Connecting Science to Practice
In this research/quality improvement project, we explore
and discuss our experience prescribing bispecific T-cell engager
therapy for patients with multiple myeloma throughout
the health network. Although many patients are unable
to receive these therapies for socioeconomic reasons, our
experience serves as a real-world template to provide these
treatments in the community setting.
Multiple myeloma (MM) is an incurable hematologic malignancy characterized by the monoclonal proliferation of plasma cells.1 MM’s disease course is often characterized by treatment nonresponse and multiple relapses.1 According to the International Myeloma Working Group, relapsed or refractory MM is defined as the progression of disease despite receiving therapy or being within 60 days of the end of the last treatment.2 Patients who do not respond to 3 different classes of therapy, including proteasome inhibitors, immunomodulatory drugs, and anti-CD38 monoclonal antibodies, have triple class–refractory disease and have an overall survival of <1 year.3,4 Despite recent treatment advances, patients with relapsed or refractory MM, particularly those with triple-class–refractory disease, face poor outcomes. There is a pressing need for novel therapies to improve outcomes in this population.
Bispecific T-cell engagers (BiTEs) are a type of monoclonal bispecific antibodies (BsAbs) that represent a novel class of immunotherapeutic agents that simultaneously target tumor cells and T cells, enhancing T-cell–mediated cytotoxicity.5 Teclistamab, talquetamab, and elranatamab are BsAbs with different molecular targets on malignant plasma cells. B-cell maturation antigen (BCMA), a member of the tumor necrosis factor receptor family, is primarily expressed on plasma cells and mature B lymphocytes and is minimally present on other tissue.4 This has made BCMA an ideal target for drugs in the treatment of MM. BCMA-targeted treatments include antibody–drug conjugates, CAR T cells, and BiTEs.6 In the past several years, CAR T-cell therapies have been routinely used as therapy; the FDA approved idecabtagene vicleucel in 2022 and ciltacabtagene autoleucel in 2021 for the treatment of relapsed or refractory MM. GPRC5D, which is expressed on malignant plasma cells in patients with MM, is the target antigen for the BsAb talquetamab.7
Cytokine release syndrome (CRS) is an exaggerated systemic reaction associated with inflammatory cytokines mediated by T-cell–engaging therapies.8 CRS is triggered by the release of inflammatory cytokines, such as TNF-α, IFN-γ, interleukin (IL)-6, and IL-10. The massive immune cell activation leads to clinical symptoms of fever, hypoxemia, and hypotension.5 The prominent role of IL-6 in the pathogenesis of CRS is displayed by the rapid reversal of clinical symptoms when IL-6 signaling is interrupted using the IL-6 receptor antagonist tocilizumab. Immune effector cell–associated neurotoxicity syndrome (ICANS) is another cytokine-mediated systemic complication of CAR-T and BsAbs that can result in endothelial dysfunction and subsequent blood–brain barrier disruption.9 This can lead to serious clinical complications, such as encephalopathy and confusion, cerebral edema, and seizures, and even death.9
Although BsAbs offer improved outcomes, their use is associated with CRS, a potentially life-threatening inflammatory response. The MajesTEC-1 and MonumenTAL-1 trials demonstrated high overall response rates (63% and 70%, respectively), but they also reported CRS in 72% to 75% of patients, emphasizing the need for careful monitoring.3,6 Reassuringly, the MonumenTAL-1 trial noted the incidence of high-grade (grade ≥3) CRS only in a very small (0.6%) population of patients.6 After these promising results, teclistamab and talquetamab were approved by the FDA for use in patients with relapsed or refractory MM who have received 4 lines of systemic therapy; however, because of the high risk for CRS and its potential to progress to catastrophic outcomes, such as systemic inflammatory response, shock, and multiorgan failure,9 the FDA has mandated a Risk Evaluation and Mitigation Strategy (REMS) program before treatment with teclistamab or talquetamab to facilitate the safe prescribing and dispensing of these novel immunotherapeutic agents.10,11
Because the median time to onset of CRS is 2 days, which most often occurs after receiving the first 2 or 3 doses of a BiTE, patients are recommended to be hospitalized for 48 hours after the administration of each of the step-up doses for teclistamab or talquetamab.10,11 As a result of CRS identification and management, transplant and cellular therapy academic centers are often the most equipped to administer and monitor patients for BsAbs-related adverse events (AEs). Despite these locations oftentimes being inconvenient and at far distances from patients’ residences, patients typically continue outpatient treatment well past the window of CRS risk because they were previously established at the academic institution and because community centers may be unfamiliar with managing CRS. Administering BsAbs in the community setting could greatly improve access to care and patients’ quality of life by avoiding the need to travel long distances, and may lower healthcare costs and ease the burden on tertiary centers.
