Connecting Science to Practice
As indications for immune checkpoint inhibitors (ICIs)
continue to expand, additional patients have become eligible
to receive these drugs as part of their cancer treatment.
The increased use of ICIs may increase the number of patients
who have immunotherapy-related adverse events.
This case report reviews the presentation, diagnosis, and
treatment of ICI-mediated hepatitis in a patient who received
dostarlimab as treatment for endometrial cancer.
The patient’s workup revealed a diagnosis of nonalcoholic
fatty liver disease, which has been previously recognized as
an emerging risk factor for ICI-mediated hepatitis. Nonalcoholic
fatty liver disease has become increasingly common,
and clinicians should be aware that it may increase the risk
for ICI-mediated hepatitis, in the same way that preexisting
liver disease does.
Dostarlimab-gxly is an immune checkpoint inhibitor (ICI) that targets PD-1 and is FDA approved as a single agent for the treatment of patients with recurrent or advanced, platinum-resistant, mismatch repair–deficient endometrial cancer, and, in combination with carboplatin and paclitaxel, for the treatment of patients with endometrial cancer.1 These approvals were based on the results of the GARNET and RUBY trials, respectively.2,3 The GARNET and RUBY trials also enrolled patients with proficient mismatch-repair (pMMR)/microsatellite-stable endometrial cancer; responses to dostarlimab were less common and of shorter duration in these patients.2-5 Notably, the FDA has granted the accelerated approval of dostarlimab for the treatment of patients with recurrent or advanced solid tumors with mismatch repair–deficient disease based on another cohort of the GARNET trial.1,4
As with other ICIs, dostarlimab can cause immune-related adverse events (irAEs) that result from weakened autoimmune tolerance. One such irAE, ICI-mediated hepatitis, occurred in 0.5% to 6.6% of patients who received treatment with dostarlimab in the GARNET and RUBY trials.1-6 Common Terminology Criteria for Adverse Events (CTCAE) grade ≥3 ICI-mediated hepatitis (elevated aspartate aminotransferase [AST] or alanine aminotransferase [ALT] or both) was reported in up to 1.9% of patients and was a common reason for the discontinuation of treatment with dostarlimab.2-5 Although irAEs can occur at any time after the administration of ICIs, ICI-mediated hepatitis typically develops after 6 to 12 weeks of treatment.7-10 The reported onset of ICI-mediated hepatitis with pembrolizumab, nivolumab, and ipilimumab treatment ranges widely from 7 days to 21 months.8,11
We report a case of ICI-mediated hepatitis in a patient with recurrent pMMR endometrial squamous-cell carcinoma 2 days after the administration of the first dose of dostarlimab.
Case Report
A 59-year-old woman presented to our hospital for abdominal pain and an inability to tolerate oral intake of liquids and solids. She had an acute small bowel obstruction and was recently diagnosed with recurrent, stage IIIa pMMR endometrial squamous-cell carcinoma at the vaginal cuff. Her medical history included stage III uterine carcinoma, for which she had a total abdominal hysterectomy and bilateral salpingo-oophorectomy and received adjuvant chemotherapy; hypertension; type 2 diabetes; morbid obesity; osteoarthritis; obstructive sleep apnea; cholecystectomy; and recurrent bowel obstructions. The patient received conservative care with bowel rest and a venting nasogastric tube.
Two days before admission, she received cycle 1, day 1 of dostarlimab 500 mg intravenously. Single-agent dostarlimab was selected by her oncologist because the patient was deemed a poor surgical candidate as a result of extensive pelvic recurrence; the oncologist wanted to avoid myelosuppressive chemotherapy given her recurrent hospitalizations for small bowel obstruction and the preliminary evidence of responses in pMMR endometrial cancer. The patient gave informed consent to proceed with dostarlimab treatment before it was administered.
