Invasive fungal infections (IFIs) are a major cause of morbidity and mortality in patients with hematologic malignancies.1-3 Although the rates of IFIs are lower in patients with acute lymphoblastic leukemia (ALL) than in patients with other acute leukemias, those with ALL have prolonged neutropenia, receive corticosteroids, and can have mucositis, which increase the risks for IFIs and invasive candidiasis.1-5 According to the National Comprehensive Cancer Network (NCCN) guidelines, ALL is a malignancy that has a high overall infection risk6; however, there is a lack of studies that have examined the incidence of fungal infections in adults with ALL. Most published data that evaluate the incidence rate of IFIs in patients with ALL include pediatric populations or lack specificity between the different types of acute leukemia or hematologic malignancies.
The incidence rates of IFIs in pediatric patients with ALL are highly variable, ranging from 3% to 24%.7-13 A study in the United Kingdom that assessed the incidence of IFIs in pediatric patients and adolescents and young adults (AYAs) with ALL demonstrated incidence rates of 2.5% for proven IFIs and 1.5% for probable IFIs.8 These patients did not routinely receive antifungal prophylaxis.8 Little is known about the incidence of IFIs in adults with ALL.
Mariette and colleagues conducted a large retrospective study that evaluated the incidence of IFIs during induction treatment in 969 adults with ALL.1 The study’s results showed an incidence rate of 6.7% (n=65) for IFIs during induction chemotherapy. However, only patients receiving intense chemotherapy were included in the study.1 Many adults with ALL do not receive intense chemotherapy because of performance status and/or age14; therefore, the risk for IFIs in this population remains unstudied. No study has evaluated the incidence of IFIs across the continuum of adults and pediatric patients.
Little is known about the risk factors for IFIs in adults with ALL. In contrast, 2 studies of pediatric patients with ALL have identified higher rates of proven, probable, and possible IFIs in patients receiving more-intense chemotherapy regimens versus less-intense regimens.12,15 In the study by Wang and colleagues, pediatric patients with relapsed ALL had a higher risk for IFIs than patients without relapsed disease.15 Conflicting evidence has been published regarding age as a risk factor for IFIs. Some studies have shown higher rates of IFIs in younger patients, whereas other studies have shown higher rates in AYAs as a result of receiving increased cumulative steroid doses.7,8,12,15
Numerous host-related factors may increase the risk for IFIs, including other immunosuppressive causes (eg, autoimmune disease, asplenia or functional asplenia resulting from disorders such as sickle cell disease), anemia, chronic kidney disease, Down syndrome–related immune polymorphisms, diabetes, chronic obstructive pulmonary disease (COPD) and other structural lung diseases, total parenteral nutrition (TPN)/lipid administration, and smoking.16-22 Recent abdominal surgeries, hemodialysis, intensive care admission, and the presence of central venous catheters have also been attributed to an increased risk for IFIs.16,17,22,23 To our knowledge, there are limited-to-no data about the cumulative effect of these predisposing factors on the incidence of IFIs in patients with ALL.
Given the designation of high infection risk, the NCCN recommends considering antifungal prophylaxis with fluconazole or an echinocandin until the resolution of neutropenia in adults.6 Despite this recommendation, azole antifungals can inhibit the metabolism of vinca alkaloids that are frequently used for the treatment of ALL through the inhibition of CYP3A4, leading to an increased risk for adverse events and delays in treatment.6,24 As a result of this drug–drug interaction, prophylactic antifungals are not routinely administered to adults with ALL in clinical practice.1,25 This practice varies from other hematologic malignancies, such as acute myeloid leukemia, for which antifungal prophylaxis is the standard of care.2-4
Similar to treatment in adults, the use of antifungal prophylaxis in pediatric and AYA patients with a mold-active azole or echinocandin is recommended per the Children’s Oncology Group (COG) guidelines, but use in clinical practice depends on patient- and center-specific preferences and practices rather than guideline-directed therapy.26 When azole antifungals are used for prophylaxis in pediatric patients, evidence shows an increased risk for neurotoxicity with vinca alkaloids.24,26,27 For this reason, the use of azole antifungals in pediatric patients with ALL is limited.
The purpose of this study was to determine the incidence rate of probable and proven IFIs in patients who are receiving chemotherapy across the continuum of adults and pediatric patients with ALL at a single center.
Methods
This study received a determination of exempt status by the institutional review board. All patients with a diagnosis code of C91.0, indicating ALL and chemotherapy received at our institution between July 1, 2014, and June 1, 2019, were included in the study. Patients who received previous hematopoietic stem-cell transplant (HSCT) or chimeric antigen receptor T-cell (CAR-T) therapy were excluded from the study; however, if a patient received HSCT or CAR-T therapy during the study period, they could be included in the study until they had a transplant or until the start of therapy. No data were collected after having a transplant or after receiving CAR-T therapy. In addition, patients who received antifungal prophylaxis during the study period were excluded from the study. Data collection was terminated the day before the relapse of ALL, and a new record was created to characterize the risk for IFIs and the outcomes at that time.
The primary end point was the incidence of IFIs in adults and pediatric patients with ALL who were receiving intense or nonintense chemotherapy. The secondary end points included the incidence of invasive candidiasis, IFIs in patients receiving intense chemotherapy, IFIs in patients receiving nonintense chemotherapy, IFIs based on disease status, and IFIs while receiving induction, consolidation, and maintenance chemotherapies.
To observe the impact of additional risk factors and any trends in IFIs, we collected information regarding several comorbidities or predisposing factors that have been associated with IFIs. The information we collected included autoimmune diseases, anemia, sickle cell disease, asplenia, Down syndrome, diabetes, COPD, TPN/lipid administration, and smoking history.
The age ranges of the pediatric and AYA patients with ALL overlap, and some studies of pediatric patients have included those aged up to 18 years, whereas the NCCN has defined AYAs as patients aged 15 to 39 years. Thus, we used our institutional practices to classify patients aged <18 years as pediatric patients, aged 18 to 39 years as AYAs, and aged ≥40 years as adults.
