Connecting Science to Practice
Patients with multiple myeloma receiving daratumumab
have an increased risk for infections, yet listeriosis remains
an underrecognized complication. This case report highlights
a patient who had Listeria monocytogenes meningoencephalitis
after daratumumab therapy. Our findings underscore
the potential immunomodulatory effects of anti-CD38
therapy and its role in increasing susceptibility to L. monocytogenes.
Given the severity of listeria meningitis, clinicians
should maintain a high index of suspicion for this infection
in daratumumab-treated patients, particularly those presenting
with neurologic symptoms or unexplained sepsis. In
addition, patient education on avoiding high-risk foods may
serve as a simple yet effective preventive measure. Increased
awareness and early recognition can improve outcomes by
facilitating timely diagnosis and appropriate antimicrobial
therapy.
Multiple myeloma (MM) is an incurable hematologic malignancy characterized by abnormal clonal plasma-cell proliferation in the bone marrow that results in extensive skeletal destruction.1 The primary therapy for newly diagnosed active MM is dependent on whether patients are candidates for autologous hematopoietic stem-cell transplant. Daratumumab in combination with lenalidomide and dexamethasone is recommended by the National Comprehensive Cancer Network as a preferred regimen in patients who are ineligible for transplant.1 This 3-drug regimen induces higher response rates and a greater depth of response in clinical trials compared with the 2-drug regimen of lenalidomide and dexamethasone.1,2 However, this daratumumab-based regimen has also been associated with a higher incidence of grade ≥3 infections (41% vs 29%, respectively) and neutropenia (54% vs 37%, respectively) than treatment with lenalidomide and dexamethasone alone.2 The results of the MAIA trial showed various causes of infection, including pneumonia (19% vs 11%, respectively), oral candidiasis (4% vs 5%, respectively), herpes zoster (3% vs 5%, respectively), and other infections, such as upper respiratory infections and urinary tract infections.2 Although some infectious etiologies were identified in this trial,2 there is a paucity of data addressing listeria infection in patients who have received daratumumab.
Below, we describe a patient with notable potential dietary exposure to Listeria monocytogenes who had meningoencephalitis after receiving daratumumab treatment for MM.
Case Report
An 82-year-old woman with MM received 8 cycles of daratumumab plus lenalidomide and dexamethasone, which included daratumumab weekly for 2 cycles, every 2 weeks for 5 cycles, then once monthly thereafter; lenalidomide 25 mg daily on days 1 to 21 of each cycle; and dexamethasone 20 mg on days 1, 8, 15, and 22 of each cycle. The daratumumab was initially received as 1100 mg intravenously (IV) for the first 6 doses, but this dosage was subsequently changed to 1800 mg subcutaneously thereafter. Twelve days after completing treatment cycle 8, the patient was brought to the emergency department at the local community hospital after sustaining a fall from which she was unable to get up. The patient’s husband reported that she had recent changes in her mental status, limited oral intake, increased loose stools, as well as an active fever.
On arrival at the emergency department, she was febrile to 38.6 °C, her creatinine kinase was elevated to 4500 U/L (normal range, 26-192 U/L), and she had mild leukocytosis at 10.1×109 cells/L (normal range, 3.4-9.6×109 cells/L) with an elevated neutrophil count of 8.29×109 cells/L (normal range, 1.56-6.45×109 cells/L). Blood cultures were drawn, and the patient was initiated on broad-spectrum antibiotics, including vancomycin at a loading dose of 25 mg/kg followed by a dose of 20 mg/kg administered IV every 12 hours, and cefepime 2000 mg every 12 hours based on a serum creatinine level of 0.78 mg/dL (baseline, approximately 0.8 mg/dL) with an estimated creatinine clearance of 56 mL/min.
On hospital day 2, the patient was transferred to a tertiary care center where she was verbally unresponsive, was unable to follow commands, and demonstrated nuchal rigidity on arrival. Given concern for meningoencephalitis, her antimicrobial therapy was broadened to include ampicillin 2000 mg IV every 4 hours and acyclovir 10 mg/kg IV every 8 hours, whereas treatment with dexamethasone and lenalidomide were held. A lumbar puncture was performed, and the cerebrospinal fluid (CSF) results were consistent with bacterial meningitis and showed 1980 total nucleated cells (normal range, 0-5 cells/µL), 73% neutrophils (normal range, 2%-4%), 403 mg/dL of protein (normal range, 0-35 mg/dL), and <20 mg/dL of glucose. The CSF Gram stain showed the presence of white blood cells but no bacterial organisms; however, a multiplex meningitis and encephalitis polymerase chain reaction panel was positive for L. monocytogenes. Peripheral blood cultures drawn in the emergency department grew L. monocytogenes in 1 of 3 bottles in 2 of 2 draws at 18 and 22 hours. Streptococcus pneumoniae urine antigen, cryptococcus CSF antigen, CSF aerobic bacterial cultures, and a fungal smear were all negative.
