Connecting Science to Practice
Use of a modified AAVD (doxorubicin, brentuximab vedotin,
vinblastine, and dacarbazine) regimen that incorporates pegylated
liposomal doxorubicin (PLD) in place of conventional doxorubicin
may be a treatment option in younger (aged 18-69 years) understudied
patients with newly diagnosed classical Hodgkin lymphoma
(cHL) with cardiac comorbidities or poor ejection fraction,
circumstances that would preclude use of conventional doxorubicin.
With 4 cycles of this regimen plus consolidative radiation, our
patient achieved a complete response and showed no significant
adverse events. Further study regarding the optimal dosing of PLD
in younger patients newly diagnosed with cHL is warranted.
Classical Hodgkin lymphoma (cHL) is an uncommon hematologic malignancy that most frequently affects young adults, who are primarily diagnosed between the ages of 20 years and 34 years.1 Overall, cHL is highly curable, with a 5-year relative survival rate of 88.9%.1 Thus, maintaining chemotherapy intensity and minimizing treatment-related adverse events (AEs) are critical to managing this patient population. The current National Comprehensive Cancer Network (NCCN) guidelines recommend modalities such as chemotherapy with or without radiation depending on the stage of disease at diagnosis, as well as the patient’s age, sex, family history of cancer or cardiac disease, comorbidities, and site of involvement.2
Chemotherapy with ABVD (doxorubicin, bleomycin, vinblastine, and dacarbazine) remains the backbone of frontline therapy for patients with early-stage cHL without underlying pulmonary comorbidities.2 ABVD is extremely efficacious, with approximately 97% of patients achieving complete remission by the end of therapy.3 However, the use of ABVD is limited by baseline cardiopulmonary function resulting from cardiotoxicity associated with doxorubicin and pulmonary toxicity associated with bleomycin.2 A chemotherapy alternative to ABVD in patients with early-stage cHL and baseline pulmonary dysfunction is AAVD (doxorubicin, brentuximab vedotin, vinblastine, and dacarbazine), which replaces bleomycin with brentuximab vedotin, a CD30-targeting antibody–drug conjugate.2,4 Because ABVD and AAVD contain doxorubicin, they are not recommended by the NCCN guidelines for patients with baseline cardiac dysfunction.2
Although the NCCN guidelines acknowledge that the BV-DTIC (brentuximab vedotin and dacarbazine) regimen can be used in newly diagnosed patients with cHL and a low baseline ejection fraction, BV-DTIC has only largely been studied in an older (aged ≥60 years), more frail patient population that was ineligible for or unwilling to receive standard frontline therapy.2,5 Hence, the management of younger, fit patients with newly diagnosed cHL and baseline cardiac dysfunction remains a challenge. The addition of the cardioprotective agent dexrazoxane administered before conventional doxorubicin is an option recognized by the NCCN guidelines, and the use of a liposomal formulation of doxorubicin in place of conventional doxorubicin has also been studied.2,6
One study investigated the use of nonpegylated liposomal doxorubicin (non-PLD) in patients with previously untreated cHL who were aged >69 years or who were between the ages of 18 years and 69 years with concomitant cardiac disease.7 In this study, treatment with a non-PLD regimen was associated with improved preservation of left ventricular ejection fraction (LVEF) and lower rates of other cardiac AEs when it replaced conventional doxorubicin in the standard ABVD regimen.7 In addition, patients in the non-PLD arm had a complete response rate of 100%, with all patients alive and disease-free at the median follow-up time of 40 months.7
Currently, treatment with non-PLD is not approved in the United States. The PLD formulation is approved; however, there is currently no recommendation on the dosing conversion between the PLD and non-PLD formulations. PLD has been safely substituted for conventional doxorubicin in combination with vincristine, bleomycin, and dacarbazine (similar to ABVD) in patients with newly diagnosed Hodgkin lymphoma with a history of or risk factors for cardiovascular disease.6 However, to our knowledge, no published evidence supports the substitution of conventional doxorubicin with pegylated doxorubicin in the AAVD regimen. We herein present a report of PLD as a replacement for conventional doxorubicin in a modified AAVD regimen to treat a patient with newly diagnosed cHL, congenital hypoplastic left heart syndrome, and pulmonary comorbidities.
Case Report
A 24-year-old man was newly diagnosed with nonbulky stage IIA cHL of mixed cellularity subtype with excellent performance status (Eastern Cooperative Oncology Group score, 0). His medical history is significant for hypoplastic left heart syndrome (repaired via the Norwood, bidirectional Glenn, and fenestrated lateral tunnel Fontan procedures during childhood) and mild cirrhosis of the liver that was presumed to be of cardiac etiology.
