Although breast cancer is the most common cancer diagnosed in female patients in the United States, it has a considerably lower incidence in male patients.1,2 According to the American Cancer Society, approximately 2800 men were estimated to be diagnosed with breast cancer in the United States in 2023, representing <1% of all cancers in men.1,2 Breast cancer cases in male patients are more likely to be positive for estrogen receptor (ER) or progesterone receptor (PR) expression than in female patients, with approximately 91% of cases of breast cancer in male patients being ER-positive and 81% being PR-positive.1
The prognosis for male patients with breast cancer is similar to that of female patients; however, the overall survival rate is lower in male patients with breast cancer.3 Compared with female patients, male patients with breast cancer are typically diagnosed at an older age and with more advanced disease.3 On average, breast cancer is diagnosed up to 10 years later in male patients than in female patients.4 Although the detection and treatment of breast cancer in female patients has significantly improved over the past several decades, male patients are not routinely screened for breast cancer.4 Male patients have been infrequently included in breast cancer trials, and therefore treatment recommendations for male patients are extrapolated from data in female patients.5
Because male patients can be diagnosed with prostate and breast cancers, there is a considerably small population of patients who may have both malignancies concurrently. The incidence of concurrent breast and prostate cancers in men is not well defined, with some reports suggesting incidence rates of 4% to 17%.6-8 Although little is known about the risk factors for synchronous or concurrent breast and prostate cancers, there are overlapping risk factors for each malignancy, including age, family history, and the presence of germline BRCA2 mutations.9-11
Treatment modalities for prostate cancer and ER-positive, PR-positive breast cancer heavily involve hormonal agents.5 For patients with advanced prostate cancer, the standard of care includes androgen-directed therapies, with a backbone of androgen deprivation therapy and a luteinizing hormone-releasing hormone agonist or antagonist.12 Along with androgen deprivation therapy, the treatment of patients with metastatic castration-sensitive prostate cancer includes the addition of a secondary hormonal agent, such as abiraterone, enzalutamide, or apalutamide, or the use of a triplet therapy with docetaxel and either darolutamide or abiraterone.12 For male patients with breast cancer and ER-positive, PR-positive disease, the current guidelines recommend hormonal therapy with tamoxifen because of the reported inferior estrogen suppression in male patients with aromatase inhibitor monotherapy.5 If male patients with breast cancer begin hormonal therapy with an aromatase inhibitor, the concomitant use of a luteinizing hormone-releasing hormone analog is recommended to fully suppress estrogen production.5 There are no established guidelines for the treatment of male patients with concurrent prostate and ER-positive, PR-positive breast cancers. Systemic treatment considerations should target androgen and estrogen production with a combination of a luteinizing hormone-releasing hormone analog and either tamoxifen or an aromatase inhibitor.
Herein, we present a patient with concurrent prostate and ER-positive, PR-positive breast cancer and the treatment considerations for this unique patient population.
Case Report
An 81-year-old man presented to his primary care provider in June 2019 for an annual checkup. At the visit, he reported a left breast lump with nipple inversion. A breast ultrasound was performed, showing a mass measuring 2.9×3.7×2.1 cm. A biopsy of the breast mass in July 2019 was positive for invasive ductal carcinoma of the breast that was grade 3, ER positive, PR positive, and HER2 equivocal by immunohistochemical staining. Germline genetic testing was negative for BRCA1 or BRCA2 mutations. In addition, a prostate examination was performed that revealed a smooth, enlarged, irregular surface with firm nodularity at the posterior aspect. A prostate-specific antigen (PSA) test was performed, which resulted in a level of 9.78 ng/mL. Although routine screening for prostate cancer is not recommended by major organizations for a patient aged 81 years,13,14 our patient was offered screening, which he accepted. After discovery of the elevated PSA and irregular prostate examination, the patient opted to pursue a further workup of his potential prostate cancer, as well as his confirmed breast cancer.
