Connecting Science to Practice
Patients with hematologic malignancies or those undergoing
stem-cell transplant are at high risk for illness or death
from fungal infections. Posaconazole is often used in this
patient population for yeast- and mold-infection prophylaxis,
but it can result in a number of potential adverse events,
including hypokalemia. This article describes 2 patient cases
with severe posaconazole-related hypokalemia resulting
from presumed pseudohyperaldosteronism and discusses the
recognition and management of this condition.
Posaconazole is a triazole antifungal that targets the enzyme 14 alpha-demethylase, thereby inhibiting ergosterol production.1 Posaconazole has a broad spectrum and is used for the treatment and prevention of fungi and mold species, such as Mucor and Aspergillus, that are resistant to other azole antifungals.2 Patients with hematologic malignancies have a high risk for illness or death from fungal infections. The mortality rates are approximately 50% to 90% for patients with hematologic malignancies and Candida or Aspergillus infection.3-5 Fungal infections can be difficult to diagnose in a timely manner, putting patients at an increased risk for mortality.6 Because of this, posaconazole is the preferred agent recommended by the National Comprehensive Cancer Network guidelines for the prophylaxis of fungal infections in patients with myelodysplastic syndromes, acute myeloid leukemia (AML), and significant graft-versus-host disease (GVHD) who are receiving immunosuppressive therapy.6 In addition, posaconazole is frequently used in the setting of hematologic malignancies in patients who are receiving cellular therapies, such as stem-cell transplant (SCT) and chimeric antigen receptor T-cell therapy, or in patients who are receiving a maintenance treatment and are not eligible for transplant.6
The standard prophylactic dosage for posaconazole is 300 mg orally twice daily on the first day followed by 300 mg orally once daily starting on the second day for delayed-release tablets, or 200 mg orally 3 times daily for the suspension formula.7 Posaconazole trough levels are drawn 30 to 60 minutes before the next scheduled dose approximately 7 to 14 days once the steady-state concentrations are anticipated.8 A target trough of ≥0.7 mg/L is the currently accepted threshold for infection prophylaxis.8 This recommendation originated from early pharmacokinetic studies, one of which demonstrated invasive fungal infections in 6.5% of patients with posaconazole trough levels of <0.7 mg/L compared with only 1.9% of patients who achieved a level ≥0.7 mg/L.8,9 In addition, a trough upper limit of 3.75 mg/L for posaconazole has been supported by early preclinical data, which show that posaconazole is more likely to interfere with steroidogenesis in patients who have levels exceeding this threshold.10,11
The adverse events (AEs) associated with posaconazole treatment include nausea, vomiting, rash, diarrhea, transaminitis, and hypokalemia.7 Posaconazole-related hypokalemia results from pseudohyperaldosteronism, which is discussed later in further detail.12 The management of posaconazole-induced hypokalemia is not clearly defined at this time, and providers may not even consider posaconazole as a causative medication. We herein describe 2 patient cases with severe posaconazole-related hypokalemia and outline the strategies for the recognition and management of this condition.
Patient 1
A 64-year-old woman who had chronic myeloid leukemia without cardiac comorbidities and was in remission after an allogeneic SCT (ASCT) 4 years earlier had hypokalemia, with a serum potassium of 2.3 mmol/L (reference range, 3.6-5.3 mmol/L), during a routine follow-up visit. An electrocardiogram performed in the setting of hypokalemia revealed a prolonged corrected QT Fridericia (QTcF) formula interval of 592 milliseconds. On further investigation, she reported having diarrhea over the last few weeks, with a minimum of 4 yellow, watery stools daily. She was directed to the emergency department, where she had a brief episode of bradycardia.
At home, the patient was receiving posaconazole 300 mg daily for fungal infection prophylaxis, and ruxolitinib and sirolimus for the treatment of chronic oral and ocular GVHD. Of note, she had transitioned from treatment with fluconazole to posaconazole 4 weeks earlier to provide broader fungal coverage, because she had several risk factors for mold infections, such as the ongoing use of immunosuppression in the setting of chronic GVHD. When she initiated treatment with posaconazole, the dosing for sirolimus was empirically reduced from 0.5 mg every 48 hours to 0.5 mg every 72 hours because posaconazole is a strong cytochrome P3A4 inhibitor.