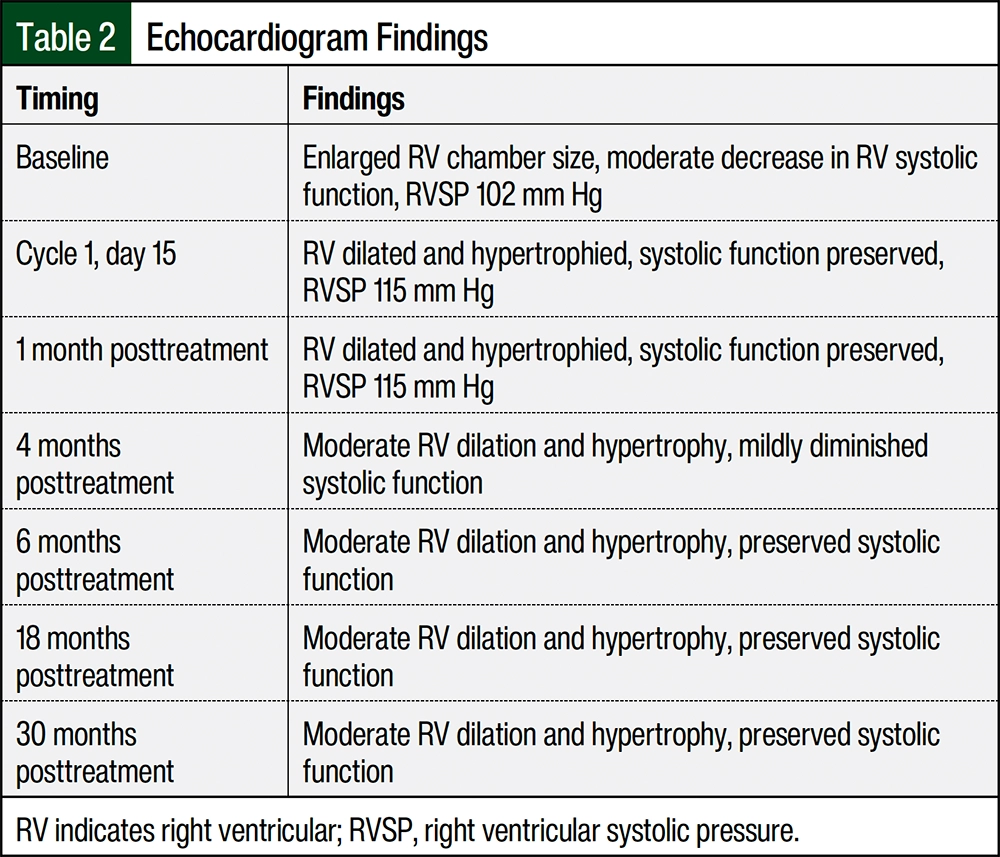
At the time of diagnosis, the patient presented with a mass on the left side of his neck and intermittent low-grade fevers. A baseline positron emission tomography (PET) scan reported a Deauville score of 5 with left cervical and subcarinal hypermetabolic adenopathy and splenomegaly presumed to result from underlying portal hypertension. The relevant baseline laboratory results include estimated glomerular filtration rate and liver function enzymes within normal limits, no evidence of cytopenia, and an erythrocyte sedimentation rate that was not elevated at 4 mm per hour. A baseline echocardiogram was significant for enlarged right ventricular chamber size, a moderate decrease in right ventricular systolic function, and an elevated right ventricular systolic pressure of 113 mm Hg, which is concerning for pulmonary arterial hypertension (PAH). Because of the patient’s history of hypoplastic left heart syndrome, no LVEF was documented.
