Oral oncolytic therapy is increasingly prescribed for patients with hematologic malignancies, but several barriers prevent or delay medication initiation, including insurance approval, financial assistance, limited distribution drug networks, and off-label use.1-4 Many oral therapies require insurance approval before they can be dispensed, and patients frequently need financial assistance for copays once a medication is approved.1-4
Because of serious potential safety concerns, the US Food and Drug Administration (FDA) requires some medications to enroll in the Risk Evaluation and Mitigation Strategy (REMS) program, which can create additional delays in medication access.5 Oral oncolytic agents are often being used off label (ie, beyond their FDA-approved indications), as increasing numbers of oral agents are becoming available and the evidence for their use is expanding. However, off-label prescriptions are often denied by the patient’s insurance company, which requires an appeal with supporting documentation for approval.6
These medication access requirements (ie, prior authorizations, appeals, financial assistance enrollment) are time-consuming for the clinic staff. Because of these barriers, patients often experience delays of more than 1 week before starting therapy after the prescription is generated.1 Patients using oncology clinics without help in navigating this process are subject to further delays and high out-of-pocket costs, which can lead to higher rates of medication abandonment.7 Although few studies have explored the association between the time to treatment initiation and mortality for patients with hematologic malignancies, delaying the initiation of oral oncolytic therapy for those with solid tumors is associated with adverse mortality and clinical outcomes.8-11
To navigate insurance requirements and facilitate timely access to therapy, some health systems have developed integrated specialty pharmacies, in which a clinical pharmacist is embedded in a specialty clinic as a member of the healthcare team.12 With access to patients’ electronic health records, the pharmacist completes documentation requirements and safety monitoring for patients’ prescribed specialty agents.12 Medications are then shipped overnight from the on-site pharmacy to patients in most states in the United States. However, some oral oncolytic agents are limited distribution drugs, which restricts their fulfillment to be completed by designated specialty pharmacies.13 Thus, the clinic pharmacist and staff must send limited distribution drug prescriptions to external pharmacies to dispense and to send to the patient. More information is needed to understand the impact of this distribution model on the time to treatment initiation.
Our objective was to compare the time to access oral oncolytic therapy between limited distribution drugs (which our health system was unable to dispense because of manufacturer or insurance restrictions) and non–limited distribution drugs (which were dispensed from our pharmacy) at a large academic health system in the southeast United States. We also tested whether access time to limited distribution drug prescriptions decreased after integrating a clinical pharmacist into the hematology clinic.
Method
This study was conducted at the outpatient hematology clinic at Vanderbilt University Medical Center (VUMC), Nashville, TN, a large academic health system. The hematology clinic treats patients with benign and malignant hematologic conditions; patients with solid tumors and other cancers are treated at the separate oncology clinic at VUMC, which also has an integrated pharmacist and technician.
An integrated specialty pharmacy currently services patients prescribed specialty medication from 23 outpatient clinics at VUMC. Pharmacists and pharmacy technicians are embedded in these clinics, and salaries are paid by the specialty pharmacy. Before specialty pharmacist integration, no outpatient oncology/hematology presence existed at the cancer center. The specialty pharmacy services approximately 300 to 400 patients with hematologic malignancies who are receiving oral oncolytic therapy at any given time.
