Connecting Science to Practice
In this case study, we show a potential new treatment for
signet ring cell breast cancer (SRCBC) using ribociclib,
fulvestrant, and denosumab. Chemotherapy has been previously
shown to be ineffective for SRCBC. This case study
follows a single patient with stage IV SRCBC with metastasis
to the bones who received these medications for 2 years.
She has had stable disease 2.5 years after her first pain
symptoms and 24 months after her official diagnosis of
metastatic breast cancer. This regimen may provide a new
treatment option for SRCBC patients, potentially increasing
the lifespan and quality of life of those affected with
this disease.
Signet ring cell breast carcinoma (SRCBC) is a rare and aggressive type of adenocarcinoma, occurring in only 0.02% of cases of breast cancer.1 Although signet ring cell cancers are predominantly present in the gastrointestinal tract,2 this is a glandular cancer that can occur in any part of the body where glandular cells are present, such as in the breast.2 Of all types of signet ring cell cancer, breast cancer makes up 1.5% of samples.3 SRCBC cells are named for the cancer’s nucleus moving to the side of the cell by being pushed by cellular mucin, causing a resemblance to a signet ring.2 More than 80% of SRCBC is initially diagnosed at stage II or higher, with an average survival rate of 26% over 5 years in patients with stage IV SRCBC compared with 43% in patients with other non–triple-negative breast cancer types.1 These percents can be compared with those of patients with metastatic breast cancer, which represents approximately 3% to 6% of all breast cancers.4
Surgical treatment can be effective in patients with SRCBC if the disease has not metastasized; however, chemotherapy has not had any benefit in these patients.5 The typical treatments for patients with SRCBC include lumpectomy (49.5%), mastectomy (50.5%), radiation (40.7%), and chemotherapy (50%).1 The lack of response to chemotherapy in patients with SRCBC can be compared with the more than 86% disease-free 5-year survival rate for overall HER2-negative hormone-positive breast cancer for all oncotype scores.6
Patients with estrogen receptor (ER)-positive SRCBC have a higher survival rate than patients with ER-negative SRCBC; however, receiving antiestrogen therapy does not change the outcome in ER-positive patients with SRCBC.1 Per the National Cancer Database, the incidence of SRCBC is only 528 (0.02%) of almost 2.7 million cases of breast cancer.1 Bone-only SRCBC is extremely rare, with only 1 case reported in the literature to date.7 In that case, the patient had a radical mastectomy and received radiotherapy and chemotherapy. Despite receiving these treatments, the patient died after 14 months.7
Case Report
Our patient is a 63-year-old woman with stage IV SRCBC with metastasis to the bones. She was diagnosed with stage I SRCBC in May 2018 via a lumpectomy after having a suspicious finding in a screening mammogram in April 2018. The lumpectomy resected a 9-mm, grade 3 tumor with atypical cells that were focal SRCBC with mucinous features from the ductal origin. The pathology results showed that the patient had a Ki-67 level of 52.24, a HER2 level of 0, an ER positivity rate of 98.5%, a progesterone receptor (PR) positivity rate of 6.6%, a Ki-67 stain score of >20%, and an Oncotype DX test score of 25.
The patient was determined to have stage I SRCBC as a result of the patient’s sentinel node biopsy having no signs of cancerous cells. No chemotherapy was recommended based on the TAILORx report.6 Data from the TAILORx study showed no benefit to the addition of chemotherapy to hormone therapy in patients with ER-positive, HER2-negative disease and an oncotype score of ≤25.6 The patient received radiation therapy from June 2018 to July 2018, and anastrozole 1 mg daily was started in August 2018. The patient did not tolerate the treatment with anastrozole and had insomnia, irritability, and generalized achiness, which led her to stop receiving the therapy in June 2022. The patient’s choice to stop treatment with anastrozole was also attributed to her concerns about decreasing bone density and a lack of documented evidence about anastrozole benefiting patients with SRCBC.
Approximately 1 month after stopping treatment with anastrozole, the patient had back pain after trying to break the fall of a heavy elderly man. Several months later, the patient had continued pain and went to her provider, who ordered a plain-film x-ray in November 2022 that showed no lytic lesions. Although there were no signs of metastasis at that time, the patient still suspects that the pain was connected to her cancer.
The patient contacted her provider again when the back pain worsened and began to radiate down her left leg. Magnetic resonance imaging (MRI) of the spine was performed in March 2023, which showed a total spine diffuse marrow abnormality that was suspicious for widely metastatic disease. The MRI also showed a probable right rib fracture, L2 disc compression, and posterior L4-L5 disc degeneration on the left lateral side. The patient returned to the oncology clinic based on the MRI findings. The patient stated that she was not convinced that she had metastatic breast cancer because all of her mammograms after the biopsy were unremarkable from 2018 to 2023. A breast MRI in May 2023 also showed no signs of breast malignancy.
