Connecting Science to Practice
This case report sheds light on the effect of continuous
renal replacement therapy on the efficacy and safety of calaspargase-
pegol, a key component of induction therapy for
acute lymphoblastic leukemia in children. In the setting of
tumor lysis syndrome, renal replacement therapy is often
indicated, coinciding with the timing of calaspargase-pegol.
There is no published literature that shows whether dose
adjustments are needed for this medication to ensure safe
and effective administration. In this case report, the therapeutic
effect of calaspargase-pegol was monitored via asparaginase
activity levels, which were at goal after infusion.
Thus, calaspargase-pegol may have similar activity without
dose adjustment for pediatric patients with acute lymphoblastic
leukemia who require continuous renal replacement
therapy.
Acute lymphoblastic leukemia (ALL) is a malignant disorder of lymphoblasts that constitutes 25% of cancers affecting children aged <15 years.1 The antineoplastic effect of asparaginase stems from L-asparagine being an essential amino acid for lymphoblasts, which lack asparagine synthetase and depend on extracellular sources of L-asparagine to survive.2 Asparaginase is an enzyme that hydrolyzes L-asparagine into aspartic acid and ammonia-depleting serum L-asparagine, leading to the starvation of lymphoblasts and an inability to synthesize proteins, DNA, and RNA, leading to cell death.2 Asparagine depletion with asparaginase therapy is an essential component of the multiagent regimen that is currently used for the treatment of pediatric ALL and lymphoblastic lymphoma. Calaspargase-pegol is used for asparagine depletion in pediatric patients. It is dosed per body surface area at 2500 IU/m2 every 3 weeks.3 Because it is a pegylated moiety, it undergoes proteolytic degradation, with a half-life elimination of approximately 16 days.3 Therapeutic drug monitoring of asparaginase’s activity levels can be performed 3 to 7 days after each dose to ensure adequate asparagine depletion and the absence of neutralizing antibodies.4
Patients presenting with a new diagnosis of high-risk ALL or lymphoblastic lymphoma who are receiving induction chemotherapy have an elevated risk for tumor lysis syndrome (TLS).5 TLS can lead to severe electrolyte derangements, such as hyperkalemia, hyperphosphatemia, hypocalcemia, and hyperuricemia.5 These imbalances can lead to life-threatening complications, such as acute kidney injury, arrythmias, seizures, and death.5 Dialysis may be required to help correct these electrolyte abnormalities and prevent serious complications if more conservative management strategies, such as hyperhydration, the dietary restriction of phosphorous, and the administration of phosphate binders, are unsuccessful.5 Treatment with calaspargase-pegol in a pediatric patient receiving dialysis or continuous renal replacement therapy (CRRT) has not been previously described.
Case Report
A previously healthy 8-year-old boy who weighed 41.5 kg presented with a 1-month history of sore throat, intermittent fever, a 3.2-kg weight loss, and an enlarging neck mass. He was admitted to the pediatric intensive care unit as a result of acute respiratory failure requiring emergent mechanical ventilation. The complete blood count with differential demonstrated hyperleukocytosis with increased blasts, thrombocytopenia, and anemia. A computed tomography scan of his head and neck with intravenous (IV) contrast showed bilateral cervical lymphadenopathy. Peripheral blood flow cytometry confirmed a diagnosis of T-cell ALL. He started receiving an induction chemotherapy regimen of (a modified Berlin-Frankfurt-Münster chemotherapy regimen that includes dexrazoxane and calaspargase-pegol) IV vincristine 1.5 mg/m2 on days 1, 8, 15, and 22; IV daunorubicin 25 mg/m2 with dexrazoxane 250 mg/m2 on days 1, 8, 15, and 22; IV calaspargase-pegol 2500 IU/m2 on day 4; and oral dexamethasone 3 mg/m2 twice daily on days 1 to 28.6 Intrathecal cytarabine was initially deferred because of the patient’s instability. The patient received intrathecal methotrexate on days 8, 15, 22, and 29 because an initial lumbar puncture was not obtained and he was therefore treated as if he were CNS3 status.
The patient quickly had TLS as defined by laboratory and clinical Cairo-Bishop classifications soon after the start of therapy, with signs of acute renal failure associated with multiple electrolyte derangements, despite multiple preventive therapies, including hyperhydration, rasburicase, and allopurinol.7 He initially received intermittent hemodialysis on day 1 of therapy followed by CRRT for a total of 4 days (days 2-5 of chemotherapy) and had improvement in renal function. The average effluent flow rate for his CRRT was 29 mL/kg/hr, with a range of 22 to 32 mL/kg/hr. The patient was eventually extubated, stabilized, and transferred to the pediatric hematology/oncology service.