Given these benefits and because most patients with MM receive care in the community setting, there has been increased interest in analyzing and addressing the barriers to accessing BsAb therapy elsewhere than at academic centers. Major concerns that are often cited as limiting factors and cause patients to hesitate whether to receive these therapies in the outpatient setting include reduced monitoring capabilities; a lack of higher acuity support for life-threatening AEs, such as CRS and ICANS; and resource constraints for recognition and prompt treatment of delayed AEs, such as prolonged cytopenias and infections.12,13 For example, the Bispecific Outpatient Safe Step-up (BOSS) program at Massachusetts General Hospital sought to enable the safe administration of elranatamab in patients with relapsed or refractory MM using standardized therapy administration and monitoring, and has shown promise in the safe delivery of treatment in the ambulatory setting.14
This study evaluates the safety and feasibility of administering teclistamab or talquetamab in community infusion centers within our network, which is supported by a tailored monitoring protocol.
Methods
This retrospective cohort study was conducted at our network infusion centers and included adults with relapsed or refractory MM who received teclistamab or talquetamab between January 2023 and May 2024.
All patients were evaluated by a physician at the academic center before beginning treatment to confirm all supportive care was started appropriately, including infection prophylaxis (eg, antiviral and Pneumocystis jirovecii pneumonia prophylaxis), prophylactic intravenous immunoglobin, and cytopenia management, if applicable.
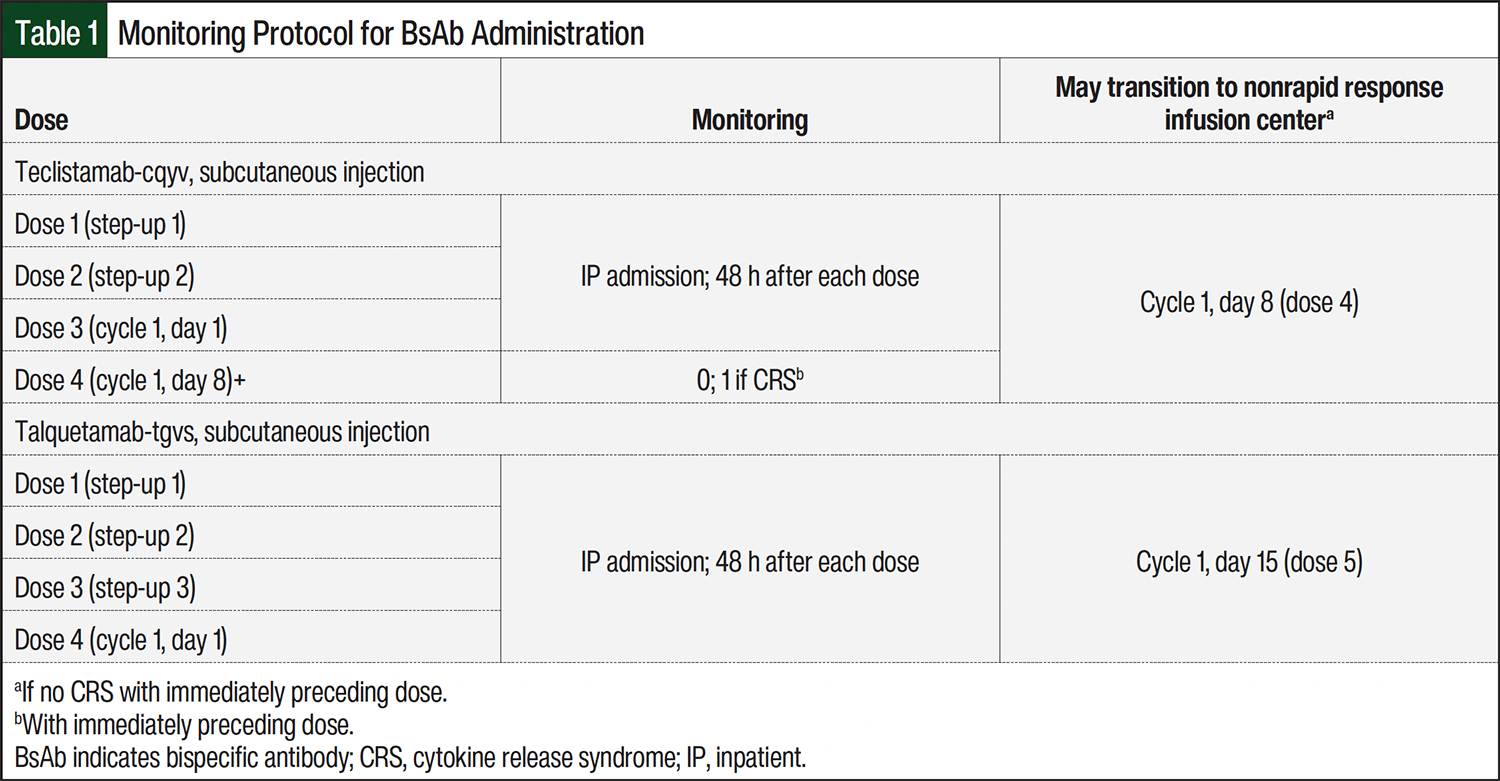
Based on the REMS and prescribing information recommendations,10,11,15 a monitoring protocol was developed to facilitate discharge from the academic hospital to receive subsequent outpatient treatment at local community infusion centers when safe and feasible (Table 1). Patients were hospitalized for 48 hours after receiving each step-up dose within the respective step-up dosing schedule to monitor for CRS and neurotoxicity. Patients were hospitalized for 48 hours after receiving the first 3 doses of teclistamab or first 4 doses of talquetamab after the biweekly talquetamab step-up schedule. Starting with the fourth dose of teclistamab or the fifth dose of talquetamab, patients were allowed to receive further treatment doses at a community infusion center if no CRS events were reported with the immediately preceding dose.
