Vincristine interferes with mitotic spindle formation in healthy and malignant cells and is a crucial component in the backbone of a variety of chemotherapy regimens for children with cancer.1 In addition to its cytotoxic effects, vincristine causes damage to myelinated and unmyelinated nerve fibers leading to significant clinical neuropathies, including peripheral and autonomic neuropathy and constipation.1,2 The risk factors for vincristine-induced neuropathy include race, increasing age, genetic polymorphisms, and cumulative vincristine dose.3,4 Drug interactions, such as those that result from the concurrent use of vincristine and cytochrome (CY)P3A4 inhibitors, may also increase the risk for adverse events.5-7
Children who are undergoing treatment for malignancies are at risk for fungal infections resulting from cytotoxic treatment regimens, impaired host immune response, and prolonged neutropenia.8 Voriconazole, a strong CYP3A4 inhibitor, provides protection against Aspergillus and Candida species, making it an ideal agent for the prophylaxis and treatment of fungal infections in children with cancer.9 Although its spectrum of activity is broader than fluconazole, voriconazole increases the area under the curve (AUC) of CYP3A4 metabolized drugs by ≥10-fold compared with moderate inhibitors (eg, fluconazole), which increase AUC by ≥2- to <5-fold.10,11
Other classes of antifungal agents, such as amphotericin B and echinocandins, provide relatively broad coverage against fungal infections, but intravenous administration may not always be feasible for patients because of cost, geographic limitations, and access to home care resources. In addition, amphotericin B is associated with significant adverse events, such as nephrotoxicity, electrolyte abnormalities, and serious infusion-related reactions.12
Despite a variety of literature demonstrating the general pharmacokinetic interaction between vincristine and azoles,1,6,7 the evidence describing the clinical significance of the concurrent administration of voriconazole and vincristine is limited.
A retrospective analysis of 130 pediatric patients with acute lymphoblastic leukemia (ALL) or Hodgkin lymphoma assessed the incidence of the concurrent use of vincristine and azole antifungals and the clinical impact on neuropathy.1 No difference was found in the incidence of grade ≥2 neuropathy in patients who received an azole simultaneously with vincristine (P = .35). However, most patients received fluconazole (78%) as opposed to voriconazole (10%), and the investigators stated that the results might be different in a patient population that is receiving more potent antifungals.1
In a comprehensive review, Moriyama and colleagues identified 47 patients who had an adverse drug event after receiving concomitant azole antifungals and vincristine.6 None of the cases were associated with the use of fluconazole, but the concomitant use of voriconazole and vincristine caused significant peripheral neuropathy in 1 patient.6
In a retrospective study, Yang and colleagues reviewed 136 cases of drug–drug interactions resulting from treatment with vincristine and azole antifungals, including itraconazole, fluconazole, and voriconazole.13 The investigators observed an increased incidence of adverse drug reactions with itraconazole and voriconazole compared with fluconazole and the control group that did not receive azole antifungal drugs. Of note, only 6 patients in this study received voriconazole.13
Vincristine-associated neuropathy, which may be exacerbated by interaction with voriconazole, can be debilitating and prolonged, leading to decreased quality of life.6,7,14 Most pediatric treatment protocols for ALL require the omission or dose reduction of vincristine for severe (grade ≥3) neuropathic adverse events.
The strategies for managing the drug interaction between voriconazole and vincristine are determined based on benefit versus risk, discussion with the clinical team (pharmacists, physicians, advanced practice providers), and an understanding of the pharmacokinetics of the medications involved. These strategies include proceeding with the concurrent administration of voriconazole and vincristine, holding voriconazole treatment before and after the administration of vincristine, or using an alternative antifungal treatment, such as micafungin.15 However, voriconazole is a desirable agent for the prophylaxis and treatment of fungal infections in pediatric patients with cancer because of its spectrum of activity, ease of administration, and availability.15
Holding voriconazole therapy based on the administration of vincristine may decrease the efficacy of the antifungal therapy by altering steady-state concentrations of voriconazole.16 The optimal time to hold treatment with voriconazole before the administration of vincristine and the true duration of the inhibitory effects of voriconazole on metabolizing enzymes remain unknown.
The aim of this retrospective analysis was to assess the incidence and severity of neuropathy associated with the concurrent administration of voriconazole and vincristine to characterize the clinical significance of this specific interaction in pediatric patients with cancer.
Methods
This retrospective study was conducted using data from electronic medical records to identify pediatric patients aged 31 days to <22 years with a cancer diagnosis who received vincristine alone or in combination with voriconazole at a single institution, Children’s Hospital Colorado, in Aurora, from January 1, 2007, to July 31, 2018. This study was approved by the Colorado Multiple Institutional Review Board.
