Connecting Science to Practice
Antibody–drug conjugates are potent therapies for patients
with cancer, but they may cause infusion-related reactions
that interrupt treatment or require dose reduction. This case
report describes a patient with a history of infusion-related
reactions who developed a severe rash while receiving
fam-trastuzumab deruxtecan-nxki and was able to continue
therapy through development of a desensitization protocol.
Desensitization uses gradual, stepwise dosing to reduce hypersensitivity
and has been effective with other medications.
Our findings suggest that desensitization should be more
routinely considered in oncology as a standard approach to
managing infusion-related reactions, minimize treatment interruptions,
and improve patient outcomes.
HER2 protein, a primary driver of tumorigenesis and cancer metastasis, is overexpressed or amplified in several subsets of cancer.1 Combining chemotherapy and immunotherapy, monoclonal antibody–drug conjugates (ADCs) are an emerging class of potent medications that target HER2-positive cancers. In May 2022, the FDA approved fam-trastuzumab deruxtecan-nxki, a conjugate of the antibody trastuzumab and the topoisomerase I inhibitor deruxtecan, for the treatment of advanced or metastatic HER2-positive breast cancer.2 Trastuzumab targets and binds to the extracellular domain of HER2 receptors on cancer cells. Once bound, the antibody is internalized along with the conjugated anticancer drug deruxtecan and subsequently causes cell death. By having a higher drug-to-antibody ratio, fam-trastuzumab deruxtecan-nxki markedly reduced the risk for cancer progression by 72% compared with trastuzumab emtansine and more than doubled the overall response rate to existing therapies.3
Fam-trastuzumab deruxtecan-nxki is infused once every 3 weeks, with an initial 90-minute infusion time that may be reduced to 30 minutes in subsequent transfusions if treatment is well tolerated.4 Because fam-trastuzumab deruxtecan-nxki is classified as a highly emetogenic chemotherapy,5 premedication with antiemetics, such as a 5-HT3 receptor antagonist (eg, fosaprepitant) and dexamethasone, is recommended before infusion. According to the prescribing information for fam-trastuzumab deruxtecan-nxki for the treatment of breast cancer, the recommended starting dose is 5.4 mg/kg, with reductions to 4.4 mg/kg and 3.2 mg/kg if severe adverse events (AEs) occur. Treatment should be discontinued if further dose reductions are necessary.4
Infusion-related reactions after receiving fam-trastuzumab deruxtecan-nxki are relatively uncommon, occurring in 0.5% to 2.6% of patients.4 Although skin rash was reported in a higher percentage of patients (8%-13%) than infusion-related reactions,4 not all instances of rash may be infusion-related. Nevertheless, managing these AEs through dose reductions, temporary treatment delay, or permanent treatment discontinuation may not be ideal for patients with advanced-stage invasive breast cancer. A desensitization procedure, which slows the infusion of the drug and allows it to be administered in structured, incremental doses may present an alternative to managing hypersensitivity symptoms and improving tolerance without interrupting the patient’s treatment.
Previous literature has reported on the development and use of successful desensitization protocols for other HER2-targeting therapies, such as trastuzumab and ado-trastuzumab emtansine.6 Here, we present the development and use of the first desensitization protocol for fam-trastuzumab deruxtecan-nxki in a 67-year-old woman with metastatic breast cancer.
Case Report
A 67-year-old woman was initially diagnosed with early-stage, HER2-positive breast cancer in 2009 at the age of 54. She underwent 6 cycles of neoadjuvant chemotherapy with docetaxel, carboplatin, and trastuzumab, followed by a mastectomy, and she responded well to treatment. In 2018, a positron emission tomography scan revealed that the patient had metastatic breast cancer. After this cancer recurrence, she received trastuzumab and palbociclib for approximately 18 months and then transitioned to treatment with ado-trastuzumab emtansine (an ADC drug), fulvestrant, and denosumab for another 24 months. In December 2021, the patient began receiving fam-trastuzumab deruxtecan-nxki intravenous infusions every 3 weeks, which she continued receiving for 10 months.