Of note, the prescribing information for posaconazole lists sirolimus as a contraindication for administration with posaconazole because of the increased risk for AEs.7 Clinically, sirolimus and posaconazole are often used together for treatment. When used together, an empiric dose reduction of 50% is recommended for sirolimus.13 Sirolimus levels should be monitored closely to ensure that they are within the desired range to avoid AEs.13
On admission to the hospital, our patient received aggressive potassium replacement (approximately 80 mEq orally daily), and treatment with posaconazole was immediately withheld considering the prolonged QTcF interval. In addition, sirolimus therapy was also held because of a supratherapeutic level of 24.6 ng/mL, which was unexpectedly high for the patient’s home dosage of 0.5 mg every 72 hours. This was believed to result from supratherapeutic posaconazole levels; hence, a random posaconazole level was drawn 6 days into admission, which was 7.5 mcg/mL. Incidentally, on the discontinuation of treatment with posaconazole, the patient’s diarrhea and hypokalemia resolved. Of note, an infectious workup for diarrhea at that time was negative.
The patient was discharged with at-home potassium supplementation and was instructed to stop receiving treatment with posaconazole indefinitely. A few days after discharge, the patient returned to the outpatient clinic for follow-up and again had hypokalemia, with a potassium level of 2.5 mmol/L. The patient had accidentally restarted treatment with posaconazole. A morning cortisol level was checked during this visit, which was on the upper limit of normal at 20.2 µg/dL (normal range, 10-20 µg/dL). Treatment with posaconazole was discontinued, and the patient’s potassium level normalized with replacement potassium in the outpatient infusion setting. When posaconazole serum concentrations were <1 mg/L approximately 2 weeks later, treatment with fluconazole was initiated for ongoing fungal prophylaxis. Our patient’s serum potassium levels remained within normal limits, with no further evidence of wasting in the absence of potassium supplementation.
Patient 2
The second patient, a 46-year-old woman with AML, had a disease relapse 20 years after having an ASCT. Her medical history was significant for mild systolic congestive heart failure, which was likely secondary to induction chemotherapy, and hypertension. At home, she was receiving posaconazole 300 mg daily and potassium chloride 40 mEq twice daily. During a hospital admission for neutropenic fever after a treatment cycle of decitabine and venetoclax, the patient had hypokalemia with a potassium level of 2.8 mmol/L. During her inpatient stay, potassium was replaced intravenously while she was receiving cardiac monitoring. The patient required a substantial amount of potassium replacement (a total of 180 mEq orally and 100 mEq intravenously on day 1, and 120 mEq orally and 120 mEq intravenously on day 2 of admission) and was discharged with a potassium level of 3.9 mmol/L. A posaconazole trough was drawn and was supratherapeutic at 7.5 mg/L.
After suspicion of posaconazole-related potassium wasting, a morning cortisol level was drawn, which was within the normal limits at 14.1 mcg/dL. Nonetheless, her 11-deoxycortisol level was elevated at 971 mg/L (normal range, ≤62 mg/L). A renin activity level was drawn and was low at 0.24 ng/mL/hour (normal range, 0.5-4 mg/mL/hr). An electrocardiogram revealed a normal QTcF calculated at 407 milliseconds. During admission, the patient’s blood pressure (BP) was consistently elevated, with her systolic BP reaching 150 to 159 mm Hg. Of note, spironolactone was our agent of choice to manage hypertension in this patient because of its potassium-sparing effect. On normalization of serum potassium levels, the patient was discharged with at-home potassium supplementation, spironolactone, and a reduced posaconazole dosage of 200 mg once daily. Two weeks later, a repeat posaconazole trough resulted at 1.9 mg/L; hence, the reduced dosage of 200 mg daily was continued. Our patient has not had hypokalemia to this degree since the posaconazole dosing was decreased, and she was able to be successfully weaned off of potassium supplementation and treatment with spironolactone.