Patients were excluded from the study if they had baseline neuropathy, spinal cord compression, or spinal hematoma; received another azole antifungal drug during the time of analysis; or if they died while receiving induction therapy for ALL. Most of the patients in the non-voriconazole treatment arm did not receive an antifungal drug, but if they did, it was micafungin or amphotericin B. These agents do not interact with vincristine and should not affect patient outcomes.
The patients were divided into 2 cohorts of those who received vincristine alone and those who received vincristine and voriconazole concurrently. Concurrent use was defined as voriconazole administered 1 day before treatment with vincristine or within 7 days after receiving vincristine. This definition was determined based on the half-life of voriconazole, which is approximately 6 hours, as well as the prolonged terminal half-life of vincristine, which ranges from 19 to 155 hours (median, 85 hours).1 These definitions align with previously published literature.1
The cohort that received vincristine alone and the cohort that received vincristine plus voriconazole were matched in a 2:1 ratio based on age, self-reported race (White, Black, other), and cancer diagnosis. The treatment data, including the chemotherapy protocol, phase in therapy at the time of the drug interaction, phase in therapy during the clinical manifestation of neuropathy, and the date of death, if applicable, were extracted from the electronic medical records.
The additional data collection points included a history of relapsed or refractory disease; the dates of all vincristine treatment administrations; the dose of vincristine; the dates of all inpatient administrations of voriconazole; the dates of active outpatient prescriptions of voriconazole; the dose, duration, and indication for treatment with voriconazole; the average of all trough levels of voriconazole during cancer treatment; the number of concurrent administrations of vincristine and voriconazole; the onset and severity of neuropathy; and the concurrent administration of strong CYP3A4 inducers or inhibitors.
The investigators reviewed the progress notes and/or roadmaps to assess the presence and severity of neuropathic adverse events based on the documented grade. The initial and highest grades of neuropathy were extracted. If no grade was available in the patient’s chart, a grade was assigned based on the modified Balis grading criteria for motor and sensory neuropathies or the Common Terminology Criteria for Adverse Events v5.0 to categorize constipation (grade ≥3), paralytic ileus, ptosis, and vocal paralysis. The grades were determined by an experienced physical therapist and another investigator who was a physician or pharmacist.