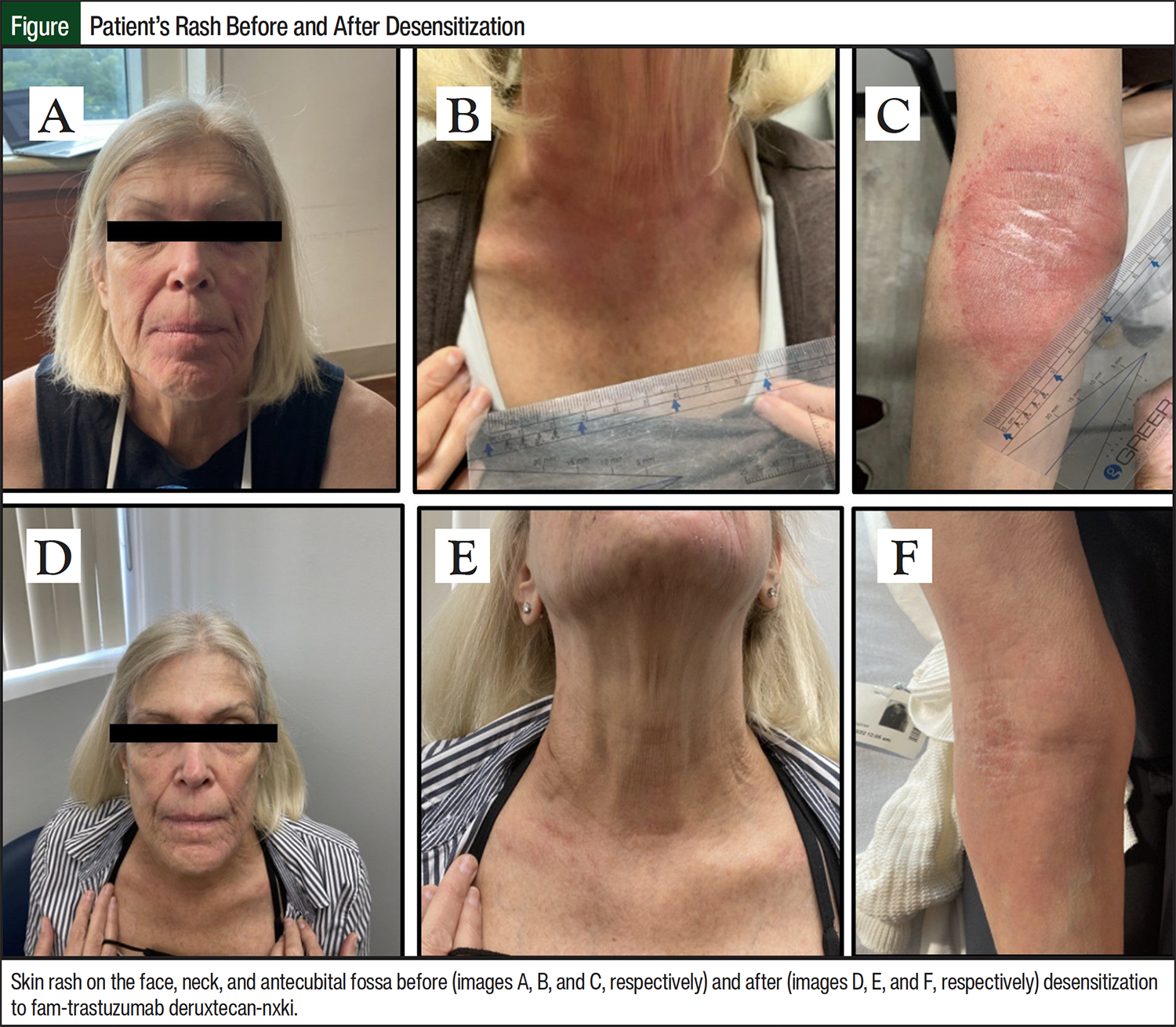
The treatment was well tolerated until the sixth month, when the patient began having a skin rash. She first noticed the rash on her face 1 day before receiving an infusion of fam-trastuzumab deruxtecan-nxki. On the day of the infusion, the rash became worse and subsequently spread to the neck and right antecubital fossa region during the subsequent infusion cycles. After the third infusion cycle, the patient presented to the allergist/immunologist with a rash on the face (Figure, image A), neck (Figure, image B), and right antecubital fossa (Figure, image C), which she described as itchy and tingly. The rash subsided between her infusion cycles, but they flared up during each infusion and persisted for several days.
To relieve the AEs, the infusion dose of fam-trastuzumab deruxtecan-nxki was reduced twice to the lowest dose of 3.2 mg/kg, and the patient received premedication with famotidine and cetirizine, but the skin rash persisted. The rash was also not responsive to treatment with diphenhydramine, loratadine, or famotidine, including other treatments such as dexamethasone or crisaborole topical ointment. Sulfacetamide sodium-sulfur topical cleanser and prednisone provided only temporary symptom relief between infusion cycles. While receiving previous cancer treatments, the patient had mild infusion-induced rosacea with platinum-based chemotherapy and fever during the first infusion cycle of trastuzumab; however, the latter resolved after receiving premedication with acetaminophen. Hives were not present on examination of the skin rash.