A baseline positron emission tomography/computed tomography (CT) scan on August 13, 2019, showed hypermetabolic uptake of the left breast mass, left axillary lymphadenopathy, prostatomegaly with infiltration along the left seminal vesicles, and vertebral involvement in the C5, T6, and L4 vertebrae. A biopsy of the L4 lesion was performed on August 27, 2019, with results that were consistent with prostate adenocarcinoma. Consequently, the patient was diagnosed with metastatic castrate-sensitive prostate adenocarcinoma. His breast cancer was diagnosed as stage IIB in August 2019. The patient declined surgical resection of his breast lesion. He was started on systemic hormonal therapy with intramuscular (IM) leuprolide 7.5 mg and oral letrozole 2.5 mg once daily for the breast and prostate cancers. For future androgen deprivation therapy, the patient received IM leuprolide 22.5 mg every 3 months. Because of his comorbidities, the concurrent diagnosis of breast cancer, and his low-volume disease, the addition of docetaxel or a secondary hormonal agent for the treatment of metastatic castration-sensitive prostate cancer was not recommended at that time.
More than 2 years later, on December 1, 2021, the patient returned to the breast cancer clinic to review the imaging results. At that time, the patient was tolerating treatment with letrozole with no reported hot flashes or arthralgia. Surveillance breast imaging performed on November 29, 2021, showed an increased size of the breast mass. The treatment options were reviewed with the patient, which included surgery and switching hormone therapy. Because the patient continued to decline surgical intervention, the decision was made to switch hormonal therapy from letrozole to oral exemestane 25 mg once daily. Repeat breast imaging would be performed in 3 months. The patient started treatment with exemestane on December 1, 2021.
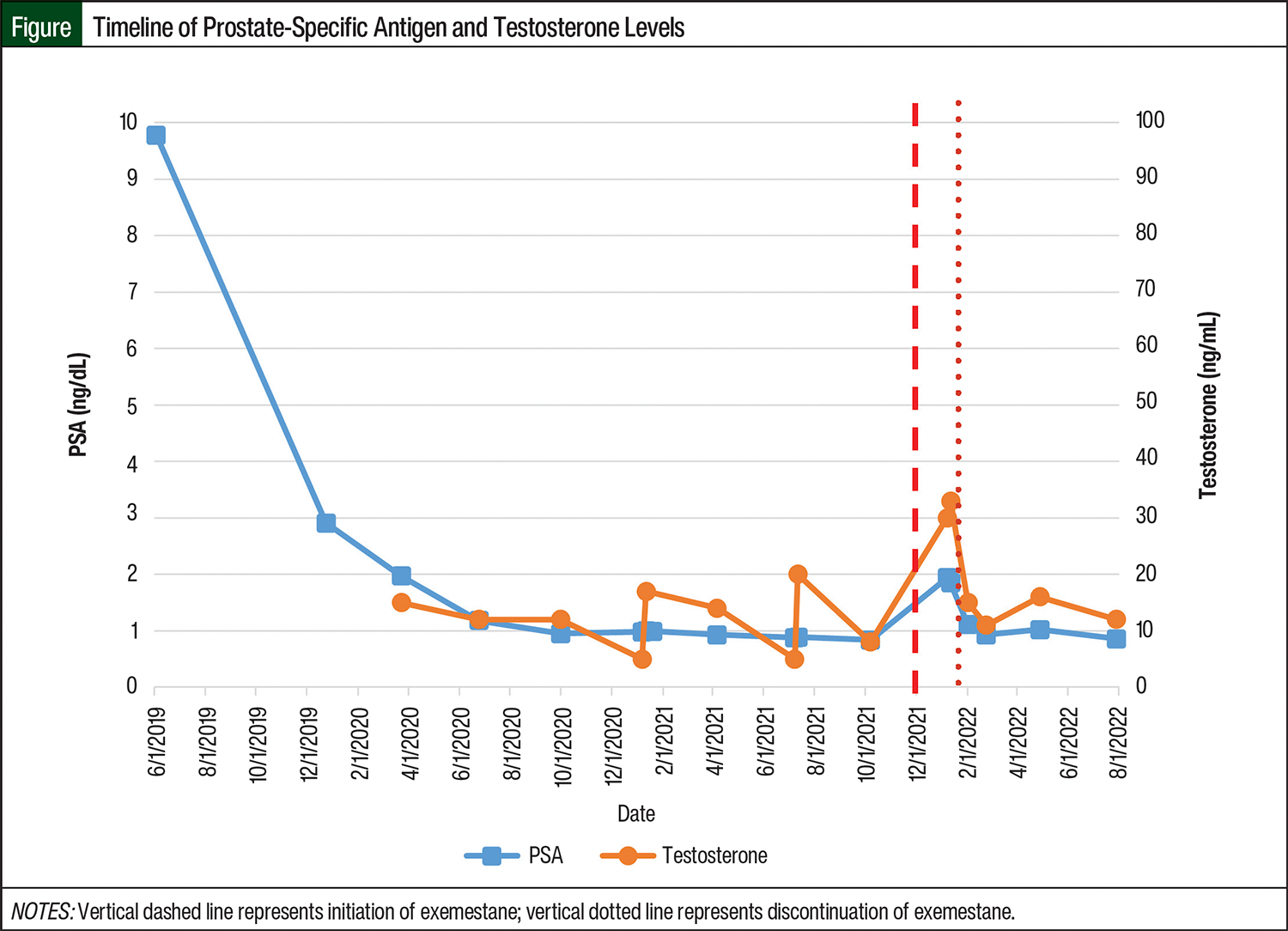
The patient returned to the genitourinary cancer clinic for a routine follow-up visit on January 12, 2022. His measured PSA values since 2019 are shown in the Figure. During this time, the patient’s PSA rose from 0.84 ng/mL on October 7, 2021, to 1.94 ng/mL on January 7, 2022. A repeat, confirmatory PSA was drawn on January 11, 2022, before his scheduled follow-up visit, which resulted in a level of 1.85 ng/mL. The patient’s testosterone levels increased during this time as well.
On investigation, the only medication change since October 2021 until January 2022 was a treatment switch from letrozole to exemestane. Because of a concern that treatment with exemestane may be causing the rise in PSA, the genitourinary oncology team recommended to the breast oncology team that the patient discontinue receiving exemestane. The patient then subsequently discontinued treatment with exemestane and started receiving therapy with oral anastrozole 1 mg once daily on January 13, 2022.
