Connecting Science to Practice
Methotrexate is a common treatment for multiple cancer
types. Methotrexate is given at high, potentially toxic doses
followed by leucovorin rescue, and high drug levels can result
in acute kidney injury. Glucarpidase is a recombinant
enzyme approved for the management of toxic methotrexate
levels in patients with impaired renal function and delayed
methotrexate clearance. Per the prescribing information,
the glucarpidase dose is 50 units/kg in a single dose
with no defined dosage maximum. At our institution, the
standard-of-care protocol caps doses at 1 vial (1000 units) of
glucarpidase because there is a lack of evidence to prove
that higher doses of glucarpidase result in greater clinical
benefit. Due to the high cost of glucarpidase, capping the
dose at 1 vial provides cost-savings for the patient and the
institution. This single-center, retrospective chart review assesses
the effectiveness of using a capped dose of glucarpidase
for methotrexate clearance in patients with renal impairment
and toxic drug levels.
Methotrexate is a common treatment for multiple cancer types. Methotrexate is given at high, potentially toxic doses followed by leucovorin rescue, and high drug levels can result in acute kidney injury (AKI).1 This results from methotrexate accumulation in renal tubules that causes crystallization and subsequent obstruction and/or direct tubular adverse events (AEs) due to oxygen radical formation in the kidney.2 Damage to the kidney decreases the clearance of methotrexate, which can lead to further nephrotoxicity, hepatotoxicity, myelosuppression, and multiorgan failure.3 The risk factors for methotrexate AEs include higher doses (≥3.5 g/m2), rapid infusions, preexisting kidney impairment, older age (aged ≥50 years), and the concurrent use of medications that are nephrotoxic or that impair methotrexate clearance (eg, proton pump inhibitors, aminoglycosides, nonsteroidal anti-inflammatory drugs, penicillin and penicillin derivatives, amphotericin, probenecid).4
Glucarpidase is a recombinant enzyme approved for the management of toxic methotrexate levels in patients with impaired renal function who have delayed methotrexate clearance.5 Glucarpidase hydrolyzes the terminal residue from extracellular methotrexate into inactive metabolites that can be excreted regardless of renal function.5 Glucarpidase is dosed per the prescribing information at 50 units/kg in a single dose with no defined maximum dosage.5 This approval was based on 2 single-arm, open-label, multicenter studies.5 A retrospective study examined glucarpidase treatment in dosages ranging from 13 to 90 units/kg in 26 patients with elevated methotrexate levels.6 In that study, there was no significant relationship between the percent decrease in methotrexate plasma concentration and glucarpidase dosage. The time to serum creatinine (SCr) recovery was also not correlated with glucarpidase dosing.6
At our institution, the standard-of-care protocol caps doses at 1 vial of glucarpidase (1000 units) because there is a lack of evidence to prove that higher doses of glucarpidase result in greater clinical benefit. Due to the high cost of glucarpidase, capping the dose at 1 vial may provide cost-savings for the patient and the institution. This study assessed the effectiveness of using a capped dose of glucarpidase for methotrexate clearance in patients with renal impairment and toxic drug levels.
The objective of this study was to assess the effectiveness of using a single vial (1000-unit) capped dose of glucarpidase for methotrexate clearance in patients with toxic drug levels and renal impairment.
Methods
This study is a retrospective case series. Patients were included in the study if they were aged ≥1 year, received high-dose methotrexate, and received a single 1000-unit dose of glucarpidase between January 1, 2012, and April 1, 2023. Patients were excluded from the study if they were enrolled in a clinical trial of glucarpidase. The patients’ baseline demographic and clinical characteristics were collected through Epic electronic health record inquiries, including the associated Beacon (Epic Systems; Verona, WI) chemotherapy plan.
The primary study outcome was the clearance of methotrexate after glucarpidase administration, which was defined as a methotrexate level of <0.05 µmol/L before the return of serum levels secondary to tissue release of nonmetabolized methotrexate. The secondary outcomes included renal recovery (defined as the return of SCr to baseline), AEs related to receiving methotrexate and/or glucarpidase, survival to hospital discharge, and cost-savings.