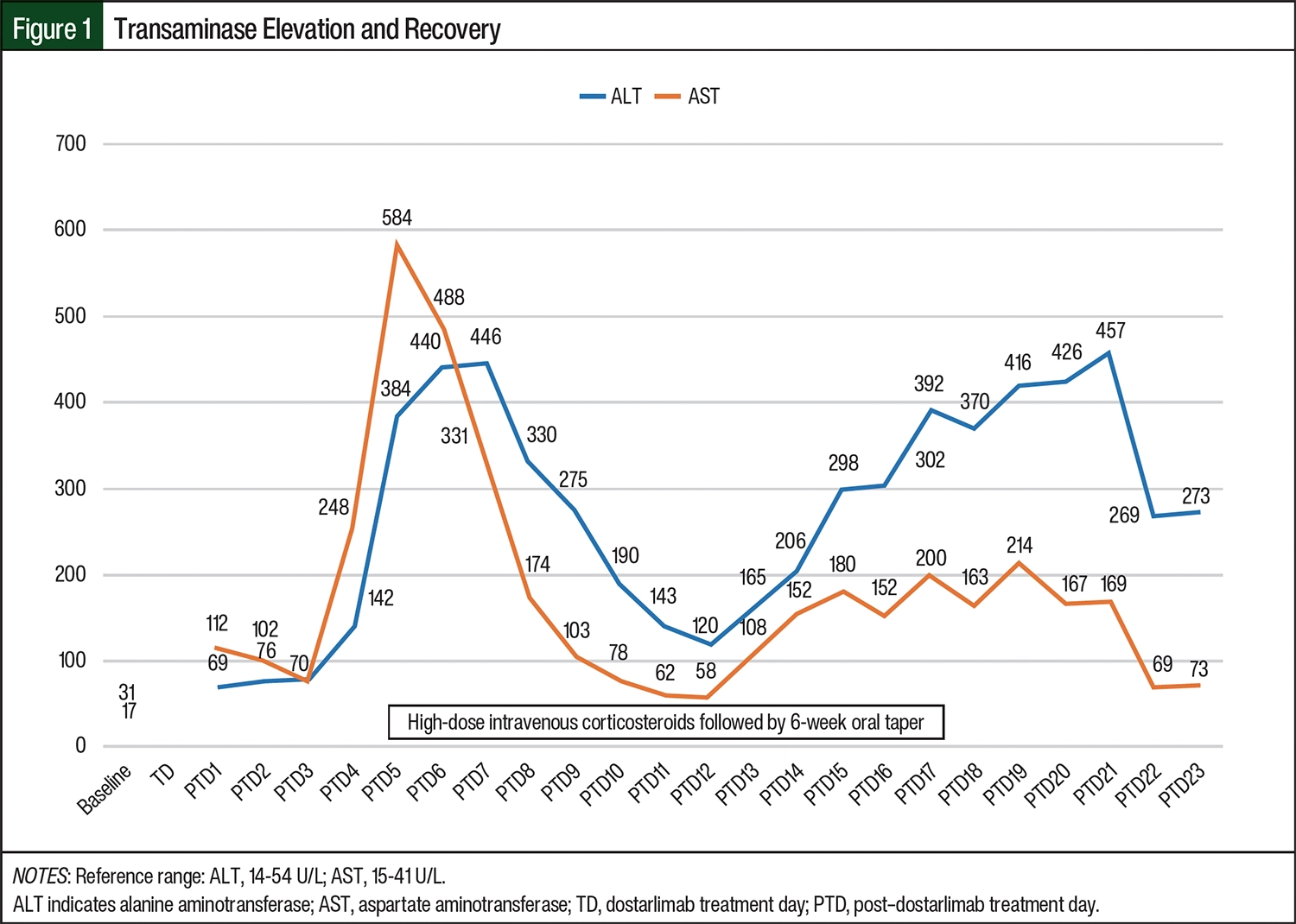
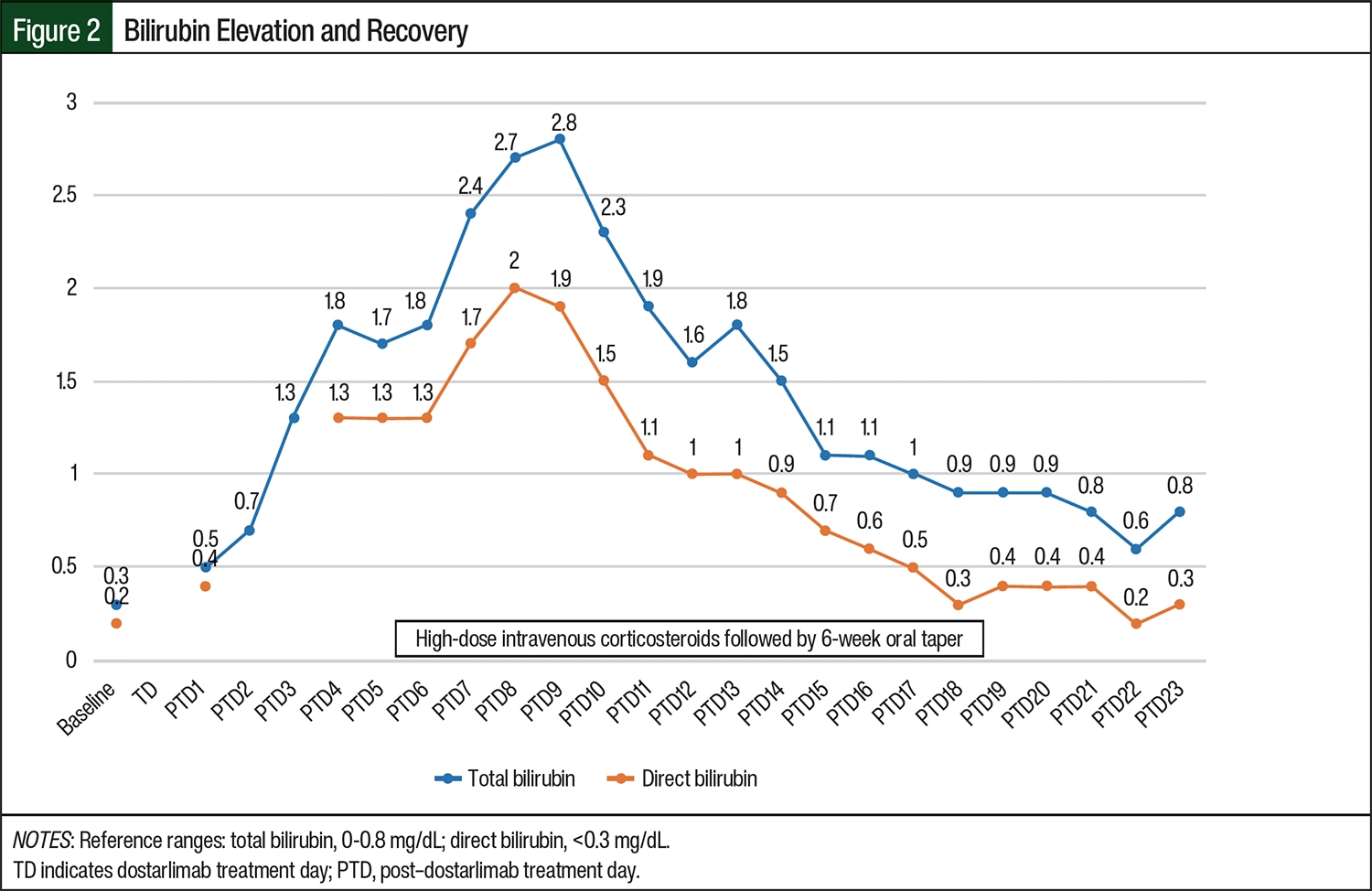
Daily laboratory testing was obtained during admission, and an uptrend of her liver function tests (LFTs) from baseline AST and ALT levels of 31 U/L and 17 U/L, respectively, to 102 U/L and 76 U/L was noted on posttreatment day 2. By posttreatment day 4, the patient’s AST and ALT levels had increased to 248 U/L and 142 U/L, respectively, and her bilirubin also started to increase from a baseline level of 0.3 to 1.8 mg/dL. Concern was raised by the attending physician (OAB) and the clinical oncology pharmacist (LLG) about whether the patient had ICI-mediated hepatitis when her LFT levels rose even higher to 584 U/L and 384 U/L for AST and ALT, respectively, on posttreatment day 5 (Figure 1 and Figure 2). Treatment with intravenous methylprednisolone 1 mg/kg daily was started, and a gastroenterologist was consulted for assistance with further diagnostic workup.
