Connecting Science to Practice
This case describes a patient who developed rhabdomyolysis
after starting palbociclib, a first-line treatment for estrogen
receptor–positive, HER2-negative breast cancer, while on
simvastatin for hypercholesterolemia. Palbociclib is a weak
CYP3A4 inhibitor, and CYP3A4 metabolizes simvastatin.
Thus, this combination can increase simvastatin levels, raising
the risk for serious adverse events (AEs) such as rhabdomyolysis.
Although simvastatin’s prescribing information
does not list concomitant use of palbociclib as contraindicated,
this case and a review of the literature show the potential
risk with palbociclib and other cyclin-dependent kinase
(CDK)4/6 inhibitors, highlighting the need for evaluating
the risks versus benefits of using statins and CDK4/6 inhibitors
and monitoring these patients for AEs.
Letrozole and palbociclib are currently approved as first-line systemic therapy for estrogen receptor (ER)–positive, HER2-negative, recurrent unresectable or metastatic breast cancer, and this combination was initiated as first-line therapy in our patient.1 Alternatively, palbociclib may also be used with other aromatase inhibitors, such as anastrozole or exemestane, or with fulvestrant and remains a viable option in subsequent lines of therapy if not used in the first-line setting.1
The PALOMA-2 trial showed significantly longer progression-free survival with palbociclib combined with letrozole compared with letrozole alone in postmenopausal patients with advanced, ER-positive, HER2-negative breast cancer.2 Letrozole is an aromatase inhibitor approved in 2001; common adverse events (AEs) include hot flashes, fatigue, weight gain, hypercholesterolemia, bone mineral density loss, ischemic cardiovascular events, and musculoskeletal effects such as arthralgia, ostealgia, and myalgia.3,4 Palbociclib is a selective cyclin-dependent kinase (CDK)4/6 inhibitor approved in 2015; common AEs include dermatologic and gastrointestinal AEs, transaminitis, neuro-musculoskeletal effects (such as asthenia), and pancytopenia (in particular, leukopenia and neutropenia). Palbociclib is considered a weak cytochrome P450 3A4 (CYP3A4) inhibitor.5,6
Simvastatin is a 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor; it is approved for hypercholesterolemia, and AEs include hepatotoxicity and musculoskeletal effects, such as myalgia, myopathy, and rhabdomyolysis.7 As a CYP3A4 substrate, simvastatin is similar to other drugs in its class, and there is currently a precaution for concomitant use of simvastatin with strong CYP3A4 inhibitors because they increase the serum concentration of statins and subsequently the risk for AEs.7 When using weak CYP3A4 inhibitors, it is crucial to monitor for significant interactions and AEs because they can still cause severe or life-threatening AEs despite their perceived potency.
We describe the case of a 65-year-old woman who has a long history of simvastatin use for hypercholesterolemia. She received palbociclib and letrozole for her breast cancer and subsequently presented with rhabdomyolysis 2 months later. The patient’s condition improved slightly after a 7-day hospital stay, but the patient died 2 months later as a result of acute liver failure, hyponatremia, and leukocytosis, all of which may have resulted from rhabdomyolysis, although progression of disease or other factors could not be ruled out.
Case Report
The patient was a 65-year-old woman with a medical history of hypertension, hypercholesterolemia, and generalized anxiety disorder. Her medications included 100 mg of losartan daily, 40 mg of simvastatin daily, and 0.5 mg of alprazolam as needed for sleep; she had been taking all of these medications for several years. In June 2021, she presented with postmenopausal bleeding for 3 months and light menstrual cramping. The patient was scheduled for a total abdominal hysterectomy and bilateral salpingo-oophorectomy and was diagnosed with grade 1 endometrial adenocarcinoma. A computed tomography (CT) scan of her abdomen and pelvis confirmed the presence of a diffusely heterogeneous liver with multiple faint nodular hypodense regions scattered throughout, which appeared to be metastases. Liver biopsy testing showed metastatic carcinoma suggestive of primary breast cancer. Final pathology testing showed stage IIIC1 grade 2 endometrial adenocarcinoma and probable stage IV breast cancer. A positron emission tomography/CT scan showed an irregular hypermetabolic mass in the upper outer quadrant of the left breast, abnormal left axillary lymph nodes, and 2 hyperbolic bone lesions in the left iliac bone and 1 in the right femoral neck, suggesting metastatic bone disease. A bilateral mammogram with breast tomosynthesis was conducted and revealed a dominant irregular mass and ≥1 additional mass with multifocal disease in the left breast, abnormal level I left axillary lymph nodes, and a benign cyst on the right breast. In July 2021, our final diagnosis was ER-positive, HER2-negative stage IV metastatic breast cancer.