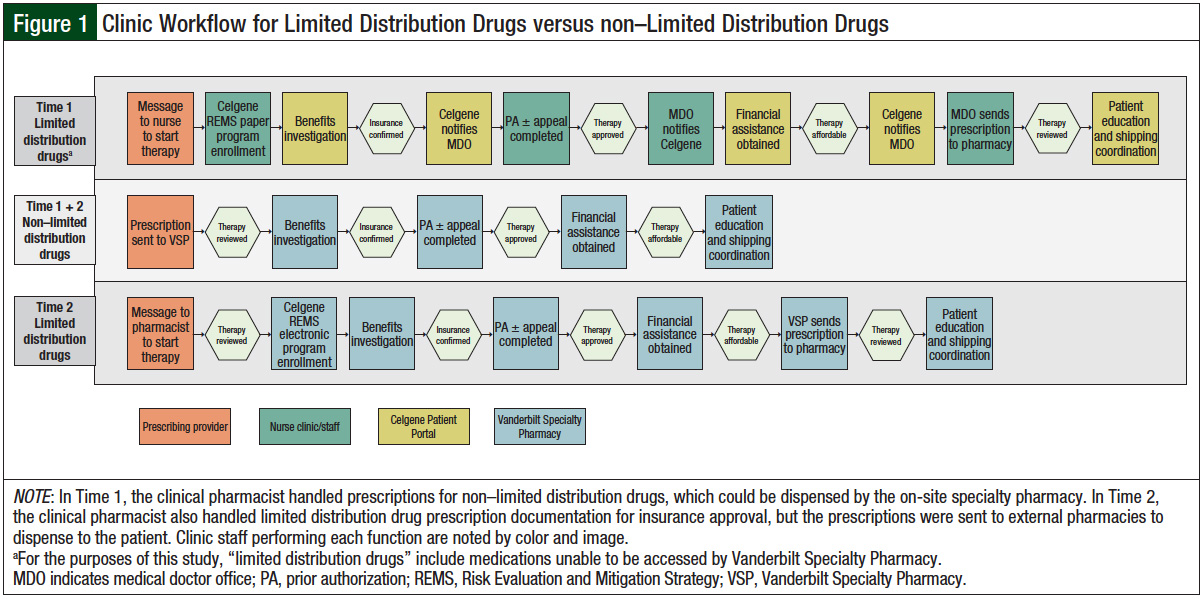
In September 2015, a clinical pharmacist and a pharmacy technician were integrated into the hematology clinic to manage the prescriptions for drugs that can be dispensed by the integrated Vanderbilt Specialty Pharmacy (ie, non–limited distribution drug prescriptions). From September 2015 to May 2016, the pharmacist and pharmacy technician completed prior authorizations, appeals, and patient financial assistance for non–limited distribution drug prescriptions.
During that period, for the 2 hematology specialty drugs that could not be dispensed by the Vanderbilt Specialty Pharmacy because of manufacturer or insurance restrictions (ie, limited distribution drug prescriptions), clinic nurses handled these tasks, including REMS-required documents, which were completed on paper forms. Nurses were dependent on outside pharmacies or on manufacturer patient assistance programs to assist with conducting benefit investigations for insurance approval. This led to significant delays between the decision to treat a patient and the date of insurance approval of prescribed therapies.
Beginning in June 2016, the clinical pharmacist and the pharmacy technician managed prescription and REMS requirement enrollment for limited distribution drug prescriptions as well, which were completed on the manufacturer’s online portal. After completing the access and safety requirement documentation for limited distribution drugs, electronic prescriptions were sent to outside specialty pharmacies to dispense and ship the medication to the patient. The clinic workflow for limited distribution drug versus non–limited distribution drug prescriptions is shown in Figure 1.
We conducted a retrospective review of oral oncolytic agents that were prescribed to adult patients by providers at the VUMC hematology clinic between September 2015 and September 2017. All prescriptions were eligible for inclusion unless they met the exclusion criteria below. Prescriptions were excluded if the shipment date was unobtainable, if the patient was uninsured, received free drug samples, or received medication solely through the manufacturer’s patient assistance program. We collected data from pharmacy claims and electronic health records; additional information for limited distribution drug prescriptions was transcribed from the manufacturer’s REMS portal into the patient’s medical record. Vanderbilt University Institutional Review Board approved the study.
We collected the age, sex, and race of patients who met the study eligibility criteria. For each prescription included in the study, we collected the drug name, specific indication, insurance type, use of financial assistance, whether the drug was prescribed in combination with other drugs and/or for an off-label indication, and whether the prescription required an insurance appeal (and if so, whether the appeal was successful). We also collected the date of the treatment decision, date the prior authorization was complete, date of insurance approval, date of medication shipment to patient, and date of an insurance appeal if needed. Shipment dates were unavailable for some prescriptions dispensed by external pharmacies; therefore, those were excluded from the analysis.