Statistical Analysis
Descriptive statistics were used to assess the primary and secondary study outcomes. Continuous variables were summarized by median (range) values, and categorical variables were summarized as frequencies (percentages). All statistics were calculated using Microsoft Excel 365.
Results
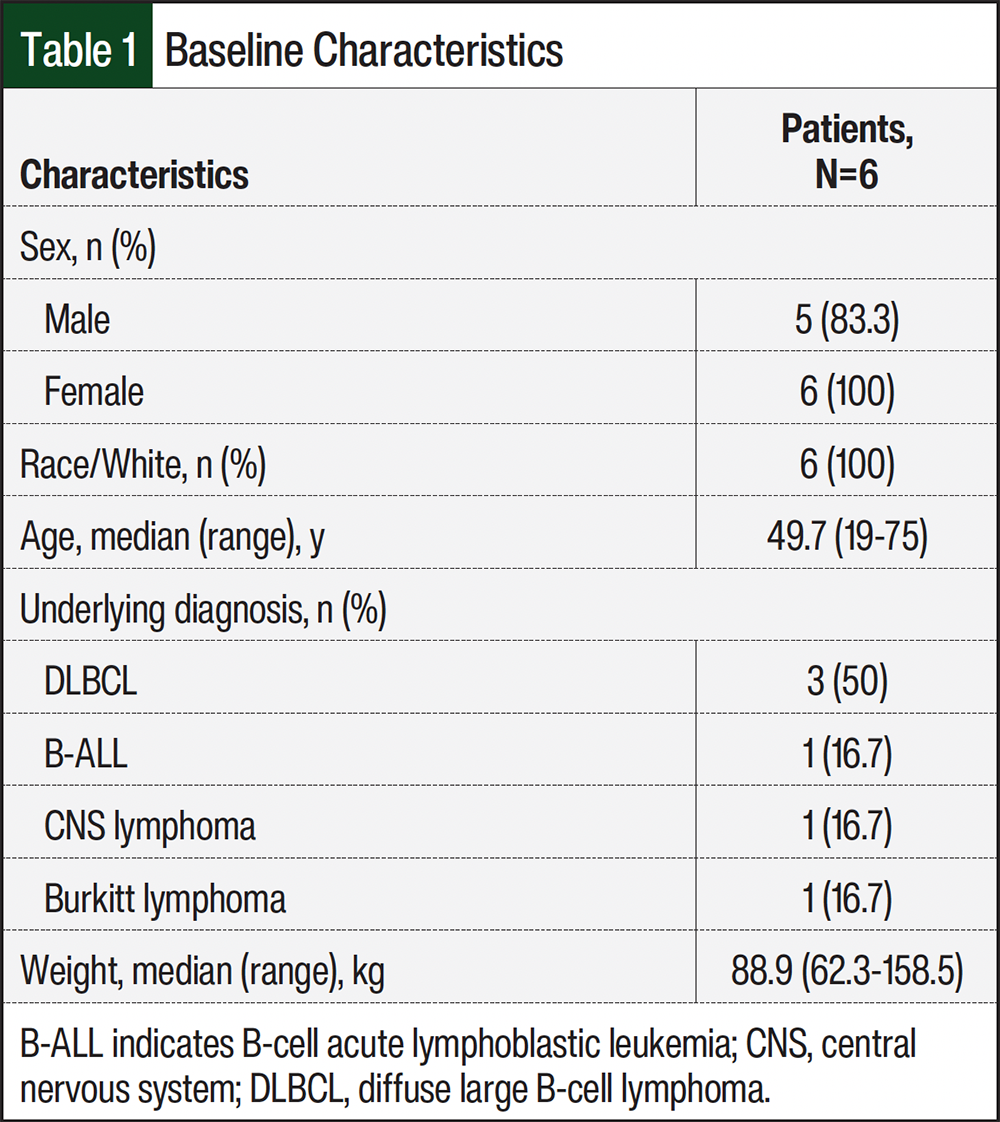
A total of 6 patients who received a flat dose of glucarpidase between January 2012 and April 2023 were included in the analysis. The patients’ baseline characteristics are shown in Table 1. The median patient age was 49.7 years, most of the patients were male (n=5, 83.3%), and all of the patients were White (n=6, 100%). The most common underlying diagnosis was diffuse large B-cell lymphoma (n=3, 50%). The median patient weight was 88.9 kg (range, 62.3-158.5 kg; Table 1).