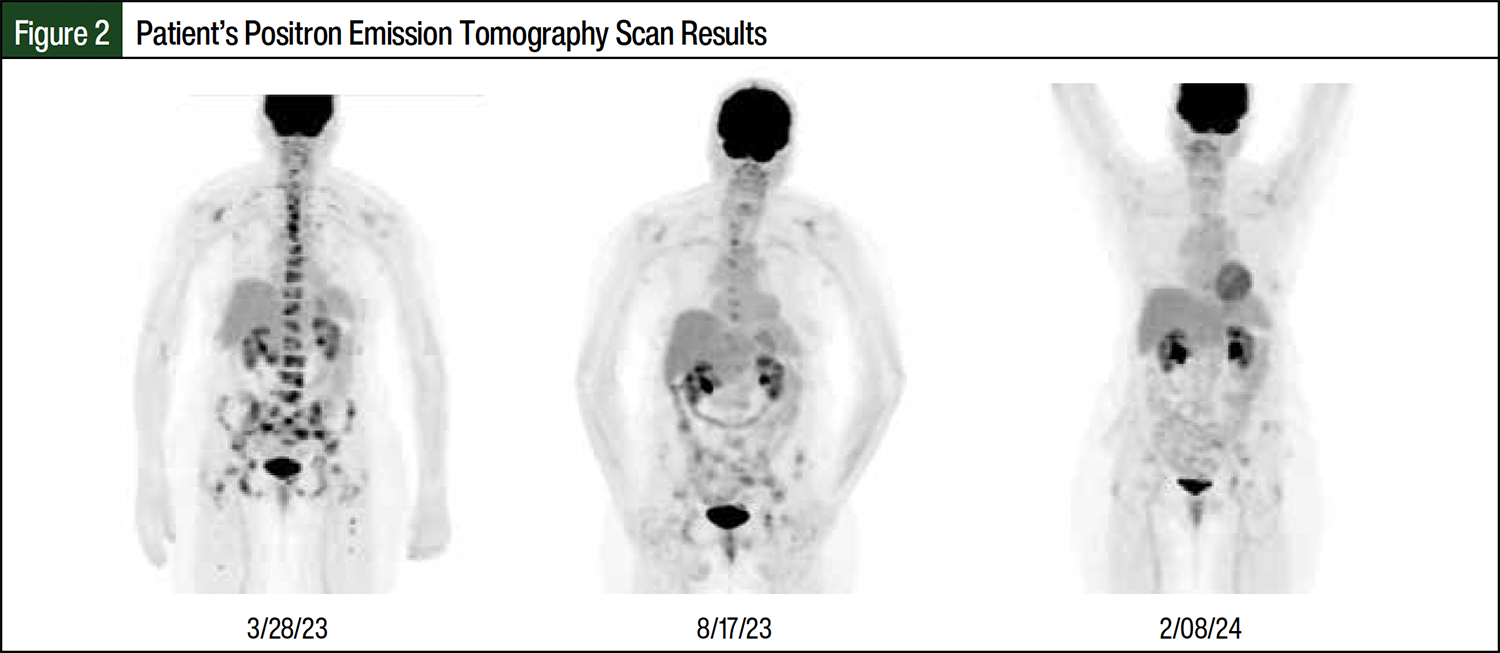
In March 2023, a positron emission tomography (PET) scan showed diffuse hypermetabolic osseous metastatic disease, which confirmed the findings of the spinal MRI report. Metastatic lesions were present in all parts of the spine, ribs, acetabulum, pelvis, shoulders, femurs, and skull. A breakthrough lesion was seen in the central spinal canal at the L3-L4 vertebrae, which was pressing on the spinal column and could explain the radiating leg pain. A bone biopsy was ordered to determine if this was a recurrence of SRCBC or a new primary cancer. In April 2023, an interventional radiology-guided bone lesion biopsy showed that metastatic carcinoma was present and was consistent with the patient’s history of ductal carcinoma. The patient had an ER positivity rate of 70%, a PR positivity rate of 0%, a HER2 level of +1, and a Ki-67 stain score of 30%. A brain MRI was also performed in April 2023 as a result of enhancing skull lesions on a PET scan, which was negative for brain lesions. The brain MRI supported the cervical spine lesions that were already discovered on the previous PET scan and cervical MRI.
The patient received 2 courses of 5-day palliative radiation for pain. The first course in April 2023 was for her left hip and leg pain, which was most likely coming from her L3 vertebra. On imaging, the left side of the L3 vertebra was encroaching on the nerve roots. The second session of palliative radiation was in June 2023 for pain in the left pubic bone area, which corresponded with the lesion on the inferior left ischium as seen on the March 2023 PET scan. Both radiation treatments resulted in marked improvement in the patient’s pain symptoms after intervention.