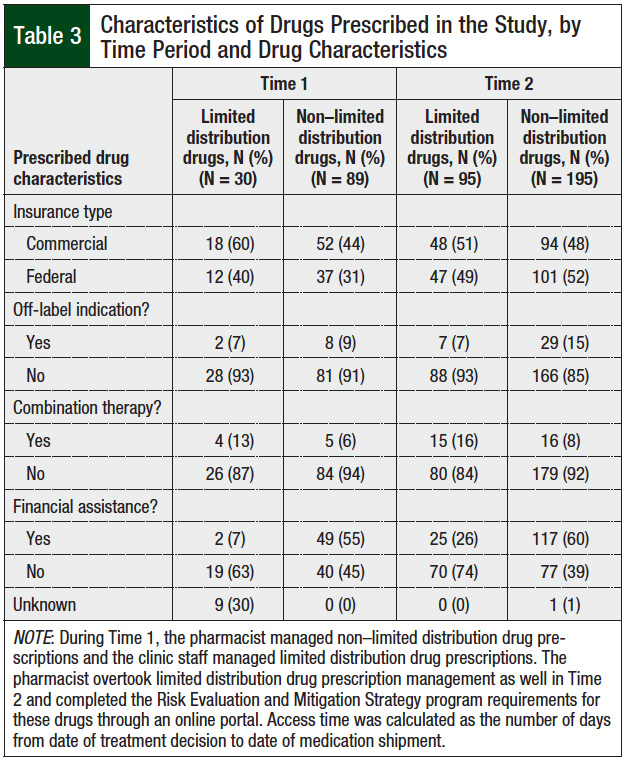
The prescriptions were stratified by the time period, with Time 1 representing September 2015 to May 2016 and Time 2 representing June 2016 to September 2017; and by the drug type, defined as limited distribution drug or non–limited distribution drug. Lenalidomide and pomalidomide were classified as limited distribution drugs; these are the only 2 hematologic specialty drugs that Vanderbilt Specialty Pharmacy does not have access to dispense, because of manufacturer restrictions; all other drugs were considered non–limited distribution drugs and could be dispensed by Vanderbilt Specialty Pharmacy.
Many specialty pharmacies are not authorized to dispense many limited distribution drugs. We evaluated the impact of only 2 limited distribution drugs; the impact of multiple limited distribution drugs may have a larger negative impact on access time. The study data were stored and managed using REDCap (Research Electronic Data Capture), hosted at Vanderbilt University.14
The primary outcome measure was time to medication access, defined as the number of days between the treatment decision date and the medication shipment date. Secondary outcome measures were the number of days from the treatment decision date to the date of prior authorization submission and date of insurance approval.
Statistical Analysis
Categorical variables were described using frequency distributions, and continuous variables were summarized using mean, standard deviation, median, and interquartile range (IQR). We calculated the median of time (in days) from the date of treatment decision to the date of prior authorization completion, the date of insurance approval, and the date of medication shipment.
Proportional odds logistic regression analysis was used to test whether medication access time was associated with a specific time period (Time 1 or Time 2), drug type (limited distribution drug or non–limited distribution drug), financial assistance use (yes or no), and off-label indication (yes or no). We hypothesized that the time to medication access was longer for limited distribution drug than for non–limited distribution drug prescriptions, and that the access time would decrease more rapidly from Time 1 to Time 2 for limited distribution drugs than for non–limited distribution drugs; therefore, we tested for an interaction between drug type and time period. The data were analyzed using the programming language R version 3.5.1 (R Foundation for Statistical Computing, Vienna, Austria).