If the 2 reviewers differed on the patient’s grade of neuropathy, a third investigator reviewed the patient’s data. Additional data including the date of the first documented neuropathy, date of most severe neuropathy, cumulative dose of vincristine received at the first documentation, and the time of the most severe neuropathy were collected. Secondary outcomes included the frequency and type of medical intervention required for the treatment of neuropathy, such as dose reduction or omission, physical therapy consultation, surgery, orthotics, serial casting, or neuropathic pain medications.
Before data collection, a power calculation revealed that 82 patients per cohort were needed to reach 80% power to detect a 40% difference between the groups. Frequency tables were used to generate the matched samples for the vincristine monotherapy cohort, and the patients were randomly selected from the sampling pool. A conditional logistic regression model was built for the incidence of neuropathy and for the incidence of severe neuropathy as outcomes.
Potential confounders, including race, age, and cancer diagnosis, were accounted for by the matched design. An alpha level of 0.05 was used to determine statistical significance. All calculations were performed using R version 3.5.1 (R Core Team, R Foundation for Statistical Computing; Vienna, Austria), and logistic regression was performed using the clogit function from the Survival package in R version 2.43.1.
Results
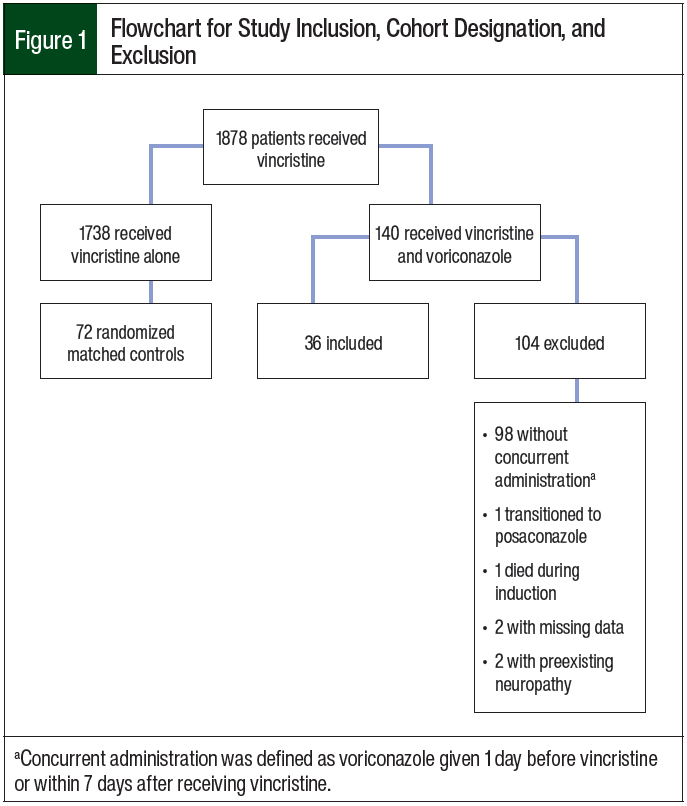
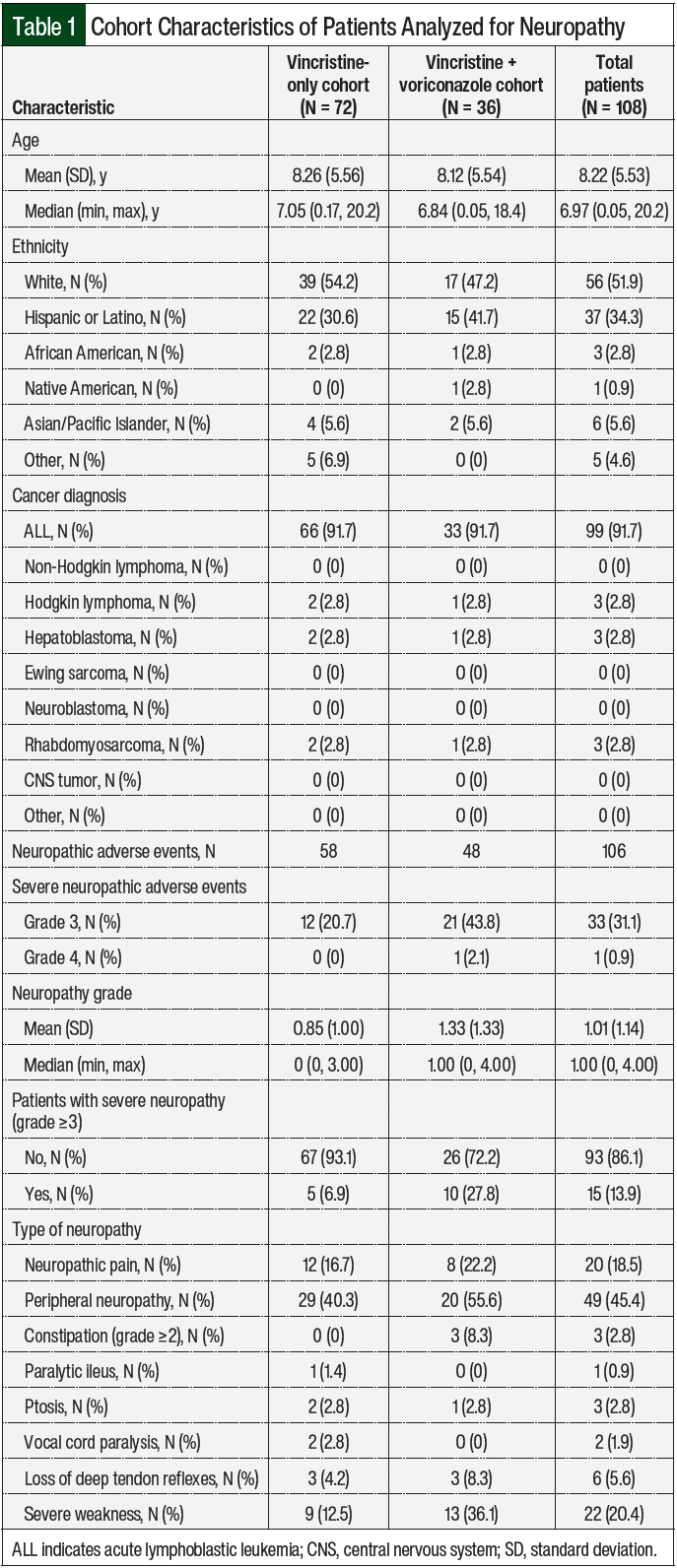
We identified 1878 patients who received vincristine during the study period. A total of 36 patients met the inclusion criteria for the voriconazole plus vincristine cohort (Figure 1). These patients were matched to 72 randomly selected patients from the cohort that received vincristine alone, leading to a total study population of 108 patients. Table 1 outlines the patients’ demographics according to the study cohort. The median patient age was 7 years (range, 0.05-20.2 years), and most patients had ALL (N = 99; 91.7%).
In the group that received vincristine only, 68 (94.4%) patients received treatment for their initial diagnosis, compared with 16 (44.4%) patients in the group that received vincristine plus voriconazole who had relapsed disease.
