Connecting Science to Practice
ALK mutations are most commonly reported in patients with
non–small cell lung cancer (NSCLC) but are known to be
present in other solid tumors. Despite evidence of these mutations
outside of the lung, ALK-directed tyrosine kinase inhibitors
(TKIs) have not been approved in any other disease
states. This report demonstrates durable clinical response to
ALK-targeted therapy in a patient with breast cancer harboring
an EML4-ALK rearrangement. The patient received alectinib
after disease progression on multiple standard-of-care
regimens. She maintained response to ALK-directed therapy
for ≥1 year, thus demonstrating the potential for beneficial
activity of ALK TKIs outside of NSCLC.
ALK is a receptor tyrosine kinase frequently activated by genetic rearrangements, resulting in ALK-fusion proteins. Activation of ALK will induce downstream signaling pathways, including JAK-STAT, RAS-MAPK, and PI3K-AKT, leading to amplified cell proliferation and malignant transformation.1 ALK is overexpressed or somatically activated in a variety of cancer types, most often, non–small cell lung cancer (NSCLC).1 It is estimated that 4% to 5% of all advanced NSCLC cases harbor an activating ALK rearrangement, with the highest rates observed in adenocarcinoma pathology, younger patients, and nonsmokers.1,2 ALK-directed therapy with tyrosine kinase inhibitors (TKIs) such as alectinib, lorlatinib, and brigatinib have become options for metastatic NSCLC with an ALK rearrangement,3 but these agents do not yet have approval outside of this setting.
ALK rearrangements are known to be present in other tumor types; for example, EML4-ALK and STRN-ALK fusions have been identified in breast cancer.4,5 Overall, an estimated 1.8% to 2.4% of breast cancer cases will have an ALK rearrangement, with higher rates observed in aggressive subtypes, such as triple-negative breast cancer (TNBC).4 Two case reports have demonstrated the benefits of ALK-targeted therapy in patients with breast cancer, with response to crizotinib and alectinib.5,6 Both patients were pretreated with traditional chemotherapy and had a documented activating ALK fusion. Treatment with alectinib demonstrated a duration of response of 2 months,5 whereas the patient receiving crizotinib had a near-complete response but died as a result of a COVID-19 infection after 4 months of therapy.6