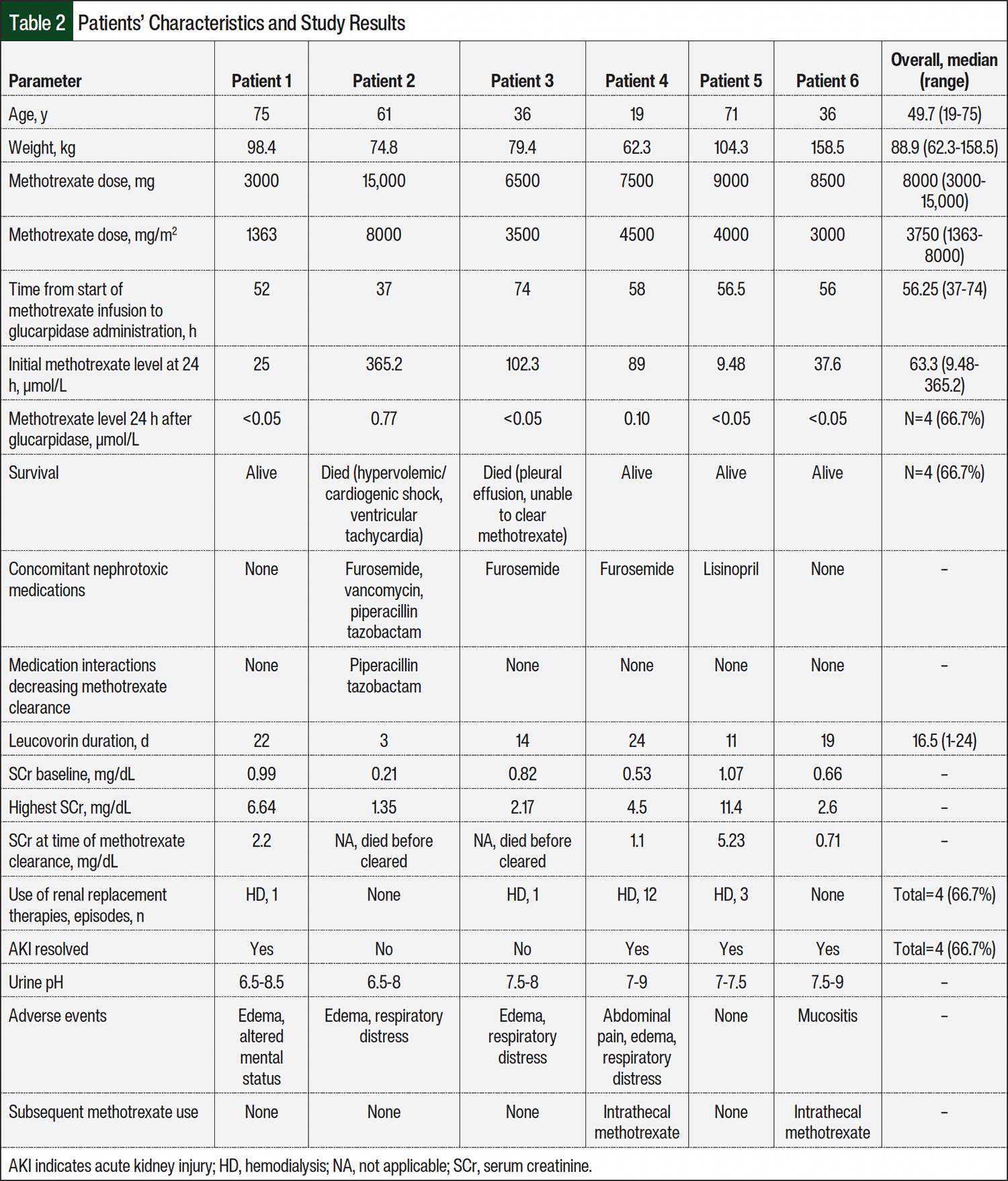
Methotrexate doses ranged from 3000 mg to 15,000 mg, with a median dose of 8000 mg (overall range, 1363-8000 mg/m2 with an overall median dose of 3750 mg/m2; Table 2). All patients received algorithm-guided intravenous leucovorin until methotrexate was cleared to <0.05 µmol/L.
