Connecting Science to Practice
Central nervous system (CNS) lymphoma is a rare but
highly aggressive complication of diffuse large B-cell lymphoma
(DLBCL). Despite the significant morbidity and
mortality associated with CNS lymphoma, the optimal
CNS prophylaxis method remains controversial. This
study assessed the efficacy and safety of CNS prophylaxis
in patients with DLBCL and determined whether one
route was more effective than the other in the prevention
of CNS relapse. This retrospective analysis evaluated patients
with DLBCL who received treatment at the University
of North Carolina (UNC) Medical Center or UNC
Health Rex between June 20, 2014, and August 1, 2021.
The primary outcome was CNS relapse rates at 2 years in
patients who received CNS prophylaxis versus no CNS
prophylaxis. The secondary outcomes included the CNS
relapse rates among patients who received intrathecal
methotrexate with or without cytarabine versus high-dose
methotrexate and all-cause mortality in all of the patients.
Diffuse large B-cell lymphoma (DLBCL) accounts for 25% to 30% of non-Hodgkin lymphoma worldwide.1 Primary central nervous system (CNS) lymphoma refers to lymphoma that has disseminated to the CNS, such as the parenchyma, dura, leptomeninges, cranial nerves, spinal cord, or intraocular compartment. Secondary CNS lymphoma, also known as CNS relapse, is a rare but highly aggressive complication of DLBCL with a median survival of 2.2 months after diagnosis.2 The CNS-International Prognostic Index (CNS-IPI) score is a widely used tool that predicts the risk for CNS relapse at 2 years in patients with DLBCL who are receiving the chemotherapy regimen of rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone.3 The CNS-IPI model categorizes patients into low risk, intermediate risk, or high risk based on 6 clinical risk factors. The risk factors include aged >60 years, elevated lactate dehydrogenase (LDH), ECOG Performance Status Score >1, stage III or IV DLBCL, extranodal involvement of >1 site, and kidney and/or adrenal gland involvement. Low-risk and intermediate-risk patients have a <5% risk for CNS relapse at 2 years, whereas patients with a high-risk score have a >10% risk for CNS relapse at 2 years.3
The 2025 NCCN B-cell lymphoma treatment guidelines recommend considering the use of CNS prophylaxis in high-risk patients (CNS-IPI score of 4-6 or adrenal gland involvement) to prevent progression to CNS lymphoma.4 In addition to the CNS-IPI score, the treatment guidelines state that CNS prophylaxis can be considered in patients with testicular lymphoma, chromosomal alterations in MYC and BCL2 and/or BCL6, primary cutaneous DLBCL leg type, stage IE DLBCL of the breast, and kidney or adrenal involvement.4 Lymphoma involvement in the bone marrow and paranasal sinus have also been associated with a high risk for CNS relapse, but treatment guidelines do not include these 2 involvement sites for the consideration of CNS prophylaxis.5 The optimal route of administration of CNS prophylaxis remains unclear, but the NCCN’s treatment guidelines suggest using either intrathecal methotrexate and/or cytarabine (4-8 doses during or after a treatment course) or systemic high-dose methotrexate (3-3.5 g/m2 for 2-4 cycles).4
Intrathecal methotrexate and high-dose methotrexate have benefits and challenges. Intrathecal methotrexate may not adequately penetrate the brain parenchyma, where approximately 73% of CNS lymphoma occurs.6 Systemic high-dose methotrexate crosses the blood-brain barrier, but is associated with significant adverse events (AEs), including acute kidney injury, which occurs in 2% to 12% of patients.7 Renal AEs lead to delayed methotrexate clearance, which further worsens renal function and increases the risk for hepatotoxicity, myelosuppression, mucositis, and dermatologic AEs.7,8 As a result of these AEs, high-dose methotrexate requires renal monitoring and methotrexate levels with concurrent hydration, urinary alkalinization, and leucovorin.7 Intrathecal methotrexate is associated with less renal AEs than high-dose methotrexate, but may result in CNS AEs, such as headache, back pain, and fever.8,9 Because of the potential for methotrexate-related AEs and a lack of strong efficacy data, the preferred method and route of prophylaxis remain controversial, despite the significant morbidity and mortality associated with CNS lymphoma.