The patient was educated regarding multiple treatment options and opted to proceed with 500 mg of fulvestrant intramuscularly on days 1, 15, and 29, followed by 500 mg of fulvestrant intramuscularly once a month and zoledronic acid every 12 weeks. Another positron emission tomography/CT scan was obtained in February 2023, and it showed progressive osseous metastatic disease in the spine, sacrum, and hips. Increased uptake associated with the right axillary lymph nodes was also noted.
Because the patient’s disease progressed, in March 2023, her therapy was changed to 2.5 mg of letrozole daily and 100 mg of palbociclib daily on days 1 to 21, in 28-day cycles (palbociclib was reduced to 100 mg instead of 125 mg because of the patient’s frailty and hepatic involvement), and 4 mg of zoledronic acid every 12 weeks was continued.
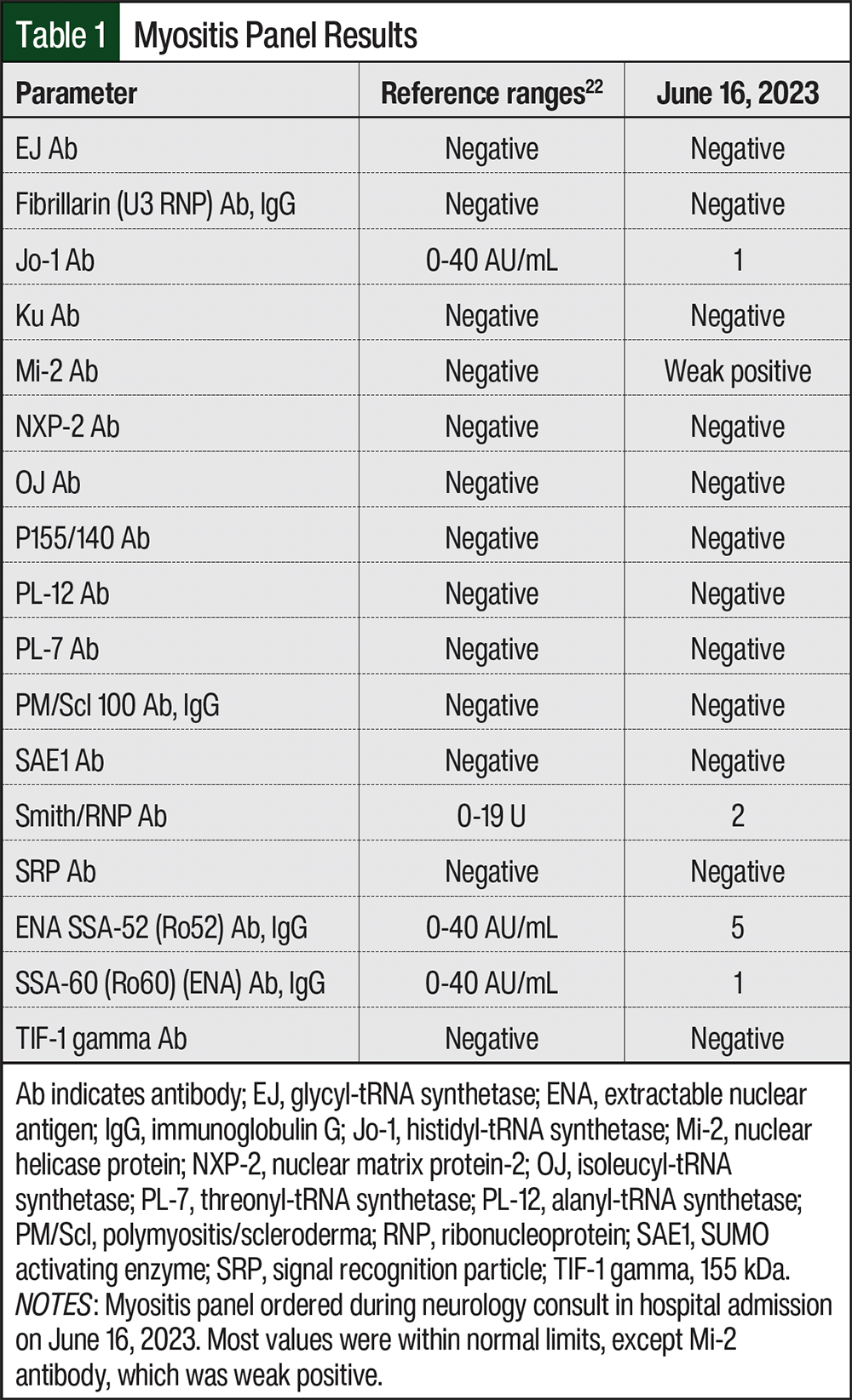
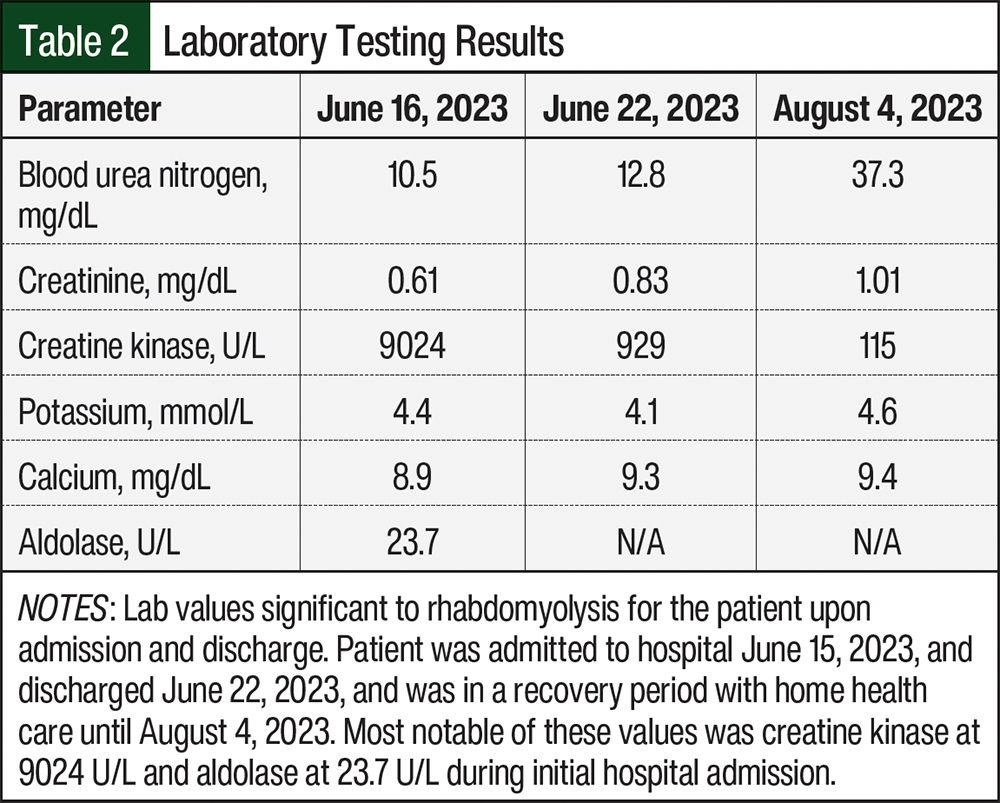
On June 15, 2023, the patient was admitted to the hospital for acute myopathy. She presented with rapid progression of weakness in the proximal muscles of her lower extremities that began 5 days before hospitalization. She reported having difficulty standing and almost immediate fatigue when walking, although before being hospitalized, the patient was able to walk independently. Laboratory studies revealed a creatine phosphokinase (CPK) level of 9024 U/L and an aldolase level of 23.7 U/L, suggestive of rhabdomyolysis. A myositis panel was also done to confirm that the muscle weakness was not caused by an autoimmune disease, specifically necrotizing myopathy, which was high on their differential diagnosis because of the rapidly progressive nature of her muscle weakness (Table 1). A neurology consulting team determined that although our patient had diffuse spondylosis and bony metastasis, she did not have critical stenosis of the central canal or nerve roots to explain this syndrome; and with a CPK level >5000 U/L, her diagnosis was