Notably, 2 weeks before presenting to the allergist/immunologist with skin rash, the patient was bit by a lone star tick, Amblyomma americanum, for the second time in 2 years. Serial blood tests after the bite indicated normal values for total immunoglobulin (Ig)E level, as well as IgE to galactose-α-1,3-galactose, beef, pork, and lamb.
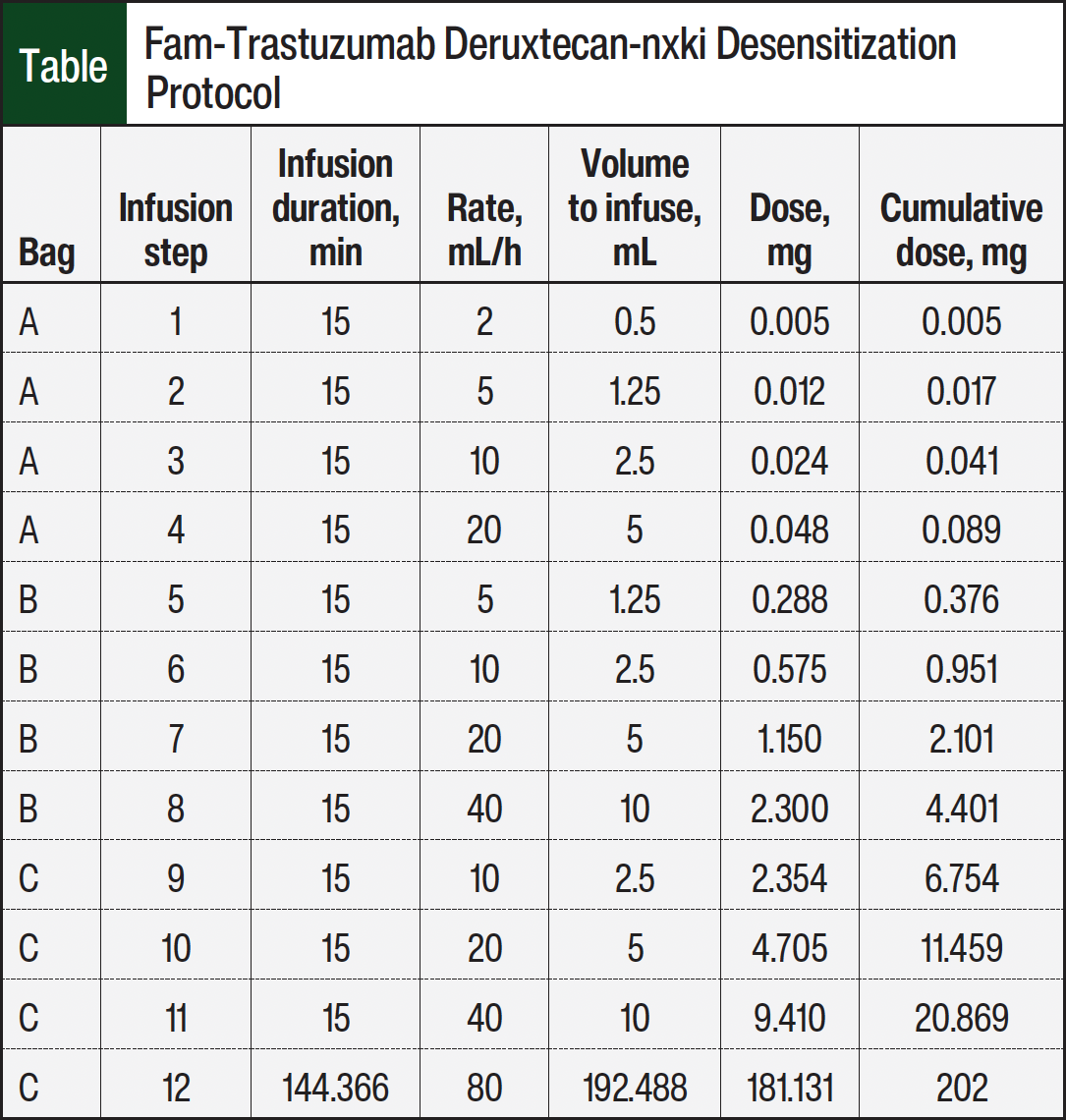
Given the patient’s history of infusion-related reactions and the worsening nature of the skin rash during and after treatment, we conducted a desensitization protocol to fam-trastuzumab deruxtecan-nxki during her next infusion cycle. Desensitization is a slowed, stepwise administration of a medication in a series of diluted concentrations up to a well-tolerated final dose. A 3-bag, 12-step desensitization protocol was used based on existing protocols of desensitization to HER2-targeting therapeutics.6 Skin testing was not performed before the desensitization because of the lack of availability of fam-trastuzumab deruxtecan-nxki at the time of consultation.
