Connecting Science to Practice
This article describes a pharmacist-driven outpatient oncology
workflow designed to improve the timely completion of
laboratory monitoring and supportive care prescribing for
patients with breast cancer receiving cyclin-dependent kinase
4/6 inhibitors. By developing standardized oral chemotherapy
treatment plans, the workflow ensures recommended
laboratory monitoring, and supportive care prescriptions are
incorporated to help prevent adverse events (AEs), prioritizing
patient safety and medication tolerability. By standardizing
supportive care prescriptions and laboratory monitoring
parameters at predefined frequencies, this workflow reduces
the need for pharmacists to reach out to the care team regarding
omissions of laboratory parameters or supportive
care prescriptions, streamlining the process. In addition, by
proactively managing AEs and improving tolerability, the
workflow has the potential to enhance patient adherence to
oral chemotherapy regimens. This approach supports improved
treatment outcomes and continuity of care in the
outpatient oncology setting with the goal of expansion to
other disease states.
Breast cancer remains the most frequently diagnosed cancer in women, with an estimated lifetime risk of 1 of 8 women having an invasive subtype of breast cancer.1 In 2021, 93.1 cases per 100,000 patients were diagnosed as hormone receptor (HR)-positive, HER2-negative breast cancer.2 This incidence was significantly higher than all other breast cancer subtypes, which had fewer than 15 cases per 100,000 patients.2 Although chemotherapy and radiation remain treatment options for HR-positive, HER2-negative breast cancer, the emergence of targeted therapies has expanded treatment options. Abemaciclib, palbociclib, and ribociclib are FDA-approved cyclin-dependent kinase (CDK)4/6 inhibitors indicated, in combination with an aromatase inhibitor, for the treatment of advanced or metastatic, HR-positive, HER2-negative breast cancer.3-5 These agents have become the preferred standard of care because of the prolonged survival benefit in patients with advanced or metastatic, HR-positive breast cancer. In addition, abemaciclib and ribociclib are indicated in the adjuvant setting in combination with endocrine therapy for adults with stage II or III, HR-positive, HER2-negative early breast cancer with a high risk for recurrence. In the adjuvant setting, both agents showed a significant improvement in invasive disease-free survival.6,7
Although CDK4/6 inhibitors have transformed the management of advanced and metastatic breast cancer, the various clinical trial results revealed various adverse events (AEs) that necessitate close monitoring of laboratory parameters to prevent treatment discontinuation.3-11 The common AEs include, but are not limited to, neutropenia, leukopenia, anemia, elevated liver enzymes, fatigue, nausea, vomiting, diarrhea, infection, arthralgias, and headache.3-5,12-14 To ensure the patient’s safety and tolerability, it is crucial that the recommended laboratory monitoring guidelines in the prescribing information are followed and that appropriate supportive care is prescribed to prevent AEs and ensure their early detection.
Abemaciclib’s prescribing information recommends monitoring a complete blood count (CBC) with differential and liver function tests at baseline, every 2 weeks for the first 2 months, monthly for the next 2 months, and then as clinically indicated.12 Palbociclib’s prescribing information recommends monitoring CBC with differential and liver function tests at baseline, every 2 weeks for the first 2 cycles, before each cycle, and then as clinically indicated.13 Ribociclib’s prescribing information recommends monitoring CBC with differential and liver function tests at baseline, every 2 weeks for the first 2 cycles, at the beginning of each subsequent 4 cycles, and then as clinically indicated. In addition, ribociclib had previous recommendations to complete electrocardiograms (EKGs) before initiation, on day 14 of the first cycle, before the start of the second cycle, and then as clinically indicated.14 In October 2024, ribociclib’s EKG monitoring parameters were updated to remove the recommendation to monitor with an EKG before the second treatment cycle.7,15
Although the recommended timing of monitoring varies across the 3 agents, ongoing monitoring is critical because of the risk for hematologic, hepatic, and cardiac AEs.12-14,16 Of the 3 agents, palbociclib carries the highest risk for hematologic AEs.16 According to a review of several trials by Lam and colleagues, in patients receiving palbociclib, the risk for neutropenia of any grade ranged from 78% to 84%.17 Of those patients, as many as 70% had grade 3 or 4 neutropenia. Similarly, the incidence of all-grade leukopenia in patients receiving palbociclib ranged from 39% to 60%, with up to 38% of patients having grade 3 or 4 leukopenia.17 For patients receiving abemaciclib, any-grade neutropenia occurred in 41.3% of patients, whereas grade ≥3 neutropenia occurred in 21.1%.5 For ribociclib, 74.3% of patients had any-grade neutropenia, with 49.7% having grade 3 neutropenia.4 Approximately 47% and 36%% of patients who received abemaciclib had any-grade alanine aminotransferase (ALT) and aspartate aminotransferase (AST) elevations, respectively.5 In addition, 7% of abemaciclib patients had ALT elevations and 3.8% had AST elevations that were grade ≥3.5 Ribociclib showed a similar incidence with 68% of any-grade liver enzyme elevations, and grade ≥3 ALT or AST elevations at 32% and 18%, respectively.8 Last, 52% of patients who received palbociclib had any-grade AST elevations and 43% had any-grade ALT elevations, with grade 3 ALT or AST elevations occurring in approximately 3% of patients.13
Ribociclib carries a warning for QTc prolongation that necessitates EKG monitoring, as outlined in the prescribing information.8,14 The results of the MONALEESA trials showed that up to 12.5% of patients receiving ribociclib had any-grade QTc prolongation.4,8-11 QT interval changes occurred within the first 4 weeks of treatment and were reversible with treatment interruption.4,8-11 Abemaciclib is associated with a significant incidence of gastrointestinal AEs, including nausea, vomiting, and diarrhea.16 The MONARCH 3 trial’s results show that 41.9% and 32.9% of patients receiving abemaciclib had any-grade nausea or vomiting, respectively.5 In addition, 81.3% of patients receiving abemaciclib had diarrhea, including 44.6% with grade 1 and 27.2% with grade 2.5 With the proper use of antiemetics, antidiarrheals, and dose adjustments for AEs, patients were able to manage their symptoms and continue with treatment. Overall, the safety profiles vary across the 3 CDK4/6 inhibitors and have the potential to negatively impact patients’ adherence if not addressed in a timely manner.