The repeat PSA values since starting treatment with anastrozole decreased to 0.93 ng/mL as of February 23, 2022, which has been consistent with the patient’s baseline value. His testosterone level has also decreased and most recently was 11 ng/dL. Repeat breast imaging on March 2, 2022, showed stable disease. At the time of this writing, the patient continues receiving treatment with leuprolide and anastrozole as well as having routine PSA and testosterone monitoring with surveillance breast imaging every 3 months. A repeat CT imaging in April 2022 showed stable metastatic, castration-sensitive prostate cancer.
Discussion
This case report highlights a unique clinical case of concurrent breast and prostate cancers in a male patient, as well as treatment considerations for this niche patient population.
Our patient was initiated on treatment with letrozole for ER-positive, PR-positive breast cancer along with systemic hormonal suppression with leuprolide, with the plan that leuprolide would provide androgen suppression for his concurrent prostate cancer. The breast and genitourinary medical oncology teams agreed with his initial treatment plan. The patient continued treatment unchanged for more than 2 years, with continued disease control of his prostate and breast cancers.
After breast imaging in December 2021 showed disease progression, the patient was switched from treatment with letrozole to exemestane because of his resistance to surgical intervention. As a structural analog of androstenedione, exemestane is classified as a steroidal aromatase inhibitor, which irreversibly inhibits the aromatase enzyme.15 Conversely, letrozole and anastrozole are nonsteroidal aromatase inhibitors that reversibly inhibit aromatase.15 Despite this pharmacologic difference, all 3 agents (ie, anastrozole, letrozole, and exemestane) are considered clinically equivalent in the treatment of ER-positive, PR-positive breast cancers.5 However, switching between aromatase inhibitors may be done clinically if patients have adverse events or disease progression.5,16
Because of its unique structure as an analog of androstenedione, exemestane was considered as a possible cause for the increases in PSA and testosterone. In males, androstenedione is normally produced by the adrenal glands and testes, where it is converted into testosterone by 17β-hydroxysteroid dehydrogenase.17 As a precursor to testosterone, androstenedione supplementation in healthy males has been shown to increase testosterone production.18 Because of its structural similarity to androstenedione, treatment with exemestane may have contributed to the increase in testosterone levels in this patient.