Results
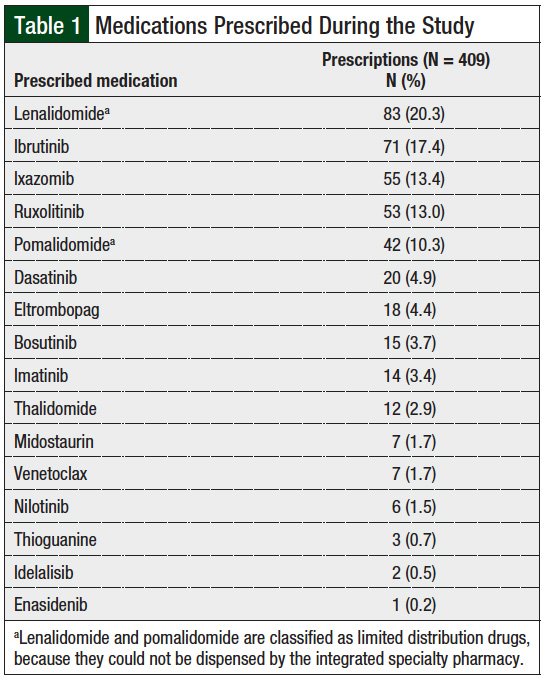
The study included 366 patients, most of whom were white (86%) women (54%), with a median age of 63 years. A total of 37 patients had prescriptions for multiple drugs during the study period, resulting in a total of 409 prescriptions (Table 1). In all, 69% of the prescriptions were for non–limited distribution drugs, and the most common prescriptions across the 2 time periods were for lenalidomide (20%), ibrutinib (17%), ixazomib (13%), ruxolitinib (13%), and pomalidomide (10%).
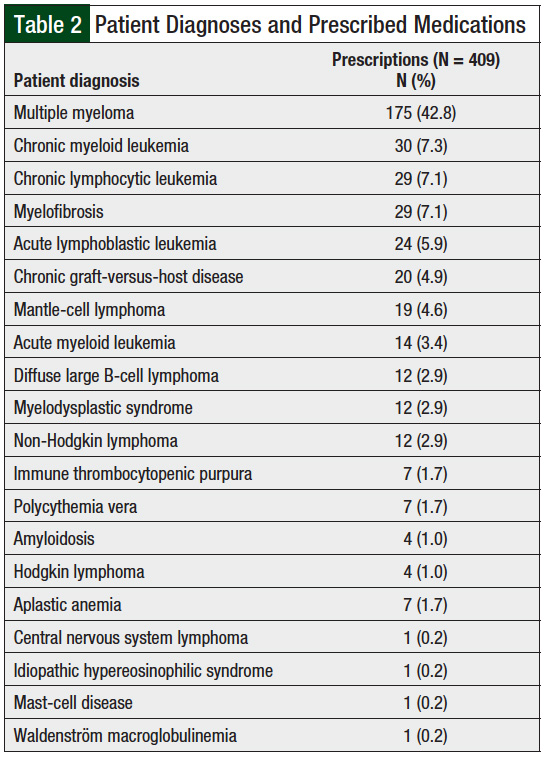
Of all the prescriptions, 10% were prescribed in combination therapy and each drug in those combinations was counted (ie, 2 oral oncolytic agents used concurrently were counted as 2 prescriptions). The most common indications for these prescriptions were multiple myeloma and chronic myeloid leukemia; 9% of prescriptions were for off-label indications (Table 2).
Federal- or state-funded insurance was used for 48% of all prescriptions in the study, and commercial insurance for 52% of the prescriptions. For the initial prescription fill, 47% of patients received financial assistance for their prescriptions. Table 3 shows the specific prescription characteristics during each of the 2 study periods.
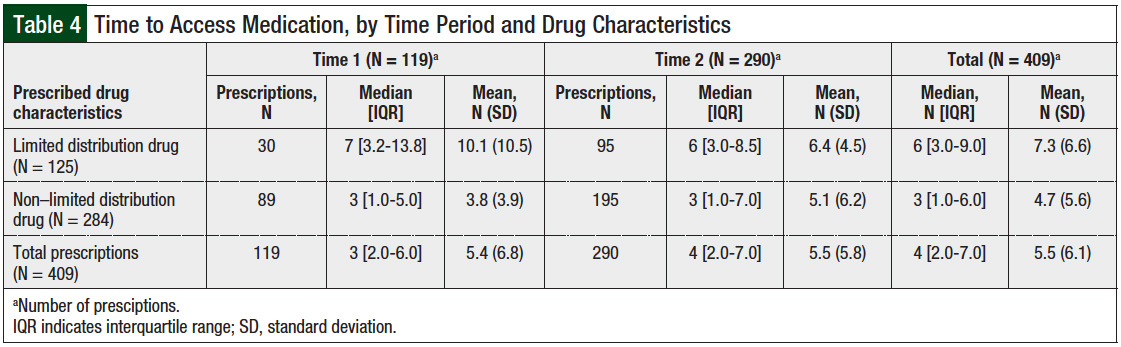
The clinical pharmacist appealed 7% of the prescriptions to obtain insurance approval—5 prescriptions in Time 1 and 23 in Time 2; all the appeals were successful. All patients in the study accessed their medication through insurance approval. Collapsing across both time periods, the median time to medication access was 6 days for limited distribution prescriptions (IQR, 3-9 days) and 3 days for non–limited distribution drugs (IQR, 1-6 days). The median access time for limited distribution medications decreased from 7 days in Time 1 to 6 days in Time 2. Table 4 shows the median and mean access time for the 2 time periods and the drug characteristics.