Ensuring adherence to oral chemotherapy treatment is essential for overall prognosis and survival. Interventions made by healthcare personnel, such as pharmacists, have positive impacts on patients’ outcomes and reduce AEs.18-21 Studies have indicated that electronic workflows for oral chemotherapy agents led by clinical pharmacists allowed for timely interventions, improved the tolerability of medications, and enhanced the documentation of interventions and medication errors.18,19 However, there is limited published research on the impact of pharmacists’ interventions and oral chemotherapy workflow implementation in outpatient oncology settings.
This study aimed to evaluate the outcomes after the implementation of pharmacist-driven oral chemotherapy treatment plans (OCTPs) within the outpatient oncology setting. OCTPs are prebuilt treatment plans developed by our institution’s clinical oncology pharmacists. The plans are added within a patient’s electronic health record (EHR) and include laboratory monitoring orders at defined frequencies based on prescribing information recommendations and supportive care prescriptions as indicated for nausea, vomiting, and diarrhea prevention when applicable. Patients are then educated on the AEs of the medication and the necessary monitoring parameters during teaching visits with nurses and clinical pharmacy specialists.
Once treatment has begun, nurses and advanced practitioners follow up to remind patients of the required laboratory monitoring, as well as to assess the patient’s tolerability and adherence. For eligible patients filling oral chemotherapy prescriptions through our specialty pharmacy, pharmacists review the required monitoring parameters before dispensing any first fill or refill. If laboratory testing is incomplete or if results are outside the acceptable range, a documented intervention is made and the oncology team is immediately consulted regarding the appropriateness of dispensing. OCTPs are designed to proactively incorporate necessary monitoring and supportive care at treatment initiation, thereby avoiding delays and reducing the need for pharmacist outreach to providers. These plans are key in preventing and managing AEs, thus potentially improving medication adherence and tolerability, which in return may reduce sick visits, emergency department visits, and hospitalizations.
The primary objective of our study was to assess the impact of pharmacist-driven OCTP implementation on the inclusion of appropriate supportive care prescriptions and laboratory monitoring for patients with breast cancer who were initiating CDK4/6 inhibitor therapy in the outpatient oncology setting, with a focus on evaluating the frequency of pharmacists’ interventions. The secondary objectives included comparing sick visits, hospitalizations, and adherence rates within the first 3 months of CDK4/6 therapy initiation between the pre- and post-OCTP cohorts.
Methods
We conducted this retrospective cohort study at our institution and its affiliated cancer institutes from June 1, 2022, to June 30, 2024. The research team identified patients with a breast cancer diagnosis if they received a new prescription for abemaciclib, palbociclib, or ribociclib from an internal provider and filled the prescription at our institution’s specialty pharmacy during the study period. We excluded patients who were aged <18 years, received concurrent intravenous chemotherapy, were prescribed a CDK4/6 inhibitor by an external provider, transferred care from an external pharmacy to the institution’s specialty pharmacy, or received a CDK4/6 inhibitor as part of an investigational trial. We identified 2 patient cohorts at the conclusion of the data collection. Patients who did not have an OCTP in their EHR were categorized as pre-OCTP, and patients who did have an OCTP in their EHR were categorized as post-OCTP.
We collected data by patient chart reviews at baseline and during the first 3 months of treatment initiation. We reviewed various platforms, including EHRs, medication dispensing systems, and specialty pharmacy patient management software. During the chart reviews, the data collected included medication dispense records from pharmacies; laboratory values, including CBCs with differential and complete metabolic panels; EKGs; discharge summaries; office visit notes; and documented pharmacist interventions in the EHR.
We reviewed laboratory monitoring frequency based on prescribing information recommendations. At the time of this study, our institution adhered to the previous ribociclib monitoring guidelines; thus, our data include an evaluation for EKG completion before the second cycle. Regarding supportive care, unlike abemaciclib and ribociclib, palbociclib does not mandate specific supportive care because of its lower risk for gastrointestinal AEs; therefore, this outcome was not evaluated in the patients receiving palbociclib.
A pharmacist intervention was defined as a clinical pharmacist contacting the oncology care team (oncologist, nurse practitioner, or physician assistant) to address or clarify issues related to the omission of any of the following: baseline laboratory tests, laboratory tests taken during treatment at the recommended intervals, antidiarrheal prescriptions, antiemetic prescriptions, or EKGs. Interventions were identified with the use of intervention documentation within the EHR and the specialty pharmacy patient management software. Descriptive statistics were used to report the primary and secondary outcomes as counts and percentages. This study was approved by the institutional review board.