CRS was graded using the American Society for Transplantation and Cellular Therapy (ASTCT) consensus criteria.16 For patients with symptoms of CRS, immediate interventions, such as tocilizumab or corticosteroids, were readily available. If a CRS event occurred after the immediately preceding dose of teclistamab or talquetamab, the patient was closely monitored at a facility equipped with rapid response capabilities for 1 hour after the subsequent dose until the patient successfully received a dose without further CRS events, indicating that the treatment was well tolerated and could proceed without intensive observation. These treatments occurred either at 1 of the 2 main hospital infusion center locations or at 1 of the 5 community infusion centers equipped with rapid response capabilities.
If postinjection monitoring was indicated, vital signs were checked immediately before the teclistamab or talquetamab injection and immediately after the injection, and then every 30 minutes for 1 hour after the injection. ICANS, including the immune effector cell–associated encephalopathy (ICE) score, and CRS assessments were completed before the injection and were repeated only as clinically indicated by the infusion center nurse. Physician assessments were only required if patients had grade ≥1 CRS or ICANS. Corticosteroids and fluids were readily assessable; if a patient required tocilizumab for grade ≥2 CRS, the patient would be transferred to the primary academic hospital after receiving corticosteroids and other supportive care at the infusion center. After discharge, patients were seen at the academic center monthly via telehealth or in-person visits.
Training on CRS and neurotoxicity identification and management was proactively provided to all nursing and pharmacy staff at the community infusion center locations via nursing and pharmacy personnel and medical science liaisons, as able. Training included ASTCT CRS and ICANS grading, including ICE scoring, administration and monitoring criteria for teclistamab or talquetamab based on the prescribing information’s recommendations and a network-specific monitoring protocol (Table 1), and documenting within the electronic health record to accurately record any AEs and CRS and ICANS scores, as applicable. All outpatient monitoring for BiTEs is managed by infusion center nursing after protocols to ensure patient safety. Vital signs are monitored before and after each step-up dose, in accordance with the unit protocols, which require assessments every 4 hours and as needed based on clinical indications.