Discussion
Invasive fungal infections are a major cause of illness and death in patients with myelosuppression in the setting of hematologic malignancy.5 As previously mentioned, posaconazole is often prescribed as an antifungal prophylaxis in patients after an SCT because of its wide spectrum of activity against mold species, such as Aspergillus. To date, a handful of case reports have shown that posaconazole can cause severe hypokalemia and hypertension.11,12,14 Posaconazole can inhibit 11ß-hydroxysteroid dehydrogenase type 2 and 11ß-hydroxylase.15,16 The inhibition of 11ß-hydroxysteroid dehydrogenase type 2 by azole antifungals is a key component of their therapeutic mechanism, which leads to reduced integrity of the fungal cell membrane.15 This same enzyme also protects mineralocorticoid receptors from exorbitant amounts of cortisol by catalyzing the conversion of active cortisol to inert cortisone. When azole antifungals inhibit this enzyme, cortisol levels increase, leading to the activation of mineralocorticoid receptors. This stimulation of mineralocorticoid receptors is aldosterone-independent and is known as posaconazole-induced pseudohyperaldosteronism (PIPH). PIPH leads to increased absorption of renal sodium, which causes water retention, volume expansion, and activation of the RAS.15
The RAS tightly controls adrenal aldosterone secretion.15 Excessive secretion of aldosterone results in increased BP and potassium wasting. The production of aldosterone and renin is controlled by a negative feedback loop, which leads to decreased levels of both hormones.15 This may explain why our second patient had a slightly reduced renin activity level. When we considered starting an agent to manage hypertension in the second patient, we chose spironolactone because of its mechanism of action as an aldosterone antagonist. We theorized that spironolactone would help to manage excess aldosterone and potassium wasting.
Posaconazole can also inhibit 11ß-hydroxylase, the enzyme that endogenously converts deoxycorticosterone to corticosterone and 11-deoxycortisol to cortisol.16 The inhibition of 11ß-hydroxylase leads to the accumulation of 11-deoxycortisol and deoxycorticosterone, which can directly activate mineralocorticoid receptors.16 This might explain why an elevated 11-deoxycortisol level was observed in our second patient. It is difficult to interpret cortisol levels in this setting, because posaconazole’s inhibition of 11ß-hydroxysteroid dehydrogenase type 2 is expected to increase endogenous cortisol levels, whereas posaconazole’s inhibition of 11ß-hydroxylase is expected to decrease cortisol levels.16 Both patients had normal cortisol levels, although the cortisol level in our first patient was on the upper limit of normal.
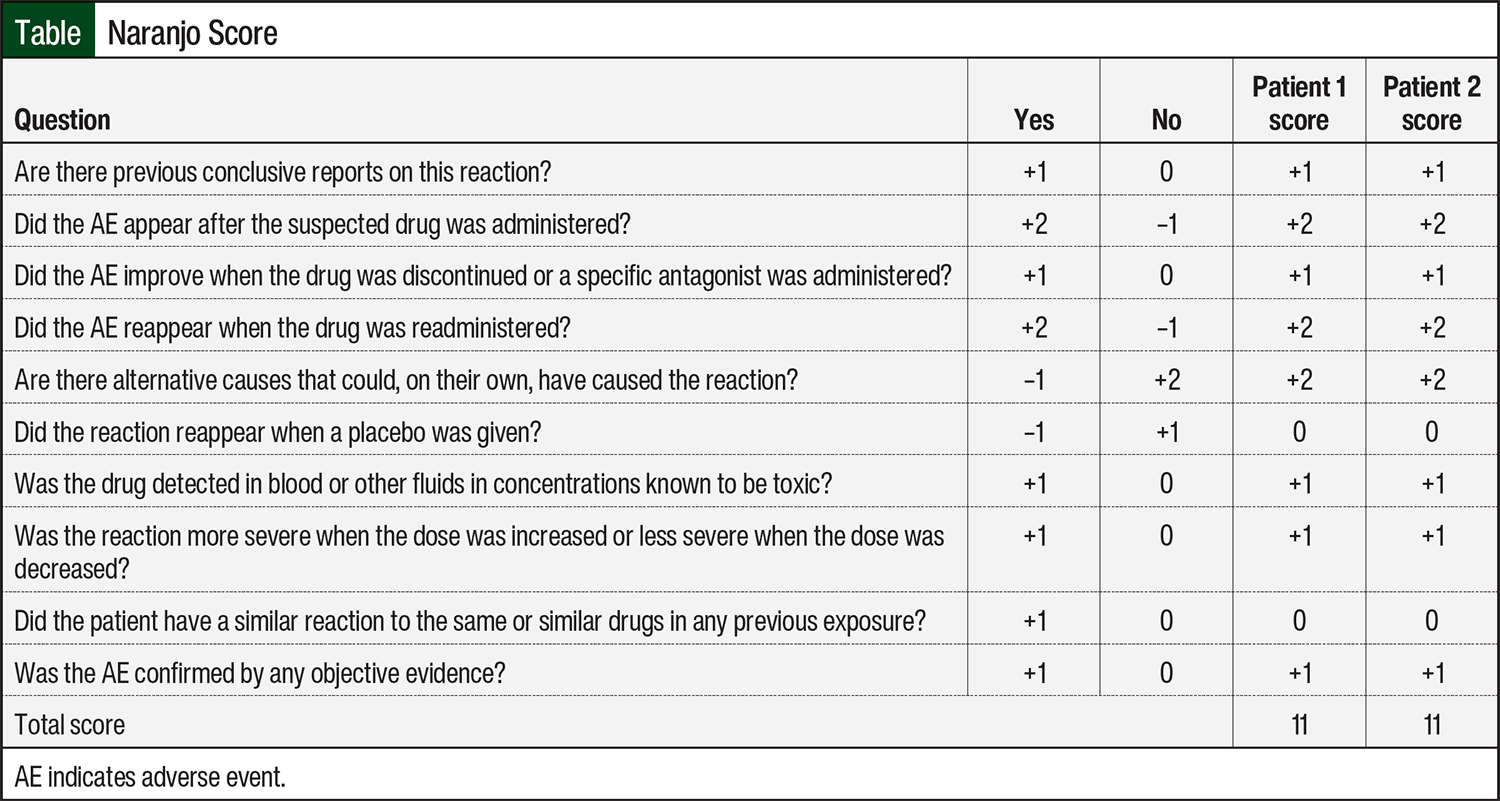