The patient’s course was further complicated by acute, altered mental status and posterior reversible encephalopathy syndrome. He also had hand tremors and peripheral neuropathy, which were likely a result of treatment with vincristine and intrathecal methotrexate. In addition, he had significant abdominal pain and was diagnosed with neutropenic enterocolitis, which eventually resolved without surgical intervention.
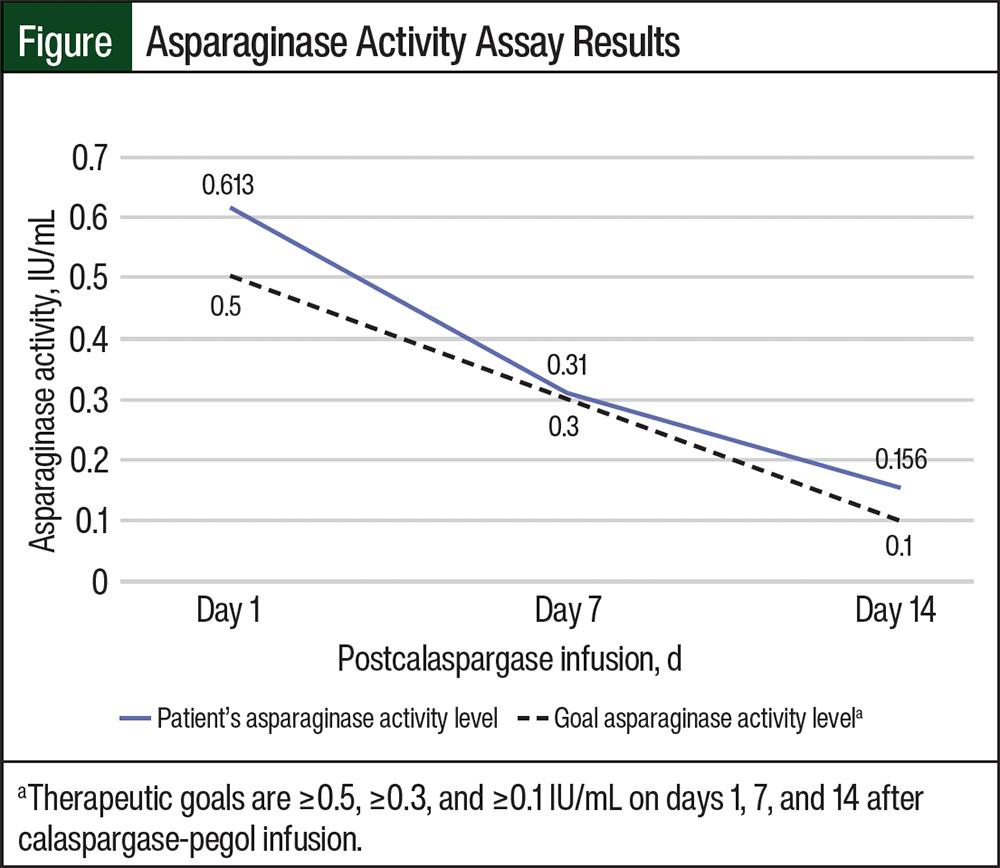
During the patient’s prolonged hospitalization, serum asparaginase activity assays were collected on days 1, 7, and 14 after calaspargase-pegol infusion (days 5, 11, and 18 of chemotherapy); the results of this testing were 0.613 IU/mL, 0.31 IU/mL, and 0.156 IU/mL, respectively (Figure). He was receiving CRRT during the time of his calaspargase-pegol infusion, and none of his complications were believed to be caused by calaspargase-pegol. The patient continued to receive CRRT for the collection of the first asparaginase activity assay, but he no longer received CRRT at the subsequent level collections.
Discussion
Asparagine depletion with asparaginase is essential for the management of pediatric ALL.8 However, the effect of renal impairment on the pharmacokinetics of calaspargase-pegol is unknown, and there are currently no dosage recommendations provided in the prescribing information for calaspargase-pegol or in the available literature for patients receiving renal replacement therapy. An in vivo pharmacokinetic study in mice showed that asparaginase is rapidly cleared from the serum by liver, spleen, and bone marrow phagocytic cells.9 However, the clearance mechanism in humans has yet to be fully elucidated. Pediatric patients presenting with ALL may be critically ill and can have life-threatening complications, such as TLS on the initiation of therapy.5 TLS can lead to renal failure that necessitates treatment with hemodialysis plus CRRT. Treatment with calaspargase-pegol in pediatric patients with ALL and CRRT has not previously been described, and it is unknown whether patients will be able to obtain therapeutic levels while receiving CRRT.