Results
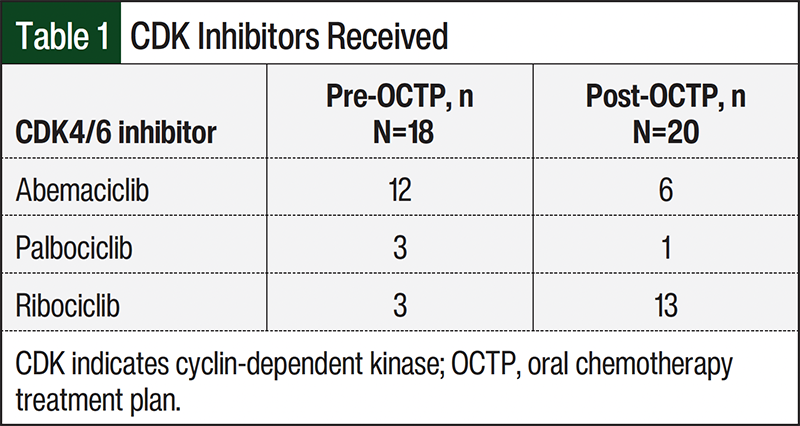
We identified 71 patients for the study, of whom 38 met the study’s inclusion criteria and 33 were excluded from the study. The excluded patients included 15 who transferred care from an external specialty pharmacy (limiting follow-up during cycles 1-3), 16 who started therapy outside the study window or who did not complete 3 treatment cycles, and 2 patients with ovarian cancer, which is not an FDA-approved indication for these agents. After the patients were excluded from the study, 18 patients receiving abemaciclib, 16 patients receiving ribociclib, and 4 patients receiving palbociclib were included in the study. The breakdown of the study patients is shown in Table 1.
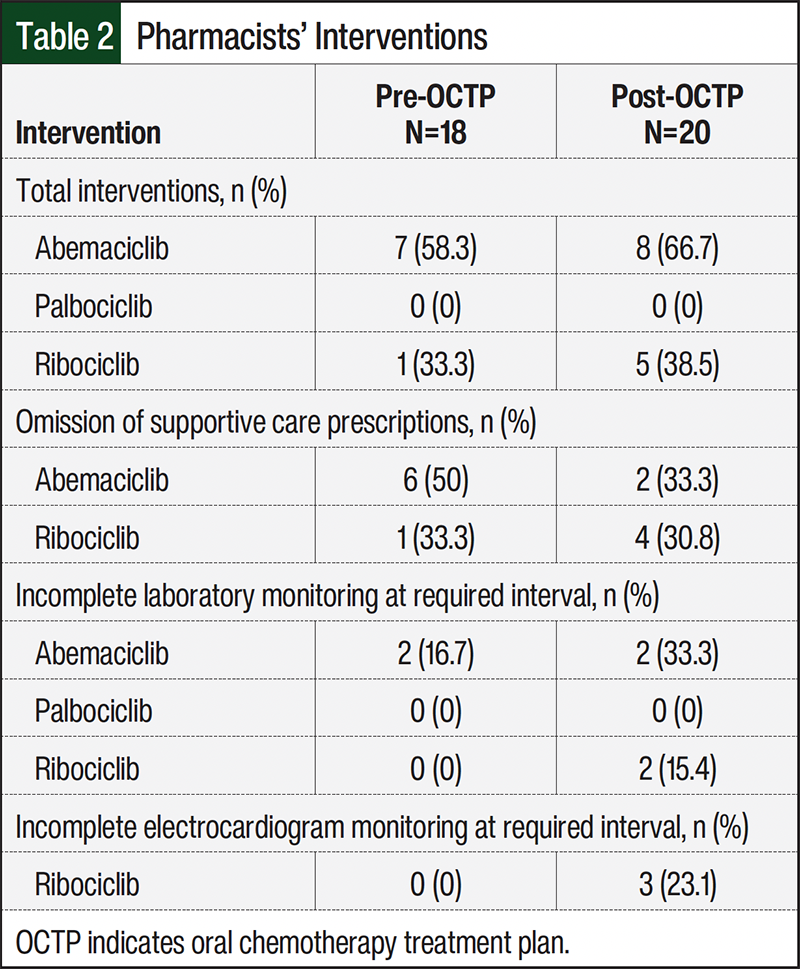
The results of our study reveal a higher percentage of pharmacist interventions in the post-OCTP cohort than in the pre-OCTP cohort for patients receiving abemaciclib (66.7% vs 58.3%, respectively) and ribociclib (38.5% vs 33.3%, respectively). None of the patients in either palbociclib cohort required any pharmacist interventions. Of the interventions we identified, most were related to the omission of supportive care prescriptions. For the omission of antiemetic prescriptions, 4 patients in the post-OCTP ribociclib cohort required interventions compared with 1 patient in the pre-OCTP ribociclib cohort. Two patients in each abemaciclib cohort (pre- and post-OCTP) also required such interventions. Table 2 provides a complete breakdown of the interventions.