The patients in the voriconazole plus vincristine group received a median of 2 (range, 1-15) concurrent doses. Most patients received all doses of voriconazole treatment in the inpatient setting (N = 17; 47.2%), although 10 (27.8%) patients received voriconazole in the inpatient and outpatient settings. A total of 9 (25%) patients received voriconazole solely in the outpatient setting, but because of this study’s retrospective design, we could not confirm outpatient treatment compliance.
The median voriconazole trough concentration was 3.4 mcg/mL (interquartile range [IQR], 2.2-5.2 mcg/mL), and the median duration of therapy was 25.5 days (IQR, 9.5-81.75 days). Most patients were prescribed voriconazole for a suspected or confirmed fungal infection (N = 22; 61%) rather than as a prophylaxis (N = 14; 38.9%).
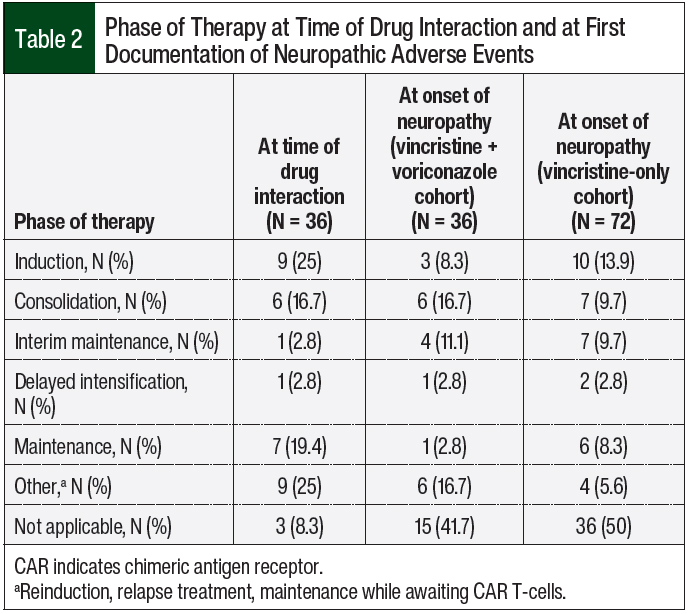
A drug interaction between voriconazole and vincristine was most frequent during induction (N = 9), consolidation (N = 6), or maintenance (N = 7) therapy for patients with ALL (Table 2). A median of 16 (IQR, 5.5-34) doses of vincristine per patient were administered before treatment with voriconazole was initiated.
A total of 36 (50%) patients in the vincristine monotherapy group had neuropathy compared with 21 (58.3%) patients in the voriconazole plus vincristine group. For the patients who received vincristine alone, the onset of neuropathy was most frequent during induction therapy (N = 10), followed by consolidation therapy (N = 7) and interim maintenance therapy (N = 7). In the vincristine plus voriconazole cohort, the clinical manifestations of an adverse event primarily occurred during consolidation therapy (N = 6), interim maintenance therapy (N = 4), or another phase of therapy, such as reinduction (N = 6; Table 2).
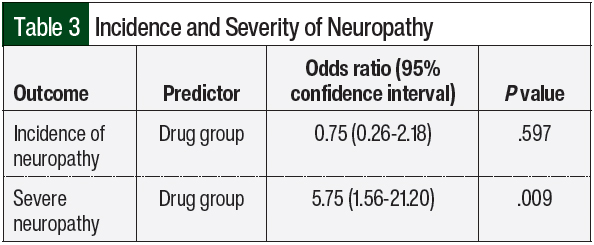
Of the 21 patients in the voriconazole plus vincristine group with neuropathy, 14 (67%) had clinical symptoms of vincristine-induced neuropathy before the initiation of voriconazole plus vincristine. These patients were shifted to the vincristine monotherapy group when analyzing the incidence of neuropathy, because neuropathy was deemed independent of the drug interaction (vincristine only, N = 86; vincristine plus voriconazole, N = 22). No statistically significant difference was found in the incidence of neuropathy between the 2 cohorts (odds ratio [OR], 0.75; 95% confidence interval [CI], 0.26-2.18; P = .597; Table 3).
A total of 58 adverse events were identified in the vincristine-only cohort. The most common types of neuropathy in both groups were peripheral neuropathy and neuropathic pain (Table 1).
Grades 3 and 4 neuropathic adverse events were considered severe because, in general, these events require a dose reduction or the omission of vincristine. In the vincristine plus voriconazole cohort, there were 21 (43.8%) grade 3 neuropathic events and 1 (2.1%) grade 4 event. In the vincristine monotherapy cohort, neuropathic events were largely less severe, with 12 (20.7%) grade 3 events and no grade 4 events. Logistic regression revealed a significantly higher incidence of grade ≥3 neuropathy in the patients who received vincristine and voriconazole concurrently versus those who received vincristine alone (OR, 5.75; 95% CI, 1.56-21.20; P = .009; Table 3).