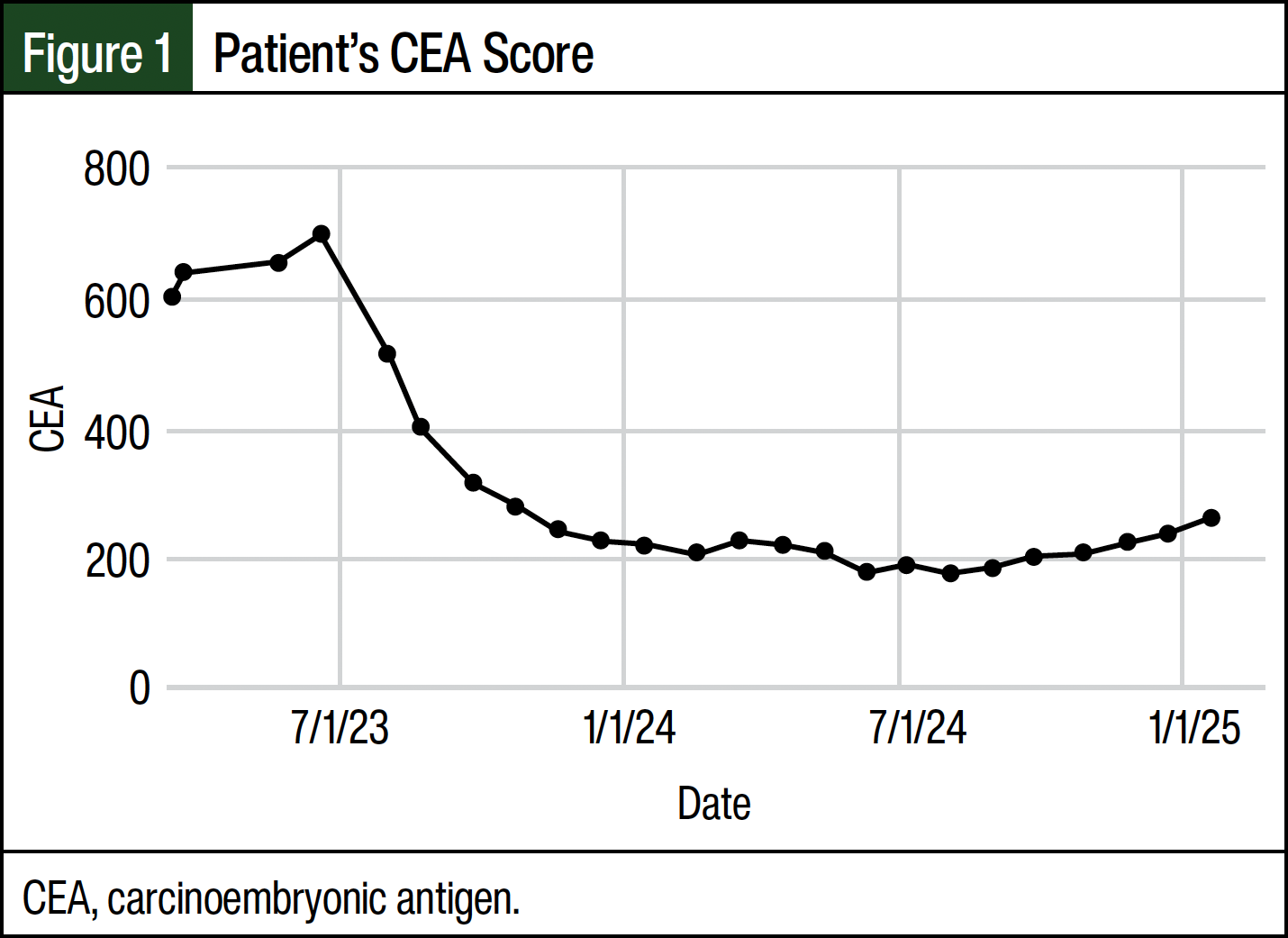
The patient’s further treatment course included abemaciclib 150 mg twice daily and monthly injections of fulvestrant 500 mg (divided into 2 injections) and denosumab 120 mg beginning April 2023. This course was determined according to the National Comprehensive Cancer Network’s treatment guidelines for ER-positive breast cancer.8 To monitor treatment, a complete blood count with differential and platelets, comprehensive metabolic panel, cancer antigen 27.29, and carcinoembryonic antigen (CEA) was drawn monthly starting in March 2023. This patient’s cancer antigen 27.29 level was normal, but her CEA level was abnormal; so, the CEA level was used to track the effectiveness of the patient’s treatments.