According to the prescribing information for exemestane, testosterone levels were not significantly increased at daily doses up to 200 mg in studies of female patients.19 However, pharmacodynamic data in healthy males aged 14 to 26 years showed that exemestane at a dose of 25 mg daily increased serum testosterone levels by 60%.20 This increase in testosterone was seen after 10 days of treatment with exemestane, but no significant difference was seen after only 1 oral dose.20 In vitro studies show an increase in androgen concentrations within breast cancer tissue after exposure to exemestane, which may be related to breast cancer cell metabolism of exemestane.21,22 The primary metabolite of exemestane, 17-hydroexemestane, binds to androgen receptors and exerts androgenic effects, with an affinity of 100-fold greater than exemestane itself.19,23
Our patient had a noticeable increase in testosterone level, from 8 ng/dL before treatment initiation to 30 ng/dL approximately 6 weeks after starting treatment with exemestane. Although the patient’s testosterone levels were still considered castrate at <50 ng/dL, the trend was concerning. This observation is congruent with reports of increased androgen concentrations and androgen receptor expression with exemestane.22 In regard to ER-positive, PR-positive breast cancer, this increase in androgens may be beneficial.22 However, in our patient, the increase in androgens was not desired and was potentially detrimental to his prostate cancer.
Evidence of the impact of exemestane on the progression of prostate cancer in male patients is sparse. The results of a phase 2 trial of patients with advanced prostate cancer showed that while receiving exemestane, 3 of 4 patients had an increase in bone pain and 3 patients had clear PSA progression, both of which resolved after the discontinuation of exemestane treatment.24 Two patients had radiologic progression of their prostate cancer while receiving exemestane. Because of these results, the investigators ended the trial early and concluded that exemestane did not have a role in the treatment of prostate cancer.24 In a second study, 31% of enrolled patients with advanced prostate cancer reported a transient increase in bone pain soon after starting treatment with formestane, a steroidal aromatase inhibitor that is structurally similar to exemestane.25
From a prostate cancer perspective, the increase in PSA in our patient in January 2022 was concerning. No other medications were changed or started since his previous PSA level in October 2021, aside from exemestane. Based on the previously discussed literature and the unique pharmacology of exemestane, the observed increase in PSA and testosterone suggested that exemestane was responsible for the increase in PSA. Further strengthening this conclusion, after discontinuing treatment with exemestane, the patient’s testosterone and PSA levels returned to baseline. There were no recurrences in PSA or testosterone elevations after switching from exemestane to anastrozole, another nonsteroidal aromatase inhibitor.
The probability of exemestane leading to the progression of this patient’s prostate cancer can be estimated using the Naranjo scale.26 The signs of prostate cancer progression occurred after the administration of exemestane (+2 on the Naranjo scale), improved after the discontinuation of treatment with exemestane (+1), occurred without an alternative cause (+2), and were confirmed by objective evidence (+1). Using this validated scoring tool, this particular scenario would receive a score of 6 on the Naranjo scale, indicating that the progression of our patient’s prostate cancer was likely related to treatment with exemestane.
Conclusion
In conclusion, our case report highlights unique therapy considerations in a man with concurrent breast and prostate cancers. The current evidence shows that treatment with exemestane increases androgen concentrations and may hasten the progression of prostate cancer. The results of our case report support these conclusions. The use of exemestane for the treatment of concurrent breast and prostate cancers in a male patient should be avoided.
Author Disclosure Statement
Dr Magnuson is an Editorial Board Member at American Society of Clinical Oncology Cancer.net; Dr Fitzgerald and Dr Fung have no conflicts of interest to report.
References
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57.
- American Cancer Society. Cancer Facts & Figures 2023. Accessed June 3, 2024. www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf
- Giordano SH. A review of the diagnosis and management of male breast cancer. Oncologist. 2005;10:471-479.