If the patient has any-grade CRS with the immediately preceding dose of teclistamab or talquetamab, vitals are assessed every 30 minutes for 1 hour, followed by additional monitoring as clinically indicated. If the patient has a fever, further CRS grading is performed. Although the nursing staff initially documents these findings, final confirmation and evaluation are performed by a healthcare provider, either an advanced practitioner or physician, as available. In addition to CRS monitoring, a neurotoxicity assessment, which includes evaluating ASTCT ICANS criteria parameters, including the ICE score, is completed by nursing staff before each dose and as clinically necessary. In addition, all community infusion center pharmacies were enrolled in the REMS programs for teclistamab and talquetamab before the patients started treatment at the academic hospital.15 The prescribing providers at the academic center and community infusion centers were also REMS certified to ensure full compliance with the REMS requirements and safety protocols.
Results
A total of 24 patients received teclistamab or talquetamab at a network facility between January 2023 and May 2024. In all, 4 patients were not discharged for further outpatient treatment because they died during hospitalization. Of the 4 patients, 2 died of disease progression, 1 died of severe sepsis, and 1 died of respiratory failure. Among the 17 patients included for analysis, 20 teclistamab or talquetamab step-up phases were included for analysis; of the 20 step-up phases, 16 patients received teclistamab only, 1 received talquetamab alone, and 3 received talquetamab and teclistamab.
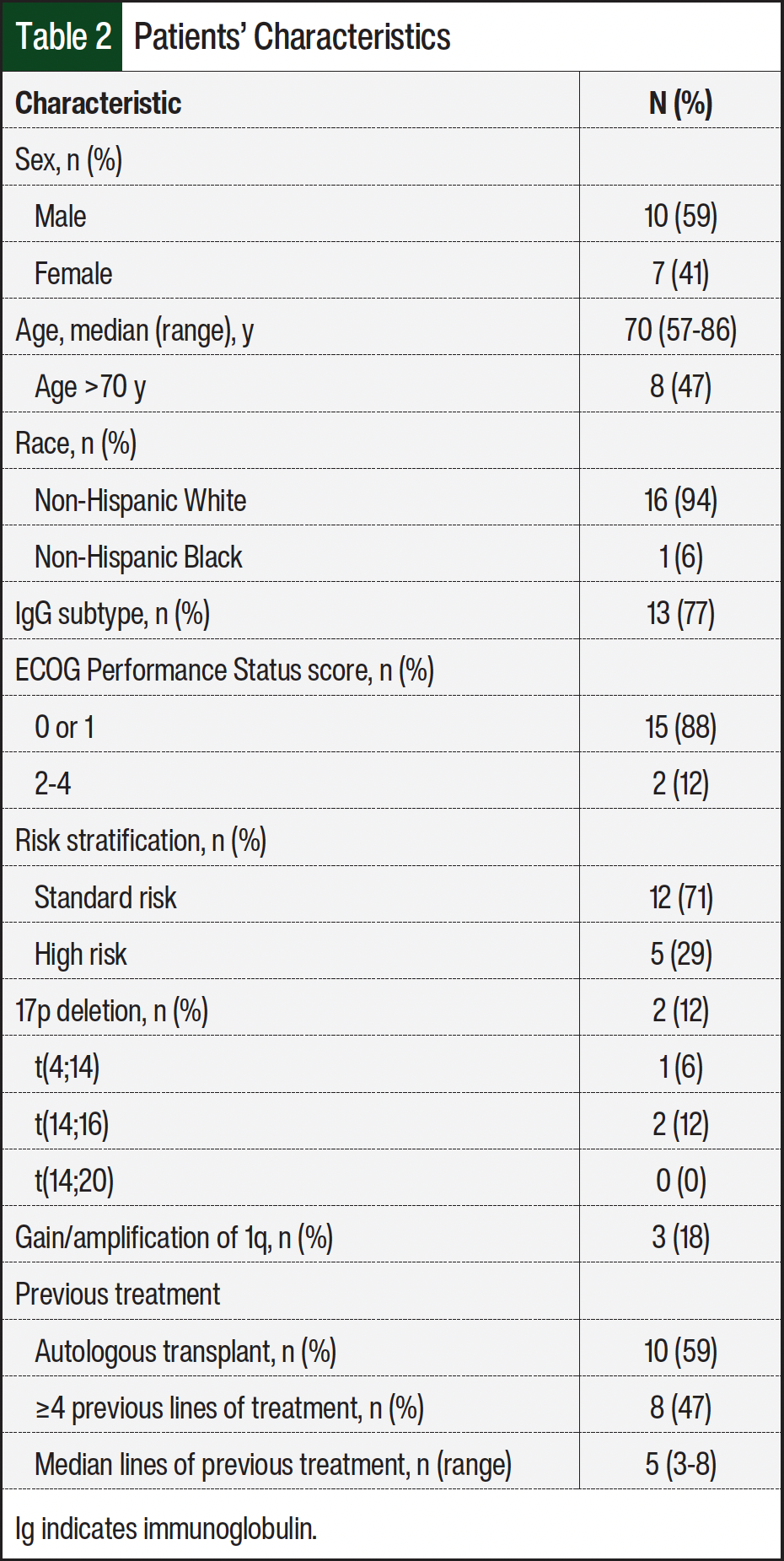