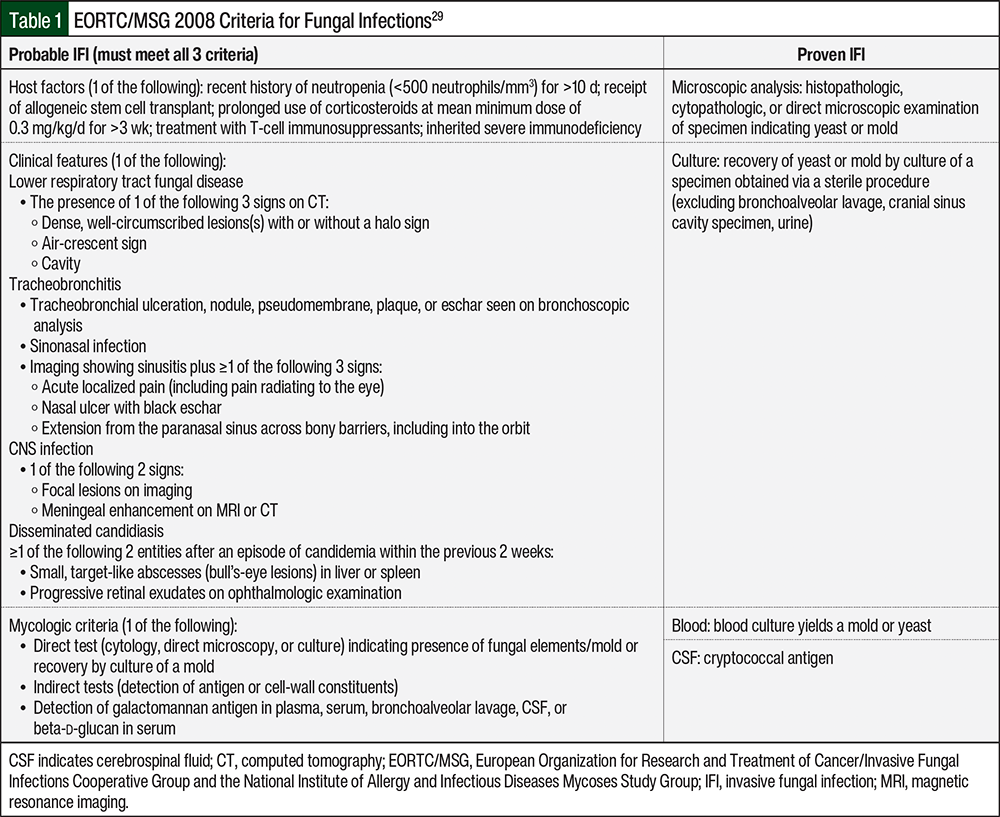
Probable and proven fungal infections were defined by the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) 2008 criteria (Table 1).29 For probable fungal infections, the clinical features were established from the chest radiograph and were confirmed by a physician. In cases when documentation was unclear, the study team adjudicated each case and worked to develop a consensus. IFIs diagnosed after the completion of chemotherapy were not included in the analysis because of the low risk for IFIs after chemotherapy, and because previous studies only assessed IFIs during induction.1,11 Possible fungal infection was defined as any patient who did not meet the definitions of probable or proven IFIs, but who received treatment for presumed fungal infection during the study period.
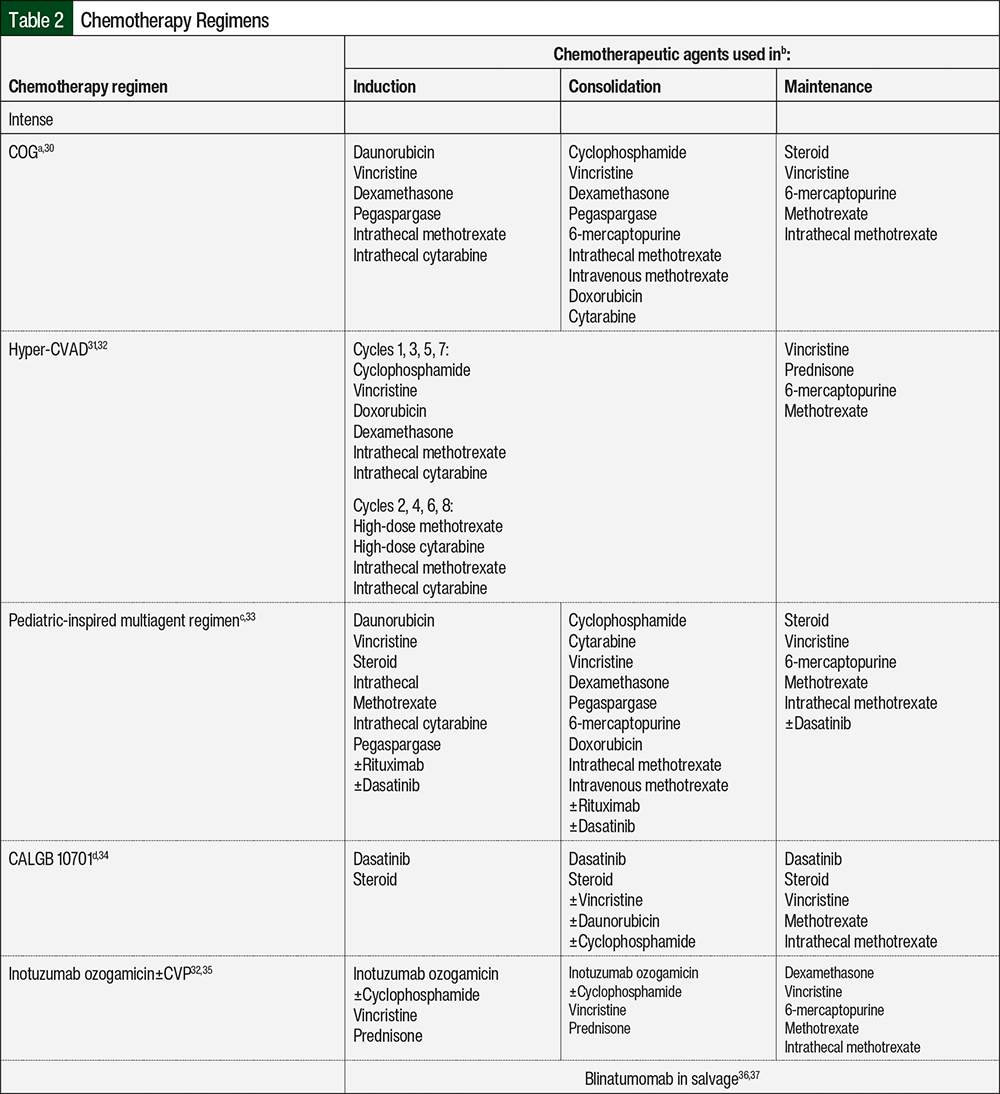
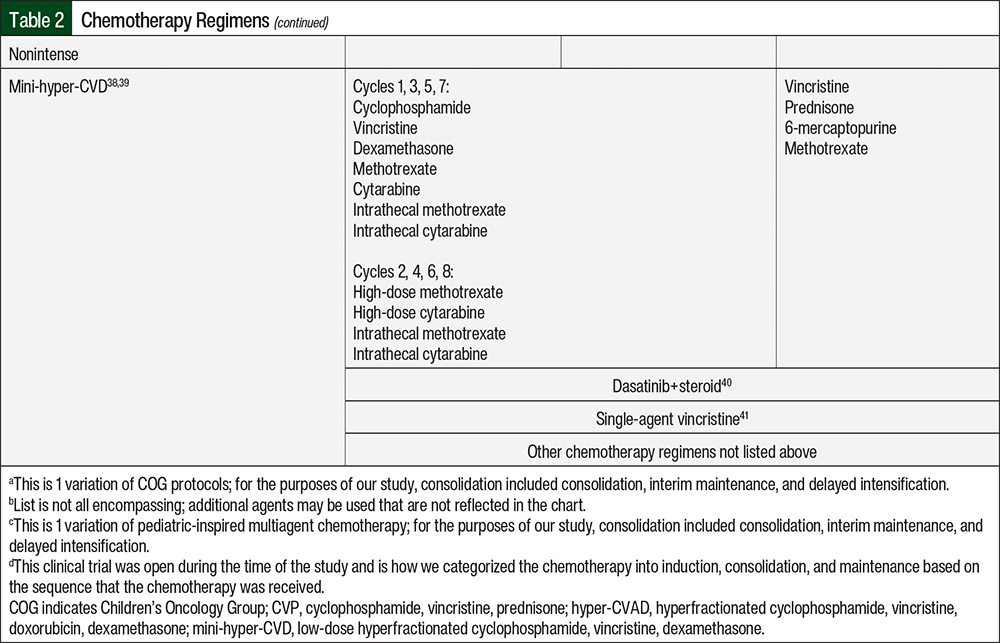
The regimens that were classified as intense chemotherapy included the COG studies; hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone (hyper-CVAD)/high-dose methotrexate plus high-dose cytarabine; pediatric-inspired multiagent regimens; inotuzumab ozogamicin with or without cyclophosphamide, vincristine, and prednisone; and blinatumomab in the salvage setting. The regimens that were classified as nonintense chemotherapy included low-dose hyperfractionated cyclophosphamide, vincristine, and dexamethasone (mini-hyper-CVD); dasatinib plus dexamethasone or prednisone; single-agent vincristine; and any other regimen that was not listed. The chemotherapy regimens included in the study are further defined in Table 2.30-41
Statistical Analysis
The patients’ characteristics were summarized using counts and proportions for the categorical variables, and medians and interquartile ranges for the continuous variables. The incidence of IFIs was calculated as the number of diagnosed IFIs divided by the number of person-years at risk to account for the varying lengths of follow-up. Person-years were defined as the time from a patient’s initiation of chemotherapy until the discontinuation of chemotherapy, transplant, death, or the end of follow-up. The incidence rates are presented with associated two-sided 95% exact Poisson confidence intervals (CIs). All analyses were descriptive in nature, and no hypothesis tests were performed because of the small event counts. SAS 9.4 (SAS Institute, Inc; Cary, NC) was used for all analyses.