The patient continued receiving ampicillin 2000 mg IV every 4 hours for 21 days. She improved and reported feeling much better by hospital day 4. After the recovery of her mental status, she noted that she had multiple possible dietary exposures to L. monocytogenes within the past 2 weeks, after consuming deli meats and unpasteurized cheese. The patient was counseled by the infectious diseases team about how to avoid future listeria infections, including recommendations for washing and preparing food and avoiding unpasteurized dairy products and delicatessen meats and cheeses. On hospital day 29, the patient was discharged from the hospital.
Discussion
Daratumumab is a human immunoglobulin G1 kappa monoclonal antibody that targets CD38, an antigen with high surface expression on MM cells. Daratumumab primarily induces cell death via complement-dependent cytotoxicity, antibody-dependent cell-mediated cytotoxicity, and antibody-dependent cellular phagocytosis.3 CD38 is also expressed on various immune cells, including macrophages, regulatory T cells, regulatory B cells, and natural killer cells, making this cell-surface receptor a necessary component for immune cell activation and proliferation.4 A study of CD38 knockout mice demonstrated that CD38 plays a crucial role in the innate immune response against L. monocytogenes.5 The absence of CD38 resulted in an increased susceptibility of mice to listeria infection via altered neutrophil and inflammatory monocyte recruitment to the site of listeria infection and inefficient macrophage uptake of listeria.5 Because CD38 induction in inflammatory macrophages is conserved from mouse to humans, there is a possibility that anti-CD38 therapy may place humans at an increased risk for listeria infection as well.6 Similar to daratumumab, isatuximab is another anti-CD38 monoclonal antibody for the treatment of MM that has an increased risk for infection.7
During clinical trials that compared daratumumab in combination with lenalidomide and dexamethasone versus lenalidomide and dexamethasone alone, neutropenia and infections, including upper respiratory tract infections, bronchitis, and pneumonia, occurred more frequently in the daratumumab group than in the control group.2,8,9 The associated organisms included viral, fungal, and typical bacterial pathogens.10 In one phase 3 clinical trial, a single case of grade 3 or 4 listeriosis was reported in an individual who received daratumumab plus lenalidomide and dexamethasone, whereas no cases were reported in the lenalidomide and dexamethasone arm.11 Herpes simplex virus and varicella zoster virus prophylaxes are routinely coadministered with daratumumab and are continued for at least 3 months beyond the end of therapy.1 The addition of prophylaxis that targets bacterial or fungal pathogens is indicated based on the absolute neutrophil count and/or corticosteroid dose; however, routine use outside of these circumstances is not recommended.1