consistent with myopathy. The neurology team continued to suspect a possible autoimmune process (eg, polymyositis or dermatomyositis), despite a negative myositis panel, and considered rhabdomyolysis, statin-induced myopathy, autoimmune myositis, and critical illness myopathy. The neurology team believed that this could have been triggered by simvastatin, even though she had been receiving it for years, and they did not recognize the relationship to her chemotherapeutic agents. All potentially implicated agents were discontinued, and the patient was given a drug holiday from palbociclib, letrozole, and simvastatin. The patient then received 4 rounds of plasmapheresis, aggressive hydration, and pulse methylprednisolone. CPK levels subsequently trended downward (Table 2). The patient’s chronic medications were cross-checked and revealed an interaction between simvastatin and palbociclib. The oncology and neurology teams concurred that the diagnosis was rhabdomyolysis precipitated by the interaction. The patient was discharged in June 2023 to a rehabilitation facility and scheduled for follow-up with an oncologist for further management; palbociclib, letrozole, and simvastatin remained on hold.
Home health services were involved per patient request 2 weeks after discharge from the hospital. Initial evaluation showed weight loss of 20 lb in the past 3 months, impaired vision, disorganized thinking, numbness in the hands and feet, lower back pain, bladder incontinence, shortness of breath after walking >20 feet, and a need for assistive equipment for gait and transfer. Physical therapy was started to improve the patient’s strength, gait, and balance. Occupational therapy was also started to address functional deficits and improve skills for activities of daily living. The patient was instructed to complete home exercises, which progressively increased her walking distance and improved standing strength.