Results
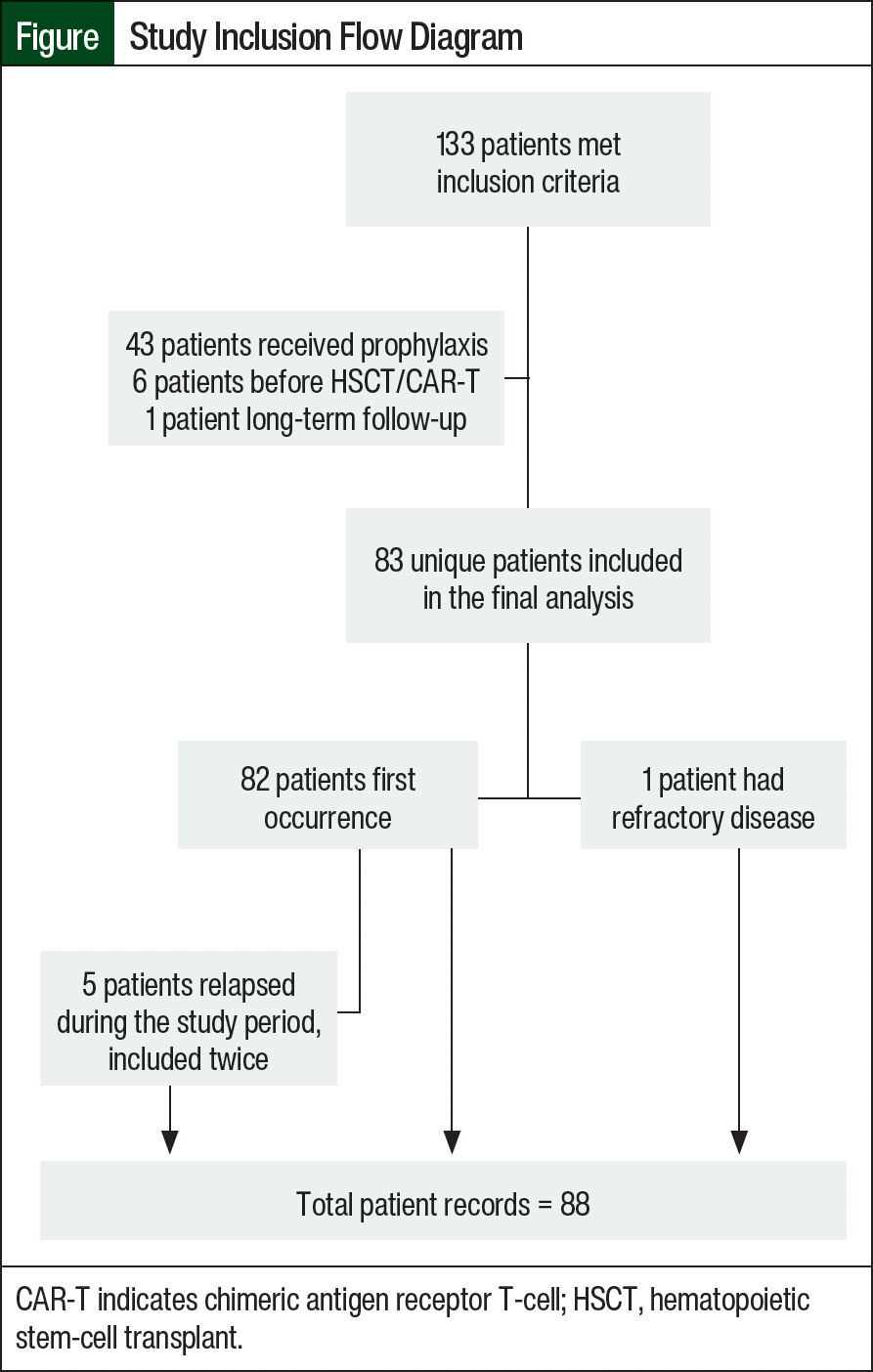
A total of 133 patients met our study inclusion criteria, of whom 43 (32%) were excluded from the study because they received antifungal prophylaxis. The study exclusions are further outlined in the Figure. A total of 88 patient records were included in the final analysis, with 82 (93%) initial treatments, 1 patient with refractory disease at the time of study initiation, and 5 patients who relapsed during the study (relapsed or refractory ALL, 7%). In all, 30 (34.1%) of the 83 patients were adults, 26 (29.6%) were AYA, and 32 (36.4%) were pediatric patients. The average age of the patients was 26 years (interquartile ratio, 13-46), and 64% of the patients were male.
In terms of ALL subtypes, 64% of the patients had Philadelphia chromosome (Ph)-negative B-cell ALL, 24% had Ph-positive B-cell ALL, and 13% had T-cell ALL. The records were reviewed during each phase of chemotherapy and included 68 records evaluated during induction, 57 during consolidation, and 62 during maintenance. Most patients received intense chemotherapy regimens, including 77 (88%) patients in any phase of chemotherapy and 57 (84%) patients in the induction phase.
Overall, patients had few comorbidities. The most common comorbidity was anemia in 8% of patients, and 6% of patients had diabetes, 2% had comorbid autoimmune disease, and 1% had asplenia, Down syndrome, or COPD.
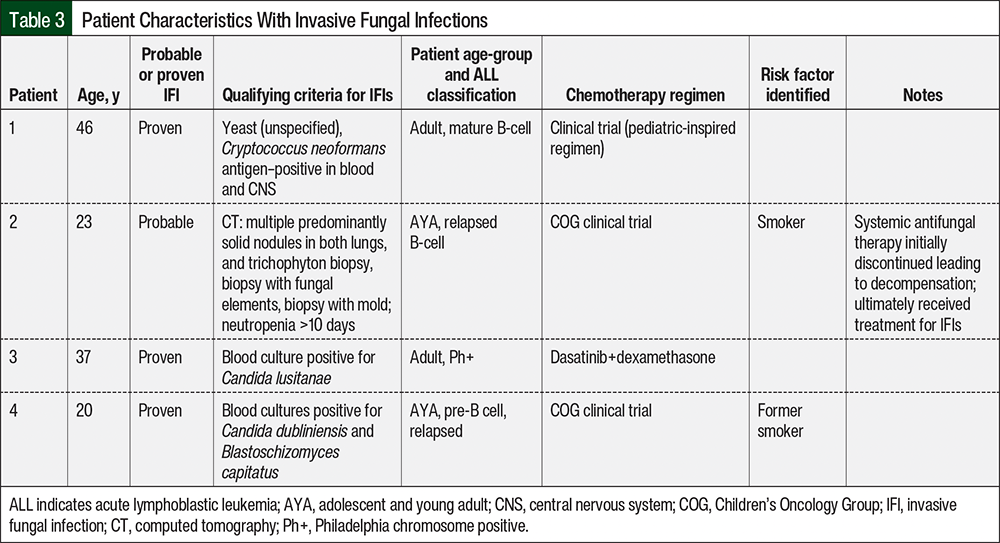
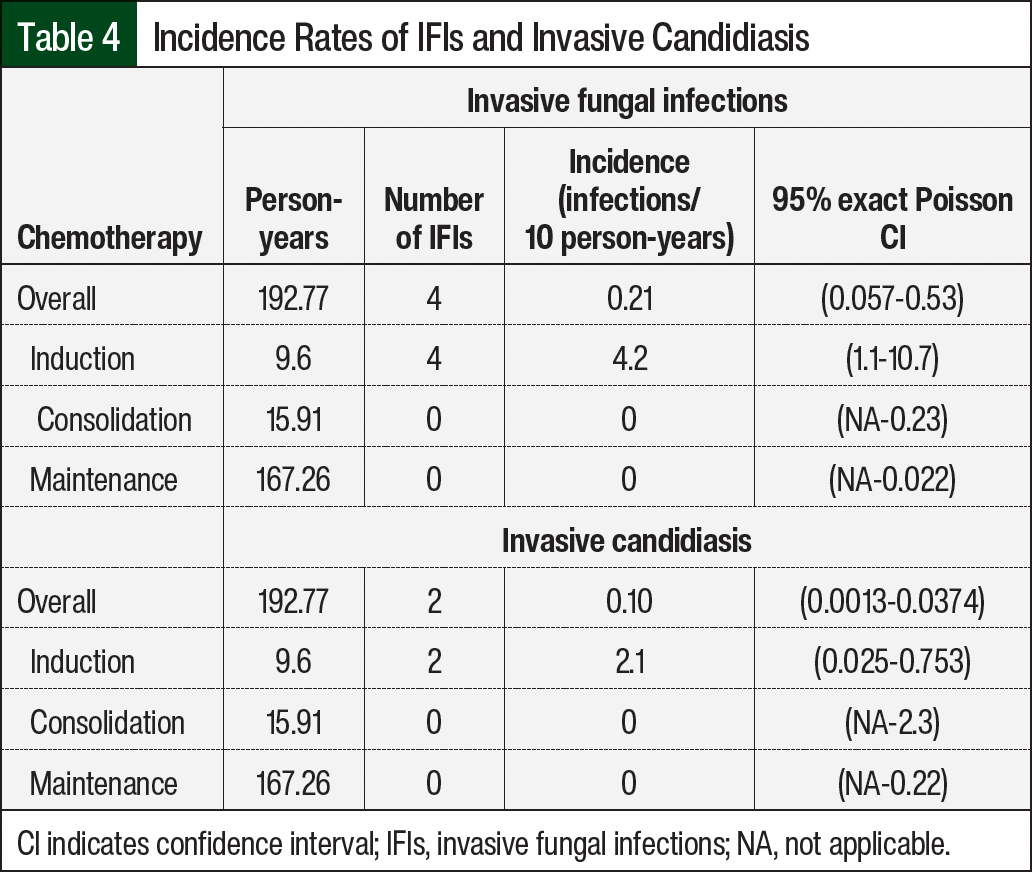