- Miao H, Verkooijen HM, Chia KS, et al. Incidence and outcome of male breast cancer: an international population-based study. J Clin Oncol. 2011;29:4381-4386.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): breast cancer. Version 2.2024. March 11, 2024. Accessed June 3, 2024. www.nccn.org/professionals/physician_gls/pdf/breast.pdf
- Lee UJ, Jones JS. Incidence of prostate cancer in male breast cancer patients: a risk factor for prostate cancer screening. Prostate Cancer Prostatic Dis. 2009;12:52-56.
- Abhyankar N, Hoskins KF, Abern MR, Calip GS. Descriptive characteristics of prostate cancer in patients with a history of primary male breast cancer—a SEER analysis. BMC Cancer. 2017;17:659.
- Satram-Hoang S, Ziogas A, Anton-Culver H. Risk of second primary cancer in men with breast cancer. Breast Cancer Res. 2007;9:R10.
- Tai YC, Domchek S, Parmigiani G, Chen S. Breast cancer risk among male BRCA1 and BRCA2 mutation carriers. J Natl Cancer Inst. 2007;99:1811-1814.
- Rudlowski C. Male breast cancer. Breast Care (Basel). 2008;3:183-189.
- American Cancer Society. Prostate Cancer Risk Factors. Accessed July 8, 2024. https://www.cancer.org/cancer/types/prostate-cancer/causes-risks-prevention/risk-factors.html
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): prostate cancer. Version 3.2024. March 8, 2024. Accessed June 3, 2024. www.nccn.org/professionals/physician_gls/pdf/prostate.pdf
- US Preventive Services Task Force. Prostate cancer: screening. May 8, 2018. Accessed January 30, 2023. www.uspreventiveservicestaskforce.org/uspstf/recommendation/prostate-cancer-screening
- Carter HB, Albertsen PC, Barry MJ, et al. Early detection of prostate cancer: AUA Guideline. J Urol. 2013;190:419-426.
- Lombardi P. Exemestane, a new steroidal aromatase inhibitor of clinical relevance. Biochim Biophys Acta. 2002;1587:326-337.
- Henry NL. Endocrine therapy toxicity: management options. Am Soc Clin Oncol Educ Book. 2014;e25-e30.
- King DS, Sharp RL, Vukovich MD, et al. Effect of oral androstenedione on serum testosterone and adaptations to resistance training in young men: a randomized controlled trial. JAMA. 1999;281:2020-2028.
- Leder BZ, Longcope C, Catlin DH, et al. Oral androstenedione administration and serum testosterone concentrations in young men. JAMA. 2000;283:779-782.
- Aromasin (exemestane) tablets, for oral use [prescribing information]. Pfizer; November 2021. Accessed April 23, 2022. www.accessdata.fda.gov/drugsatfda_docs/label/2021/020753s022lbl.pdf
- Mauras N, Lima J, Patel D, et al. Pharmacokinetics and dose finding of a potent aromatase inhibitor, aromasin (exemestane), in young males. J Clin Endocrinol Metab. 2003;88:5951-5956. Erratum in: J Clin Endocrinol Metab. 2004;89:732.
- Takagi K, Miki Y, Nagasaki S, et al. Increased intratumoral androgens in human breast carcinoma following aromatase inhibitor exemestane treatment. Endocr Relat Cancer. 2010;17:415-430.
- Gao L, Bao Z, Deng H, et al. The beneficial androgenic action of steroidal aromatase inactivators in estrogen-dependent breast cancer after failure of nonsteroidal drugs. Cell Death Dis. 2019;10:494.
- Ariazi EA, Leitão A, Oprea TI, et al. Exemestane’s 17-hydroxylated metabolite exerts biological effects as an androgen. Mol Cancer Ther. 2007;6:2817-2827.
- Bonomo M, Mingrone W, Brauchli P, et al. Exemestane seems to stimulate tumour growth in men with prostate carcinoma. Eur J Cancer. 2003;39:2111-2112.
- Shearer RJ, Davies JH, Dowsett M, et al. Aromatase inhibition in advanced prostatic cancer: preliminary communication. Br J Cancer. 1990;62:275-276.
- Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30:239-245.