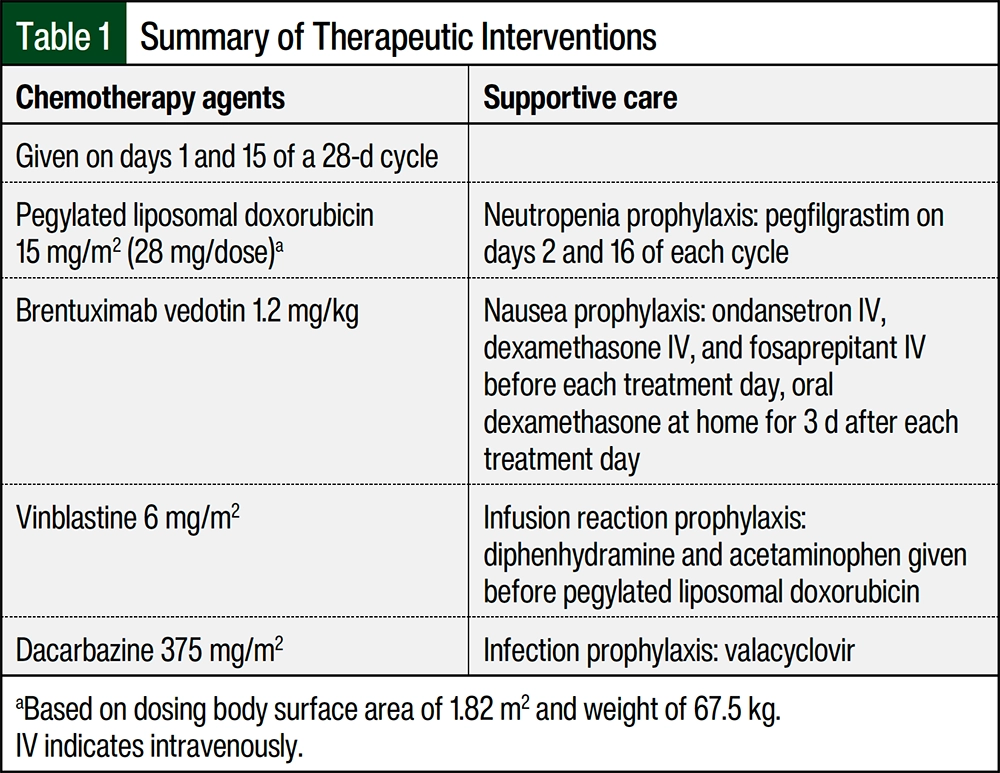
Considering the patient’s baseline congenital cardiac disease and pulmonary hypertension, cardiac and pulmonary AEs associated with ABVD therapy were concerning. After collaboration with the patient’s cardiologist and other members of the multidisciplinary team, the decision was made to initiate a modified version of the AAVD regimen and substitute PLD, a less cardiotoxic formulation, for conventional doxorubicin (thus ADVD). Although there are published safety and efficacy data for the incorporation of pegylated doxorubicin in a modified ABVD regimen,6 a bleomycin-free regimen was ultimately prioritized considering this patient’s baseline PAH. The anthracycline-free BV-DTIC regimen was not pursued considering this regimen has only been studied in an elderly patient population.5
Table 1 outlines the dosing of this modified regimen, including supportive care measures. To determine the dosing of PLD in this regimen, we considered alternative settings where PLD is safely administered. In the GVD (gemcitabine, vinorelbine, PLD) regimen used in the relapsed or refractory cHL setting, PLD is safely given at a dose of 15 mg/m2 intravenously on days 1 and 8 of a 21-day cycle, targeting a cumulative dose of 30 mg/m2 in a 21-day period.8 Considering that PLD can be safely dosed at 15 mg/m2 as frequent as every 7 days, we extrapolated the tolerability of this dose for our patient.
Because our patient had stage IIA cHL, we decided to extrapolate the PET-directed approach of the RAPID study with our modified ADVD regimen.9 Our patient received 2 cycles of ADVD followed by a restaging PET scan that demonstrated a complete response, with resolution of the hypermetabolic adenopathy initially visualized and a Deauville score of 2. The patient then received 2 additional cycles of ADVD followed by consolidative radiation (20 Gy in 10 fractions) to the left neck region. The end-of-treatment PET scan demonstrated a sustained complete response, except for mild 18F-fluoro-2-deoxy-D-glucose (FDG) avidity in the mediastinal and bilateral lymph nodes. These FDG-avid regions were attributed to a granulomatous reaction, and hence the end-of-treatment PET was reported as a Deauville score of 3X.
At the time of this report, 2.5 years after the completion of therapy, all surveillance computed tomography (CT) scans performed in 6-month intervals continue to demonstrate a sustained complete response. Overall, our patient tolerated therapy well without treatment delays, with the exception of pneumonia warranting hospitalization on one occasion after cycle 2 that led to a 2-week treatment delay. Notably, our patient had grade 3 or 4 thrombocytopenia during the expected chemotherapy nadir period, although no platelet transfusions were required. Despite the significant thrombocytopenia, his platelet counts recovered before each subsequent treatment day, and hence no dose reductions were made. No dermatologic AEs were noted throughout therapy, which is typically a concern with PLD treatment, and the patient’s renal and hepatic functions remained stable throughout chemotherapy. In addition, only very mild (grade 1) neuropathy of the fingertips was reported during therapy.
Repeat echocardiograms performed at approximately 1, 4, 6, 18, and 30 months after the completion of chemotherapy demonstrated the preservation of cardiac function compared with baseline (Table 2). Regarding liver disease, surveillance CT scans continued to demonstrate stable morphologic features of chronic liver disease or cirrhosis that were suspected to be of cardiac etiology and splenomegaly related to portal hypertension. Approximately 1.5 years after the completion of therapy, our patient successfully fathered a healthy child in the absence of any fertility preservation measures.