Two months later, in August 2023, the patient was admitted to the hospital with fulminant hepatic failure caused by disease progression, severe symptomatic hyponatremia, and leukocytosis. She appeared jaundiced and had dark yellow urine. After discussions with the oncologist and hospitalist care team, the patient decided to stop aggressive treatment and pursue hospice care.
Discussion
The combination of palbociclib and letrozole is considered standard-of-care, first-line therapy for the treatment of advanced hormone receptor–positive, HER2-negative, recurrent unresectable or metastatic breast cancer.1 Palbociclib alone is not known to cause rhabdomyolysis. Common AEs of palbociclib plus letrozole in the PALOMA-2 trial included neutropenia, leukopenia, fatigue, nausea, asthenia, arthralgia, and alopecia.2
After reviewing the medications that the patient received at home, it appeared that simvastatin was the only medication that could have led to rhabdomyolysis.8 Rhabdomyolysis comes from the breakdown of muscle, which leads to muscle weakness and an increase of creatine kinase. Statins given in higher doses have shown association with rhabdomyolysis.9 Simvastatin is metabolized by CYP3A4 in the liver.7 Some studies have shown that by inhibiting CYP3A4, an increase of statin concentration in the body has been found.10,11
Palbociclib and letrozole were the only new drugs that had been initiated before the patient developed symptoms. Palbociclib is extensively metabolized in the liver and is a weak CYP3A4 inhibitor, which may increase serum concentrations of the active metabolite(s) of simvastatin.6,7 However, the pivotal phase 3 trial of palbociclib plus letrozole excluded patients treated with any strong CYP3A4 inhibitors and inducers and select moderate CYP3A4 inhibitors and inducers, and highly discouraged use of CYP3A4-sensitive substrates.2 This could be why there is no accompanying warnings for either drug and why more frequent objective monitoring has not been recommended.