A diagnostic workup for other etiologies of her ICI-mediated hepatitis was performed. Although a right upper quadrant ultrasound showed diffuse fatty liver disease, she had no other liver abnormalities. A hepatitis panel was negative, and per gastroenterology recommendations, antinuclear antibody, alpha-1 antitrypsin, celiac panel, ceruloplasmin, cytomegalovirus, herpes simplex virus, mitochondrial antibody, anti–smooth-muscle antibodies, Epstein–Barr virus, and thyroid panels were ordered, all of which were unremarkable. Baseline iron studies were within normal limits and were not repeated. A detailed medication history was obtained, and the patient denied recently taking acetaminophen or herbal supplements. She also denied having any history of recent or remote illicit drug or alcohol use. ICI-mediated hepatitis was the most likely cause of her hepatitis given that other etiologies were ruled out.
The patient remained asymptomatic throughout her hospitalization, and she never had any other signs of acute hepatitis, such as fever, tea-colored urine, or jaundice. Her transaminase levels peaked at an AST of 584 U/L and an ALT of 440 U/L on posttreatment day 5. This degree of transaminitis is 14 times and 8 times the upper limit of normal (ULN), respectively, indicating grade 3 hepatitis per CTCAE 5.0.12 Her total bilirubin peaked at 2.8 mg/dL on posttreatment day 9, which is consistent with grade 2 hepatitis.12 Of note, her alkaline phosphatase level, which was normal (ie, 46 U/L) at baseline, peaked to 187 U/L on posttreatment day 9, and then slowly decreased to the normal range. Her transaminitis began to improve within 48 hours of starting treatment with high-dose steroids, but there was a lag in recovery of her hyperbilirubinemia.
The patient’s treatment was transitioned intravenous (IV) steroids to oral prednisone on posttreatment day 7 with a planned 6-week taper. She was started on total parenteral nutrition (TPN), and her prednisone dose was initially tapered down from 1 mg/kg daily to 0.75 mg/kg daily on posttreatment day 13. Her transaminase levels began to rise again from an AST of 120 U/L to 426 U/L and an ALT of 58 U/L to 169 U/L while she was receiving TPN, prompting the nutrition service to reduce the frequency of lipids from daily to every 72 hours on posttreatment day 20. The patient’s LFTs remained elevated at grade 3 hepatitis, despite continued treatment with oral prednisone and the reduced frequency of IV lipids, and treatment with dostarlimab was permanently discontinued. This case of ICI-related hepatitis was reported to FDA MedWatch.
Discussion
Dostarlimab is a humanized, immunoglobulin G4 isotype, monoclonal antibody, like pembrolizumab and nivolumab.13 The AEs in clinical trials of dostarlimab are similar to those with other ICIs and are mainly irAEs.1-5,13
ICI-mediated hepatitis occurs in 5% to 10% of patients who receive immunotherapy, and most cases are mild to moderate (CTCAE grade 1 and 2).7,9,10 Severe-to-fatal ICI-mediated hepatitis (grade 3-5) has been reported in 0.05% to 3% of patients.8,9,14 The incidence of hepatitis is similar with either anticytotoxic T-lymphocyte–associated antigen 4 or PD-1/PD-L1 monotherapy, and the risk increases to 13% to 30% when ICIs are used in combination.7-10 Other potential risk factors for irAEs include being female, having first-line treatment with an ICI, and having preexisting autoimmune conditions.9,10,14 Preexisting liver disease is linked to ICI-mediated hepatitis, and nonalcoholic fatty liver disease has been reported as a possible risk factor.8-11
Patients with ICI-mediated hepatitis are often asymptomatic, although they may have fatigue, fever, jaundice, anorexia, and abdominal pain.7,8,10 Transaminitis occurs more often than hyperbilirubinemia in patients with ICI-mediated hepatitis.8 The signs and symptoms of ICI-mediated hepatitis may be more pronounced in patients with grade 3 or 4 ICI-mediated hepatitis, and may include tea-colored urine, coagulopathy, and altered mental status.10 Our patient was asymptomatic; had 3 risk factors, including being female, first exposure, and fatty liver disease; and presented with grade 3 transaminitis.
