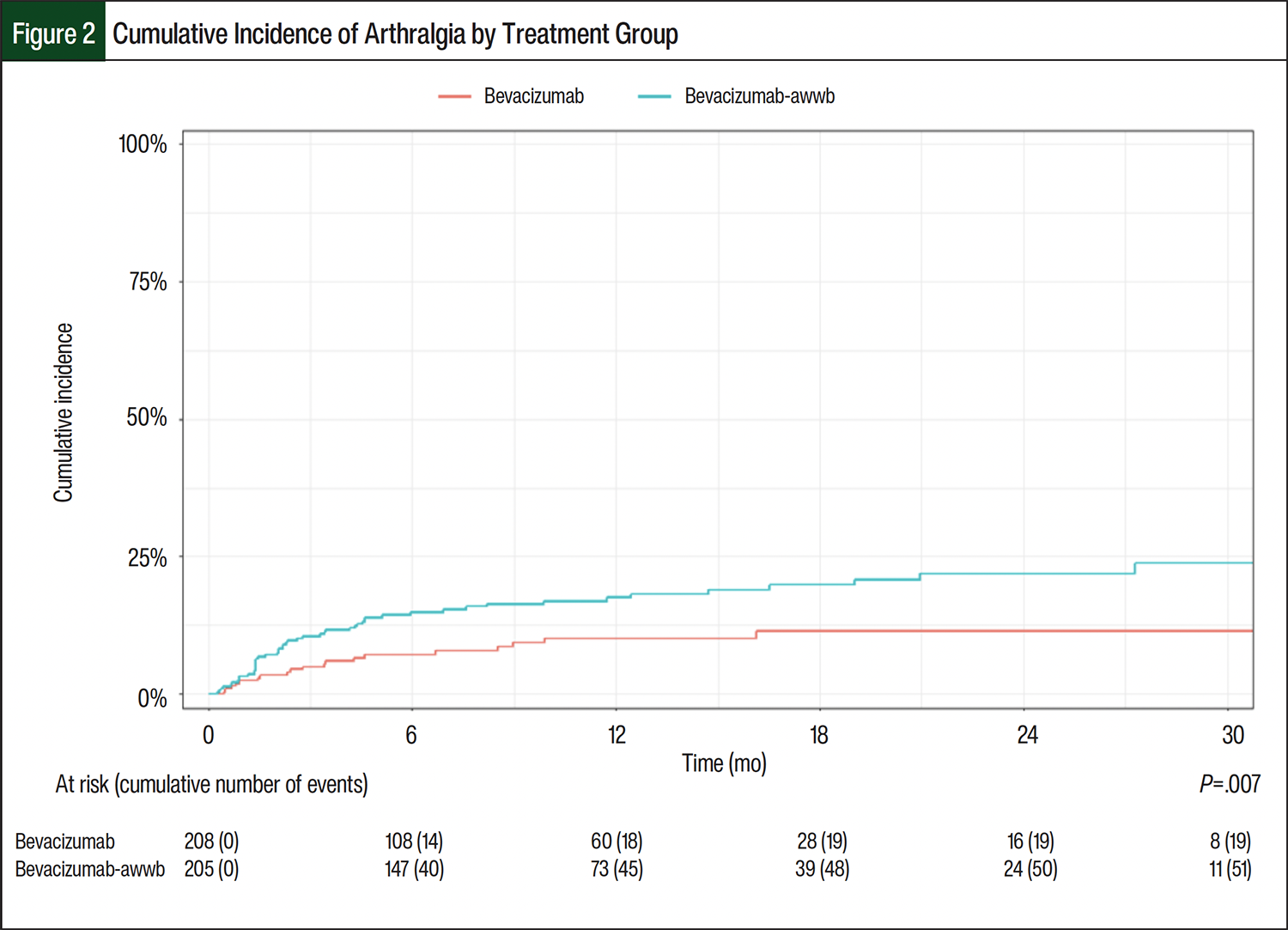
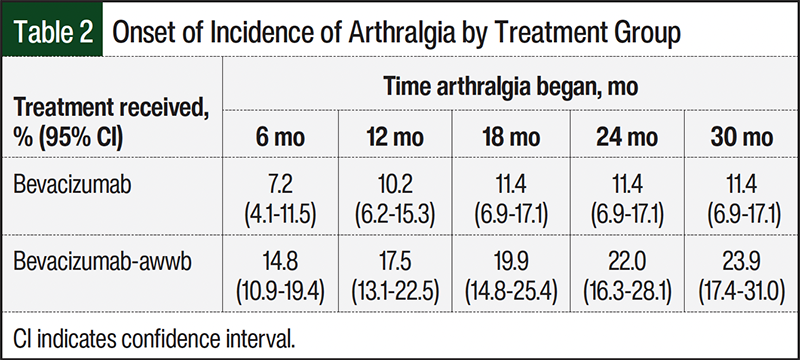
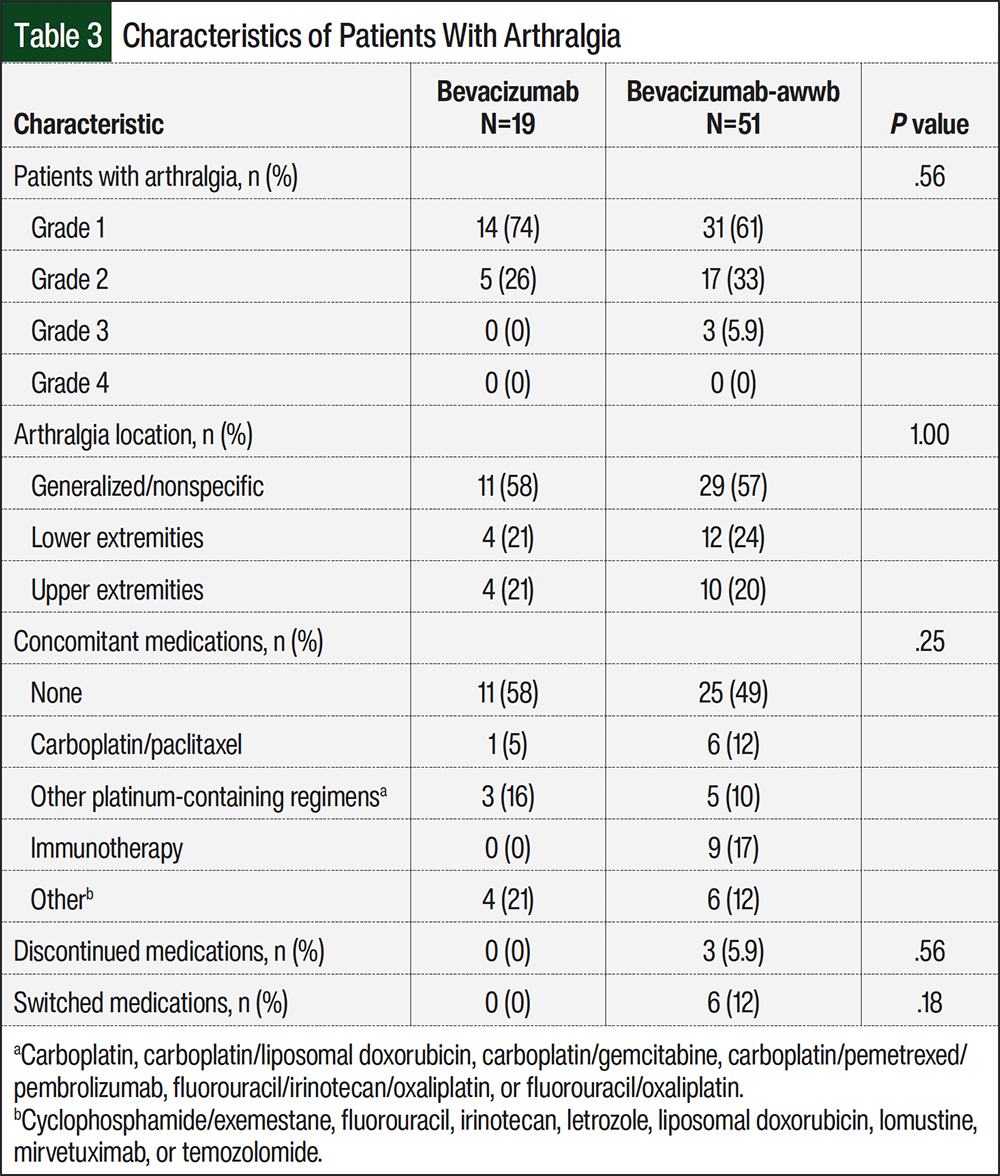
In all, 70 (14.2%) patients who were included in the overall study population had arthralgia. There was a significant difference in the percentage of patients with arthralgia in both groups (P=.007, Figure 2). The cumulative incidence of arthralgia at 6 months after treatment initiation was 7.2% (95% CI, 4.1-11.5) in the bevacizumab group and 14.8% (95% CI, 10.9-19.4) in the bevacizumab-awwb group (Table 2). By 24 months, the cumulative incidence of arthralgia had increased to 11.4% (95% CI, 6.9-17.1) in the bevacizumab group and 22% (95% CI, 16.3-28.1) the bevacizumab-awwb group (Table 2). Patients who had arthralgia were further assessed to describe the grade and location of arthralgia (Table 3). Patients most often had grade 1 arthralgia, with similar representations in each group (74% and 61% in the bevacizumab and bevacizumab-awwb groups, respectively), although approximately 6% (n=3) of patients in the bevacizumab-awwb group had grade 3 arthralgia (Table 3). The locations of arthralgia were comparable between the groups, with most patients having nonspecific or generalized arthralgia (58% and 57% of the bevacizumab and bevacizumab-awwb groups, respectively; Table 3). Discontinuation and change in therapy