We collected 192.77 person-years of follow-up on 88 occurrences of ALL. Of the 88 records reviewed, 4 adults or AYAs had an IFI; no pediatric patients had probable or proven IFIs. The details on each individual case are shown in Table 3. A total of 3 cases were classified as a proven IFI and 1 IFI was classified as probable. All IFI events occurred during induction chemotherapy. The incidence of any fungal infection was 0.21 per 10 person-years (Table 4).
Two patients with proven invasive fungal infections had invasive candidiasis (Table 4). Both patients had candidemia. The incidence of invasive candidiasis during induction therapy was 2.1 infections per 10 patient-years. The non-Candida organisms that were identified included Cryptococcus neoformans, Blastoschizomyces capitatus, and Trichophyton spp. (Table 3).
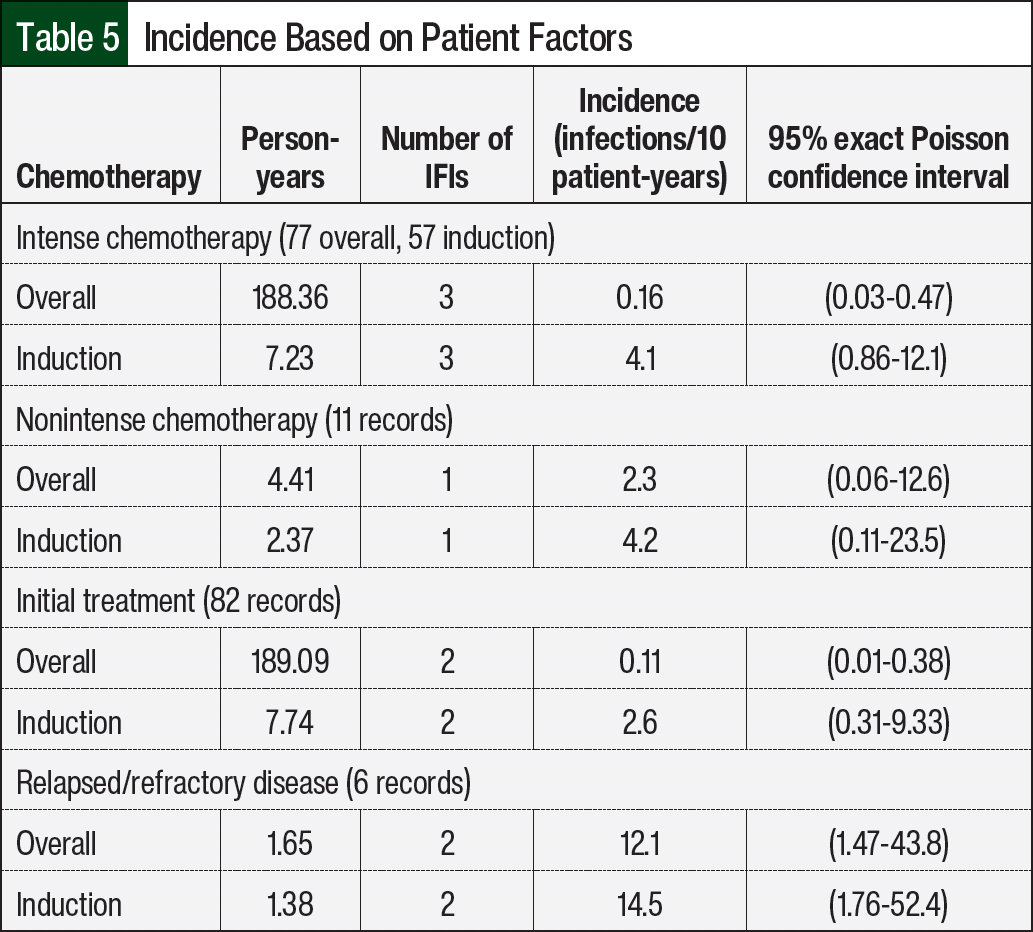
Despite most (84%) patients receiving intense chemotherapy during induction, the incidences of IFIs during induction were similar between the 3 patients who received intense chemotherapy and the 1 patient who received nonintense chemotherapy (incidence, 4.1 vs 4.2 per 10 person-years, respectively; Table 5). In contrast, when observing the overall cohort in any phase of treatment, the incidence was 0.16 infections per 10 person-years (95% CI, 0.03-0.47) in those who received intense chemotherapy and 2.3 infections per 10 person-years (95% CI, 0.06-12.6) in those who received nonintense chemotherapy.