Here we report on a patient with breast cancer with an EML4-ALK chromosomal rearrangement who later developed an ALK G1202R acquired resistance mutation. She demonstrated a durable complete clinical response to alectinib, followed by a trial of lorlatinib. We are not aware of any other published cases reporting response to ALK-directed therapy in this setting.
Case Report
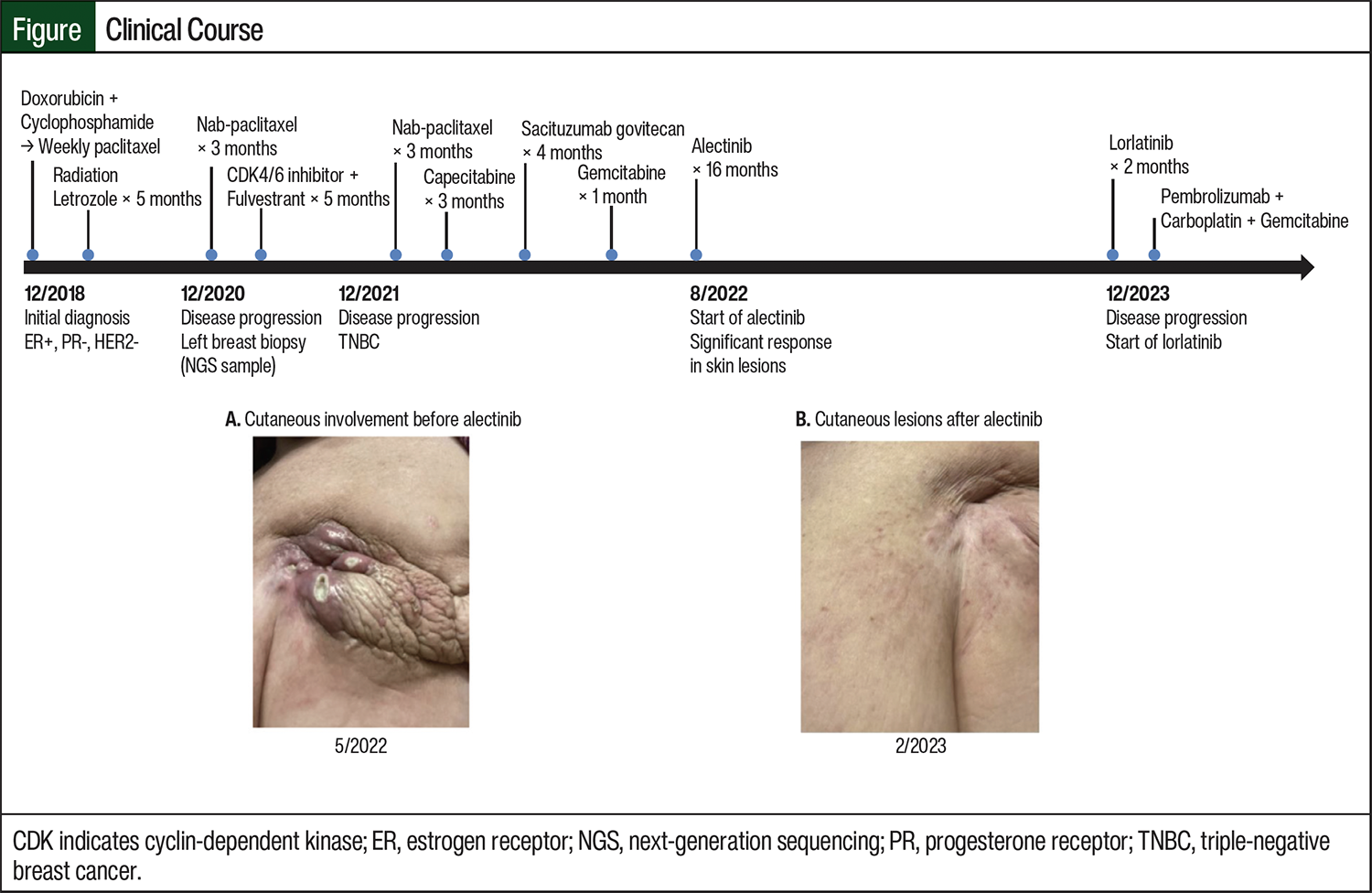
A 52-year-old woman presented with a left breast mass. A stereotactic core biopsy confirmed grade 3 ductal carcinoma in situ, which was estrogen receptor–positive, progesterone receptor–negative, and HER2-negative (immunohistochemistry score of 0). She was started on neoadjuvant treatment with 4 cycles of doxorubicin and cyclophosphamide every 3 weeks, followed by 12 cycles of weekly paclitaxel. Surgical resection revealed 3 of 7 lymph nodes positive for metastatic disease, and her final staging was ypT2N2a. She received localized radiation and was placed on hormonal therapy with anastrozole for a planned duration of 5 years.
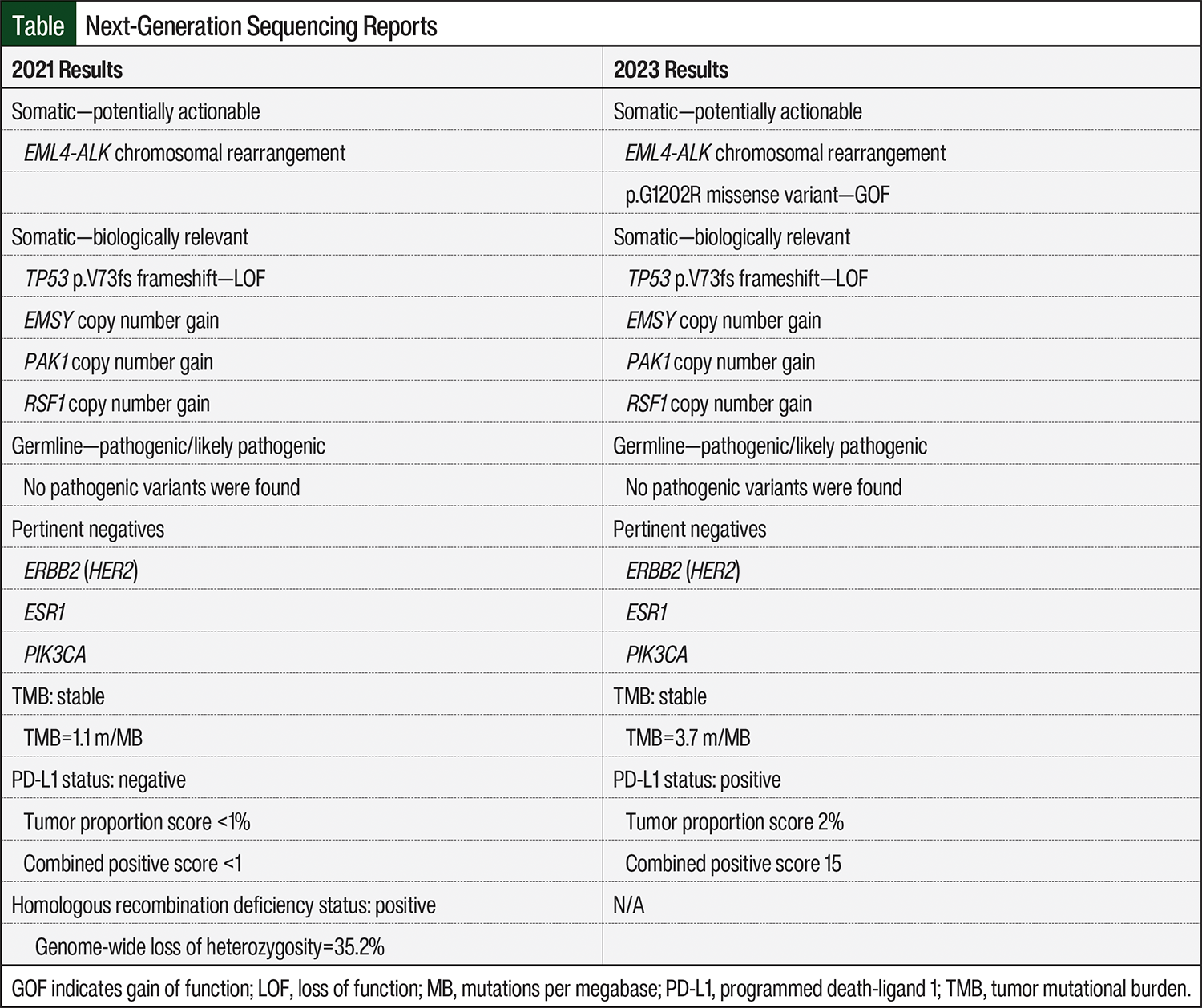
The patient returned almost 1 year later with relapsed disease in her left breast and visible skin involvement with subcutaneous nodules in the left sternal region. Tissue from the new lesion and blood samples were then sent for genomic profiling via next-generation sequencing (Tempus, Chicago, IL). Results revealed an EML4-ALK chromosomal rearrangement and mutations in TP53, EMSY, PAK1, and RSF1. The EML4-ALK fusion involved intron 2 of EML4 (including the coil-coiled domain) being fused with exon 19 of ALK. The fusion retained an intact ALK kinase domain and was predicted to be activating. A complete report of genomic profiling results is described in the Table. Our patient was subsequently treated with nab-paclitaxel