To our knowledge, CNS prophylaxis in DLBCL is limited to retrospective cohort studies. A 2021 retrospective study by Bobillo and colleagues evaluated patients with newly diagnosed DLBCL who had a high risk for CNS relapse.9 The study showed no difference in the rate of CNS relapse in patients who received intrathecal methotrexate versus systemic high-dose methotrexate.9 However, Cheah and colleagues conducted a 2014 multicenter, retrospective analysis of patients with DLBCL who had a high risk for CNS relapse.10 The study showed that the addition of systemic high-dose methotrexate with or without cytarabine was associated with a lower rate of CNS relapse compared with intrathecal methotrexate alone.10 Last, a 2015 retrospective analysis by Ferreri and colleagues stratified high-risk patients by CNS-IPI scores and extranodal involvement sites and showed that systemic high-dose methotrexate significantly reduced CNS relapse.11
The purpose of this study was to evaluate CNS relapse rates at 2 years in patients with DLBCL who received CNS prophylaxis with methotrexate compared with patients who did not receive CNS prophylaxis and to determine if intrathecal methotrexate with or without cytarabine versus high-dose methotrexate may be more effective in preventing CNS relapse.
Methods
This was a retrospective, multicenter, cohort study approved by the University of North Carolina (UNC) Institutional Review Board. Patients’ data were collected through the UNC Analytics and Outcomes department. Patients aged ≥18 years who received treatment for DLBCL at UNC Medical Center or UNC Health Rex between June 20, 2014, and August 1, 2021, were eligible for study inclusion. Patients were excluded from the study if they had CNS involvement at the time of diagnosis, other lymphoma diagnoses (Burkitt, lymphoblastic, lymphoproliferative, and gray-zone), received dual routes of prophylaxis (intrathecal and systemic), were incarcerated, or were pregnant.
The primary objective of this study was to evaluate CNS relapse rates at 2 years in patients with DLBCL who received CNS prophylaxis versus patients who did not receive CNS prophylaxis. The secondary outcomes evaluated the CNS relapse rates at 2 years in the subset of patients who received intrathecal methotrexate with or without cytarabine versus those who received high-dose methotrexate and assessed all-cause mortality at 2 years in all of the patients. The safety outcomes included the rates of renal impairment and delayed methotrexate clearance in the subset of patients who received high-dose methotrexate. Renal impairment was defined as a serum creatinine increase of >0.3 mg/dL within 10 days of the administration of systemic high-dose methotrexate. Delayed methotrexate clearance was defined as a methotrexate level of >1 μmol/L at ≥48 hours and then 0.2 μmol/L at 72 hours after the administration of systemic high-dose methotrexate.
The data collected at baseline included age, sex, ethnicity, ECOG Performance Status Score, LDH, CNS lymphoma history, number of extranodal sites, involvement of extranodal sites, stage IE primary DLBCL of breast, primary cutaneous DLBCL, leg type, translocations of MYC and BCL2 and/or BCL6 (double-hit and/or triple-hit lymphoma), HIV status, DLBCL stage, DLBCL new diagnosis or relapse, CNS-IPI score, chemotherapy backbone, CNS prophylaxis agent, number of prophylactic doses administered, and high-dose methotrexate dosing. The documentation of CNS lymphoma relapse and mortality at 2 years was obtained for the primary and secondary outcomes. Delayed methotrexate levels and serum creatinine were obtained for patients who received high-dose methotrexate.
Statistical Analysis
Continuous variables for the baseline characteristics were tested for normality and were reported as a median (interquartile range), and the 2 groups (CNS prophylaxis and no CNS prophylaxis) were compared using the Mann-Whitney U test. Categorical variables were reported as counts and percentages and were compared with the chi-square test. A multivariable logistic regression model was used to assess the relationship between the primary outcome, CNS lymphoma relapse rates at 2 years, and the treatment groups (CNS prophylaxis and no CNS prophylaxis). To adjust for potential confounding, various baseline characteristics were included in the multivariable analysis and were selected via a stepwise process. A multivariable logistic regression was also used to assess the relationship between CNS lymphoma relapse rates at 2 years and the CNS prophylactic agent (no CNS lymphoma prophylaxis, intrathecal methotrexate, intrathecal methotrexate/cytarabine, and systemic high-dose methotrexate), correcting for various baseline characteristics selected via a stepwise approach.