The primary and secondary outcomes are shown in Table 2. For the primary outcome, a total of 4 (66.7%) patients transiently cleared methotrexate to <0.05 µmol/L by 24 hours. Glucarpidase was administered from 37 to 74 hours after methotrexate administration. Renal function returned to baseline in 4 (66.7%) patients, and 4 patients also required renal replacement therapy with hemodialysis, as noted in the Appendix Table. One patient was able to return to baseline kidney function without receiving dialysis. The AEs that frequently resulted from methotrexate and its associated supportive therapies were edema (n=4, 66.7%), respiratory distress (n=3, 50%), mucositis (n=1, 16.7%), and altered mental status (n=1, 16.7%).
The overall survival rate was 66.7% (n=4); of the 2 patients who died, 1 died from unrelated cardiac complications that were present before methotrexate administration and 1 died from a pleural effusion and subsequent respiratory failure. These 2 patients who died were unable to clear methotrexate, but methotrexate-related AEs were not ruled as the cause of death. Methotrexate-related AEs and organ dysfunction could have certainly contributed to the death of both patients, but they died before the completion of care for the methotrexate-related AEs. Glucarpidase dosing was unrelated to the final outcomes of these 2 patients. The details of fluid status, urine pH, SCr levels, and the timing/dosing of methotrexate and leucovorin are shown in Table 2 and in the Appendix Table.
Discussion
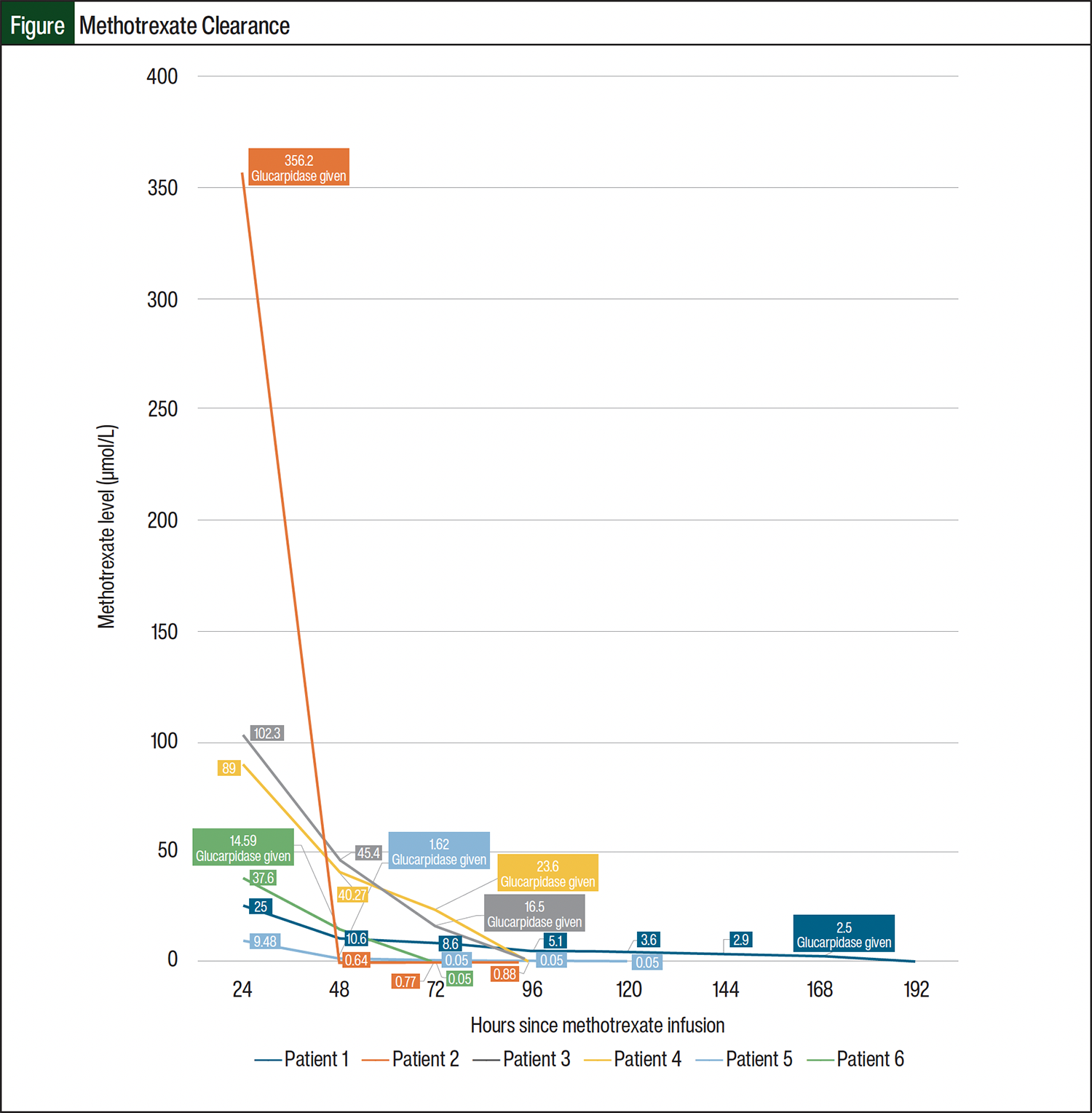