for symptom control and then switched to a cyclin-dependent kinase (CDK)4/6 inhibitor with fulvestrant. She received abemaciclib next but switched to palbociclib after 1 cycle because of uncontrolled nausea. After 5 months of CDK4/6 inhibitor therapy, her disease progressed, and she had worsening of her subcutaneous nodules. Nab-paclitaxel was again given for symptom control, and a biopsy of the sternal mass revealed TNBC. Given her new hormone receptor status (TNBC), she was initiated on capecitabine, followed by a trial of sacituzumab-govitecan. The patient’s full clinical history and treatment lines are described in the Figure.
As fifth-line therapy, the patient was started on 600 mg of alectinib orally twice daily to target her EML4-ALK rearrangement. She was noted to have a remarkable clinical response in the subcutaneous nodules, with improved skin discoloration and near-complete disappearance of the lesions. She remained on therapy for 16 months without significant adverse events (AEs) before disease progression was noted in the right breast. Biopsy results revealed TNBC and repeat next-generation sequencing on tumor tissue detected the EML4-ALK rearrangement with a new G1202R resistance mutation; therefore, her treatment was switched to lorlatinib. A complete review of new next-generation sequencing findings is in the Table. Lorlatinib was selected based on the presence of the acquired resistance ALK G1202R mutation, which is known to correlate with disease highly resistant to alectinib, brigatinib, ceritinib, and crizotinib in NSCLC.7,8 Promising clinical evidence demonstrates lorlatinib’s ability to maintain sensitivity to the ALK G1202R resistance alteration.9 This patient received only 2 cycles of lorlatinib, however, before therapy was discontinued as a result of neurologic AEs, including peripheral neuropathy and intense nightmares, which are known to occur with this agent. She was subsequently treated with chemoimmunotherapy consisting of pembrolizumab, carboplatin, and gemcitabine because of her programmed death-ligand 1 combined positive score of 15.