The Naranjo Scale is a useful tool to estimate the probability of adverse drug reactions.17 Naranjo scores range from –4 to +13, and a score of ≥9 indicates a causative relationship between medication and AE.17 Both of our patients had a score of 11, indicating a definite drug-related AE (Table). Both patients presented here had severe hypokalemia and supratherapeutic posaconazole levels. Once posaconazole was discontinued or dose-decreased, the hypokalemia resolved.
In the first patient, the choice was made to switch to a different azole antifungal because of QTc prolongation and the accompanying diarrhea. Although a mold-covering agent, such as voriconazole, was preferred considering her history of SCT, voriconazole was cost-prohibitive. Isavuconazole and itraconazole were also considered, although ultimately fluconazole was chosen because of lower rates of hypokalemia and mineralocorticoid receptor activation.15 In our second patient, reducing the posaconazole dosage from 300 mg to 200 mg daily led to the resolution of her hypokalemia, and a subsequent posaconazole trough was within the desired range of 0.7 to 3.75 mg/L.
It is unclear why both of our patients had supratherapeutic posaconazole levels on a standard daily dosage of 300 mg. Neither patient was concurrently receiving P-glycoprotein–inhibiting therapies that could have increased posaconazole exposure, and both patients had normal hepatic function. Posaconazole’s metabolism is predominately mediated through glucuronidation; hence, individual genetic variability in uridine diphosphate glucuronosyltransferase enzymatic activity may be a factor contributing to variable serum concentrations of the patients18,19; this has yet to be characterized for posaconazole specifically.
Conclusion
We summarized 2 cases of severe hypokalemia associated with treatment with posaconazole at the standard dosage of 300 mg daily after having SCT. Clinicians should consider PIPH as a differential diagnosis in patients who present with hypokalemia, hypertension, diarrhea, and supratherapeutic posaconazole levels. Clinicians may also consider ordering cortisol levels, 11-deoxycortisol levels, corticosterone levels, and renin activity levels in addition to a posaconazole trough to aid in the recognition of PIPH. In the event that PIPH is suspected, it is reasonable to reduce the dosage of posaconazole to 100 to 200 mg daily or to pursue an alternative azole antifungal.
Based on our experience, we recommend that posaconazole dose reductions be followed up with a repeat trough level to ensure that it remains at ≥0.7 mg/L for therapeutic activity. The use of an aldosterone antagonist, such as spironolactone or eplerenone, may also help rapidly resolve hypokalemia and hypertension. To our knowledge, this is the only report that focuses on PIPH in patients with hematologic malignancies who have had an SCT.