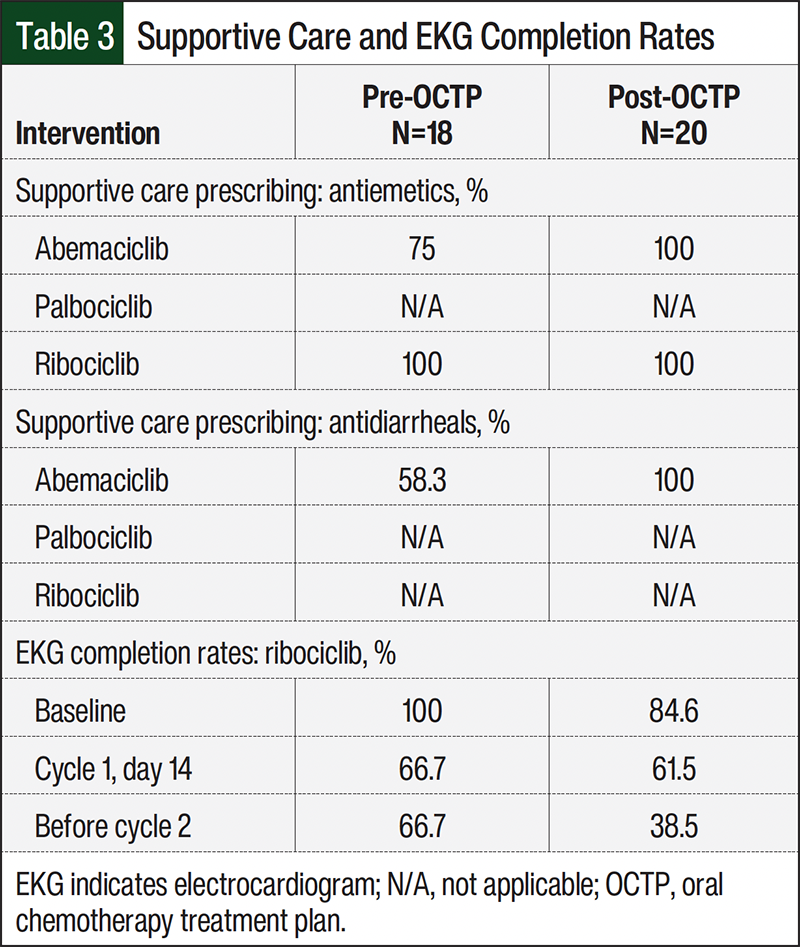
For patients receiving abemaciclib, there was a higher percentage of supportive care medications prescribed at baseline in the post-OCTP cohort than in the pre-OCTP cohort (Table 3). Of the patients receiving abemaciclib in the pre-OCTP cohort, 75% had an antiemetic medication prescribed at treatment initiation and 58.3% had an antidiarrheal medication prescribed at treatment initiation compared with 100% of patients in the post-OCTP abemaciclib cohort. All of the patients in the pre- and post-OCTP ribociclib cohorts were prescribed antiemetic medications at treatment initiation.
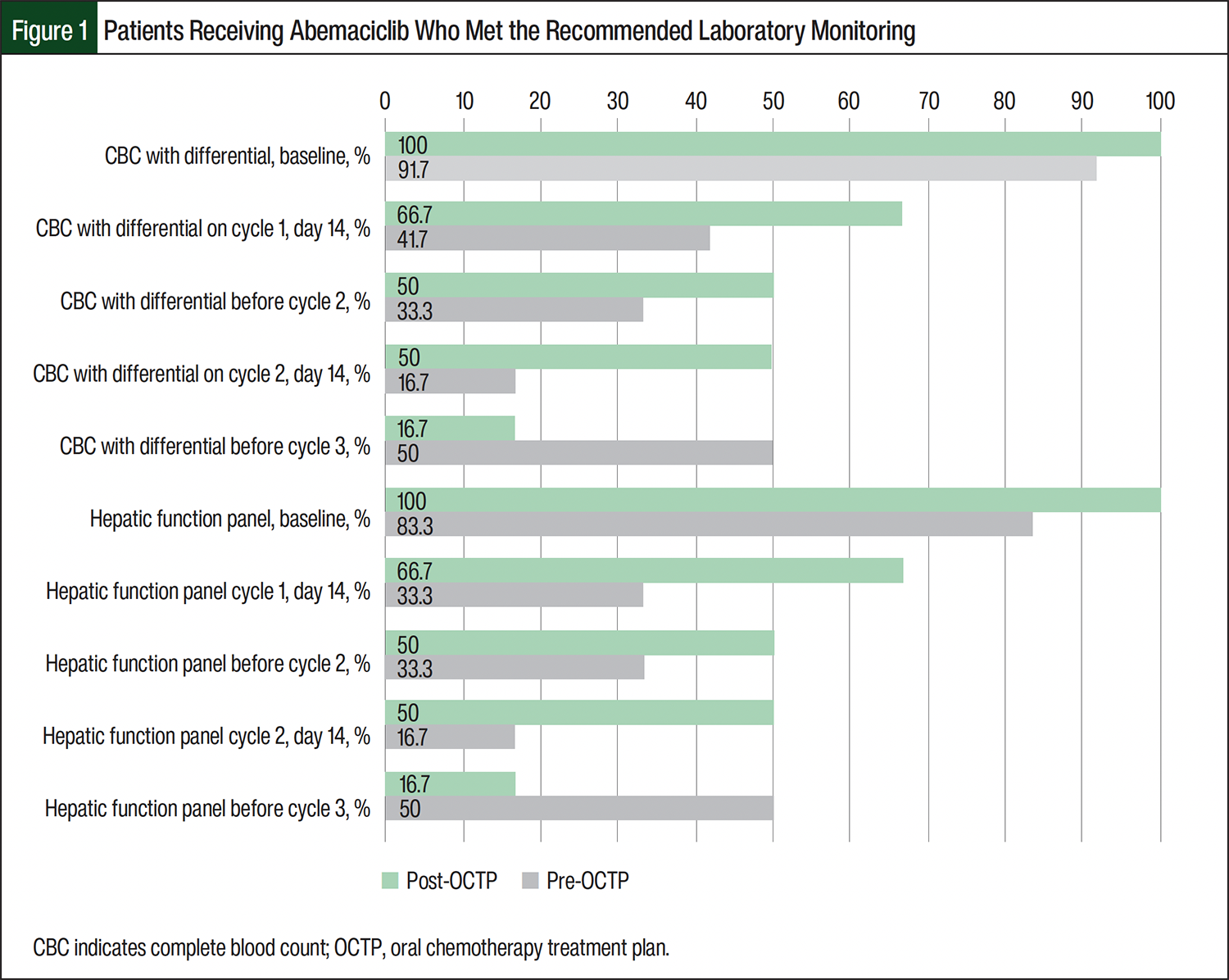
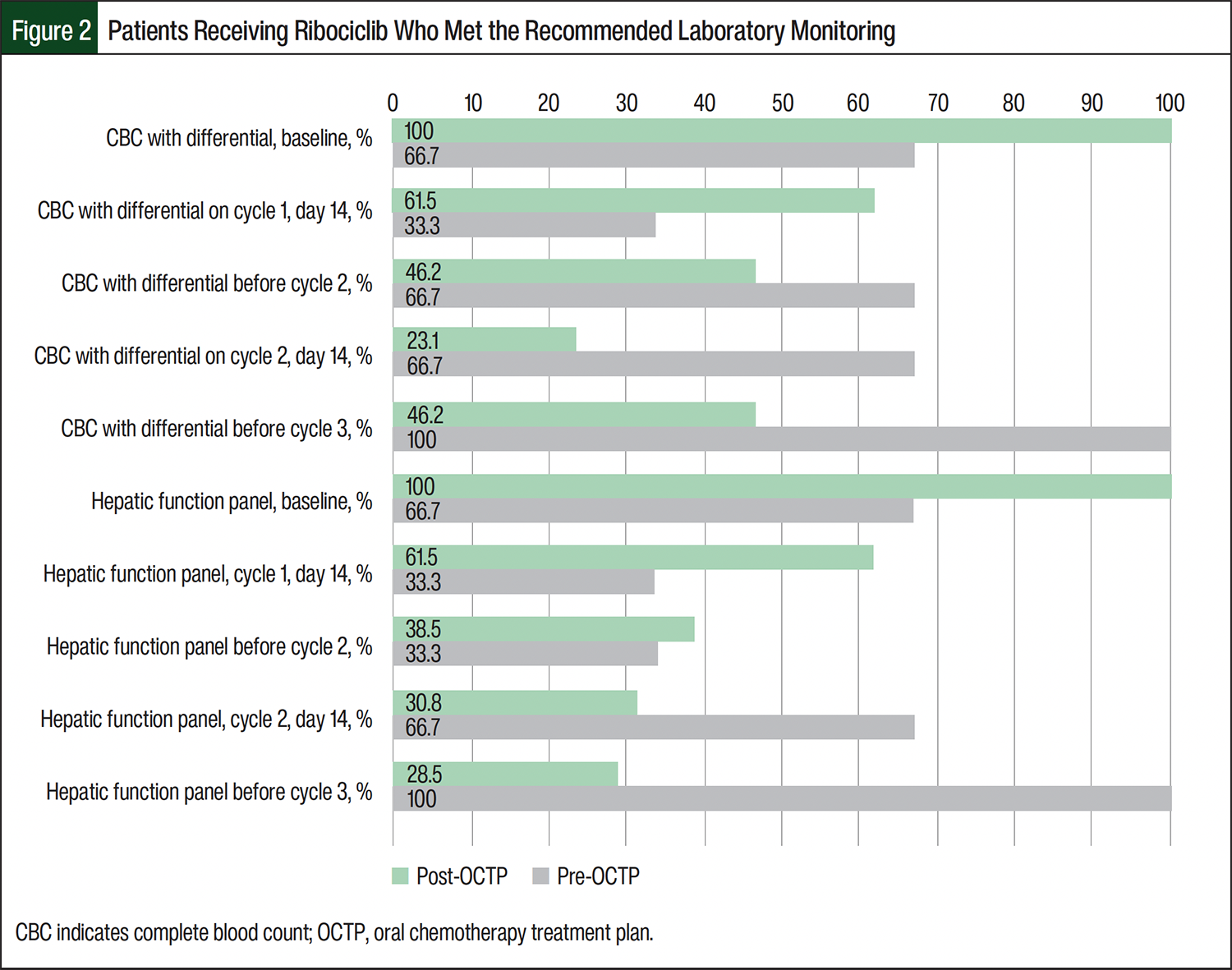
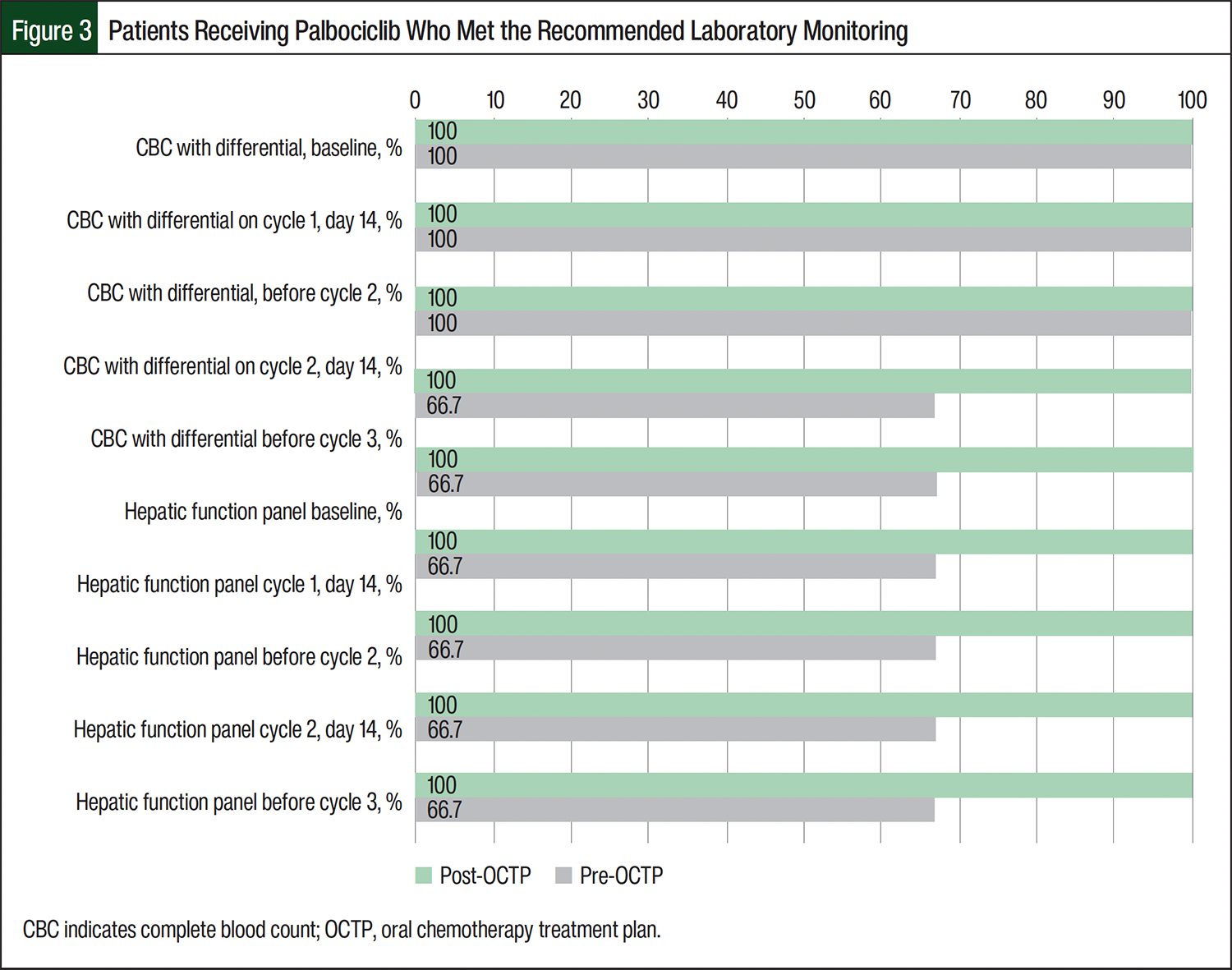
The pre-OCTP baseline laboratory testing completion rates for the patients receiving any CDK4/6 inhibitor ranged from 66.7% to 100%. Post-OCTP implementation, 100% of the patients receiving CDK4/6 inhibitors completed baseline laboratory testing on time (Figures 1-3). Among the patients receiving abemaciclib, most laboratory testing completion rates were higher after the implementation of an OCTP. For patients in the post-OCTP abemaciclib group, CBC with