A total of 2 IFIs occurred during initial treatment for ALL, and 2 IFIs occurred in patients who received treatment for relapsed ALL. We observed higher incidences of IFIs in the group with relapsed or refractory ALL than in those with treatment-naïve ALL (12.1 vs 0.11 infections per 10 patient-years overall, respectively; Table 5). No formal comparison was made because of the small event counts.
Regarding the organisms involved in IFIs (Table 3), we identified 2 cases of invasive candidiasis (Patients 3 and 4) that were confirmed by positive blood cultures. One patient (Patient 4) also had a positive blood culture for Blastoschizomyces capitatus. Patient 1 had a positive Cryptococcus neoformans antigen in the blood and central nervous system. Patient 2 had neutropenia and had radiographic imaging suggestive of fungal infection; however, no organism was identified from respiratory or blood cultures. In addition, galactomannan antigen and beta-d-glucan were negative in the serum. Patient 2 also had a concomitant disseminated rash that was positive for Trichophyton spp. elements on punch biopsy. Additional biopsies revealed fungal elements and mold elements. Because this is typically a noninvasive organism, systemic antifungal therapy was discontinued and the patient subsequently decompensated. Consequently, the patient received broad-spectrum antifungals for presumed invasive Trichophyton spp. infection and improved. Although the positive mycologic criteria were not from a respiratory source, given the positive elements on biopsy and the presumed invasive Trichophyton spp. infection, Patient 2 was considered to have probable IFI based on the clinical course and EORTC/MSG 2008 criteria.
None of the patients who had an IFI had any relevant comorbidities or TPN/lipid administration. However, 2 of the patients who had an IFI had a smoking history (Table 3).
Discussion
Overall, the rates of IFI were low in patients who received chemotherapy for ALL in the absence of antifungal prophylaxis. Our study identified only 4 IFIs. Of the 4 IFIs that were identified, all were diagnosed in AYAs or adults. The results of our study are consistent with the results of a similar study that showed low rates of IFIs in adults with ALL receiving induction chemotherapy.1 Unlike the study by Mariette and colleagues,1 our study was not restricted to only the induction phase; it also included patients in the consolidation and maintenance phases of treatment. Despite this, all patients had an IFI during induction, suggesting that the risk for IFI is highest during induction chemotherapy. It is important to note that our study also identified a high incidence of IFIs for patients with relapsed or refractory ALL. This is expected, because these patients typically receive intense chemotherapy and have prolonged durations of neutropenia. Also significant is that the sample size for our subgroup with relapsed or refractory ALL was small, and further studies must be completed to validate these results.
The overall incidences of IFIs in our study were similar between the intense and nonintense chemotherapy subgroups. This is important in that no previous study in adults has evaluated the risk for IFIs based on the intensity of chemotherapy. It might be expected that patients receiving more intense chemotherapy will have a greater risk for IFIs given the higher doses of steroids they receive and the longer durations of neutropenia. In comparison with the overall cohort, the nonintense chemotherapy group had a numerically higher incidence of IFIs (0.16 infections per 10 person-years, 95% CI, 0.03-0.47 vs 2.3 infections per 10 person-years, 95% CI, 0.06-12.6), but no formal comparisons were made because of the limited sample size in this group. Therefore, the interpretation of this outcome is limited. Given the small sample size, we can only theorize, but not draw any strong conclusions, about these data.
Although we did not collect data on all possible predisposing factors for IFIs, we did collect data on several clinically relevant risk factors. Two of the 4 patients who had an IFI had a smoking history, suggesting the risk for IFIs may be higher in patients who smoke. A large meta-analysis of 25 studies showed that smoking significantly increased the risk for invasive fungal disease compared with nonsmokers, regardless of the baseline characteristics, including immunosuppression.20 Given the increased risk for IFIs established in the literature and our findings, smoking cessation strategies should be considered in patients with ALL.
None of the pediatric patients in our study had an IFI. Although the incidence and prevalence of IFIs in pediatric patients have varied widely in previous literature, our data demonstrate a lower incidence than previous studies.7,8,12,15 This low incidence may be a result of our study’s small sample size of 32 pediatric patients and 26 AYAs, which is considerably smaller than other pediatric studies.7-13 However, the results of 2 small studies showed high rates of IFIs, with incidence rates of >20%.7,10 Sahbudak Bal and colleagues conducted a study of 125 pediatric patients with ALL who had an incidence rate of IFIs of 24%.7 Similarly, Hale and colleagues conducted a study of 113 pediatric and adolescent patients with ALL who had an incidence rate of IFIs of 21%.7,10 Of note, the studies conducted by Sahbudak Bal and colleagues (Turkey) and Hale and colleagues (Australia) were not conducted within the United States.7,10 The endemicity of fungal disease can vary based on geographic location, which can make comparing the incidence of IFIs between studies difficult. Based on the results of our study, it is unclear if pediatric patients have a lower risk for IFIs than AYAs and adults.