The results of this study demonstrate that a capped dose of 1000 units (1 vial) of glucarpidase is effective in reducing methotrexate levels to a clinically safe value with limited AEs, regardless of patient weight. If compared with the FDA-approved glucarpidase dosing of 50 units/kg, the patients in this cohort received doses ranging from 6.4 to 16.1 units/kg. Despite these lower doses of glucarpidase by weight, 4 of the 6 patients effectively cleared methotrexate to <0.05 µmol/L (Figure). The 2 other patients methotrexate levels were substantially reduced, but these levels were >0.05 µmol/L 24 hours after the administration of methotrexate.
One patient (Patient 2) cleared methotrexate to 0.77 µmol/L after glucarpidase administration (Table 2). Patient 2 started receiving methotrexate at a significantly higher level than the other patients, and their weight-based dose was equivalent to 13.4 units/kg of glucarpidase. However, there was a profound effect with the 1000-unit dose because it effectively reduced the methotrexate level from 365.2 µmol/L to 0.77 µmol/L (a 99% decrease). This patient was also receiving several other nephrotoxic medications (ie, furosemide, vancomycin, and piperacillin-tazobactam), which may have prevented the patient’s kidneys from properly clearing methotrexate. The piperacillin component of piperacillin plus tazobactam interacts with methotrexate and can result in increased methotrexate serum levels due to the competition of penicillin with methotrexate for excretion sites in renal tubules.7 The patient had an AKI, with an increased SCr of 6 times their baseline value. Although the patient’s urine output was normal, they had significant fluid overload with edema and accompanying respiratory distress.