The patient was directly admitted to the non-COVID intensive care unit. She was under the direct supervision of a registered oncology nurse and a board-certified physician in internal medicine, clinical adult and pediatric allergy/immunology, and pulmonary diseases. Loratadine and famotidine, as well as the antinausea drug palonosetron, were administered as premedications 1 hour before the procedure. The patient did not prefer to receive any steroids as premedication before the procedure. Notably, the affected areas still had a skin rash on the day of the desensitization procedure.
The Table shows the 3-bag (bags A, B, and C), 12-step protocol that was used. The desensitization was administered over 5.2 hours, reaching a target cumulative dose of 202 mg of fam-trastuzumab deruxtecan-nxki, based on the patient’s weight of 63 kg and the prescribed dose of 3.2 mg/kg. The patient was monitored for the worsening of her skin rash and other infusion-related reactions during the procedure, which did not occur. In the days after the desensitization, the rash that was present before the desensitization was markedly less severe in redness and itchiness compared with rashes that emerged after the previous infusion cycles before the desensitization (Figure images D, E, and F).
Discussion
The 3-bag, 12-step desensitization protocol used in this report was adapted from literature on a successful desensitization to the predecessor drug of fam-trastuzumab deruxtecan-nxki, ado-trastuzumab emtansine.6 It is important to note that desensitization to ADCs is limited in clinical data, unlike desensitization to traditional platinum-based chemotherapy and certain monoclonal antibodies, which are more well-documented.6-9
The key feature of desensitization is the slowed, incremental infusion of a medication over several hours, although the reason for better tolerance is unclear.10 In subsequent infusion cycles, the patient tolerated the therapy without any rash when the medication was infused over approximately 5 hours rather than the 90-minute rate used before desensitization.
The patient’s infusion-related reaction to fam-trastuzumab deruxtecan-nxki possesses several characteristics that differentiate it from other infusion-related reactions to monoclonal antibody-related treatments, such as trastuzumab. Previous literature has shown that mild infusion-related reactions frequently happen after anticancer treatments; they are the most severe during the first few infusion cycles and can often resolve with premedication in subsequent cycles.11 However, our patient had a skin rash during the ninth infusion cycle that worsened with subsequent infusions, despite dose reductions and premedications.
In addition, the patient initially had the skin rash 2 weeks before being bitten by a lone star tick for the second time in 2 years. A lone star tick bite can lead to sensitization to the sugar molecule alpha-gal.12 In certain instances, alpha-gal sensitization can cause allergic reactions to the monoclonal antibody cetuximab; these patients often present with abnormally high IgE levels to alpha-gal, beef, pork, and lamb.12 Despite being repeatedly tested for 2 years, our patient’s total serum IgE levels, as well as IgE levels to alpha-gal, beef, pork, and lamb, have been within normal limits. Moreover, to our knowledge, there are currently no cases of tick bites that have caused sensitivity to trastuzumab, the monoclonal antibody used in fam-trastuzumab deruxtecan-nxki.
Our patient’s infusion-related skin rash is a type of hypersensitivity reaction for which the mechanism of action can be difficult to elucidate. ADCs cause all 4 types of hypersensitivity reactions, as classified by Coombs and Gell.13 Thus, it is unclear whether this skin rash was caused by IgE-mediated (true allergy; hypersensitivity reaction type I) or non–IgE-mediated mechanisms (hypersensitivity reaction type II, III, or IV).14
Monoclonal ADC chemotherapy drugs are highly potent and have significant potential as treatment for a wide range of cancers, including HER2-low–expressing breast cancer and HER2-positive non–small cell lung cancer.15 Although fam-trastuzumab deruxtecan-nxki is a potent treatment for breast cancer and other cancers, the resultant AEs can be unpleasant. If these reactions are mild and recur despite premedication, a desensitization protocol could be considered an alternative to reducing treatment dose or discontinuing treatment, especially when there are less efficacious alternative therapies available.