In the proportional odds logistic regression analysis (Table 5), the time to medication access was significantly associated with the drug type (ie, limited distribution drug or non–limited distribution drug), off-label indication or on-label, and the drug by time interaction (ie, before or after pharmacist embedded in the clinic); financial assistance use and the main effect of time were not statistically significant.
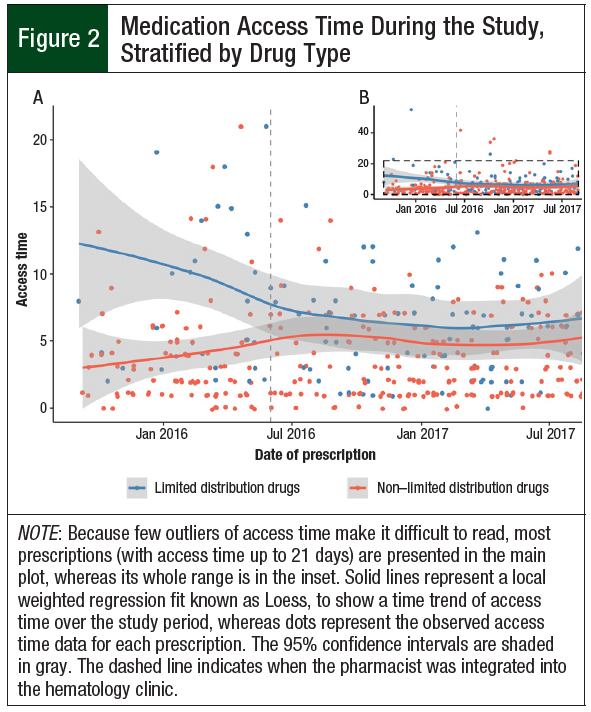
The median access time was significantly longer for limited distribution drug than to non–limited distribution drug prescriptions in Time 1 (odds ratio [OR], 6.31; P <.001), and access time for limited distribution drugs decreased from Time 1 to Time 2, but the access time for non–limited distribution drugs remained similar between the 2 time periods (ie, drug type by time period interaction = OR, 0.40; P = .039). Figure 2 shows the change in time to medication access across the 2 time periods.
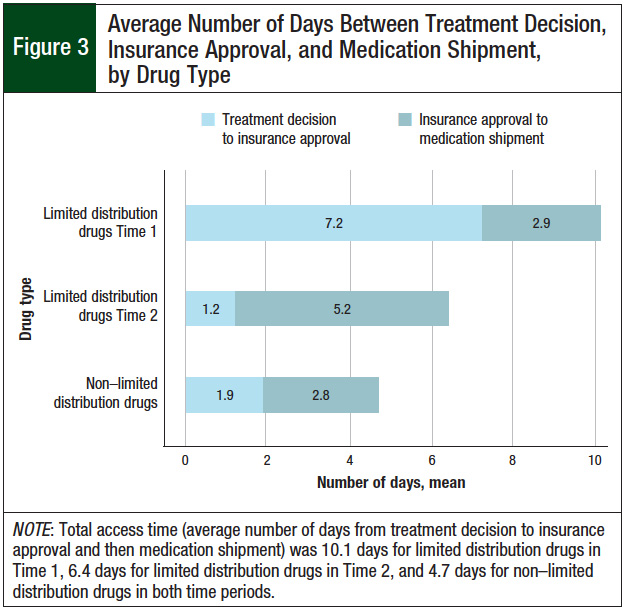
Figure 3 shows the division of the average medication access time by the number of days between treatment decision and insurance approval versus the number of days between insurance approval and medication shipment. Across the time periods, the mean time for accessing non–limited distribution drugs was 4.7 days; 1.9 days between treatment decision and insurance approval, and 2.8 days between insurance approval and medication shipment. For limited distribution drugs, the average time from treatment decision to insurance approval was 7.2 days in Time 1, which decreased to 1.2 days in Time 2; the time from insurance approval to medication shipment increased from 2.9 days in Time 1 to 5.2 days in Time 2.