Although more patients in the vincristine plus voriconazole group had relapsed disease than patients who received vincristine monotherapy, the total median doses of vincristine were similar between the 2 groups (N = 33.5 doses [IQR, 22.5-41.25] for the vincristine plus voriconazole cohort vs N = 35 doses [IQR, 23-41.25] for the vincristine cohort).
In the voriconazole plus vincristine group, the median number of vincristine doses at the first documentation of neuropathy was 9 (IQR, 4-10.5), with a median cumulative dose of 13 mg/m2 (IQR, 6-18 mg/m2), compared with a median of 7 doses of vincristine (IQR, 3.75-13) and a cumulative dose of 10.5 mg/m2 (IQR, 4.35-19.05 mg/m2) in the patients who received vincristine alone.
Of the patients who received vincristine and voriconazole concurrently, 25 (69%) received both medications within 1 day of each other. In this subgroup, there were 37 documented neuropathic adverse events, of which 17 (45.9%) were grade 3 and 1 (2.7%) was grade 4.
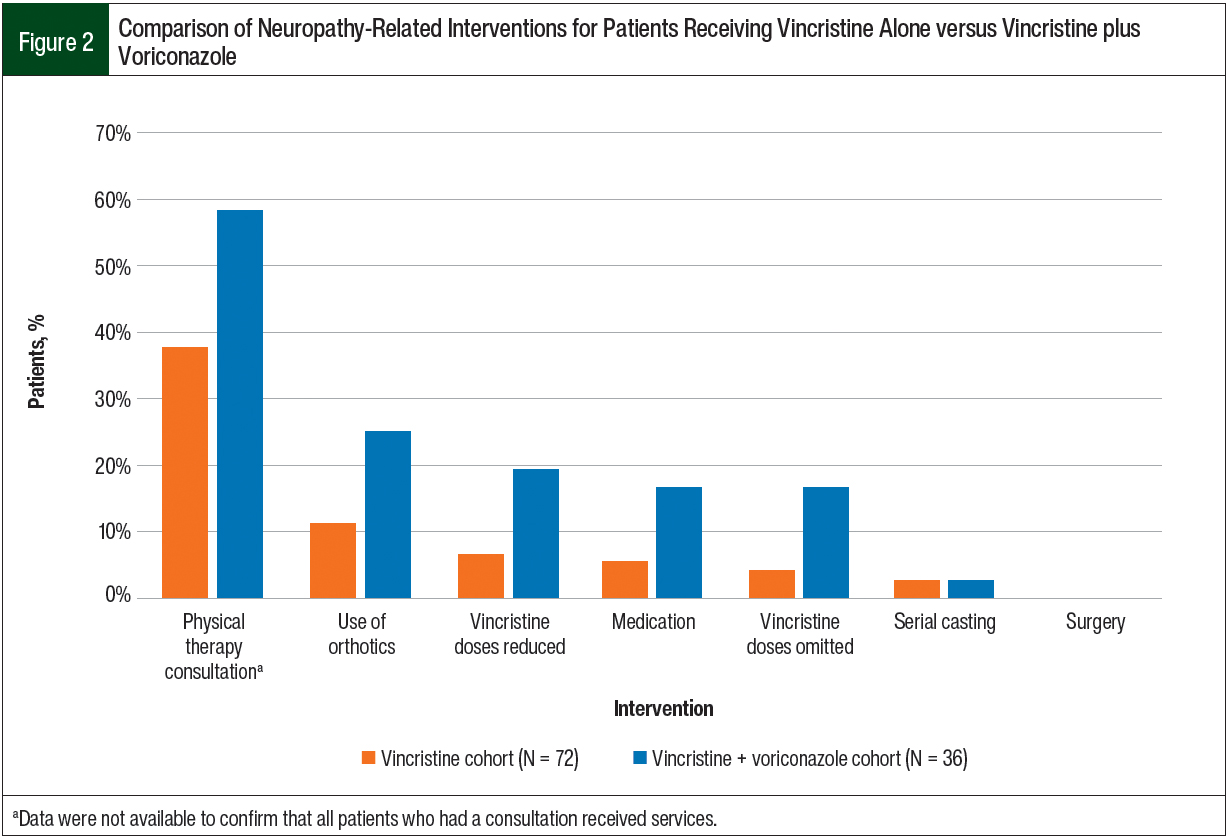
The patients in the voriconazole plus vincristine group required more interventions for the treatment of neuropathy than those in the vincristine monotherapy cohort (Figure 2). Specifically, 50 interventions were required in the 36 patients receiving voriconazole plus vincristine compared with 49 interventions in the 72 patients receiving vincristine alone.