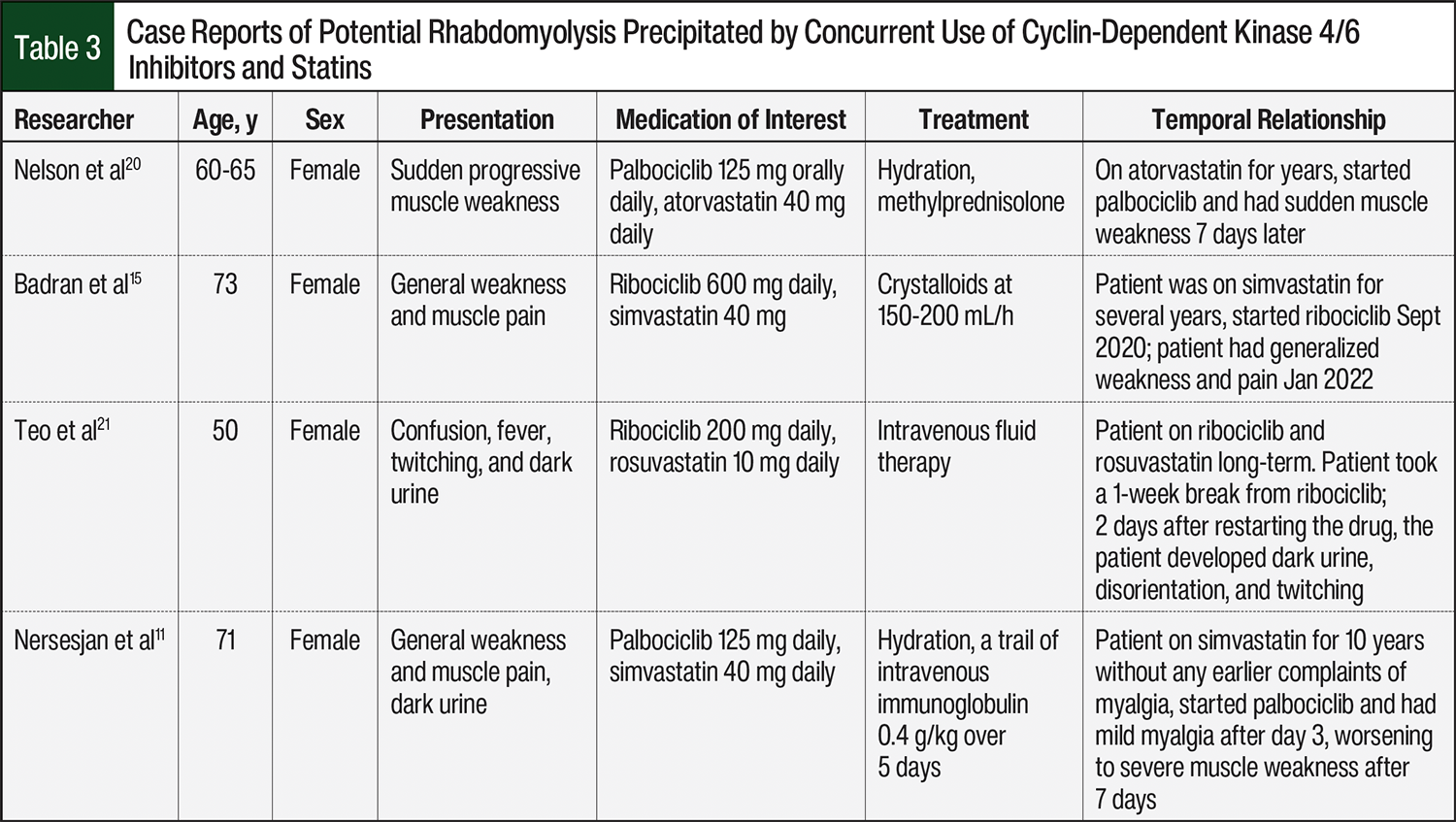
Case reports of potential rhabdomyolysis precipitated by concurrent use of CDK4/6 drugs and statins are summarized in Table 3. Our case specifically involves palbociclib, a weak CYP3A4 inhibitor, and extrapolation to other CDK4/6 inhibitors with differing CYP3A4-inhibition profiles, such as ribociclib, should be made cautiously.