For sample size, a power analysis was conducted based on an estimated odds ratio of 4.75 for the primary outcome. With a significance level of 5% and power to 80%, the minimum sample size needed for testing 1 coefficient in a logistic regression model is 162 patients, and for a chi-square test with 1 degree of freedom the minimum sample size is 153 patients. The baseline characteristics for CNS relapse rates versus no CNS relapse were stratified and were compared using Fisher’s exact test. An alpha level of 0.05 was used for all statistical tests. The statistical analyses were performed using R statistics software, version 4.4.0 (R Foundation for Statistical Computing; Vienna, Austria) and SAS version 3.82 (SAS Studio; Cary, NC).
Results
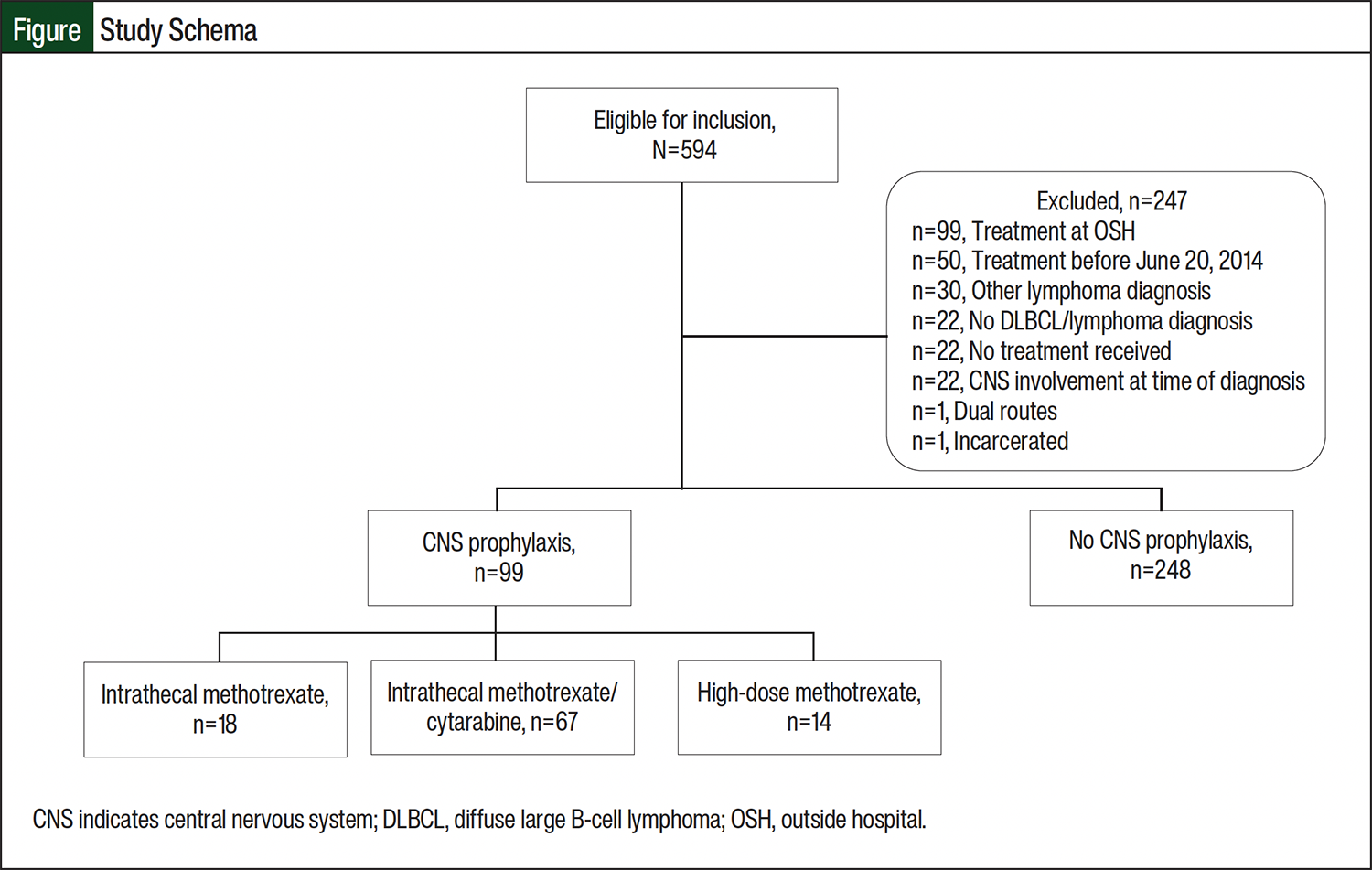
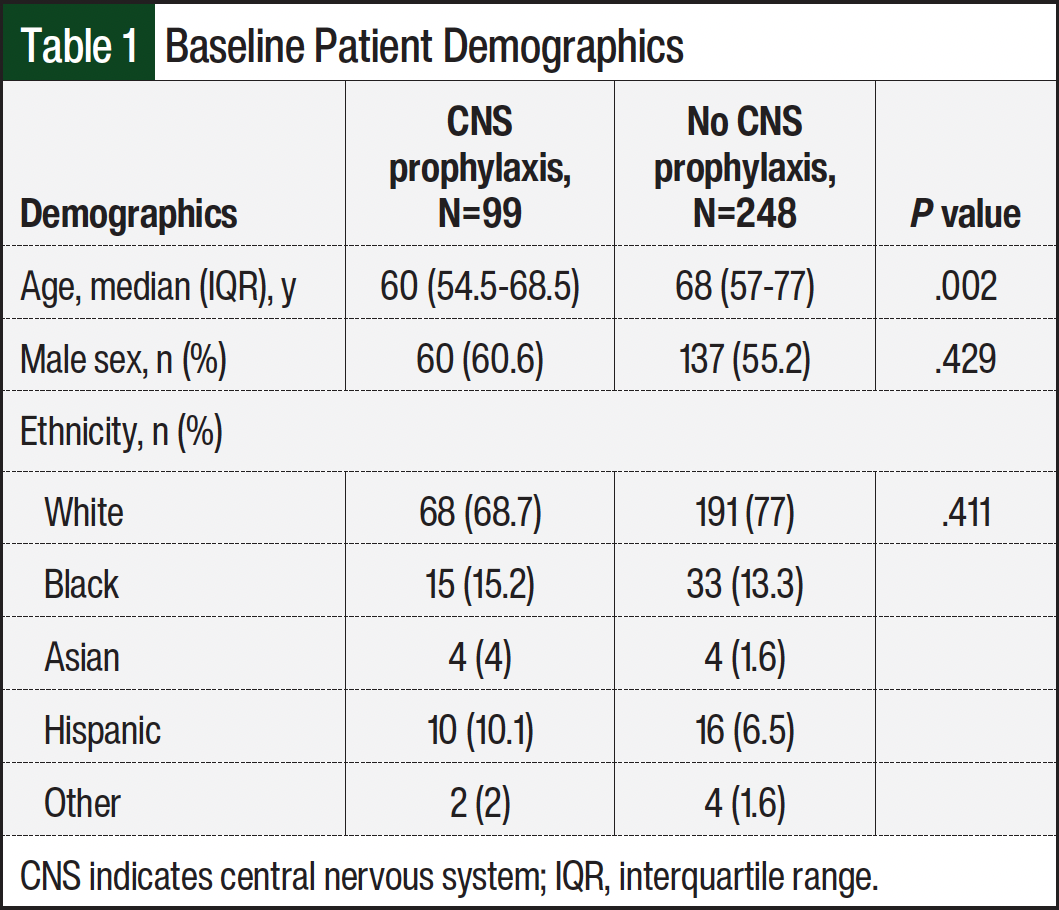
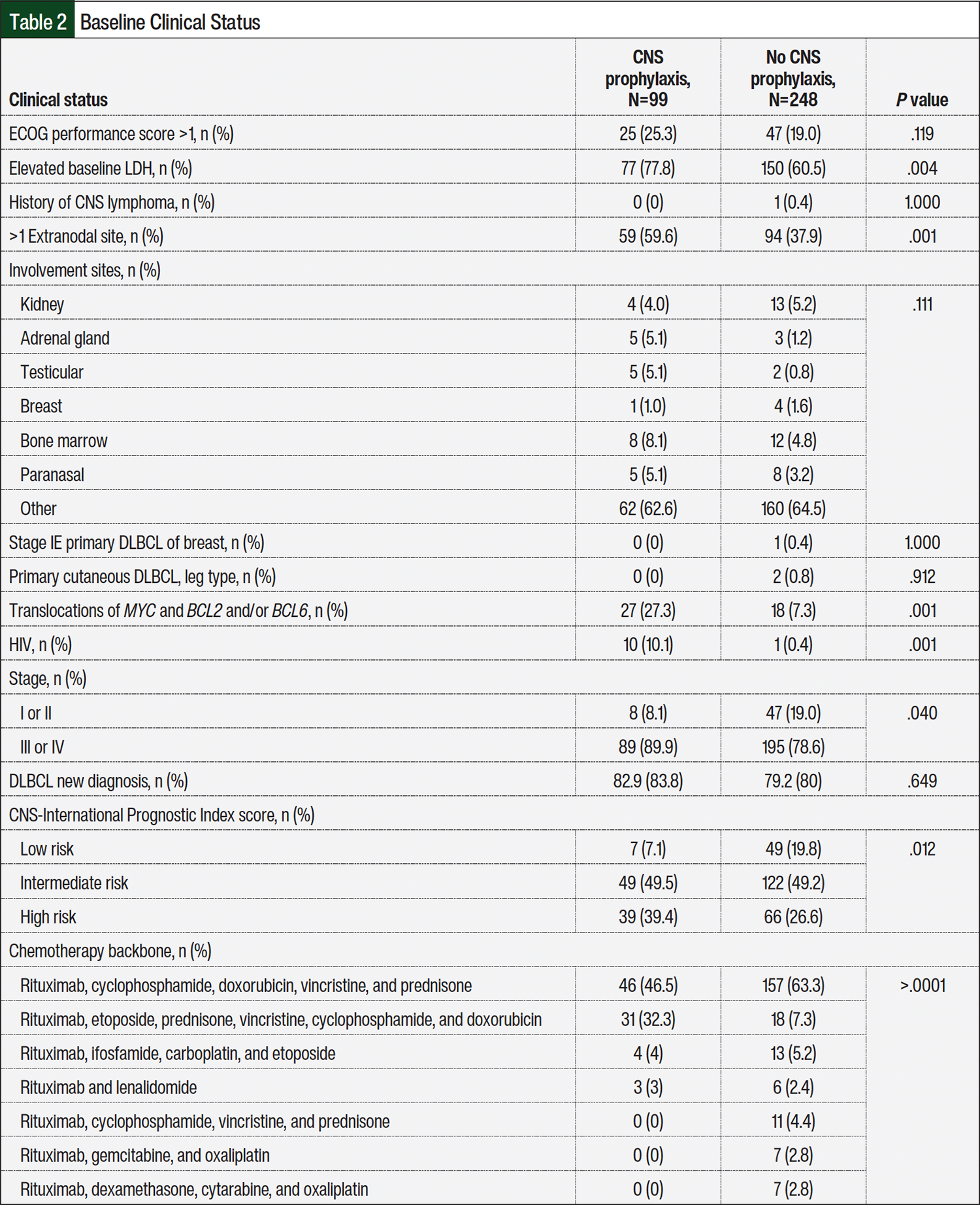
Of the 594 patients identified, 347 met the criteria for inclusion in this study, including 99 patients in the CNS prophylaxis group and 248 patients in the no CNS prophylaxis group (Figure). The baseline characteristics and clinical status differed between the 2 groups, as shown in Table 1 and Table 2. The median age was 60 years in the CNS prophylaxis group and 68 years in the no CNS prophylaxis group. The CNS prophylaxis group had higher rates of elevated baseline LDH (77.8% vs 60.5%, respectively; P=.004), >1 extranodal site (59.6% vs 37.9%; P=.001), presence of double-hit and/or triple-hit lymphoma (27.3% vs 7.3%; P=.001), HIV (10.1% vs 0.4%; P=.001), stage III or IV cancer (89.9% vs 78.6%; P=.04), and high-risk CNS-IPI score (39.4% vs 26.6%; P=.012). Rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone was the most frequently received chemotherapy backbone overall in the CNS prophylaxis and no CNS prophylaxis groups (46.5% and 63.3%, respectively); however, rituximab, etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin regimen was received by 32.3% of patients in the CNS prophylaxis group and by 7.3% of patients in the no CNS prophylaxis group (P>.0001).