Discussion
The goal of our study was to assess the impact of an integrated clinic pharmacist and limited distribution drug networks on the time to accessing oral oncolytic agents at an outpatient hematology clinic. In the first period, when the clinical pharmacist managed non–limited distribution drug prescriptions and a nurse managed limited distribution drug prescriptions, the access time was significantly longer for limited distribution drug prescriptions. After the pharmacist began to manage limited distribution drug prescriptions and REMS requirements in Time 2, the access time for limited distribution drugs significantly decreased but remained longer than non–limited distribution drugs.
Our study is among the first analyses to delineate the impact of a clinical pharmacist and limited distribution networks on time to accessing oral oncolytic medications within an integrated health system specialty pharmacy.
Accessing oral oncolytic medication is often time-consuming and burdensome. Previous studies reported a median time to access oral anticancer medications of 7 to 12 days after the prescription is generated.1-4
In clinics without integrated pharmacies (as was the case for limited distribution drugs in Time 1 in our study), clinic staff send the prescription to an external pharmacy, which notifies the clinic whether a prior authorization is required by the patient’s insurance. If so, staff members then complete the prior authorization and send it back to the insurance provider. If a prior authorization is denied, the clinic staff are notified by the pharmacy benefits manager (PBM) that an appeal is required.
Once a PBM approves the prescription (after either a prior authorization or appeal), the pharmacy notifies the patient and/or clinic of the patient copayment. If the patient needs financial assistance, the pharmacy, nurse, or patient completes paperwork to enroll in a financial assistance program, which further delays the time to medication access.1 Finally, the external pharmacy calls the patient to conduct medication counseling and safety monitoring, and then arranges the medication to be ordered and shipped to the patient.
One study showed that clinic staff spent a median of 2 hours per prescription to facilitate patient access; the pharmacy process time took a median of 6 days, accounting for more than half the time between prescription generation and medication access.2 Delays in medication access may contribute to the patient or provider stress and highlight inefficient workflow and communication among the involved parties.
We found faster access time to oral oncolytic agents than reported in previous studies,1-4 highlighting the value of integrated specialty pharmacies. After the pharmacist and pharmacy technician began to manage all prescriptions, the median time for all drugs from the treatment decision to medication shipment was 4 days, even though 10% of the prescriptions were prescribed for off-label indications, and a high proportion of patients used financial assistance programs.
Our findings support integrated health system specialty pharmacies’ ability to streamline workflow and expedite patient access to oral oncolytic medications. In this model, a clinical pharmacist is physically embedded in the clinical care and has access to the electronic health record, allowing access to patient information and real-time communication with clinic providers and with staff. Providers notify the pharmacist immediately when deciding to prescribe a drug, and the pharmacist conducts insurance benefits investigation, completes prior authorization (if needed), enrolls the patient in financial assistance, and conducts medication counseling. The prescription is then dispensed by the on-site pharmacy.
Patients and insurance providers have a single point of contact, reducing the paperwork and correspondence among multiple parties. Clinic pharmacists and pharmacy technicians in integrated specialty pharmacies streamline the process for starting the therapy for the patient, which allows the clinic staff to devote more time and resources to patient care.