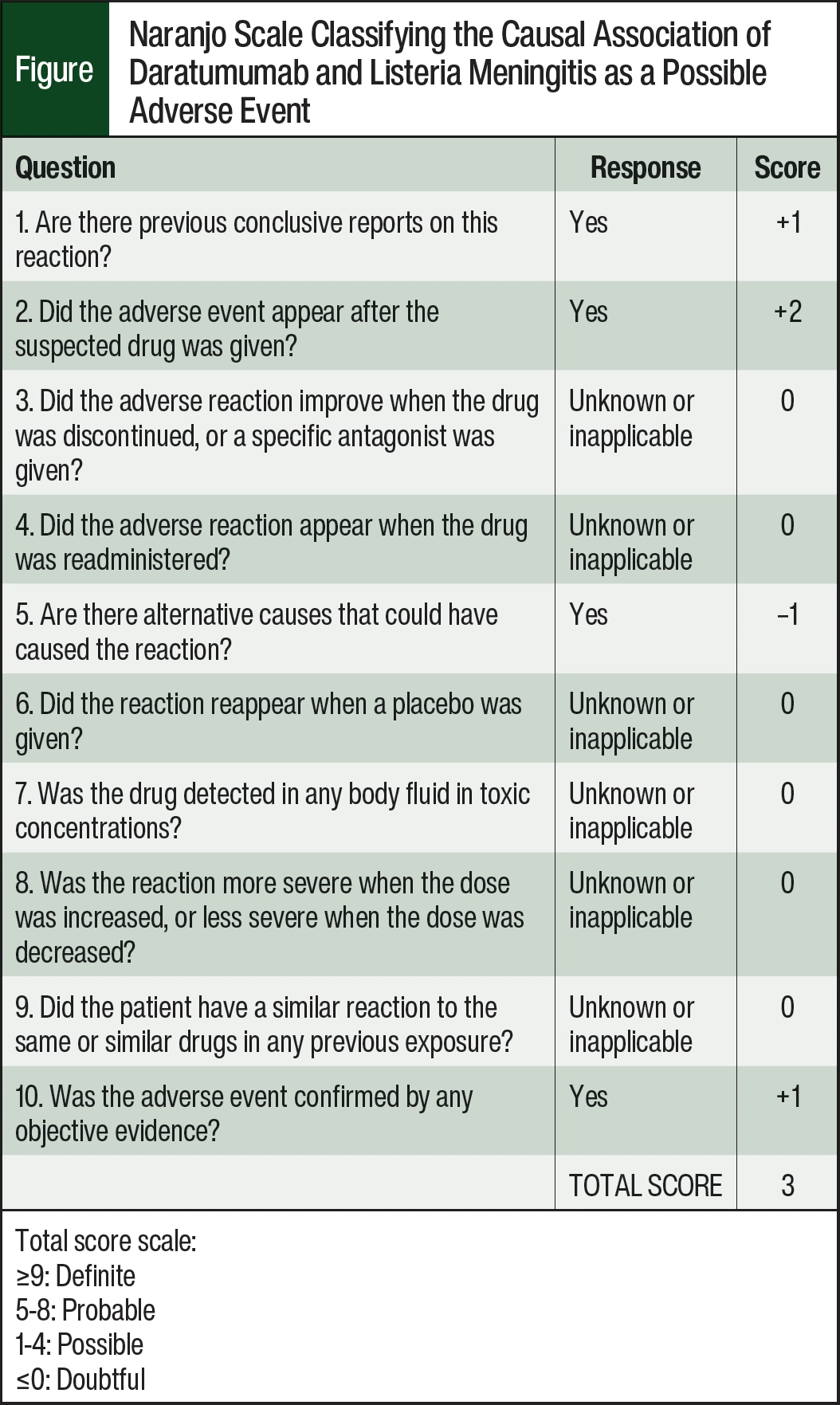
Reports of listeria meningitis in patients who receive daratumumab are rare, and, to our knowledge, this is the first reported case in the United States. The causal relationship between daratumumab and listeria meningoencephalitis in this case is classified as “possible” using the Naranjo algorithm (Figure).12 This patient presented with a reasonable temporal sequence of exposure and infection, and although her advanced age is a concomitant factor that may have served as an alternative explanation and/or risk factor for listeria meningoencephalitis, the established relationship between daratumumab exposure and listeriosis in the literature suggests that the natural decline of immunity and the administered immunotherapy may have played a role in this infection.11,13,14
A comprehensive literature review of the PubMed database with MESH terms “daratumumab” and “listeria,” “listeriosis,” or “meningitis” identified 2 published case reports of daratumumab-associated listeria meningitis, both of which were in Japan.13,14 This search also revealed a published case-control study that examined the risk for L. monocytogenes infection among patients receiving daratumumab amid an outbreak at a restaurant in a tertiary cancer center in Toronto, Ontario, Canada, over 9 months.11 Three of the 7 (43%) cases of listeriosis occurred in patients with MM who were receiving daratumumab. This resulted in patients with MM who received daratumumab having a 340-fold increased risk (95% confidence interval [CI], 68-1693; P<.001) for listeriosis compared with other patients with cancer and a 75-fold risk for listeriosis (95% CI, 8-726; P<.001) compared with all other patients with MM. Similar to the present case, patients in the case-control study received concomitant corticosteroids and an immunomodulating agent, potentially confounding the results; however, although the use of corticosteroids may increase host susceptibility to L. monocytogenes infection, there were no cases of listeriosis in patients with MM who were receiving corticosteroids without daratumumab.11 In addition, an increased risk for L. monocytogenes infection has not been associated with exposure to lenalidomide.