differential and hepatic function monitoring rates were 100% at baseline; 66.7% at cycle 1, day 14; 50% before cycle 2 and at cycle 2, day 14; and 16.7% before cycle 3. For the patients in the pre-OCTP abemaciclib group, the completion rates for CBC with differential and hepatic function monitoring parameters were, respectively, 91.7% and 83.3% at baseline; 41.7% and 33.3% at cycle 1, day 14; 33.3% before cycle 2; 16.7% at cycle 2, day 14; and 50% before cycle 3. Of note, before cycle 3, the laboratory monitoring completion rates were higher in the pre-OCTP abemaciclib cohort than in the post-OCTP abemaciclib cohort (Figure 1). Among the patients who received ribociclib, the EKG completion rates were higher in the pre-OCTP group than in the post-OCTP group. Specifically, the rates were 100% at baseline; 66.7% at cycle 1, day 14; and 66.7% before cycle 2. After the implementation of OCTP, these rates decreased to 84.6%, 61.5%, and 38.5%, respectively (Table 3).
After the implementation of OCTP, the 3-month medication adherence rates increased for patients who received ribociclib (97% vs 92% pre-OCTP). Conversely, adherence decreased for patients who received abemaciclib (96% vs 97% pre-OCTP) and for patients who received palbociclib (80% compared with 94% pre-OCTP).
There were no hospitalizations resulting from CDK4/6 inhibitor AEs in the 38 study patients, and none of the patients receiving palbociclib required sick visits for oral chemotherapy AEs. In the post-OCTP cohort, 15.4% of patients receiving ribociclib and 50% of those receiving abemaciclib required a sick visit in the cancer center related to CDK4/6 inhibitor AEs compared with 0% and 8.3%, respectively, of the pre-OCTP cohort.
Discussion
This study evaluated the impact of pharmacist-driven OCTP implementation on the use of appropriate supportive care and laboratory test monitoring for patients with breast cancer initiating CDK4/6 inhibitor therapy in the outpatient oncology setting. The study’s primary focus was to evaluate the frequency of pharmacist interventions before and after the implementation of an OCTP. The results revealed that the post-OCTP abemaciclib and ribociclib cohorts had increased percentages of interventions documented versus the pre-OCTP cohorts (Table 2). OCTPs were newly integrated into our institution’s outpatient oncology clinic workflows, requiring ongoing education to providers, nursing, and pharmacy staff.