Last, future studies may consider limitations surrounding desensitization procedures. These protocols can take several hours and must be performed in a clinical setting with close supervision by a doctor and nursing staff. As a result, desensitization procedures may be expensive. Although some studies suggest that desensitization may be less costly than alternative therapies, an economic analysis would be helpful in determining the overall costs associated with these procedures.16
Conclusion
Monoclonal ADC drugs are very effective therapies and can be considered as treatment for a variety of cancers, including HER2-low–expressing breast cancer and HER2-positive non–small cell lung cancer. However, their effectiveness comes with a risk for AEs, including infusion-related hypersensitivity reactions. These reactions can lead to dose reductions, therapy delays, or even the discontinuation of a treatment that is otherwise highly effective. In such cases, we recommend procedures such as desensitization, which involves slowing the infusion rate of the drug to help patients continue treatment and avoid therapy discontinuation that results from unmanageable AEs. This case report details the first successful use of a desensitization protocol for fam-trastuzumab deruxtecan-nxki.
Author Disclosure Statement
Dr Szema is currently employed at Northwell Health and RDS2 Solutions and has received research grants from the US Department of Defense; Centers for Disease Control and Prevention; National Institute for Occupational Safety and Health World Trade Center Health Program; National Institutes for Health National Heart, Lung, and Blood Institute; New York State; and Butterfly Ultrasound. Ms Budhan, Dr Kaplan, and Dr Kwan-Mangal have no conflicts of interest to report.
References
- Iqbal N, Iqbal N. Human epidermal growth factor receptor 2 (HER2) in cancers: overexpression and therapeutic implications. Mol Biol Int. 2014:852748.
- US Food and Drug Administration. D.I.S.C.O. Burst Edition: FDA approval of Enhertu (fam-trastuzumab deruxtecan-nxki) for adult patients with unresectable or metastatic HER2-positive breast cancer. May 24, 2022. Accessed September 22, 2025. www.fda.gov/drugs/resources-information-approved-drugs/fda-disco-burst-edition-fda-approval-enhertu-fam-trastuzumab-deruxtecan-nxki-adult-patients
- Cortés J, Kim SB, Chung WP, et al. Trastuzumab deruxtecan versus trastuzumab emtansine for breast cancer. N Engl J Med. 2022;386:1143-1154.
- Enhertu (fam-trastuzumab deruxtecan-nxki) injection, for intravenous use [prescribing information]. Daiichi Sankyo, Inc; January 2025. Accessed September 22, 2025. www.accessdata.fda.gov/drugsatfda_docs/label/2025/761139s032s035lbl.pdf
- Notini G, Naldini MM, Sica L, et al. Management of trastuzumab deruxtecan-related nausea and vomiting in real-world practice. Front Oncol. 2024;14:1374547.
- Kempke A, Kraft S, Castells M, et al. Desensitization permits continued trastuzumab and ado-trastuzumab emtansine therapy after hypersensitivity reaction. J Hematol Oncol Pharm. 2020;10:380-383.
- Castells M. Rapid desensitization for hypersensitivity reactions to medications. Immunol Allergy Clin North Am. 2009;29:585-606.
- Hong DI, Bankova L, Cahill KN, et al. Allergy to monoclonal antibodies: cutting-edge desensitization methods for cutting-edge therapies. Expert Rev Clin Immunol. 2012;8:43-54.
- Fizesan M, Boin C, Aujoulat O, et al. Successful rapid desensitization to the antibody–drug conjugate brentuximab vedotin in a patient with refractory Hodgkin lymphoma. J Oncol Pharm Pract. 2016;22:188-192.
- Vultaggio A, Matucci A, Nencini F, et al. Mechanisms of drug desensitization: not only mast cells. Front Pharmacol. 2020;11:590991.
- Roselló S, Blasco I, García Fabregat L, et al. Management of infusion reactions to systemic anticancer therapy: ESMO Clinical Practice Guidelines. Ann Oncol. 2017;28(suppl 4):iv100-iv118.
- Steinke JW, Platts-Mills TAE, Commins SP. The alpha-gal story: lessons learned from connecting the dots. J Allergy Clin Immunol. 2015;135:589-597.
- Coombs RRA, Gell PGH. Chapter 1. The classification of allergic reactions underlying disease. In: Coombs RRA, Gell PGH, eds. Clinical Aspects of Immunology. Oxford: Blackwell Scientific Publications; 1963.
- Lenz HJ. Management and preparedness for infusion and hypersensitivity reactions. Oncologist. 2007;12:601-609.
- Enhertu granted breakthrough therapy designation in the US for patients with HER2-low metastatic breast cancer. AstraZeneca. April 27, 2022. Accessed September 22, 2025. www.astrazeneca.com/media-centre/press-releases/2022/enhertu-granted-btd-her2-low-breast-cancer.html#!
- Sloane D, Govindarajulu U, Harrow-Mortelliti J, et al. Safety, costs, and efficacy of rapid drug desensitizations to chemotherapy and monoclonal antibodies. J Allergy Clin Immunol Pract. 2016;4:497-504.