One case report described a patient receiving 20 mg of simvastatin and 400 mg of danazol daily.10 Danazol is a strong inhibitor of CYP3A4.10 After a percutaneous coronary intervention, the patient’s daily dose of simvastatin was increased to 40 mg, and danazol was increased to 600 mg daily.10 The patient presented to the emergency department 21 days after initial discharge with signs and symptoms of rhabdomyolysis.10 The concomitant administration of danazol and simvastatin is now contraindicated.7
Another case study reported a patient receiving 200 mg of ketoconazole orally 3 times daily and 10/80 mg of ezetimibe/simvastatin orally daily.13 Ketoconazole is a strong inhibitor of CYP3A4.13,14 After an increase to 400 mg of ketoconazole orally 3 times daily, the patient presented to the outpatient clinic 1 month later with blood in the urine and weakness, and the patient was admitted to the hospital 2 days later and diagnosed with rhabdomyolysis.13 This case report is one of many that confirms the contraindication of simvastatin and concomitant administration of a strong CYP3A4 inhibitor in the simvastatin prescribing information.7 Although these cases involved strong CYP3A4 inhibitors, more recent case reports suggest possible interactions and rhabdomyolysis with weaker CYP3A4 inhibitors, such as the CDK4/6 inhibitors palbociclib and ribociclib, when coadministered with statins.15,16 There are several case reports of rhabdomyolysis in patients receiving ribociclib and simvastatin,15,16 and a case report by Poumeaud and colleagues involved palbociclib with simvastatin.17 In these cases, symptoms resolved after discontinuation of the CDK4/6 inhibitor and/or statin.15-17 These cases suggest a possible pharmacokinetic interaction, likely mediated by CYP3A4 inhibition, which may increase statin exposure and AEs.15-17
Genetic variation in SLCO1B1, which encodes the hepatic transporter OATP1B1, has been strongly associated with altered statin pharmacokinetics.18 Patients carrying reduced-function alleles (eg, SLCO1B1 *5/*5) exhibit higher statin plasma concentrations and are at increased risk for statin-associated myopathy.19 Although pharmacogenomic testing was not performed in our case, undiagnosed SLCO1B1 polymorphisms may have contributed to the observed rhabdomyolysis.18
Physicians and pharmacists should check for drug–drug interactions with all of a patient’s medications at the initiation of oral chemotherapy agents. With any simvastatin and anticancer therapy interactions, regardless of recommendations on the prescribing information, a baseline CPK level should be obtained because concurrent use may increase the risk for myopathy.19 Based on our case report, where even a reduced starting dose of a weak CYP3A4 inhibitor was sufficient to precipitate this interaction, we recommend that if the baseline CPK level is within normal limits, treatment should proceed with ongoing monitoring of CPK levels. Patient risk factors for myopathy should also be considered, such as age, additional interacting drugs, kidney impairment, and increased statin dose, which could compound the interaction risk.19 Patients should be educated on signs and symptoms of myopathy, such as muscle pain, weakness, fatigue, and monitor these daily.19 If the baseline CPK is elevated, clinicians should assess the risk-benefit ratio of continuing the current statin and anticancer therapy, particularly when both agents are known to affect CYP3A4 metabolism. In such situations, switching to a statin that is not primarily metabolized by CYP3A4 (eg, pravastatin or rosuvastatin) or adjusting the statin dose may reduce the risk for myopathy. Consideration should also be given to selecting anticancer therapies with minimal CYP3A4-interaction potential, when clinically appropriate.
Conclusion
This case report highlights the interaction between simvastatin and palbociclib that can cause severe rhabdomyolysis. The interaction likely stems from palbociclib being a weak CYP3A4 inhibitor, which can cause toxic plasma concentrations of simvastatin, a CYP3A4 substrate. This is one of several case reports that outline rhabdomyolysis caused by the concomitant administration of simvastatin and palbociclib. Addition of a warning to the prescribing information of simvastatin for the concomitant use of weak inhibitors of CYP3A4 should be considered. If simvastatin and palbociclib are to be used in combination, there is a need for additional baseline and subsequent monitoring.
Author Disclosure Statement
Dr Hosick, Dr Abi Karam, Dr Korah, Dr Bidne, and Dr Tamer have no conflicts of interest to report.
References
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): breast cancer. Version 4,2025. April 17, 2025. www.nccn.org/professionals/physician_gls/pdf/breast.pdf
- Finn RS, Martin M, Rugo HS, et al. Palbociclib and letrozole in advanced breast cancer. N Engl J Med. 2016;375:1925-1936.