Furthermore, a case report by Ueno and colleagues included a patient with MM who received treatment with isatuximab and had subsequent L. monocytogenes bacteremia.15 This report further supports the potential risk for listeria infections that are associated with anti-CD38 monoclonal antibodies and highlights the importance of monitoring and preventive measures in these patients.15
The current treatment guidelines for MM do not address strategies for the prevention of listeria infection in patients who receive daratumumab.1 However, treatment with daratumumab places patients at an increased risk for listeriosis, as is evidenced by preclinical data showing enhanced L. monocytogenes susceptibility in mice with CD38 knockout, as well as aftermarket case reports and case series.4,11
Conclusion
Given the severity of listeria meningitis, it is critical for healthcare providers to be aware of the potential risk for this condition and to monitor for the signs and symptoms of listeria meningitis in patients with MM who are receiving daratumumab. Counseling should prioritize key educational points resulting from the high burden of disease in patients with MM. Although it may be difficult to advise patients about making dietary changes for such a rare adverse event, educating them about the importance of avoiding certain high-risk foods, such as undercooked meats, unpasteurized dairy products, and raw produce, can be considered before and during treatment with daratumumab.
For those patients who are receiving sulfamethoxazole and trimethoprim for Pneumocystis jirovecii prophylaxis, daratumumab may serendipitously provide prophylaxis against L. monocytogenes given the overlap of spectrum of activity; however, the role of isolated antimicrobial prophylaxis against listeria in patients receiving daratumumab is currently unknown. A heightened awareness among clinicians of the increased risk for listeriosis in patients with MM who are receiving daratumumab, especially those who have gastroenteritis, unexplained sepsis, or neurologic findings that are suspicious for infection, can aid in the prompt recognition of listeria meningitis or meningoencephalitis and can help ensure the prompt initiation of appropriate empiric antimicrobial therapy.
Author Disclosure Statement
Dr Edwards, Dr Warfield, Dr Saha, Dr Tosh, Dr Stevens, and Dr Clement have no conflicts of interest to report.
References
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): multiple myeloma. Version 1.2025. Accessed January 30, 2025. www.nccn.org/professionals/physician_gls/pdf/myeloma.pdf
- Facon T, Kumar SK, Plesner T, et al. Daratumumab, lenalidomide, and dexamethasone versus lenalidomide and dexamethasone alone in newly diagnosed multiple myeloma (MAIA): overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22:1582-1596.
- Nooka AK, Kaufman JL, Hofmeister CC, et al. Daratumumab in multiple myeloma. Cancer. 2019;125:2364-2382.
- Li W, Li Y, Jin X, et al. CD38: a significant regulator of macrophage function. Front Oncol. 2022;12:775649.
- Lischke T, Heesch K, Schumacher V, et al. CD38 controls the innate immune response against Listeria monocytogenes. Infect Immun. 2013;81:4091-4099.
- Amici SA, Young NA, Narvaez-Miranda J, et al. CD38 is robustly induced in human macrophages and monocytes in inflammatory conditions. Front Immunol. 2018;9:1593.
- Vassilopoulos S, Vassilopoulos A, Kalligeros M, et al. Cumulative incidence and relative risk of infection in patients with multiple myeloma treated with anti-CD38 monoclonal antibody-based regimens: a systematic review and meta-analysis. Open Forum Infect Dis. 2022;9:ofac574.
- Dimopoulos MA, Oriol A, Nahi H, et al. Daratumumab, lenalidomide, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375:1319-1331.
- Facon T, Kumar S, Plesner T, et al. Daratumumab plus lenalidomide and dexamethasone for untreated myeloma. N Engl J Med. 2019;380:2104-2115.
- Darzalex (daratumumab for injection), for intravenous use [prescribing information]. Janssen Biotech; July 2024.
- Khan S, Vaisman A, Hota SS, et al. Listeria susceptibility in patients with multiple myeloma receiving daratumumab-based therapy. JAMA Oncol. 2020;6:293-294.
- Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30:239-245.
- Fujii T, Ohno N, Kitahara S, et al. Listeria meningitis in a patient with multiple myeloma undergoing daratumumab/bortezomib/dexamethasone therapy. Rinsho Ketsueki. 2022;63:117-120.
- Horikita F, Hashiguchi J, Nagashima T, et al. Listeria meningitis after gastrointestinal endoscopy in a patient with multiple myeloma undergoing daratumumab combination therapy. Rinsho Ketsueki. 2020;61:1611-1615.
- Ueno T, Ohta T, Imanaga H, et al. Listeria monocytogenes bacteremia during isatuximab therapy in a patient with multiple myeloma. Intern Med. 2021;60:3605-3608.