Unfortunately, the patient died from cardiac complications (ie, mixed hypervolemic and cardiogenic shock, ventricular tachycardia) before additional interventions could be performed. Thus, we were unable to collect additional methotrexate levels. It is unclear whether this patient’s diminished methotrexate clearance may have been affected by the fluid overload and the drug interaction with piperacillin plus tazobactam rather than decreased glucarpidase function. Because this patient’s methotrexate level decreased to 0.77 µmol/L after receiving 1 dose of glucarpidase, we do not believe additional doses of glucarpidase were warranted.
The other patient who did not clear methotrexate to <0.05 µmol/L was Patient 4. This patient cleared methotrexate from 89 µmol/L to 0.1 µmol/L (a 99% decrease) after receiving a capped dose of 1000 units of glucarpidase (equivalent to 16 units/kg), which is clinically similar to 0.05 µmol/L, because some institutions use 0.1 µmol/L as the threshold for methotrexate clearance.1,4 This patient required hemodialysis and additional supportive care after their SCr reached 8.5 times the baseline level, but they recovered to baseline renal function without additional methotrexate-related AEs. Despite clearing methotrexate, none of the patients in this case series received subsequent doses of intravenous methotrexate.
Patient 6 illustrated the activity of glucarpidase despite having morbid obesity. Although this patient received only 6.3 units/kg of glucarpidase, it reduced their methotrexate level from 37.6 µmol/L to the desired level of <0.05 µmol/L. The patient reported mucositis as a methotrexate-related AE, but their AKI effectively cleared and resolved after glucarpidase administration without the use of renal replacement therapies. This patient demonstrates that even in patients with a larger body mass index, receiving a flat dose of glucarpidase is effective in clearing methotrexate. If a weight-based dose of glucarpidase would have been used, this patient would have received 7925 units of glucarpidase (equivalent to 8 vials) instead of only 1 vial. The average wholesale price of glucarpidase is $46,176.00.8 Using 8 vials would have cost $369,408 compared with a cost of $46,176 for a single vial.8,9
The results of our study align with those of a recent analysis by Gupta and colleagues that compared glucarpidase use with a control arm.10 The most frequently administered dose was 50 units/kg, which is the FDA-recommended dose, with 111 (53.1%) of 207 patients receiving glucarpidase at this dose. Gupta and colleagues found that receiving glucarpidase resulted in a 2.43-fold higher adjusted odds ratio of renal recovery (95% confidence interval, 1.38-4.27) compared with not receiving glucarpidase. There was also a benefit in recovery from neutropenia and the normalization of liver function. An additional weight-based dosing strategy was evaluated by Heuschkel and colleagues in 7 patients receiving high-dose methotrexate followed by half-dose glucarpidase (25 units/kg).11 These patients all received glucarpidase between 42 and 70 hours after initiating treatment with methotrexate. Within 24 hours of glucarpidase administration, methotrexate concentrations decreased from between 3.1 and 182.4 μmol/L to between 0.02 and 2.03 µmol/L. Similar to our results, methotrexate rebound was detected, but methotrexate concentrations remained <10 µmol/L, which did not warrant any additional doses of glucarpidase.11
Schaff and colleagues investigated the use of lower doses of glucarpidase in patients with central nervous system lymphoma as prophylaxis for methotrexate AEs.12 A total of 8 patients were included in a phase 1 study after receiving high-dose methotrexate and 2000- or 1000-unit flat doses of glucarpidase repeated every 2 weeks for up to 8 total cycles. These patients did not have to have methotrexate AEs, because this was an evaluation of the use of glucarpidase for the prevention of AEs rather than as a rescue treatment. A total of 55 glucarpidase treatments were administered. The 2000-unit glucarpidase dose yielded a >95% reduction in plasma methotrexate concentrations after 33 (97.1%) of 34 doses, and glucarpidase 1000-unit doses yielded a >95% reduction after 15 (75%) of 20 doses.12 These clinical outcomes could be future areas of study in patients receiving flat doses of glucarpidase in the rescue setting rather than for the prevention of methotrexate-related AEs.