The most common intervention was a physical therapy consultation followed by the use of orthotics. Medications, including gabapentin, pregabalin, subanesthetic ketamine, and lidocaine patches, were prescribed for the treatment of neuropathy in 6 patients in the voriconazole plus vincristine group and in 4 patients in the vincristine monotherapy cohort.
A total of 7 patients in the voriconazole plus vincristine group required a 50% dose reduction, with a median of 2 reduced doses administered per patient (IQR, 1.25-8) compared with 5 patients requiring a dose reduction in the vincristine monotherapy group (four 50% dose reductions and one 75% dose reduction) and a median of 3 reduced doses administered per patient (IQR, 2.25-3.5). In all, 6 patients required a dose omission in the vincristine plus voriconazole group compared with 3 patients in the vincristine monotherapy group.
Discussion
Vincristine is a crucial component in the backbone of a variety of chemotherapy regimens for the treatment of pediatric malignancies. Patients with cancer may require the concurrent administration of voriconazole and vincristine for the prophylaxis or treatment of fungal infections during their course of chemotherapy if the benefit is determined to outweigh the risk. Voriconazole is a potent inhibitor of vincristine’s primary metabolizing enzyme, CYP3A4, potentially putting patients at higher risk for vincristine-induced neuropathy.5
Clinically significant drug interactions between vincristine and azole antifungals have been reported in the literature5-7; however, few studies assess the incidence and severity of neuropathy with the concurrent administration of vincristine and voriconazole.
This retrospective analysis did not show a significant difference in the incidence of vincristine-associated neuropathy (all grades) with the concurrent administration of vincristine and voriconazole versus with vincristine alone, but the severity of neuropathy may be increased with the combination regimen. The patients who received concurrent vincristine and voriconazole had 5.75 times (95% CI, 1.56-21.20) higher odds of grades 3 and 4 neuropathies than patients who received vincristine alone. Severe neuropathies put patients at risk for dose reductions and treatment delays, which could negatively affect patient outcomes, and prolonged neuropathy may substantially affect patient quality of life.14,17
Literature describing the drug interaction between vincristine and voriconazole is limited to small studies and case reports.1,7,18 Vincristine-induced neuropathy that is exacerbated by treatment with voriconazole was described in a case report of a 14-year-old girl with T-cell ALL and pulmonary aspergillosis.18 Voriconazole exacerbated the severity of her peripheral neuropathy pain within 24 to 48 hours after the administration of vincristine and required intensified antalgic treatment.18
Nikanjam and colleagues also analyzed a subgroup of patients who received an azole antifungal drug within 1 day after the administration of vincristine, and their study showed no significant difference in the incidence of grade ≥2 neuropathy compared with those who did not receive an antifungal drug (P = .32).1 Our study did not assess the incidence and severity of neuropathy in this subgroup; however, patients who received voriconazole within 1 day of vincristine (N = 25) had 37 neuropathic adverse events, 18 (48.6%) of which were grade 3 or 4.
The outcomes of this study are consistent with previously published literature and add to the concern that voriconazole worsens the severity of vincristine-induced neuropathy.6,7,13,18 Pediatric institutions should consider increased monitoring for neuropathic adverse events in patients who are exposed to voriconazole and vincristine concurrently.
The use of alternative antifungal agents may be warranted. For example, isavuconazole, an azole antifungal that was approved by the US Food and Drug Administration in 2015, provides broad coverage against Candida species and invasive aspergillosis with only moderate inhibition of CYP3A4.19 To our knowledge, there are currently no studies assessing the clinical impact of the drug interaction between vincristine and isavuconazole specifically.
Additional options for empiric antifungal drug coverage include echinocandins or amphotericin B.12 Echinocandins do not provide adequate fungal coverage against Zygomycetes, Cryptococcus species, or Fusarium species and must be administered intravenously.15,20 Although amphotericin B provides broad empiric coverage, it has an undesirable adverse-event profile, including nephrotoxicity, electrolyte wasting (eg, hypomagnesemia, hypokalemia), and infusion reactions (eg, chills, fever, myalgias, hypotension).12
Larger, prospective studies that assess a homogenous patient population, such as children who received treatment according to the same protocol, prespecified assessment timeline (ie, induction therapy through the end of maintenance therapy), and time to the first incident of neuropathy, are warranted. Studies that account for additional confounding factors, such as pharmacogenomic differences, would also allow for a more accurate analysis of the clinical impact of the drug interaction between voriconazole and vincristine. As pharmacogenomic testing becomes more widely available, this may provide an opportunity to identify patients who are at high risk for neuropathic adverse events.
Limitations
There were several limitations to this study. This study’s retrospective design eliminated the ability to assess patient compliance or to confirm the start dates for outpatient prescriptions for voriconazole. The investigators assumed that treatment with voriconazole was initiated on the ordered date and that complete adherence was maintained throughout the duration of therapy.