Although access time to limited distribution drugs improved after integrating the clinical pharmacist and the technician into the clinic, time to access medication remained slower for limited distribution drug prescriptions (which must be dispensed by an external specialty pharmacy) than for non–limited distribution drug prescriptions (which can be shipped by the on-site pharmacy). The mean access time to these drugs significantly decreased from 10 days to 6 days after the pharmacist managed these prescriptions (ie, from Time 1 to Time 2), but access time remained slower for limited distribution drugs than for non–limited distribution drugs in Time 2 (6 days vs 3 days).
These findings suggest that drug acquisition is more efficient after transitioning drug access responsibilities from the clinic staff to a dedicated clinical pharmacist; however, unlike non–limited distribution drug prescriptions, limited distribution drug prescriptions cannot be fully integrated into the clinic workflow, and the process of distribution through required external pharmacies is a persistent barrier to timely treatment initiation. Non–limited distribution drug prescription access can be managed internally by a healthcare system, with a single team overseeing drug access, ordering, and shipment.
By contrast, for limited distribution drug prescriptions, the clinic pharmacist compiles documentation required by the patient’s insurance, then sends this paperwork to an external pharmacy to order and dispense. After the pharmacist began managing limited distribution drug prescriptions in Time 2, prior authorizations were completed more quickly and the insurance company approved the drug more quickly, but the average time between insurance approval and medication shipment was 5.2 days (Figure 3). Thus, delays in external pharmacy processing time still existed, consistent with Niccolai and colleagues’ findings, which resulted in delayed patient drug acquisition.2
Limited distribution drug networks were developed to ensure the quality and safety of high-cost drugs, by closely monitoring the drug’s supply chain. By limiting distribution to few select pharmacies, drug manufacturers can ensure that those pharmacies use licensed pharmacists who provide high-touch care to patients through medication counseling, safety monitoring, and adverse event reporting.5,13
Integrated health system specialty pharmacies, like traditional specialty pharmacies, maintain national accreditation for safety and quality, as well as use clinical pharmacists with disease-specific drug expertise. Clinical pharmacists in integrated health system specialty pharmacies are uniquely suited to monitor patient health in collaboration with healthcare providers, with real-time communication through the shared electronic medical record.
Our data suggest that in addition to providing patient-centered care, integrated specialty pharmacies have faster processing time than external pharmacies, and this efficiency results in faster patient drug acquisition. Faster drug acquisition builds on previous evidence suggesting that this specialty pharmacy model leads to better medication adherence and rates of access to medication.12 In addition, within this model, patients can be monitored more closely for response to therapy and adverse events; dosing changes and/or drug therapy changes can be completed in the clinic in real time.
Limitations
This study has several limitations. Our study was limited to patients with drug insurance coverage, because uninsured patients receive oral oncolytic drugs through the manufacturer’s patient assistance program; therefore, the shipment dates would not be available for uninsured patients.
We also excluded patients who received free drug samples; our pharmacy dispenses free samples (if available) to patients to prevent delays in treatment initiation, another benefit of integrated pharmacies having access to dispense medications.
Time to medication access was computed using calendar days rather than business days, so weekends account for some time between treatment decision and medication shipment. Medication access was computed based on medication shipment date, not the day that the patient started therapy. For patients using non–limited distribution drug prescriptions dispensed by our on-site pharmacy, local patients sometimes receive the medication on the same day, but most patients receive the medication the following day.
We computed access time for all limited distribution drug prescriptions (which were dispensed by multiple external pharmacies) without comparing access time between specific external pharmacies, which may vary in processing speed and level of assistance with drug approvals. Medication shipment dates were unavailable for some limited distribution drugs, particularly in Time 1, and these cases were excluded from the study, which could explain why the time between insurance approval and medication shipment was faster in Time 1 than in Time 2 (when more dates were available).
Not knowing when patients start therapy is another challenge with limited distribution drugs. For non–limited distribution drugs, the pharmacist knows exactly when the patient receives the medication. For limited distribution drugs, the clinician must call the pharmacy, sometimes multiple times, to inquire about shipment dates. Finally, our primary outcome was access time, because quick treatment initiation is expected to lead to better outcomes, but future work is needed to assess the associations between access time and long-term clinical outcomes.