Furthermore, there was an unequal distribution of patients across the pre- and post-OCTP cohorts. The pre-OCTP cohorts who received abemaciclib or palbociclib were twice the size of the post-OCTP counterpart cohorts. Conversely, the post-OCTP ribociclib cohort was more than 4 times larger than the pre-OCTP cohort (Table 1).
Consistent with the prescribing information recommendations, post-OCTP patients receiving abemaciclib or ribociclib were more likely to be prescribed supportive care medications, including antiemetics and antidiarrheals (Table 3). Prescribing supportive care prescriptions up front allows patients to promptly address and mitigate any AEs that may arise. Our findings demonstrate that OCTPs can play a crucial role in minimizing the risk for AEs that could result in treatment interruption and delay.
The percentage of patients completing the recommended laboratory monitoring parameters at the recommended intervals varied across all 3 agents in pre- and post-OCTP cohorts. All patients in the post-OCTP cohorts completed CBCs with differential and hepatic function panels at baseline. Apart from the 1 post-OCTP patient who received palbociclib achieving 100% laboratory test completion at the recommended intervals, the post-OCTP cohort that received abemaciclib demonstrated the next highest rate of laboratory test completion at the recommended intervals, followed by patients who received ribociclib in the post-OCTP cohorts (Figures 1-3). The patients in the pre-OCTP cohort who received ribociclib had higher percentages of timely EKG completions (baseline, 100%; cycle 1, day 14, 66.7%; before cycle 2, 66.7%) than the post-OCTP cohort that received ribociclib (baseline, 84.6%; cycle 1, day 14, 61.5%; before cycle 2, 38.5%).
The inconsistent laboratory test monitoring and EKG completion rates observed in the post-OCTP group are likely multifactorial. Although cycle dates were collected and documented as accurately as possible within the EHRs, patients may have initiated a CDK4/6 inhibitor cycle on a different day than documented without notifying the clinic. This could lead to the misclassification of timely laboratory testing completion. The misclassification of these patients is also possible with treatment delays, because restart dates may be inaccurately documented or reported. The variability in laboratory test monitoring results suggests that there are opportunities to further improve the documentation used to track cycle data in the EHRs.
Limitations
This study has several limitations to consider when interpreting the findings. First, the distribution of sample sizes in this study are unequal across the pre- and post-OCTP cohorts, leading to skewed data. Because of the unequal sample sizes, the percentages may be biased, limiting the ability to directly compare pre- and post-OCTP results for each CDK4/6 inhibitor. Because of the study’s small sample size, there is a likelihood of type II error; therefore, we may be unable to detect significant differences between the pre- and post-implementation periods. It is also important to note that because of the varying recommended monitoring parameter frequencies and side effect profiles of CDK4/6 inhibitors, we were unable to compare the agents head-to-head, which could lead to confounded results.
The retrospective design of our study was challenging because EHR documentation regarding treatment delays, interruptions, and precise cycle start dates was often missing or unclear. These discrepancies in electronic medical records could lead to the misclassification of treatment timelines and inaccurate end point categorization.
Variability and a lack of consistency in provider OCTP use, including modification and removal of prebuilt supportive care prescriptions and laboratory orders, is another limitation. These changes may lead to unnecessary pharmacist interventions and delays in supportive care prescribing, potentially underestimating the intent and benefits of OCTPs. In addition, when the plans were newly integrated into clinic workflows, they likely received heightened initial attention and oversight. This could have contributed to an increase in interventions during the early implementation phase. Ultimately, further provider education on standardized OCTP use should be prioritized. In addition, improvements to the operational workflow processes within the EHRs should be implemented to ensure patients are appropriately monitored. However, it is important to emphasize that although the OCTPs ensure that laboratory orders are added within the EHR and workflows can be optimized, it is also a patient’s responsibility to follow through with timely laboratory monitoring.
Last, this study focuses specifically on patients with breast cancer who are receiving CDK4/6 inhibitors, which limits its generalizability to other types of cancer, given the variations in treatment regimens and patient populations. Our institution is in the process of integrating OCTPs for oral anticancer agents used in the treatment of different malignancies. Future research could explore the application of OCTPs in diverse patient populations across various malignancies.
Conclusion
For patients with advanced or metastatic breast cancer who are receiving CDK4/6 inhibitors, pharmacist-implemented OCTPs can potentially optimize the management of these treatments. In our small, single-center study, the use of OCTPs immediately resulted in all post-OCTP patients receiving necessary supportive care prescriptions at baseline. The completion of accurate laboratory monitoring at predefined intervals was completed more often on time in most of the post-OCTP medication cohorts compared with the pre-OCTP medication cohorts. By promoting timely supportive care prescribing and adherence to the recommended laboratory monitoring at treatment initiation, OCTPs aim to enhance medication tolerability and minimize treatment delays. OCTPs can also provide patient counseling opportunities and reduce the need for prescription clarification.
Although there was not an immediate decrease seen in pharmacist interventions after OCTP implementation, this is likely attributed to ongoing education and the integration of these plans into workflows. This approach has the potential to improve overall treatment success and streamline outpatient oncology workflows. Our study highlights the positive impact of pharmacist-driven outpatient workflows and supports the broader implementation of pharmacist-driven treatment plans across multiple malignancies. Further studies are warranted to assess whether the long-term use of OCTPs decreases pharmacists’ interventions.