In addition, the documentation of neuropathic adverse events was inconsistent, requiring chart review and clinical judgment for grade assignment. To improve accuracy and consistency, multiple investigators separately reviewed the patients’ charts and used validated assessment scales that were recommended by the Children’s Oncology Group protocols to assign grades. An experienced physical therapist assisted with the interpretation of the modified Balis grading scale for motor and sensory neuropathies. However, there may have been undocumented symptoms or adverse events that could not be identified through retrospective review.
At our institution, the concurrent use of voriconazole and vincristine is avoided when the risk is determined to outweigh the benefit because of the known pharmacokinetic interaction between these 2 drugs. Therefore, only a small number of patients met this study’s inclusion criteria, and the sample was not large enough to achieve full power, which may have contributed to our inability to detect differences in the incidence of neuropathy between the groups.
The sample also included a heterogeneous patient population and variable treatment protocols. To address this limitation, the patients’ diagnoses were included in the matching model and the total number of administrations and cumulative doses of vincristine were collected. The patients were also matched based on race to minimize the pharmacogenomic differences between the groups. Genetic polymorphisms in CYP enzymes and the CEP72 gene may decrease the metabolism of vincristine and increase the risk for adverse events.3,21-23 Pharmacogenomic testing has not been implemented at our study institution, so this information was unknown.
A larger number of patients in the voriconazole and vincristine cohort had relapsed or refractory disease (50%) compared with those in the vincristine monotherapy cohort (5.6%), suggesting that the patients who received voriconazole and vincristine had more severe disease. These patients may have been more susceptible to neuropathic adverse events, despite receiving a similar number of total doses of vincristine.
In addition, we did not define a specific time frame for the assessment of neuropathy after a drug interaction, and it is possible that some patients had not reached the time of peak onset. In a longitudinal study by Lavoie Smith and colleagues, the investigators assessed vincristine-induced peripheral neuropathy (VIPN) over a 12-month period in pediatric patients with newly diagnosed ALL, and the peak onset was approximately 2 to 4 months.14 However, in a review of case reports describing azole-induced VIPN, the onset of symptoms occurred between 2 and 30 days after the concurrent administration of an antifungal azole and vincristine.6 This limitation was addressed by collecting the known risk factors for vincristine-induced neuropathy, including the total number of doses of vincristine and the cumulative dose of vincristine in mg/m2.
The concomitant use of other medications that are known to result in neuropathy or constipation, such as opioids, was not reviewed in this study. The incidence of neuropathy-related adverse events may also be confounded by voriconazole’s concentration-dependent neurotoxicity, which typically occurs at concentrations of >5.5 mcg/mL.14,24 The median trough concentrations were below the recommended maximum, at 3.4 mcg/mL (IQR, 2.2-5.2 mcg/mL); however, 3 patients had average trough concentrations of voriconazole that exceeded the upper limit (maximum, 10.3 mcg/mL).
Last, 1 patient in the vincristine monotherapy cohort received rifampin, a strong CYP3A4 inducer, 4 days after the administration of vincristine. This patient was included in the final analysis because enzyme induction does not occur immediately, and vincristine was expected to have cleared before a clinically meaningful interaction occurred. One patient in the vincristine monotherapy group received clarithromycin, a strong CYP3A4 inhibitor, and vincristine concurrently.
Conclusion
This study showed no difference in the incidence of neuropathy between patients who received vincristine and voriconazole concurrently and patients who received vincristine alone. However, our results show that the patients who were exposed to the drugs concurrently may have more severe neuropathic adverse events and may require significant interventions.
Institutions that treat pediatric patients should consider increased monitoring in children who receive concomitant voriconazole and vincristine, the use of echinocandins, or the addition of alternative azole antifungals to the formulary. Although more robust studies with a larger sample size and prospective trial design are needed to assess the clinical significance of the interaction between voriconazole and vincristine, this study provides information to assist practitioners in making clinical decisions regarding the risks and benefits of the concurrent administration of these drugs.
Author Disclosure Statement
Dr Sabus, Dr Merrow, Dr Burke, Mr Kordas, Dr Williams, Dr Larson, and Dr Eisenman have no conflicts of interest to report.
References
- Nikanjam M, Sun A, Albers M, et al. Vincristine-associated neuropathy with antifungal usage: a Kaiser Northern California experience. J Pediatr Hematol Oncol. 2018;40:e273-e277.
- Carozzi VA, Canta A, Chiorazzi A. Chemotherapy-induced peripheral neuropathy: what do we know about mechanisms? Neurosci Lett. 2015;596:90-107. Erratum in: Neurosci Lett. 2015;596:108.
- Sims RP. The effect of race on the CYP3A-mediated metabolism of vincristine in pediatric patients with acute lymphoblastic leukemia. J Oncol Pharm Pract. 2016;22:76-81.