Of the 17 patients, most were male (59%), White (94%), and had standard-risk MM (71%; Table 2). The median patient age was 70 years. A total of 13 (77%) patients had the immunoglobulin G subtype of MM. Most patients had an ECOG Performance Status of 0 or 1 (88%) with only 2 (12%) patients having an ECOG score of 2 to 4. In all, 2 (12%) patients had 17p deletion on cytogenetics, 1 (6%) patient had translocation 4;14, 2 (12%) patients had translocation 14;16, none of the patients had t(14;20), and 3 (18%) patients had amplification/gain of 1q. A total of 10 (59%) patients had a previous autologous stem-cell transplant. The median lines of previous treatment was 5 (range, 3-8 lines); 8 (47%) patients received ≥4 previous lines of systemic treatment (Table 2).
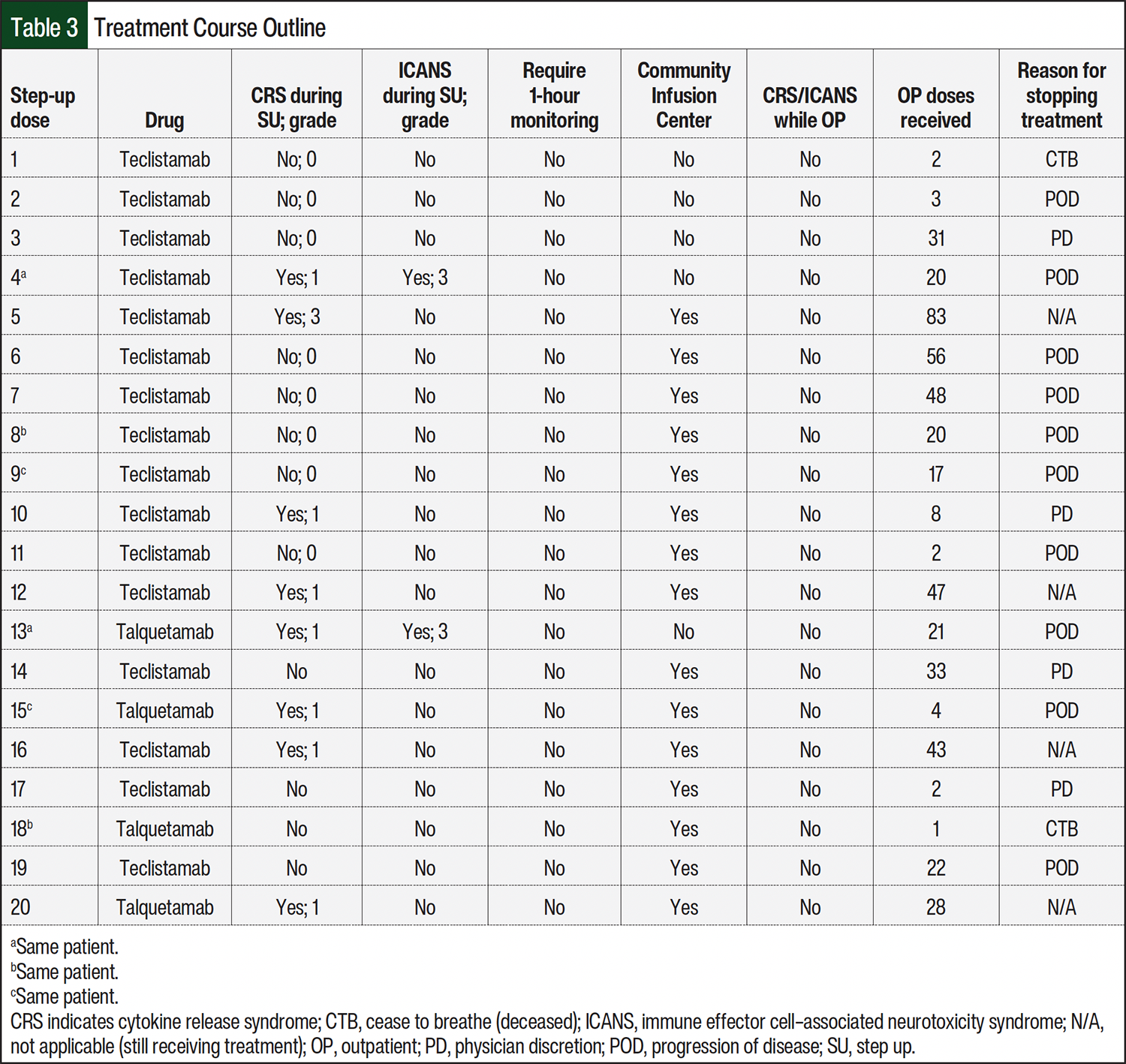
At the start of inpatient treatment, CRS occurred in 8 (40%) of the 20 step-up phases (Table 3). Most (87.5%) of the CRS cases were grade 1, with 1 (12.5%) incident of grade 3 CRS. All incidences of CRS occurred after the first step-up dose, apart from one grade 1 CRS episode that occurred after the third step-up dose of talquetamab. CRS was managed according to the ASTCT guidelines, with supportive care for grade 1 CRS and supportive care, tocilizumab, and dexamethasone for grade 3 CRS. As expected, ICANS occurred much less often, with only 2 (10%) incidences of grade 3 ICANS, which occurred in the same patient who received teclistamab and subsequent talquetamab. ICANS was managed with supportive care, levetiracetam, and dexamethasone with a prolonged taper, and the patient returned to baseline cognitive function by discharge. Because all of the patients returned to their baseline functional and cognitive statuses by discharge and tolerated subsequent doses of teclistamab or talquetamab without CRS or ICANS, including the patients with grade 3 CRS and ICANS, each patient was discharged and received outpatient teclistamab or talquetamab treatment without requiring 1 hour of additional monitoring (Table 3).