When ICI-mediated hepatitis is suspected, treatment should be started without delay and other causes of liver injury must be excluded. The treatment guidelines for ICI-mediated hepatitis provide recommendations for further workup and ongoing evaluation.15-17 A medication history should be performed to assess for the use of hepatotoxic medications, substances, and/or supplements.15-17 A workup should be conducted to assess for viral hepatitis, alcohol-related liver injury, iron overload, vascular disorders, and metastatic liver disease.15-17 When primary autoimmune hepatitis is suspected, antinuclear antibodies, anti–smooth-muscle antibodies, and antineutrophil cytoplasmic antibodies can be tested. When only the patient’s transaminases are elevated, creatine kinase testing can be obtained to rule out myositis; when only alkaline phosphatase is elevated, gamma glutamyl transferase can be checked.15,16 Of note, the NCCN’s treatment guidelines recommend this workup for patients with all grades of transaminitis, whereas ASCO’s treatment guidelines recommend the workup for patients with grade 2 to 4 transaminitis.15,16 A liver biopsy can be considered if hepatitis does not respond to systemic steroids or in cases when an alternative etiology would change the patient’s treatment; the NCCN’s guidelines reserve biopsy for patients with grade 3 or 4 hepatitis.15,16
The management guidelines for ICI-mediated hepatitis are based on the severity of presentation using CTCAE grading (Table 1).15-17 ICIs can be continued with close monitoring and the consideration of other causes for asymptomatic grade 1 elevations of transaminases and/or total bilirubin.15-17 For grade 2 elevations of LFTs without symptoms, ICIs should be temporarily held, LFTs should be monitored every 3 days, and treatment with prednisone 0.5 to 1 mg/kg daily can be started.15-17 In patients with grade 3 hepatitis, the ASCO guidelines recommend considering the permanent discontinuation of ICI therapy when the patient is asymptomatic and permanently discontinuing treatment with ICIs when the patient is symptomatic.16 The NCCN’s guidelines recommend holding ICI therapy for grade 3 hepatitis, whereas ESMO recommends the permanent discontinuation of ICI therapy regardless of the patient’s symptoms.15,17 All guidelines recommend the immediate initiation of treatment with steroids 1 to 2 mg/kg daily; methylprednisolone is recommended by NCCN and ASCO for the treatment of all cases of grade 3 hepatitis, but ESMO specifies the use of IV methylprednisolone when ALT and/or AST are >400 U/L.17 For patients with grade 4 hepatitis, treatment with an ICI should be permanently discontinued and IV methylprednisolone 2 mg/kg daily should be started immediately.15-17 Of note, dostarlimab’s prescribing information recommends the permanent discontinuation of treatment with dostarlimab when LFTs are elevated to >8 times the ULN.1
When a patient receives steroids, a lack of improvement in symptoms and/or LFTs within 2 to 4 days should prompt the addition of treatment with mycophenolate mofetil, azathioprine, or tacrolimus.15-17 Although it is indicated for the treatment of other irAEs, infliximab should be avoided in therapy for ICI-mediated hepatitis because of its propensity to cause liver injury.15-17 A steroid taper can begin once symptoms and LFTs have improved to grade ≤1 level, and a prolonged taper of at least 1 month is recommended.15-17 If needed, steroids can be titrated back up for the treatment of breakthrough LFT elevations or symptoms.15,16
Our patient started receiving steroids per the NCCN’s guideline recommendations, and her LFTs initially began to decrease. Although hepatotoxic medications were avoided, TPN was required because of her ongoing inability to eat as a result of small bowel obstruction. Her LFT results began to worsen, with her AST increasing from 120 U/L to 426 U/L and her ALT increasing from 58 U/L to 169 U/L, while she was receiving TPN; mycophenolate mofetil was not added to the steroids she was receiving and, instead, her daily lipid supplementation frequency was reduced to every 72 hours in response. The patient’s liver function and clinical condition stabilized, and she was discharged from the hospital 23 days after receiving treatment with dostarlimab.