- Mora E, Lavoie Smith EM, Donohoe C, Hertz DL. Vincristine-induced peripheral neuropathy in pediatric cancer patients. Am J Cancer Res. 2016;6:2416-2430.
- Brüggemann RJM, Alffenaar JWC, Blijlevens NMA, et al. Clinical relevance of the pharmacokinetic interactions of azole antifungal drugs with other coadministered agents. Clin Infect Dis. 2009;48:1441-1458.
- Moriyama B, Henning SA, Leung J, et al. Adverse interactions between antifungal azoles and vincristine: review and analysis of cases. Mycoses. 2012;55:290-297.
- van Schie RM, Brüggemann RJM, Hoogerbrugge PM, te Loo DMWM. Effect of azole antifungal therapy on vincristine toxicity in childhood acute lymphoblastic leukaemia. J Antimicrob Chemother. 2011;66:1853-1856.
- Pana ZD, Roilides E. Risk of azole-enhanced vincristine neurotoxicity in pediatric patients with hematological malignancies: old problem–new dilemma. Pediatr Blood Cancer. 2011;57:30-35.
- Cronin S, Chandrasekar PH. Safety of triazole antifungal drugs in patients with cancer. J Antimicrob Chemother. 2010;65:410-416.
- Townsend R, Desai A, Azie N, et al. Drug interaction profiles of isavuconazole, voriconazole and posaconazole with immunosuppressants metabolized by CYP4503A4 (CYP3A4). Poster presented at: European Congress of Clinical Microbiology and Infectious Diseases; April 25-28, 2015; Copenhagen, Denmark. Poster 216.
- US Food and Drug Administration. Drug development and drug interactions: table of substrates, inhibitors and inducers. 2017. Updated August 24, 2022. www.fda.gov/drugs/developmentapprovalprocess/developmentresources/druginteractionslabeling/ucm093664.htm. Accessed March 21, 2023.
- Hamill RJ. Amphotericin B formulations: a comparative review of efficacy and toxicity. Drugs. 2013;73:919-934.
- Yang L, Yu L, Chen X, et al. Clinical analysis of adverse drug reactions between vincristine and triazoles in children with acute lymphoblastic leukemia. Med Sci Monit. 2015;21:1656-1661.
- Lavoie Smith EM, Li L, Chiang CW, et al. Patterns and severity of vincristine-induced peripheral neuropathy in children with acute lymphoblastic leukemia. J Peripher Nerv Syst. 2015;20:37-46.
- Groll AH, Pana D, Lanternier F, et al; for the 8th European Conference on Infections in Leukaemia. 8th European Conference on Infections in Leukaemia: 2020 guidelines for the diagnosis, prevention, and treatment of invasive fungal diseases in paediatric patients with cancer or post-haematopoietic cell transplantation. Lancet Oncol. 2021;22:e254-e269.
- Soler-Palacín P, Frick MA, Martín-Nalda A, et al. Voriconazole drug monitoring in the management of invasive fungal infection in immunocompromised children: a prospective study. J Antimicrob Chemother. 2012;67:700-706.
- Pui CH, Evans WE. A 50-year journey to cure childhood acute lymphoblastic leukemia. Semin Hematol. 2013;50:185-196.
- Bennis Y, Bodeau S, Lutun A, et al. Severe neurological disorders and refractory aspergillosis in an adolescent treated by vincristine and voriconazole. J Clin Pharm Ther. 2018;43:265-268.
- Ledoux MP, Denis J, Nivoix Y, Herbrecht R. Isavuconazole: a new broad-spectrum azole. Part 2: pharmacokinetics and clinical activity. J Mycol Med. 2018;28:15-22.
- Kofla G, Ruhnke M. Pharmacology and metabolism of anidulafungin, caspofungin and micafungin in the treatment of invasive candidosis: review of the literature. Eur J Med Res. 2011;16:159-166.
- Abaji R, Ceppi F, Patel S, et al. Genetic risk factors for VIPN in childhood acute lymphoblastic leukemia patients identified using whole-exome sequencing. Pharmacogenomics. 2018;19:1181-1193.
- Renbarger JL, McCammack KC, Rouse CE, Hall SD. Effect of race on vincristine-associated neurotoxicity in pediatric acute lymphoblastic leukemia patients. Pediatr Blood Cancer. 2008;50:769-771.
- Diouf B, Crews KR, Lew G, et al. Association of an inherited genetic variant with vincristine-related peripheral neuropathy in children with acute lymphoblastic leukemia. JAMA. 2015;313:815-823.
- Jin H, Wang T, Falcione BA, et al. Trough concentration of voriconazole and its relationship with efficacy and safety: a systematic review and meta-analysis. J Antimicrob Chemother. 2016;71:1772-1785.