A total of 13 network infusion centers, including 11 community infusion centers, were used for the outpatient administration of teclistamab or talquetamab. Of the 11 infusion centers, 7 (64%) did not have rapid response capabilities. A total of 13(65%) patients received their infusions in a community infusion center. None of the patients presented with CRS or ICANS after their initial step-up doses, and all patients were managed outpatient without the need for corticosteroids, tocilizumab, or subsequent hospitalization for treatment-related AEs. The patients continued to receive outpatient treatment with teclistamab or talquetamab for a median of 20.5 doses (range, 1-83 doses). The reasons for ultimately stopping therapy were disease progression (50%), physician discretion as a result of concomitant medical issues that necessitated treatment breaks (20%), the patient was still receiving treatment (20%), and death resulting from infectious complications (10%; Table 3).
Discussion
This study demonstrates the feasibility and safety of administering BsAbs for relapsed or refractory MM in community settings after receiving initial step-up doses. Our monitoring protocol combined with staff training may serve as a model for the safe administration of BsAbs at community locations after receiving initial inpatient step-up doses.
A limitation that is often cited for the administration of teclistamab or talquetamab locally is the lack of rapid response capabilities, which we showed is safe after the patient receives initial step-up doses. These findings are significant because many patients with relapsed or refractory MM receive treatment in community centers, where access to life-extending therapies such as teclistamab or talquetamab is critical. Expanding the use of these therapies in community infusion centers has the potential to transform patients’ outcomes by improving accessibility and convenience.