- US Food and Drug Administration. Femara approval letter. January 10, 2001. Accessed January 2, 2024. www.accessdata.fda.gov/drugsatfda_docs/nda/2001/20-726S006_Femara_Approv.pdf
- Femara (letrozole) tablets, for oral use [prescribing information]. Novartis Pharmaceuticals Corporation; December 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2024/020726s043lbl.pdf
- US Food and Drug Administration. Palbociclib (Ibrance). March 31, 2017. Accessed January 10, 2024. www.fda.gov/drugs/resources-information-approved-drugs/Palbociclib-ibrance
- Ibrance (palbociclib) capsules, for oral use [prescribing information]. Pfizer Labs; September 2023. Accessed January 2, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2025/207103s020lbl.pdf
- Zocor (simvastatin) tablets, for oral use [prescribing information]. Organon & Co; August 2023. Accessed January 2, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2023/019766s104lbl.pdf
- Letrozole - Palbociclib - Simvastatin - Alprazolam - Apixaban - Ascorbic Acid - Cholecalciferol - Cyanocobalamin - Losartan - Multivitamin - Ondansetron - Oxycodone. Interactions. Lexi-Drugs. Lexicomp online. Wolters Kluwer. Accessed January 2, 2024. http://online.lexi.com
- Lagampan C, Poovorawan N, Parinyanitikul N. Lactic acidosis, a potential toxicity from drug–drug interaction related to concomitant ribociclib and metformin in preexisting renal insufficiency: a case report. Cancer Rep. 2022;5:e1575.
- Stankovic I, Vlahovic-Stipac A, Putnikovic B, et al. Concomitant administration of simvastatin and danazol associated with fatal rhabdomyolysis. Clin Ther. 2010;32:909-914.
- Nersesjan V, Hansen K, Krag T, Duno M, Jeppesen TD. Palbociclib in combination with simvastatin induce severe rhabdomyolysis: a case report. BMC Neurol. 2019;19:1-7.
- Palbociclib - Simvastatin. Interactions. Lexi-Drugs. Lexicomp online. Wolters Kluwer. Accessed March 19, 2024. http://online.lexi.com
- Watkins JL, Atkinson BJ, Pagliaro LC. Rhabdomyolysis in a prostate cancer patient taking ketoconazole and simvastatin: case report and review of the literature. Ann Pharmacother. 2011;45:e9.
- Nizoral (ketoconazole) tablets [prescribing information]. Janssen Pharmaceuticals. Accessed January 2, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2013/018533s040lbl.pdf
- Badran O, Abu Amna M, Turgeman I, Bar-Sela G. Rhabdomyolysis induced by the interaction between ribociclib and statins–case report and literature review. Breast Cancer (Dove Med Press). 2023;15:47-50.
- Streicher C, Daulange A, Madranges N, Vayre L. Severe rhabdomyolysis induced by possible drug-drug interaction between ribociclib and simvastatin. J Oncol Pharm Pract. 2021;27:722-726.
- Poumeaud F, Fontanier A, Dion J, et al. Severe toxic rhabdomyolysis under combined palbociclib and simvastatin treatment: a case report. Front Oncol. 2022;12:1026434. Erratum in: Front Oncol. 2023;13:114399.
- Ramsey LB, Johnson SG, Caudle KE, et al. The clinical pharmacogenetics implementation consortium guideline for SLCO1B1 and simvastatin-induced myopathy: 2014 update. Clin Pharmacol Ther. 2014;96:423-428.
- Simvastatin. Lexi-Drugs. Lexicomp online. Wolters Kluwer. Accessed April 7, 2024. http://online.lexi.com
- Nelson KL, Stenehjem D, Driscoll M, Gilcrease GW. Fatal statin-induced rhabdomyolysis by possible interaction with palbociclib. Front Oncol. 2017;7:150.
- Teo SW, Hayes T, Gome J. Ribociclib may potentiate rosuvastatin effect in causing late onset rhabdomyolysis. BMJ Case Rep. 2023;16:e255632.
- Mayo Clinic Laboratories. Extended Myositis Panel reference values. WMCG Laboratory Test Catalog; 2025.