Limitations
This study is limited by the relatively low incidence of high-dose methotrexate–related AEs requiring glucarpidase rescue. In addition, the retrospective design, lack of diversity in our patient population, and single-center and single-arm design make additional validation of these data important. It would also be imperative to gather data from individuals with very high levels of serum methotrexate, >200 µmol/L in the setting of AKI to assess the lower bounds of serum methotrexate levels reached with flat 1000-unit dosing of glucarpidase. It is unclear if our 2 patients who did not have AKI recovery would have eventually recovered if they lived or if they received a full weight-based rescue dose of glucarpidase. Future trials are needed to compare the clinical and economic outcomes of capped glucarpidase doses across several institutions.
Conclusion
This report provides real-world evidence that a capped 1000-unit dose of glucarpidase may be effective in rapidly reducing methotrexate concentrations to safe levels in patients of different sizes, without the need for repeat doses. We recommend continuing the use of this dosing strategy for patients requiring methotrexate rescue due to delayed renal clearance. Future research is warranted to explore this dosing strategy in larger patient populations.
Author Disclosure Statement
Dr Ertz and Dr Bubalo have no conflicts of interest to report.
References
- Widemann BC, Adamson PC. Understanding and managing methotrexate nephrotoxicity. Oncologist. 2006;11:694-703. doi:10.1634/theoncologist.11-6-694
- Howard SC, McCormick J, Pui CH, et al. Preventing and managing toxicities of high-dose methotrexate. Oncologist. 2016;21:1471-1482. doi:10.1634/theoncologist.2015-0164
- Perazella MA, Moeckel GW. Nephrotoxicity from chemotherapeutic agents: clinical manifestations, pathobiology, and prevention/therapy. Semin Nephrol. 2010;30:570-581. Erratum in: Semin Nephrol. 2011;31:317. doi:10.1016/j.semnephrol.2010.09.005
- Wiczer T, Dotson E, Tuten A, et al. Evaluation of incidence and risk factors for high-dose methotrexate-induced nephrotoxicity. J Oncol Pharm Pract. 2016;22:430-436. doi:10.1177/1078155215594417
- Voraxaze (glucarpidase) injection, for intravenous use [prescribing information]. BTG International; August 2019. Accessed February 2, 2026. www.accessdata.fda.gov/drugsatfda_docs/label/2019/125327s064lbl.pdf
- Scott JR, Zhou Y, Cheng C, et al. Comparable efficacy with varying dosages of glucarpidase in pediatric oncology patients. Pediatr Blood Cancer. 2015;62:1518-1522. doi:10.1002/pbc.25395
- Takeda M, Khamdang S, Narikawa S, et al. Characterization of methotrexate transport and its drug interactions with human organic anion transporters. J Pharmacol Exp Ther. 2002;302:666-671. doi:10.1124/jpet.102.034330
- Glucarpidase. Lexi-Drugs. Lexicomp online. Wolters Kluwer. Accessed February 5, 2024. http://online.lexi.com
- Kala J, Nelson R, Drudge C, et al. Glucarpidase for treating adults with delayed methotrexate elimination due to impaired renal function: an economic simulation analysis. Clinicoecon Outcomes Res. 2023;15:165-179. doi:10.2147/CEOR.S397154
- Gupta S, LaCasce A, Leaf RK, et al. Clinical outcomes in patients with high-dose methotrexate toxicity treated with vs. without glucarpidase. Presented at: 65th ASH Annual Meeting and Exposition. December 9-11, 2023; San Diego, CA. Abstract 268.
- Heuschkel S, Kretschmann T, Teipel R, et al. Half-dose glucarpidase as efficient rescue for toxic methotrexate levels in patients with acute kidney injury. Cancer Chemother Pharmacol. 2022;89:41-48. doi:10.1007/s00280-021-04361-8
- Schaff LR, Lobbous M, Carlow D, et al. Routine use of low-dose glucarpidase following high-dose methotrexate in adult patients with CNS lymphoma: an open-label, multi-center phase I study. BMC Cancer. 2022;22:60. doi:10.1186/s12885-021-09164-x