Author Disclosure Statement
Dr Altmaier, Dr Komerdelj, Dr De Padova, Dr Heers, Ms Davis, Ms O’Riley, and Dr Smith have no conflicts of interest to report.
References
- Hof H. A new, broad-spectrum azole antifungal: posaconazole—mechanisms of action and resistance, spectrum of activity. Mycoses. 2006;49(Suppl 1):2-6.
- Cornely OA, Maertens J, Winston DJ, et al. Posaconazole vs. fluconazole or itraconazole prophylaxis in patients with neutropenia. N Engl J Med. 2007;356:348-359.
- Viscoli C, Girmenia C, Marinus A, et al. Candidemia in cancer patients: a prospective, multicenter surveillance study by the Invasive Fungal Infection Group (IFIG) of the European Organization for Research and Treatment of Cancer (EORTC). Clin Infect Dis. 1999;28:1071-1079.
- Denning DW, Marinus A, Cohen J, et al. An EORTC multicenter prospective survey of invasive aspergillosis in hematological patients: diagnosis and therapeutic outcome. J Infect. 1998;37:173-180.
- Bow EJ, Loewen R, Cheang MS, Schacter B. Invasive fungal disease in adults undergoing remission-induction therapy for acute myeloid leukemia: the pathogenetic role of the antileukemic regimen. Clin Infect Dis. 1995;21:361-369.
- National Comprehensive Care Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): prevention and treatment of cancer-related infections. Version 3.2024. September 23, 2004. Accessed December 6, 2024. www.nccn.org/professionals/physician_gls/pdf/infections.pdf
- Noxafil (posaconazole) injection, for intravenous use [prescribing information]. Merck; January 2022. Accessed December 6, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2022/022003Orig1s027lbl.pdf
- Dekkers BGJ, Bakker M, van der Elst KCM, et al. Therapeutic drug monitoring of posaconazole: an update. Curr Fungal Infect Rep. 2016;10:51-61.
- Jang SH, Colangelo PM, Gobburu JVS. Exposure-response of posaconazole used for prophylaxis against invasive fungal infections: evaluating the need to adjust doses based on drug concentrations in plasma. Clin Pharmacol Ther. 2010;88:115-119.
- Märtson AG, Veringa A, van den Heuvel ER, et al. Posaconazole therapeutic drug monitoring in clinical practice and longitudinal analysis of the effect of routine laboratory measurements on posaconazole concentrations. Mycoses. 2019;62:698-705.
- Barton K, Davis TK, Marshall B, et al. Posaconazole-induced hypertension and hypokalemia due to inhibition of the 11β-hydroxylase enzyme. Clin Kidney J. 2018;11:691-693.
- Mahmood M, Abu Saleh O, Sohail MR. Hypokalemia and hypertension associated with supratherapeutic posaconazole levels. Antimicrob Agents Chemother. 2017;61:e00019-.
- Glotzbecker B, Duncan C, Alyea E, 3rd, Campbell B, Soiffer R. Important drug interactions in hematopoietic stem cell transplantation: what every physician should know. Biol Blood Marrow Transplant. 2012;18:989-1006.
- Hanna JJ, Guastadisegni JM, Kouma MA, et al. Blastomycosis presenting with acute airway obstruction from a retropharyngeal abscess and complicated by severe hypokalemia during posaconazole therapy: a case report and review of literature. Open Forum Infect Dis. 2022;9:ofac414.
- Ji HH, Tang XW, Zhang N, et al. Antifungal therapy with azoles induced the syndrome of acquired apparent mineralocorticoid excess: a literature and database analysis. Antimicrob Agents Chemother. 2022;66:e0166821.
- Davis MR, Nguyen MVH, Gintjee TJ, et al. Management of posaconazole-induced pseudohyperaldosteronism. J Antimicrob Chemother. 2020;75:3688-3693.
- Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30:239-245.
- Li Y, Theuretzbacher U, Clancy CJ, et al. Pharmacokinetic/pharmacodynamic profile of posaconazole. Clin Pharmacokinet. 2010;49:379-396.
- Guillemette C, Lévesque E, Harvey M, et al. UGT genomic diversity: beyond gene duplication. Drug Metab Rev. 2010;42:24-44.