In May 2023, the patient’s treatment regimen was changed to ribociclib 600 mg daily with monthly injections of fulvestrant and denosumab because abemaciclib was not tolerated as a result of severe nausea, vomiting, and diarrhea. In September 2023, the dose of ribociclib was lowered to 400 mg daily because the patient’s absolute neutrophil count was 0.69 cells/mm3. As of the writing of this article, the patient has been receiving treatment for 20 months and is currently tolerating it well. She has had a remarkable decrease in her CEA level as well as a continual regression of cancer on her PET scans (Figure 1 and Figure 2). The CEA values in Figure 1 can be compared with the patient’s CEA level of 1.1 in 2019. Her pain scores dropped from an 8 to a 1 or 2, and she was able to resume activities of daily living. Before her diagnosis and treatment, the patient received consistent nonsteroidal anti-inflammatory drug therapy, used a walker to walk any distance, and received weekly acupuncture sessions for pain management. Now, she does not require any medication for pain. Her eosinophil rate has decreased from 10.9% before treatment to 2.2% after treatment.
Discussion
To our knowledge, there was only 1 previously published case report of stage IV bone-only SRCBC.7 In patients with grade 1 ER-positive breast cancer, there is a recurrence rate of 5.8% over 5 years and 14.3% over 25 years.9 These recurrence rates can be compared with those of patients with grade 3 ER-positive breast cancer when examined over all stages of 14.1% over 5 years and 23.5% over 25 years.9 Patients with ER-positive SRCBC have had an improved outcome compared with patients with ER-negative SRCBC, but antiestrogen therapy has provided no benefit in patients with SRCBC.1
In our patient, her back pain started within 1 month of stopping treatment with anastrozole. This led the patient to suspect that her cancer was never fully eliminated and was instead suppressed by the antiestrogen therapy. The ER positivity rate of 98.5% in this patient might have been a factor in her outcomes, despite antiestrogen therapy not helping previous patients with SRCBC. To our knowledge, our case report is the first to show that the combination of ribociclib, fulvestrant, and denosumab has led to a remarkable improvement in CEA levels, the regression of cancer as seen on multiple follow-up PET scans, and a decrease in pain in a patient with SRCBC.
Conclusion
SRCBC is a very rare and aggressive form of breast cancer. To our knowledge, bone-only SRCBC has only been described in the literature 1 other time. This case report shows that ribociclib, fulvestrant, and denosumab is a potentially effective treatment regimen for patients with SRCBC. Currently, the patient in this report is alive and feels well, despite having widely metastatic bone disease. She has had stable disease 2.5 years after her first pain symptoms and 24 months after her official diagnosis of metastatic breast cancer by MRI. Her PET scans and CEA levels confirm that the cancer is regressing with treatment with immunomodulators combined with antiestrogens and monoclonal antibodies.
Dr Ford Nenadich, Ms Nenadich, and Dr Shoua have no conflicts of interest to report.
References
- Mehdi M, Kong AL, Frebault J, et al. Prognostic outcomes of signet ring cell carcinoma of the breast. J Surg Res. 2021;264:138-148.
- Zheng J, Liu J, Yang W, et al. The clinicopathological and immunohistochemical features of breast carcinomas with signet-ring-cell differentiation. World J Surg Oncol. 2023;21:181.
- Wu SG, Chen XT, Zhang WW, et al. Survival in signet ring cell carcinoma varies based on primary tumor location: a SEER database analysis. Expert Rev Gastroenterol Hepatol. 2018;12:209-214.
- Daily K, Douglas E, Romitti PA, Thomas A. Epidemiology of de novo metastatic breast cancer. Clin Breast Cancer. 2021;21:302-308.
- Wang S, Zhang Y, Yin F, et al. Prognostic analysis of primary breast signet ring cell carcinoma and mucinous breast adenocarcinoma: a SEER population-based study. Front Oncol. 2021;11:783631.
- Sparano JA, Gray RJ, Makower DF, et al. Adjuvant chemotherapy guided by a 21-gene expression assay in breast cancer. N Engl J Med. 2018;379:111-121.
- Ma S, Leckey BD Jr, Zhang WL, et al. Signet-ring cells in the bone marrow as an indication of cryptic metastasis of breast carcinoma: a case report. Medicine (Baltimore). 2019;98:e14883.
- Gradishar WJ, Moran MS, Abraham J, et al. NCCN Clinical Practice Guidelines in Oncology: Breast Cancer, Version 3.2022. J Natl Compr Canc Netw. 2022;20:691-722.
- Colleoni M, Sun Z, Price KN, et al. Annual hazard rates of recurrence for breast cancer during 24 years of follow-up: results from the International Breast Cancer Study Group Trials I to V. J Clin Oncol. 2016;34:927-935.