were higher in the bevacizumab-awwb group than in the bevacizumab group. Within the bevacizumab- awwb group, approximately 6% (n=3) of the patients discontinued the medication after noting arthralgia, and 12% (n=6) switched to an alternative agent, which was largely noted as switching from the biosimilar to the reference drug bevacizumab (Table 3). Of the patients switching from the biosimilar back to the reference drug, 50% (n=3) reported improvement in their arthralgia. None of the patients in the bevacizumab group discontinued or switched drugs because of arthralgia.
An evaluation of the concomitant medications showed that 58% of patients in the bevacizumab group and 49% of patients in the bevacizumab-awwb group were receiving that therapy alone at the time that they had arthralgia. Carboplatin in combination with paclitaxel was received concomitantly in 1 (5%) and 6 (12%) patients in the bevacizumab and bevacizumab-awwb groups, respectively (Table 3).
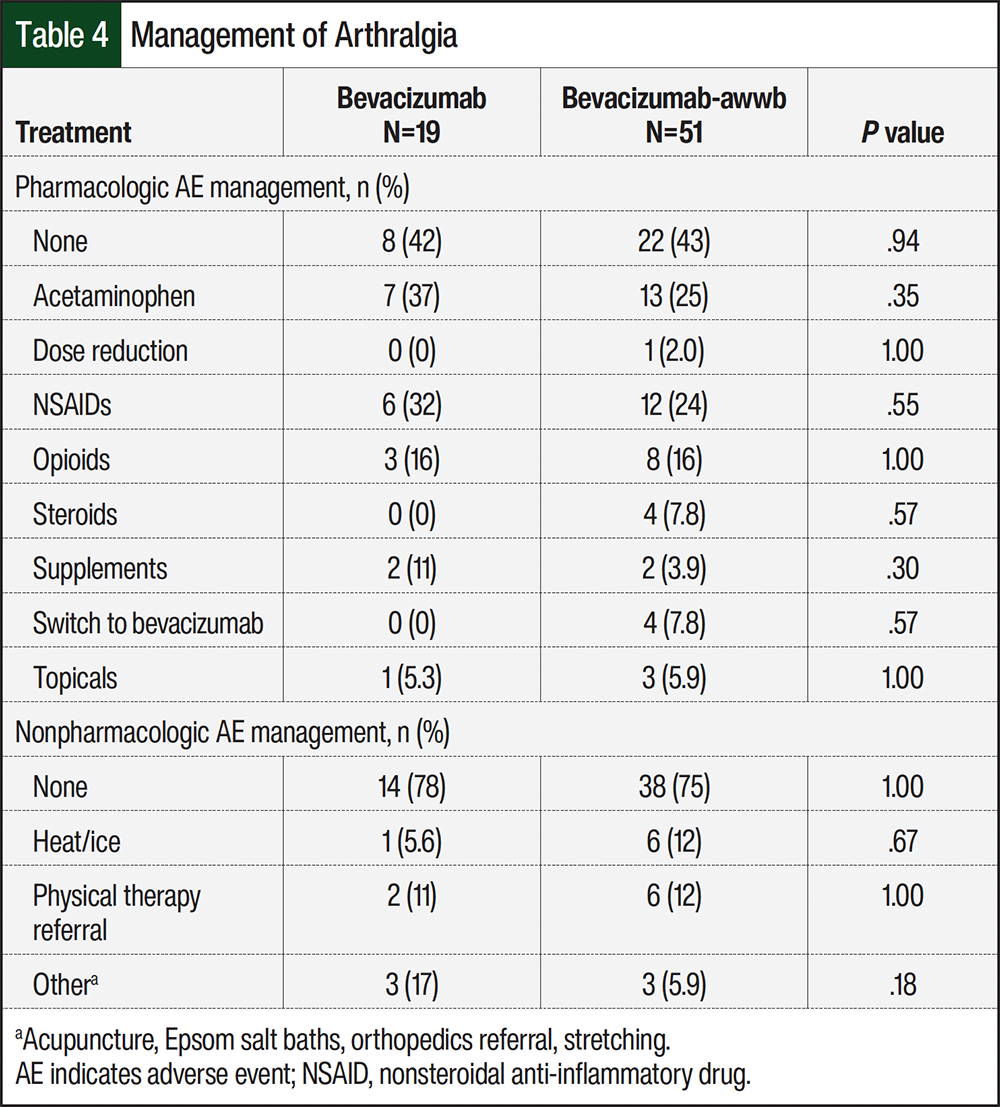
Pharmacologic AE management was recommended in 58% and 57% of patients who had arthralgia in the bevacizumab and bevacizumab-awwb groups, respectively (Table 4). Of those who received pharmacologic management strategies, most patients were recommended to use acetaminophen or a nonsteroidal anti-inflammatory drug. Opioids were prescribed for