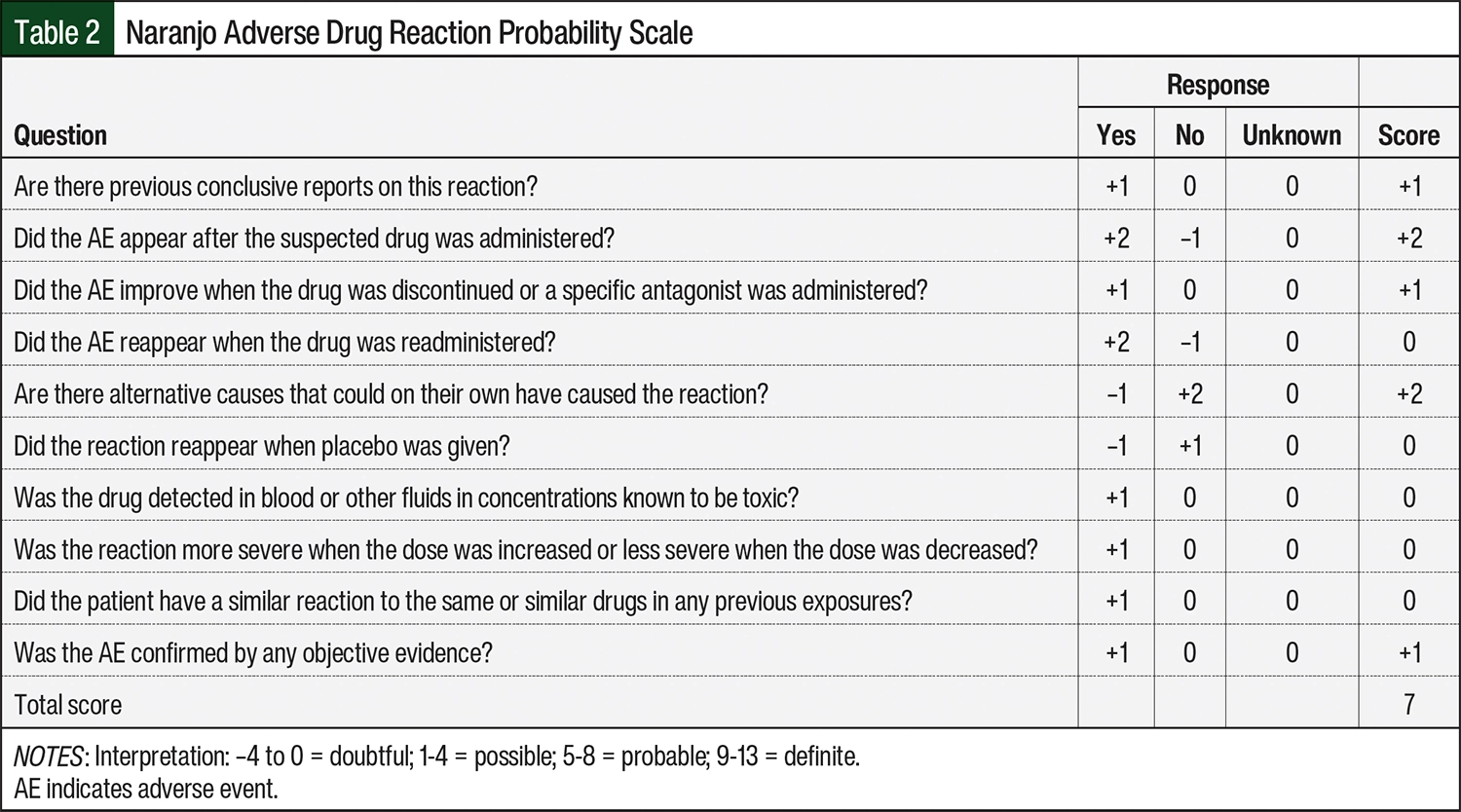
To better characterize the likelihood that dostarlimab was the cause of immune-mediated hepatitis, the Naranjo scale was used to calculate an adverse drug reaction probability score.18 A score of 7 was calculated for our patient, indicating that dostarlimab was the probable cause of her hepatitis (Table 2). Amid concerns about the underreporting of irAEs in clinical trials, it is important to report these AEs to the FDA via its MedWatch program.19
Conclusion
Treatment with dostarlimab can result in ICI-mediated hepatitis, with an incidence and presentation that is similar to other ICIs. Clinicians should be aware that, in addition to preexisting liver disease, nonalcoholic fatty liver disease has been identified as a possible risk factor for ICI-mediated hepatitis. As demonstrated by this case, ICI-mediated hepatitis can occur quickly, and a high index of suspicion along with prompt treatment may improve patients’ outcomes.
Author Disclosure Statement
Dr O’Brien, Dr Gingo, and Dr Olawaiye have no conflicts of interest to report.