Discussion
Activating ALK rearrangements have been well-studied in NSCLC since the discovery of the EML4-ALK fusion in 2007.2 ALK fusions are known to be oncogenic drivers and may correlate with a poorer prognosis.2 More than 20 ALK-fusion partner genes have now been identified, but EML4 remains the most common and has also been reported in other solid tumors, such as breast cancer.2,4 The detection of ALK rearrangements has led to an evolution in the first-line treatment of advanced NSCLC to include ALK-targeted TKIs, offering an option for targeted agents in a disease historically treated with cytotoxic chemotherapy.1
Three generations of ALK TKIs have been approved to treat patients with NSCLC who develop resistance and to optimize pharmacokinetics. The first ALK inhibitor approved was crizotinib, followed by second-generation agents (alectinib, ceritinib, brigatinib), and the third-generation agent lorlatinib.10 Patients will eventually acquire resistance to ALK therapy, most often caused by secondary mutations such as L1196M, G1269A, and G1202R.11 In the case of G1202R, clinical studies have established resistance to both first- and second-generation ALK inhibitors, but sensitivity is retained to lorlatinib.9 For this reason, the NCCN recommends lorlatinib as second-line ALK-targeted therapy for high-risk resistant mutations, such as ALK G1202R.3
Activating ALK rearrangements in other solid tumor subtypes, including breast cancer, are rare, and limited evidence exists to support the use of ALK-targeted TKIs outside of NSCLC. This case report shows potential clinical benefit with alectinib in patients with EML4-ALK rearranged breast cancer. Our patient was heavily pretreated and had a durable response to alectinib for more than 1 year. Disease progression while using alectinib was likely caused by the presence of the ALK G1202R acquired resistance mutation, which is known to be associated with resistance to ALK-targeted therapies. Many patients with NSCLC with resistant ALK mutations relapse within 1 year of treatment initiation, which follows the pattern observed in our patient case.
Conclusion
Our patient showed evidence of disease control and a durable response with alectinib in previously treated EML4-ALK rearranged breast cancer. She progressed through multiple lines of chemotherapy, with a short duration of response to traditional breast cancer regimens. She was noted to have persistent subcutaneous involvement with skin discoloration and dimpling, and multiple sternal nodules. She demonstrated remarkable clinical improvement in her cutaneous lesions with alectinib and maintained response for more than 1 year. On disease progression, genomic profiling revealed an acquired ALK G1202R resistance mutation, and her treatment was switched to lorlatinib. The third-generation TKI was specifically chosen to target her newly acquired resistance mutation, but response could not be adequately assessed, as lorlatinib was prematurely discontinued for neurologic AEs.
This case demonstrates activity of alectinib in a patient with refractory, EML4-ALK rearranged, breast cancer. Further investigation into the significance of ALK activation in solid tumors other than lung cancer and treatment implications is needed.
Author Disclosure Statement
Dr Roman is on the Advisory Board of Astellas and Daiichi Sankyo; Dr Hilton is on the Advisory Board of AstraZeneca, Gilead, and Biotheranostics, and is on the Speaker’s Bureau of AstraZeneca and Daiichi Sankyo; Dr Eross, Dr Shockley, and Dr Miller have no conflicts of interest to report.
References
- Chevallier M, Borgeaud M, Addeo A, Friedlaender A. Oncogenic driver mutations in non-small cell lung cancer: past, present and future. World J Clin Oncol. 2021;12:217-237.
- Chia PL, Mitchell P, Dobrovic A, John T. Prevalence and natural history of ALK positive non-small-cell lung cancer and the clinical impact of targeted therapy with ALK inhibitors. Clin Epidemiol. 2014;6:423-432.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): non-small cell lung cancer. Version 7.2025. July 10, 2025. Accessed August 11, 2025. www.nccn.org/professionals/physician_gls/pdf/nscl.pdf
- Shreenivas A, Janku F, Gouda MA, et al. ALK fusions in the pan-cancer setting: another tumor-agnostic target? NPJ Precis Oncol. 2023;7:101.
- Kelly AD, Wiklund T, Kononen J, Creeden J. STRN-ALK fusion-positive case of breast cancer with response to alectinib. JCO Precis Oncol. 2021;5:1281-1284.
- Kus T, Aktas G, Oktay C, et al. Dramatic response to crizotinib in a breast cancer patient with ALK gene rearrangement. Anticancer Drugs. 2022;33:400-405.
- Yoda S, Lin JJ, Lawrence MS, et al. Sequential ALK inhibitors can select for lorlatinib-resistant compound ALK mutations in ALK-positive lung cancer. Cancer Discov. 2018;8:714-729.
- Katayama R, Shaw AT, Khan TM, et al. Mechanisms of acquired crizotinib resistance in ALK-rearranged lung cancers. Sci Transl Med. 2012;4:120ra17.
- Shaw AT, Solomon BJ, Besse B, et al. ALK resistance mutations and efficacy of lorlatinib in advanced anaplastic lymphoma kinase-positive non-small-cell lung cancer. J Clin Oncol. 2019;37:1370-1379.
- Wu J, Savooji J, Liu D. Second- and third-generation ALK inhibitors for non-small cell lung cancer. J Hematol Oncol. 2016;9:19.
- Pan Y, Deng C, Qiu Z, et al. The resistance mechanisms and treatment strategies for ALK-rearranged non-small cell lung cancer. Front Oncol. 2021;11:713530.