pharmacologic management of arthralgia in 16% of each group (Table 4). Among the patients who had arthralgia, 42% and 43% of patients in the bevacizumab and bevacizumab-awwb groups, respectively, did not receive pharmacologic management strategies. Furthermore, 78% of patients in the bevacizumab group and 75% in the bevacizumab-awwb group did not receive nonpharmacologic management strategies (Table 4). Those who received nonpharmacologic management recommendations were instructed to apply heat or ice or received a physical therapy referral.
Discussion
Biosimilars are often deemed as acceptable alternatives to reference drugs. This study shows that clinically meaningful differences in AE profiles may exist between biosimilars and reference drugs, specifically in the incidence of arthralgia between bevacizumab and bevacizumab-awwb. Previous safety analyses lacked comparison between bevacizumab and bevacizumab-awwb when considering arthralgia as an AE, making this study a novel comparison of the reference and biosimilar drugs.10,11 In general, the lack of analysis presents a potential gap in safety analysis when considering and comparing a biosimilar with a reference drug. To further complicate matters, insurance also dictates the use of specific biosimilars. Further comparison and analysis between a reference drug and a biosimilar may be necessary in choosing formulary-preferred drugs.
Data regarding the incidence and severity of arthralgia as an AE in patients receiving bevacizumab or bevacizumab-awwb are lacking. Although none of the patients in the bevacizumab group had grade 3 arthralgia, 3 patients in the bevacizumab-awwb cohort had grade 3 arthralgia, which shows that patients have more arthralgia with the biosimilar than the reference drug and that they are also having a higher severity of the AE. Early and sustained separation in the cumulative incidence curves for arthralgia represents an earlier onset of arthralgia in the bevacizumab-awwb group versus the bevacizumab group as well. With higher grading and severity of arthralgia, patients’ activities of daily living are further affected. AEs that negatively affect quality of life are of greater clinical significance and need proper management. An analysis completed within this study concerning the pharmacologic and nonpharmacologic management of arthralgia points to the need for guidance for the treatment of arthralgia, given that national guidelines do not have recommendations regarding arthralgia treatment related to cancer or chemotherapy. Guidance at an institutional or national level may lead to improved outcomes for patients and an ability to continue the proper treatment for their malignancy.