Author Disclosure
Dr Felitte, Dr Kisla, Dr Brady, Dr Tat, Dr Mills, and Dr McDonald have no conflicts of interest to report.
References
- National Breast Cancer Foundation, Inc. Breast Cancer Facts & Stats. Updated September 29, 2025. Accessed March 22, 2026. www.nationalbreastcancer.org/breast-cancer-facts
- National Cancer Institute. SEER cancer stat facts: female breast cancer subtypes. Accessed March 21, 2026. https://seer.cancer.gov/statfacts/html/breast-subtypes.html
- Finn RS, Martin M, Rugo HS, et al. Palbociclib and letrozole in advanced breast cancer. N Engl J Med. 2016;375:1925-1936. doi:10.1056/NEJMoa1607303
- Hortobagyi GN, Stemmer SM, Burris HA, et al. Ribociclib as first-line therapy for HR-positive, advanced breast cancer. N Engl J Med. 2016;375:1738-1748. Erratum in: N Engl J Med. 2018;379:2582. doi:10.1056/nejmoa1609709
- Goetz MP, Toi M, Campone M, et al. MONARCH 3: abemaciclib as initial therapy for advanced breast cancer. J Clin Oncol. 2017;35:3638-3646. doi:10.1200/jco.2017.75.6155
- Rastogi P, O’Shaughnessy J, Martin M, et al. Adjuvant abemaciclib plus endocrine therapy for hormone receptor-positive, human epidermal growth factor receptor 2-negative, high-risk early breast cancer: results from a preplanned monarchE overall survival interim analysis, including 5-year efficacy outcomes. J Clin Oncol. 2024;42:987-993. Errata in: J Clin Oncol. 2024;42:2111; J Clin Oncol. 2025;43:113. doi:10.1200/JCO.23.01994
- Slamon D, Lipatov O, Nowecki Z, et al. Ribociclib plus endocrine therapy in early breast cancer. N Engl J Med. 2024;390:1080-1091. doi:10.1056/NEJMoa2305488
- Hortobagyi GN, Stemmer SM, Burris HA, et al. Overall survival with ribociclib plus letrozole in advance breast cancer. N Engl J Med. 2022;386:942-950. doi:10.1056/nejmoa211463
- Slamon DJ, Neven P, Chia S, et al. Ribociclib plus fulvestrant for postmenopausal women with hormone-receptor-positive, human epidermal growth factor receptor 20 negative advanced breast cancer (MONALEESA-3). Lancet Oncol. 2018;19:904-915. doi:10.1016/S1470-2045(18)30292-4
- Slamon DJ, Neven P, Chia S, et al. Overall survival with ribociclib plus fulvestrant in advanced breast cancer. N Engl J Med. 2020;382:514524. doi:10.1056/NEJMoa1911149
- Tripathy D, Im SA, Colleoni M, et al. Ribociclib plus endocrine therapy for premenopausal women with hormone-receptor-positive, advanced breast cancer (MONALEESA-7). N Engl J Med. 2018;379:2294-2306. doi:10.10 56/NEJMoa1810527
- Verzenio (abemaciclib) tablets, for oral use [prescribing information]. Eli Lilly and Company; February 2025. Accessed March 22, 2026. www.accessdata.fda.gov/drugsatfda_docs/label/2025/208716s019lbl.pdf
- Ibrance (palbociclib) capsules, for oral use [prescribing information]. Pfizer Inc; September 2025. Accessed March 22, 2026. www.accessdata.fda.gov/drugsatfda_docs/label/2025/207103s023lbl.pdf
- Kisqali (ribociclib) tablets, for oral use [prescribing information]. Novartis Pharmaceuticals Corporation; September 2025. Accessed March 22, 2026. www.novartis.com/us-en/sites/novartis_us/files/kisqali.pdf
- NCCN Guidelines Insights: Breast Cancer, Version 5.2025. J Natl Compre Canc Netw. 2025;23:426-436. doi:10.6004/jnccn.2025.0053
- Braal CL, Jongbloed EM, Wilting SM, et al. Inhibiting CDK4/6 in breast cancer with palbociclib, ribociclib, and abemaciclib: similarities and differences. Drugs. 2021;81:317-331. doi:10.1007/s40265-020-01461-2
- Lam SY, Liu WS, Lee CS. A review of CDK4/6 inhibitors. US Pharm. 2020;45:3-8.
- Finn A, Bondarkena C, Edwards K, et al. Evaluation of electronic health record implementation on pharmacist interventions related to oral chemotherapy management. J Oncol Pharm Pract. 2017;23:563-574. doi:10.1177/1078155216665247
- De Raya J, Modlin J. Evaluation of pharmacist-driven approach to monitoring for neutropenia related to CDK4/6 inhibitors in women with advanced breast cancer. J Hematol Oncol Pharm. 2024;14:115-120.
- Colombo LRP, Aguiar PM, Lima TM, Storpirtis S. The effects of pharmacist interventions on adult outpatients with cancer: a systematic review. J Clin Pharm Ther. 2017;42:414-424. doi:10.1111/jcpt.12562
- Battis B, Clifford L, Huq M, et al. The impacts of a pharmacist-managed outpatient clinic and chemotherapy-directed electronic order sets for monitoring oral chemotherapy. J Oncol Pharm Pract. 2017;23:582-590. doi:10.1177/1078155216672314