Conclusions
Our findings suggest that integrating a clinical pharmacist and a pharmacy technician into a hematology clinic can reduce, but not eliminate, barriers to timely access to limited distribution drugs, warranting a review of the benefit of these networks and the criteria for selecting in-network pharmacies for health systems. Health system pharmacies and drug manufacturers must collaborate to overcome barriers to drug access and to ensure patient safety. It is worth considering a hybrid model of care in which limited distribution drugs are integrated within health system pharmacies to ensure best timely access and safety of therapy, thus maximizing the clinical value of specialty drugs for patients with hematologic malignancies.
Acknowledgments
This study was supported by the VICTR Learning Healthcare System Platform under CTSA award No. UL1 TR002243 from the National Center for Advancing Translational Sciences (NCATS). Its contents are solely the responsibility of the authors and do not necessarily represent official views of the NCATS or the National Institutes of Health.
Author Disclosure Statement
Dr Zuckerman has received research funding from Sanofi and Gilead. Dr Jagasia is a consultant to Incyte, Kadmon, and Genentech, and has received research funding from Mallinckrodt and Janssen. Dr Wyatt, Dr Peter, Mr DeClercq, Dr Choi, Dr Starks, and Dr Maulis have no conflicts of interest to report.
Correction
In the print version of this article, in Figure 2, the limited-distribution drugs and the non–limited-distribution drugs graphs were labeled incorrectly, showing the wrong color for the 2 categories. This has been corrected online and in the PDF.
References
- Geynisman DM, Meeker CR, Doyle JL, et al. Provider and patient burdens of obtaining oral anticancer medications. Am J Manag Care. 2018;24:e128-e133.
- Niccolai JL, Roman DL, Julius JM, Nadour RW. Potential obstacles in the acquisition of oral anticancer medications. J Oncol Pract. 2017;13:e29-e36.
- Wang AA, Tapia C, Bhanji Y, et al. Barriers to receipt of novel oral oncolytics: a single-institution quality improvement investigation. J Oncol Pharm Pract. 2020;26:279-285.
- O’Neil D, Accordino MK, Wright JD, et al. Delay in receipt of newly prescribed oral anticancer drugs. J Clin Oncol. 2019;37(15_suppl):Abstract 6541.
- US Food and Drug Administration. What’s in a REMS? January 26, 2018. www.fda.gov/drugs/risk-evaluation-and-mitigation-strategies-rems/whats-rems. Accessed July 23, 2019.
- Kalis JA, Pence SJ, Mancini RS, et al. Prevalence of off-label use of oral oncolytics at a community cancer center. J Oncol Pract. 2015;11:e139-e143.
- Streeter SB, Schwartzberg L, Husain N, Johnsrud M. Patient and plan characteristics affecting abandonment of oral oncolytic prescriptions. J Oncol Pract. 2011;7(3 suppl):46s-51s.
- Khorana AA, Tullio K, Elson P, et al. Increase in time to initiating cancer therapy and association with worsened survival in curative settings: a U.S. analysis of common solid tumors. J Clin Oncol. 2017;35(15_suppl):Abstract 6557.
- Bhandari S, Ngo P, Mudra S, et al. Treatment delays in localized breast cancer: a NCDB analysis. J Clin Oncol. 2019;37(15_suppl):Abstract e18023.
- Sheinson D, Wong WB, Wu N, Mansfield AS. Is a delay in ALK inhibitor initiation associated with poorer survival? A retrospective analysis based on real-world data. J Clin Oncol. 2019;37(15_suppl):Abstract e18240.
- Sheinson D, Wong WB, Wu N, Mansfield AS. Impact of delaying initiation of anaplastic lymphoma kinase inhibitor treatment on survival in patients with advanced non-small-cell lung cancer. Lung Cancer. 2020;143:86-92.
- Bagwell A, Kelley T, Carver A, et al. Advancing patient care through specialty pharmacy services in an academic health system. J Manag Care Spec Pharm. 2017;23:815-820.
- Karas L, Shermock KM, Proctor C, et al. Limited distribution networks stifle competition in the generic and biosimilar drug industries. Am J Manag Care. 2018;24:e122-e127.
- Harris PA, Taylor R, Thielke R, et al. Research electronic data capture (REDCap) - a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377-381.