This case describes a patient who received CRRT immediately before, during, and for 1 day after calaspargase-pegol infusion. Therapeutic drug monitoring of asparaginase activity levels was completed to ensure adequate asparagine depletion. The levels were obtained on days 1, 7, and 14 after infusion. The results, which are shown in the Figure, demonstrate the ability to maintain minimum asparaginase activity thresholds for efficacy at the 3 time points of ≥0.5 IU/mL, ≥0.3 IU/mL, and ≥0.1 IU/mL, respectively, per the Children’s Oncology Group’s standard recommendations.10
Published literature also suggests a single asparaginase activity level of ≥0.1 IU/mL at 14 days after calaspargase-pegol infusion provides adequate asparaginase depletion.11,12 In addition to maintaining efficacy, the patient did not have any adverse events related to calaspargase-pegol therapy, which includes hypersensitivity reactions, pancreatitis, thrombosis, bleeding, or hepatotoxicity.13
This case provides evidence that calaspargase-pegol activity is not dramatically affected by CRRT. However, this patient was only receiving CRRT for a brief period of time and was no longer receiving renal replacement therapy 1 day after infusion with calaspargase-pegol. The findings of this case may not be applicable to patients who are receiving CRRT for a longer duration of time or to those who are receiving intermittent hemodialysis. Future studies are needed to determine the true effect of CRRT on calaspargase-pegol activity in pediatric patients.
Conclusion
We report a case of treatment with calaspargase-pegol for ALL in a pediatric patient receiving CRRT. This case demonstrates that the therapeutic effect of calaspargase-pegol was maintained via the monitoring of asparaginase activity levels, which were at goal after infusion. This case provides evidence that calaspargase-pegol may have similar activity without dose adjustment in pediatric patients with ALL who require CRRT.
Author Disclosure Statement
Dr Mace, Dr Dalal, and Dr Lynch have no conflicts of interest to report.
References
- National Cancer Institute. Childhood Acute Lymphoblastic Leukemia Treatment (PDQ®): Health Professional Version. Revised April 1, 2025. Accessed November 11, 2024. www.cancer.gov/types/leukemia/hp/child-all-treatment-pdq
- Schore RJ, Devidas M, Bleyer A, et al. Plasma asparaginase activity and asparagine depletion in acute lymphoblastic leukemia patients treated with pegaspargase on Children’s Oncology Group AALL07P4. Leuk Lymphoma. 2019;60:1740-1748.
- US Food and Drug Administration. Center for Drug Evaluation and Research. BLA Multidisciplinary Review and Evaluation: BLA 761102. December 21, 2017. Accessed April 18, 2025. www.accessdata.fda.gov/drugsatfda_docs/nda/2018/761102Orig1s000MultidisciplineR.pdf
- Salzer W, Bostrom B, Messinger Y, et al. Asparaginase activity levels and monitoring in patients with acute lymphoblastic leukemia. Leuk Lymphoma. 2018;59:1797-1806.
- Russell TB, Kram DE. Tumor lysis syndrome. Pediatr Rev. 2020;41: 20-26.
- Teachey DT, Devidas M, Wood BL, et al. Children’s Oncology Group trial AALL1231: a phase III clinical trial testing bortezomib in newly diagnosed T-cell acute lymphoblastic leukemia and lymphoma. J Clin Oncol. 2022;40:2106-2118.
- Cairo MS, Bishop M. Tumour lysis syndrome: new therapeutic strategies and classification. Br J Haematol. 2004;127:3-11.
- Silverman LB, Gelber RD, Dalton VK, et al. Improved outcome for children with acute lymphoblastic leukemia: results of Dana-Farber Consortium Protocol 91-01. Blood. 2001;97:1211-1218.
- van der Meer LT, Terry SYA, van Ingen Schenau DS, et al. In vivo imaging of antileukemic drug asparaginase reveals a rapid macrophage-mediated clearance from the bone marrow. J Nucl Med. 2017;58:214-220.
- Children’s Oncology Group. Inotuzumab ozogamicin and post-induction chemotherapy in treating patients with high-risk B-ALL, mixed phenotype acute leukemia, and B-LLy. ClinicalTrials.gov identifier: NCT03959085. Updated May 23, 2025. Accessed November 11, 2024. https://clinicaltrials.gov/study/NCT03959085
- van der Sluis IM, Vrooman LM, Pieters R, et al. Consensus expert recommendations for identification and management of asparaginase hypersensitivity and silent inactivation. Haematologica. 2016;101:279-285.
- Bleyer A, Asselin BL, Koontz SE, Hunger SP. Clinical application of asparaginase activity levels following treatment with pegaspargase. Pediatr Blood Cancer. 2015;62:1102-1105.
- Vrooman LM, Blonquist TM, Stevenson KE, et al. Efficacy and toxicity of pegaspargase and calaspargase pegol in childhood acute lymphoblastic leukemia: results of DFCI 11-001. J Clin Oncol. 2021;39:3496-3505.