Studies have shown that patients from economically disadvantaged backgrounds and underserved areas have less access to innovative therapies for MM.17-19 In addition, Black and other minority patients have disproportionately reduced access to these advanced treatments. Research also indicates that patients in rural areas may face limited availability of autologous transplantation or participation in clinical trials.17,18 These socioeconomic and geographic factors contribute to significant disparities in care.19 By enabling the safe administration of BsAbs in community infusion centers, healthcare systems can bridge these gaps and provide underserved populations with more equitable access to effective therapies.
Surveys of oncologists in community settings have identified several common barriers to the use of BsAbs, including transitioning patients from inpatient to outpatient care, managing remote patients, and trepidations regarding addressing AEs without rapid response capabilities and specially trained in-house staff.20 Socioeconomic barriers, such as transportation and financial constraints, further complicate access to BsAbs for many patients.21 Administering these therapies in local infusion centers helps to alleviate these challenges by reducing the burden of travel. Health literacy is another critical factor, because underserved patients may struggle to recognize AEs, such as CRS, that require prompt intervention to prevent severe outcomes.22 To address this, our institution mandates caregiver education for all patients receiving teclistamab or talquetamab, including recognizing the early signs of CRS and infection, knowing when to seek medical attention, and understanding the importance of adherence to follow-up protocols. Such initiatives are vital in ensuring the safety and success of BsAb administration in community settings.
Because of overlapping AE profiles and similar administration strategies, the monitoring protocol for teclistamab and talquetamab can be extrapolated and may be applicable to other BsAbs in other disease states. Implementing tailored monitoring protocols, training staff at community infusion centers, and providing comprehensive education for caregivers could help expand access to these therapies as well. Our findings align with previous real-world studies, including those from Mayo Clinic sites in Phoenix/Scottsdale, AZ, Jacksonville, FL, and Rochester, MN, which also support the safety of outpatient BsAb administration.23 However, a key limitation of the Mayo Clinic study lies in its limited generalizability, because its outcomes reflect practices within highly resourced centers.
In contrast, our study emphasizes the successful implementation of a standardized protocol for BsAb delivery across community infusion centers, settings that more closely resemble the treatment environment, and more limited infrastructure available to the broader patient population. As the treatment landscape for relapsed or refractory MM and other hematologic malignancies continues to evolve, it is essential to prioritize equitable access to BsAbs. The safe administration of BsAbs in community settings will facilitate the expansion of access to novel therapies for patients residing in remote areas without academic or tertiary care centers, allow patients to stay within their support systems and receive care within familiar environments, and improve the overall efficiency of healthcare systems by cutting down on logistical delays and costs.
Limitations
Our study has limitations, which include a small sample size, a lack of CRS and ICANS events occurring after the initial ramp up to evaluate the 1 hour postinjection monitoring protocol, and the use of a fully integrated network with academic and community sites that share an electronic health record.
Conclusion
The incidences of CRS and ICANS after the initial teclistamab or talquetamab step-up doses are complete are rare. Our findings support that there were no CRS or ICANS events that occurred beyond the initial ramp-up period for teclistamab or talquetamab. BiTEs, such as teclistamab and talquetamab, may be safely administered in community infusion centers with appropriate monitoring protocols, but they need to be evaluated in a larger patient population. Having a protocol for the administration of teclistamab or talquetamab in the community setting would expand access to effective therapies for relapsed or refractory MM and thereby address disparities in care and improve patients’ outcomes. Future research should focus on scaling this approach to diverse settings and including other BsAb therapies.
Author Disclosure Statement
Dr Jiwani, Dr Maples, Dr Rajarajan, Dr Babu, Dr Sadashiv, and Dr Mewawalla have no conflicts of interest to report.
References
- Malard F, Neri P, Bahlis NJ, et al. Multiple myeloma. Nat Rev Dis Primers. 2024;10:45.
- Relapsed refractory multiple myeloma (RRMM) glossary. International Myeloma Foundation. 2023. Accessed June 16, 2025. www.myeloma.org/publications-videos/relapsed-refractory-multiple-myeloma-rrmm-glossary
- Schinke CD, Touzeau C, Minnema MC, et al. Pivotal phase 2 MonumenTAL-1 results of talquetamab (tal), a GPRC5DxCD3 bispecific antibody (BsAb), for relapsed/refractory multiple myeloma (RRMM). J Clin Oncol. 2023;41(16 suppl):8036.