Another consideration when comparing our incidence rates to those from previous studies is the inclusion of possible infections in the definition of IFI. According to the EORTC/MSG criteria, possible fungal infections are defined as “cases that meet the criteria for a host factor and a clinical criterion but for which mycological criteria are absent.”29 Five studies conducted in pediatric and/or adolescent populations included possible infections.8-12 In our study, we identified an additional 3 patients who were not characterized as having proven or probable fungal infections, but who did receive antifungal therapy (pediatric patients, n=0; AYAs, n=2; adults, n=1). Although our results may have been different had we designed our study to include possible infections in our incidence rate, we chose to design our study to include strictly proven and probable infections in the definition of IFIs to prevent the inaccurate diagnosis of invasive fungal infections, and in alignment with the study design used by Mariette and colleagues in adults.1
The EORTC/MSG research consortium have long recognized the issues with and controversy surrounding possible IFI.29,42 In fact, shortly after we completed our study, the EORTC/MSG criteria revised its definition of IFI, diminishing the scope of the category of possible IFI.42 In addition, the new definition had more specific and expansive diagnostic criteria for probable IFI. The new definition added the requirement of a reverse halo sign for non-aspergillosis pulmonary fungal infection and removed the signs of disseminated candidiasis from the clinical criteria. The section on mycologic criteria was expanded to include new galactomannan antigen thresholds and removed beta-d-glucan.42 If we had used the 2020 EORTC/MSG criteria as opposed to the 2008 criteria, Patient 2 would not have met the definition for probable or even possible IFI, because the patient did not have a reverse halo sign on chest x-ray. Thus, the new definition would have impacted the results of our study.
Interestingly, we excluded 32% of patients with ALL because they received prophylaxis, which was higher than we anticipated. Although we did not collect any specific information about these patients, we suspect that this percent was driven by the pediatric cohort and/or likely consisted mainly of echinocandin prophylaxis to avoid an interaction with vincristine. Nevertheless, most patients did not receive prophylaxis, which is consistent with the standard clinical practice at our institution.
Limitations
This study has limitations, including its retrospective nature and small sample size. The study’s results rely on documentation in the electronic health record, which may be incomplete or missing key pieces of clinical information. Examination of the subgroups within this study, including nonintense chemotherapy and relapsed or refractory disease, are only exploratory because of the small sample size and imprecise results demonstrated by the wide CIs.
In addition, because standard clinical practice at our institution is not to use prophylaxis, our study did not include a comparator group, so a true estimation of risk reduction remains unknown. Although we did not make this comparison, our study represents the incidence of IFIs in standard clinical practice.
Conclusion
Despite prolonged neutropenia and overall high infection risk, many patients with ALL do not receive antifungal prophylaxis. The results of this single-center study indicate a low overall risk for IFIs in patients with ALL across the age spectrum, which suggests that, even in the absence of antifungal prophylaxis, patients do not have a high risk for IFIs.
Patients with relapsed or refractory ALL and those with a smoking history may have a higher risk for IFIs than patients without these risk factors. Because of the potential higher risk for IFIs in these subsets of patients with ALL in our study, future studies are warranted in these patient populations. In addition, further studies are necessary to determine if there is a benefit to antifungal prophylaxis in patients with ALL.
Acknowledgment
The authors would like to thank Juliette Miller for her assistance with data collection.
Author Disclosure Statement
Dr Martin, Dr Treptow, Dr Delibert, Dr Melaragno, Ms Baran, and Dr O’Dwyer have no conflicts of interest to report.
References
- Mariette C, Tavernier E, Hocquet D, et al. Epidemiology of invasive fungal infections during induction therapy in adults with acute lymphoblastic leukemia: a GRAALL-2005 study. Leuk Lymphoma. 2017;58:586-593.
- Lee C-H, Lin C, Ho C-L, Lin J-C. Primary fungal prophylaxis in hematological malignancy: a network meta-analysis of randomized controlled trials. Antimicrob Agents Chemother. 2018;62:e00355-18.
- Bhatt VR, Viola GM, Ferrajoli A. Invasive fungal infections in acute leukemia. Ther Adv Hematol. 2011;2:231-247.
- Lien M-Y, Chou C-H, Lin C-C, et al. Epidemiology and risk factors for invasive fungal infections during induction chemotherapy for newly diagnosed acute myeloid leukemia: a retrospective cohort study. PLoS One. 2018;13:e0197851.
- Oh S-M, Byun JM, Chang E, et al. Incidence of invasive fungal infection in acute lymphoblastic and acute myelogenous leukemia in the era of antimold prophylaxis. Sci Rep. 2021;11:22160.
- Baden LR, Bensinger W, Angarone M, et al. Prevention and treatment of cancer-related infections. J Natl Compr Canc Netw. 2012;10:1412-1445.
- Sahbudak Bal Z, Yilmaz Karapinar D, Karadas N, et al. Proven and probable invasive fungal infections in children with acute lymphoblastic leukaemia: results from a university hospital, 2005-2013. Mycoses. 2015;58:225-232.
- O’Reilly MA, Govender D, Kirkwood AA, et al. The incidence of invasive fungal infections in children, adolescents and young adults with acute lymphoblastic leukaemia/lymphoma treated with the UKALL2011 protocol: a multicentre retrospective study. Br J Haematol. 2019;186:327-329.
- Das A, Oberoi S, Trehan A, et al. Invasive fungal disease in pediatric acute leukemia in the nontransplant setting: 8 years’ experience from a tertiary care center in North India. J Pediatr Hematol Oncol. 2018;40:462-467.
- Hale KA, Shaw PJ, Dalla-Pozza L, et al. Epidemiology of paediatric invasive fungal infections and a case-control study of risk factors in acute leukaemia or post stem cell transplant. Br J Haematol. 2010;149:263-272.
- Mor M, Gilad G, Kornreich L, et al. Invasive fungal infections in pediatric oncology. Pediatr Blood Cancer. 2011;56:1092-1097.
- Mann G, Attarbaschi A, Schrappe M, et al. Invasive fungal infections in pediatric acute lymphoblastic leukemia: incidence, characterization, outcome and risk analysis of Study ALL-BFM 2000. Blood. 2014;124:3658-3658.
- Rosen GP, Nielsen K, Glenn S, et al. Invasive fungal infections in pediatric oncology patients: 11-year experience at a single institution. J Pediatr Hematol Oncol. 2005;27:135-140.
- Sawalha Y, Advani AS. Management of older adults with acute lymphoblastic leukemia: challenges & current approaches. Int J Hematol Oncol. 2018;7(1):Ijh02.