Discussion
To our knowledge, this is the first case report that describes the use of this ADVD regimen. As previously mentioned, this ADVD regimen versus a PLD-based ABVD regimen was selected for this patient because of concerns about the use of bleomycin-based therapy with his underlying PAH. We felt that the use of the anthracycline-sparing BV-DTIC regimen was less suitable for our otherwise young, fit patient considering the BV-DTIC regimen has largely been evaluated in an elderly patient cohort. We acknowledge that the use of dexrazoxane before conventional doxorubicin would have also been a suitable alternative cardiac-sparing treatment approach. However, ADVD was ultimately chosen after an in-depth discussion of both options with the patient.
The dosing of PLD in our ADVD regimen (15 mg/m2 on days 1 and 15) was largely based on the dosing of this therapy in other non-cHL settings, as previously mentioned, given the absence of literature on the use of PLD with ABVD or AAVD at the time of our case study. Of note, in the study by Liu and colleagues, which was published after the time that this patient was receiving treatment, PLD was safely dosed at 25 mg/m2 on days 1 and 15 of an ABVD-inspired regimen.6 Hence, the question of the ideal dosing of PLD in ABVD- and AAVD-based regimens remains unanswered at this time. This case report is also novel from the standpoint of using a PET-directed approach to therapy akin to the RAPID trial in a patient with early-stage cHL who received a modified AAVD regimen.9 Because our patient had favorable early-stage disease, we felt that 6 cycles of this modified AAVD regimen (as traditionally studied in advanced-stage cHL in the ECHELON-1 study) would result in undue AEs.10
Conclusion
Given the absence of literature investigating the use of PLD in an AAVD-based regimen for the treatment of newly diagnosed cHL, this case report may provide preliminary insights on the safety and efficacy of substituting PLD for conventional doxorubicin in patients with early-stage cHL and cardiac and pulmonary comorbidities. Overall, our patient tolerated 4 cycles of ADVD without significant AEs or treatment delays and remains in complete response to date with stable cardiac function. He only had grade 1 neuropathy, which improved within approximately 3 to 6 months after treatment cessation, and he did not have any hand-foot dermatologic AEs, which are associated with treatment with liposomal doxorubicin. To date, the patient has not had any worsening of PAH. Further studies regarding the optimal dosing of PLD in patients newly diagnosed with cHL are warranted.
Dr Lee, Dr Heers, Ms Lundquist-Crabbe, Dr Stanford, and Dr Klueppelberg have no conflicts of interest to report.
References
- National Cancer Institute. SEER cancer stat facts: Hodgkin lymphoma. Accessed June 10, 2024. https://seer.cancer.gov/statfacts/html/hodg.html
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines): Hodgkin lymphoma. Version 3.2024. March 18, 2024. Accessed March 19, 2024. www.nccn.org/professionals/physician_gls/pdf/hodgkins.pdf
- Engert A, Plütschow A, Eich HT, et al. Reduced treatment intensity in patients with early-stage Hodgkin’s lymphoma. N Engl J Med. 2010;363:640-652.
- Ansell SM, Radford J, Connors JM, et al. Overall survival with brentuximab vedotin in stage III or IV Hodgkin’s lymphoma. N Engl J Med. 2022;387:310-320.
- Friedberg JW, Forero-Torres A, Bordoni RE, et al. Frontline brentuximab vedotin in combination with dacarbazine or bendamustine in patients aged ≥60 years with HL. Blood. 2017;130:2829-2837.
- Liu W, Yang M, Ping L, et al. Chemotherapy with a pegylated liposomal doxorubicin-containing regimen in newly diagnosed Hodgkin lymphoma patients. Cardiovasc Toxicol. 2021;21:12-16.
- Salvi F, Luminari S, Tucci A, et al. Bleomycin, vinblastine and dacarbazine combined with nonpegylated liposomal doxorubicin (MBVD) in elderly (≥70 years) or cardiopathic patients with Hodgkin lymphoma: a phase-II study from Fondazione Italiana Linfomi (FIL). Leuk Lymphoma. 2019;60:2890-2898.
- Bartlett NL, Niedzwiecki D, Johnson JL, et al. Gemcitabine, vinorelbine, and pegylated liposomal doxorubicin (GVD), a salvage regimen in relapsed Hodgkin’s lymphoma: CALGB 59804. Ann Oncol. 2007;18:1071-1079.
- Radford J, Illidge T, Counsell N, et al. Results of a trial of PET-directed therapy for early-stage Hodgkin’s lymphoma. N Engl J Med. 2015;372:1598-1607.
- Connors JM, Jurczak W, Straus DJ, et al. Brentuximab vedotin with chemotherapy for stage III or IV Hodgkin’s lymphoma. N Engl J Med. 2018;378:331-344. Erratum in: N Engl J Med. 2018;378:878.