- Danis R, Bujarski S, Yashar D, et al. Outcomes of triple-class refractory multiple myeloma patients at a single clinic: a retrospective study. Leuk Res. 2025;151:107677.
- Dahlén E, Veitonmäki N, Norlén P. Bispecific antibodies in cancer immunotherapy. Ther Adv Vaccines Immunother. 2018;6:3-17.
- Moreau P, Garfall AL, van de Donk NWCJ, et al. Teclistamab in relapsed or refractory multiple myeloma. N Engl J Med. 2022;387:495-505.
- Chari A, Minnema MC, Berdeja JG, et al. Talquetamab, a T-cell-redirecting GPRC5D bispecific antibody for multiple myeloma. N Engl J Med. 2022;387:2232-2244.
- Shimabukuro-Vornhagen A, Gödel P, Subklewe M, et al. Cytokine release syndrome. J Immunother Cancer. 2018;6:56.
- Markouli M, Ullah F, Unlu S, et al. Toxicity profile of chimeric antigen receptor T-cell and bispecific antibody therapies in multiple myeloma: pathogenesis, prevention and management. Curr Oncol. 2023;30:6330-6352.
- Talvey (talquetamab-tgvs) injection, for subcutaneous use [prescribing information]. Janssen Biotech, Inc; October 2025. Accessed January 16, 2026. www.accessdata.fda.gov/drugsatfda_docs/label/2025/761342s016lbl.pdf
- Tecvayli (teclistamab-cqyv) injection, for subcutaneous use [prescribing information]. Janssen Biotech, Inc; August 2025. Accessed January 16, 2026. www.accessdata.fda.gov/drugsatfda_docs/label/2025/761291s013lbl.pdf
- Garfall AL, Banerjee R, Frenzel L, et al. A roadmap to implementing outpatient administration of bispecific antibodies in multiple myeloma. Front Oncol. 2025;15:1630146.
- Mahmoudjafari Z, Ali A, Davis J, et al. Seamless navigation of bispecific therapies: optimizing management and outpatient access with a focus on coordination. J Adv Pract Oncol. 2024:1-16.
- Cirstea D, Kim EB, O’Neill S, et al. Implementing the bispecific outpatient safe step-up (BOSS) program for elranatamab in ambulatory treatment of multiple myeloma. J Clin Oncol. 2025;43:e19507.
- Tecvayli and Talvey REMS. Tec-talrems.com. Published 2025. Accessed June 16, 2025. www.tec-talrems.com/#Main
- Lee DW, Santomasso BD, Locke FL, et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol Blood Marrow Transplant. 2019;25:625-638.
- Ai S, Thind A, Parmar G. Differences in survival of patients with multiple myeloma in rural versus metropolitan regions: analysis of population data of an Australian local health district. Aust J Rural Health. 2024;32:1207-1215.
- Mateos MV, Ailawadhi S, Costa LJ, et al. Global disparities in patients with multiple myeloma: a rapid evidence assessment. Blood Cancer J. 2023;13:109.
- Ebrahimi H, Megally S, Plotkin E, et al. Barriers to clinical trial implementation among community care centers. JAMA Netw Open. 2024;7:e248739.
- Killmurray C. Utilization challenges hold back the adoption of bispecific therapies. February 23, 2024. Accessed January 20, 2026. www.targetedonc.com/view/utilization-challenges-hold-back-the-adoption-of-bispecific-therapies
- Kim J, Cho J, Lee MH, et al. Comparison of CAR T-cell vs. bispecific antibody as third- or later-line large B-cell lymphoma therapy: a meta-analysis. Blood. 2024;144:629-638.
- Holden CE, Wheelwright S, Harle A, Wagland R. The role of health literacy in cancer care: A mixed studies systematic review. PLoS One. 2021;16:e0259815.
- Sandahl TB, Soefje SA, Fonseca R, et al. Real-world safety and health care resource utilization of teclistamab under an outpatient model for step-up dosing administration. JCO Oncol Pract. 2025;21:702-709.