Limitations
A limitation to this study includes the nature of a retrospective chart review constraining the ability to capture the incidence of arthralgia specific to the drug of interest versus other causes of arthralgia. Evaluation of concomitant oncology medications showed that approximately 50% of the patients in each group were receiving bevacizumab or bevacizumab-awwb monotherapy, which mitigates this limitation to some extent, but it still has shortcomings regarding a full representation of contributing factors to arthralgia. Patient comorbidities, pain scoring and baseline pain before treatment, as well as other nononcology medications and interventions may be contributing factors to arthralgia. In addition, patients may have had arthralgia, but documentation was not provided or the patient did not disclose the AE to their provider. Confirmation bias may have influenced this study, because gynecologic oncology providers increasingly noticed and discussed arthralgia in patients receiving bevacizumab-awwb, suggesting that provider hyperawareness of this AE grew as the study progressed. Finally, most of the patients identified as White women and had a diagnosis of gastrointestinal or gynecologic cancer, which may limit the generalizability of these findings to other populations and disease sites.
Conclusion
The use of biosimilars should be carefully considered when approaching patient care and the potential for any AEs. This study’s results showed a significantly higher incidence of arthralgia in patients who received bevacizumab-awwb, a biosimilar, than in those who received bevacizumab. This study showed that transitioning to bevacizumab may be an appropriate clinical solution in patients with arthralgia related to bevacizumab-awwb, especially in those with grade 3 arthralgia. The development of institutional or national guidance regarding the treatment of arthralgia may provide patients with improved clinical outcomes and the ability to detect this AE and manage it properly, thereby allowing for optimal treatment without interruption or discontinuation. Because bevacizumab biosimilars are continuously being added to the market, further comparative literature investigations may be warranted when choosing a biosimilar and comparing AE profiles to a reference drug.
Author Disclosure Statement
Dr Grahl is a consultant to AstraZeneca and Daiichi Sankyo and is on the Speaker’s Bureau of AstraZeneca and Merck; Dr Duyar-Ayerdi has received honoraria from Wolters Kluwer; Dr Schneider, Dr Brazauskas, Dr Canales, and Dr McGurty have no conflicts of interest to report.
References
- Garcia J, Hurwitz HI, Sandler AB, et al. Bevacizumab (Avastin®) in cancer treatment: a review of 15 years of clinical experience and future outlook. Cancer Treat Rev. 2020;86:102017.
- Mvasi (bevacizumab-awwb) injection, for intravenous use [prescribing information]. Amgen, Inc; June 2025. Accessed January 8, 2026. www.pi.amgen.com/-/media/Project/Amgen/Repository/pi-amgen-com/Mvasi/mvasi_pi_hcp_english.pdf
- Avastin (bevacizumab) injection, for intravenous use [prescribing information]. Genentech, Inc; September 2022. Accessed January 8, 2026. www.gene.com/download/pdf/avastin_prescribing.pdf
- Thatcher N, Goldschmidt JH, Thomas M, et al. Efficacy and safety of the biosimilar ABP 215 compared with bevacizumab in patients with advanced nonsquamous non-small cell lung cancer (MAPLE): a randomized, double-blind, phase III study. Clin Cancer Res. 2019;25: 2088-2095. Erratum in: Clin Cancer Res. 2019;25:3193.
- US Food and Drug Administration. Scientific considerations in demonstrating biosimilarity to a reference. April 2015. Accessed August 1, 2023. www.fda.gov/media/82647/download
- Nabhan C, Valley A, Feinberg BA. Barriers to oncology biosimilars uptake in the United States. Oncologist. 2018;23:1261-1265.
- Kaparelou M, Liontos M, Katsimbri P, et al. Retrospective analysis of bevacizumab-induced arthralgia and clinical outcomes in ovarian cancer patients. Single center experience. Gynecol Oncol Rep. 2022;40:100953.
- Ventriglia J, Paciolla I, Pisano C, et al. Arthralgia in patients with ovarian cancer treated with bevacizumab and chemotherapy. Int J Gynecol Cancer. 2021;31:110-113.
- Vauléon E, Behal H, Lebellec L, et al. Does bevacizumab increase joint pain in patients with cancer? Results of the prospective observational BEVARTHRALGIA study. Cancer Chemother Pharmacol. 2021;87:533-541.
- Alhaja M, Tsourounis C, Ho H, Fong R. Real-world tolerability of biosimilar bevacizumab-awwb compared to bevacizumab in patients with cancer at an academic medical center. J Clin Oncol. 2022;40(28 suppl):Abstract 322.
- Alhaja M, Tsourounis C, Ho H, Fong R. Real-world tolerability and financial impact of biosimilar bevacizumab-awwb compared with bevacizumab in patients with cancer. J Hematol Oncol Pharm. 2024;14:70-78.