References
- Jemperli (dostarlimab) injection, for intravenous use [prescribing information]. GlaxoSmithKline; August 2024. Accessed February 24, 2025. https://gskpro.com/content/dam/global/hcpportal/en_US/Prescribing_Information/Jemperli/pdf/JEMPERLI-PI-MG.PDF
- Oaknin A, Gilbert L, Tinker AV, et al. Safety and antitumor activity of dostarlimab in patients with advanced or recurrent DNA mismatch repair deficient/microsatellite instability-high (dMMR/MSI-H) or proficient/stable (MMRP/MSS) endometrial cancer: interim results from GARNET—a phase I, single-arm study. J Immunother Cancer. 2022;10:e003777.
- Mirza MR, Chase DM, Slomvitz BM, et al. Dostarlimab for primary advanced or recurrent endometrial cancer. N Engl J Med. 2023;388:2145-2158.
- Oaknin A, Pothuri B, Gilbert L, et al. Safety, efficacy, and biomarker analyses of dostarlimab in patients with endometrial cancer: interim results of the phase I GARNET study. Clin Cancer Res. 2023;29:4564-4574.
- André T, Berton D, Curigliano G, et al. Antitumor activity and safety of dostarlimab monotherapy in patients with mismatch repair deficient solid tumors: a nonrandomized controlled trial. JAMA Netw Open. 2023;6:e2341165.
- Oaknin A, Tinker AV, Gilbert, L, et al. Clinical activity and safety of the anti-programmed death 1 monoclonal antibody dostarlimab for patients with recurrent or advanced mismatch repair-deficient endometrial cancer: a nonrandomized phase 1 clinical trial. JAMA Oncol. 2020;6:1766-1772.
- Martins F, Sofiya L, Sykiotis GP, et al. Adverse effects of immune-checkpoint inhibitors: epidemiology, management and surveillance. Nat Rev Clin Oncol. 2019;16:563-580.
- Grover S, Rahma OE, Hashemi N, Lim RM. Gastrointestinal and hepatic toxicities of checkpoint inhibitors: algorithms for management. Am Soc Clin Oncol Educ Book. 2018;38:13-19.
- Hercun J, Vincent C, Bilodeau M, Lapierre P. Immune-mediated hepatitis during immune checkpoint inhibitor cancer immunotherapy: lessons from autoimmune hepatitis and liver immunology. Front Immunol. 2022;13:907591.
- Mok K, Wu C, Chan S, et al. Clinical management of gastrointestinal and liver toxicities of immune checkpoint inhibitors. Clin Colorectal Cancer. 2024;23:4-13.
- Sawada K, Hayashi H, Nakajima S, et al. Non-alcoholic fatty liver disease is a potential risk factor for liver injury caused by immune checkpoint inhibitor. J Gastroenterol Hepatol. 2020;35:1042-1048.
- US Department of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0. November 27, 2017. Accessed July 15, 2024. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcae_v5_quick_reference_5x7.pdf
- Yadav R, Mathur I, Haokip HR, et al. Dostarlimab: review on success story and clinical trials. Crit Rev Oncol Hematol. 2024;198:104374.
- Miah A, Tinoco G, Zhao S, et al. Immune checkpoint inhibitor-induced hepatitis injury: risk factors, outcomes, and impact on survival. J Cancer Res Clin Oncol. 2023;149:2235-2242.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): management of immunotherapy-related toxicities. Version 1.2025. December 20, 2024. Accessed February 24, 2025. www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf
- Schneider BJ, Naidoo J, Santomasso BD, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: ASCO guideline update. J Clin Oncol. 2021;39:4073-4126.
- Haanen J, Obeid M, Spain L, et al. Management of toxicities from immunotherapy: ESMO clinical practice guideline for diagnosis, treatment, and follow-up. Ann Oncol. 2022;33:1217-1238.
- LiverTox: Clinical and Research Information on Drug-Induced Liver Injury (Internet). Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Adverse Drug Reaction Probability Scale (Naranjo) in Drug Induced Liver Injury. Updated May 4, 2019. Accessed December 19, 2022. www.ncbi.nlm.nih.gov/books/NBK548069/
- Tapiavala S, Shenouda MA, Patel VG, et al. Reporting of immune-related adverse events (irAEs) in US Food and Drug Administration (FDA) approvals of immune checkpoint inhibitors. J Clin Oncol. 2024;42(16 suppl):e14693.