- Wang SS, Kotecha RS, Bernard A, et al. Invasive fungal infections in children with acute lymphoblastic leukaemia: results from four Australian centres, 2003-2013. Pediatr Blood Cancer. 2019;66:e27915.
- Nucci M, Anaissie E. How we treat invasive fungal diseases in patients with acute leukemia: the importance of an individualized approach. Blood. 2014;124:3858-3869.
- Anaissie E, Grazziutti M, Nucci M. Invasive fungal infections in cancer patients. In: Anaissie E, McGinnis M, Pfaller M, eds. Clinical Mycology. 2nd ed. Elsevier; 2009:431-471.
- Guinea J, Torres-Narbona M, Gijón P, et al. Pulmonary aspergillosis in patients with chronic obstructive pulmonary disease: incidence, risk factors, and outcome. Clin Microbiol Infect. 2010;16:870-877.
- Verweij PE, Kerremans JJ, Voss A, Meis JF. Fungal contamination of tobacco and marijuana. JAMA. 2000;284:2875.
- Pourbaix A, Lafont Rapnouil B, Guéry R, et al. Smoking as a risk factor of invasive fungal disease: systematic review and meta-analysis. Clin Infect Dis. 2020;71:1106-1119.
- Dare JM, Moppett JP, Shield JP, et al. The impact of hyperglycemia on risk of infection and early death during induction therapy for acute lymphoblastic leukemia (ALL). Pediatr Blood Cancer. 2013;60:E157-E159.
- Maertens J, Vrebos M, Boogaerts M. Assessing risk factors for systemic fungal infections. Eur J Cancer Care (Engl). 2001;10:56-62.
- Gamaletsou MN, Walsh TJ, Zaoutis T, et al. A prospective, cohort, multicentre study of candidaemia in hospitalized adult patients with haematological malignancies. Clin Microbiol Infect. 2014;20:O50-O57.
- Nikanjam M, Sun A, Albers M, et al. Vincristine-associated neuropathy with antifungal usage: a Kaiser Northern California experience. J Pediatr Hematol Oncol. 2018;40:e273-e277.
- Nicolato A, Nouér SA, Garnica M, et al. Invasive fungal diseases in patients with acute lymphoid leukemia. Leuk Lymphoma. 2016;57:2084-2089.
- Lehrnbecher T, Fisher BT, Phillips B, et al. Clinical practice guideline for systemic antifungal prophylaxis in pediatric patients with cancer and hematopoietic stem-cell transplantation recipients. J Clin Oncol. 2020;38(27):3205-3216.
- Teusink AC, Ragucci D, Shatat IF, Kalpatthi R. Potentiation of vincristine toxicity with concomitant fluconazole prophylaxis in children with acute lymphoblastic leukemia. Pediatr Hematol Oncol. 2012;29(1):62-67.
- Coccia PF, Pappo AS, Beaupin L, et al. Adolescent and young adult oncology, version 2.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16:66-97.
- De Pauw B, Walsh TJ, Donnelly JP, et al. Revised definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) Consensus Group. Clin Infect Dis. 2008;46:1813-1821.
- Maloney KW, Devidas M, Wang C, et al. Outcome in children with standard-risk B-cell acute lymphoblastic leukemia: results of Children’s Oncology Group trial AALL0331. J Clin Oncol. 2020;38:602-612.
- Thomas DA, O’Brien S, Cortes J, et al. Outcome with the hyper-CVAD regimens in lymphoblastic lymphoma. Blood. 2004;104:1624-1630.
- Kantarjian HM, O’Brien S, Smith TL, et al. Results of treatment with hyper-CVAD, a dose-intensive regimen, in adult acute lymphocytic leukemia. J Clin Oncol. 2000;18:547-561.
- Stock W, Luger SM, Advani AS, et al. A pediatric regimen for older adolescents and young adults with acute lymphoblastic leukemia: results of CALGB 10403. Blood. 2019;133:1548-1559.
- Wieduwilt MJ, Yin J, Wetzler M, et al. Dasatinib and dexamethasone followed by hematopoietic cell transplantation for adults with Ph-positive ALL. Blood Advances. 2021;5:4691-4700.
- Advani AS, Moseley A, Liedtke M, et al. SWOG 1312 final results: a phase 1 trial of inotuzumab in combination with CVP (cyclophosphamide, vincristine, prednisone) for relapsed/refractory CD22+ acute leukemia. Blood. 2019;134(suppl 1):227.
- Kantarjian H, Haddad FG, Jain N, et al. Results of salvage therapy with mini-hyper-CVD and inotuzumab ozogamicin with or without blinatumomab in pre-B acute lymphoblastic leukemia. J Hematol Oncol. 2023;16:44.
- Kantarjian H, Stein A, Gökbuget N, et al. Blinatumomab versus chemotherapy for advanced acute lymphoblastic leukemia. N Engl J Med. 2017;376:836-847.
- Jabbour E, Ravandi F, Kebriaei P, et al. Salvage chemoimmunotherapy with inotuzumab ozogamicin combined with mini-hyper-CVD for patients with relapsed or refractory Philadelphia chromosome-negative acute lymphoblastic leukemia: a phase 2 clinical trial. JAMA Oncol. 2018;4:230-234.
- Rausch CR, Jabbour EJ, Kantarjian HM, Kadia TM. Optimizing the use of the hyperCVAD regimen: clinical vignettes and practical management. Cancer. 2020;126:1152-1160.
- Foà R, Vitale A, Vignetti M, et al. Dasatinib as first-line treatment for adult patients with Philadelphia chromosome–positive acute lymphoblastic leukemia. Blood. 2011;118:6521-6528.
- Hardisty RM, McElwain TJ, Darby CW. Vincristine and prednisone for the induction of remissions in acute childhood leukaemia. Br Med J. 1969;2:662-665.
- Donnelly JP, Chen SC, Kauffman CA, et al. Revision and update of the consensus definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin Infect Dis. 2020;71:1367-1376.