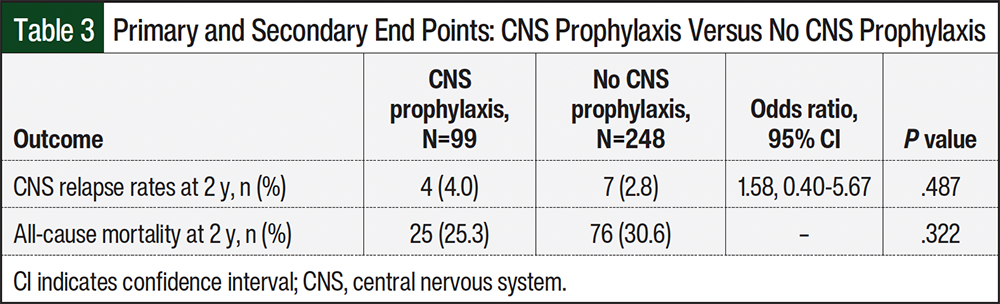
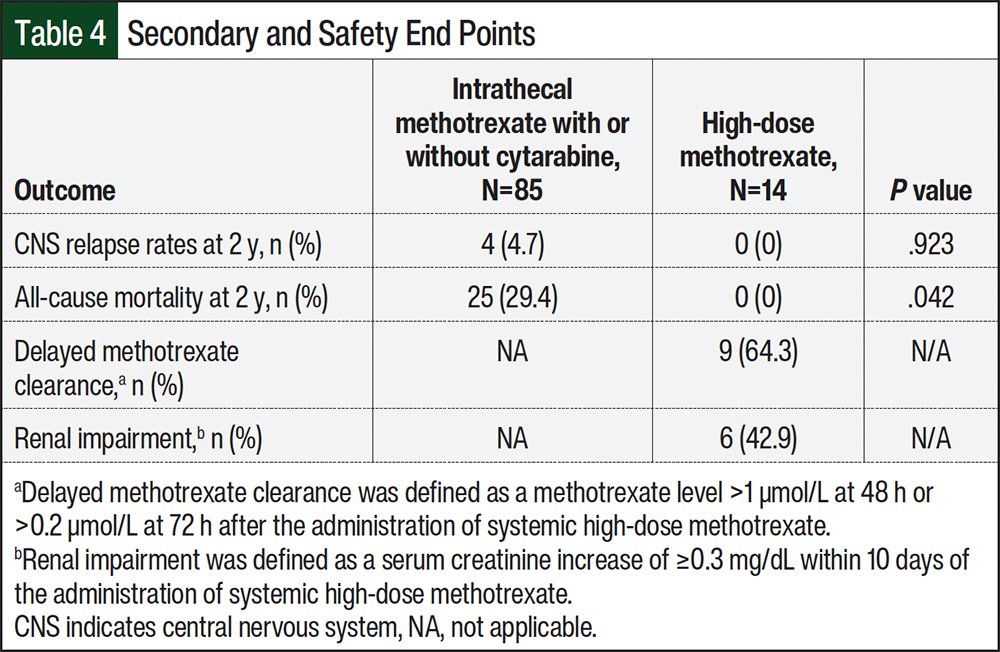
The logistic regression showed no significant difference in CNS relapse rates at 2 years between the CNS prophylaxis group and the no CNS prophylaxis group (4% vs 2.8%, respectively; P=.487) or the CNS prophylaxis agent (4.7% vs 0%, respectively; P=.923; Table 3 and Table 4). However, in the no CNS prophylaxis group, there were 2 patients who likely had CNS lymphoma based on imaging, but they died before a CNS lymphoma diagnosis was made; therefore, they were not included in the CNS relapse primary outcome. There were no significant differences in baseline clinic status between the patients who had CNS relapse and those who did not have CNS relapse (Appendix Table S1).
There was no significant difference in all-cause mortality at 2 years when comparing the CNS prophylaxis and no CNS prophylaxis groups (25.3% vs 30.6%, respectively; P=.322; Table 3). However, there was a significant difference in the all-cause mortality rate at 2 years between the patients who received intrathecal methotrexate with or without cytarabine and high-dose methotrexate (29.4% vs 0%, respectively; P=.042; Table 4). Last, in patients who received high-dose methotrexate, 64.3% of patients had delayed methotrexate clearance and 42.9% had renal impairment (Table 4).
Discussion
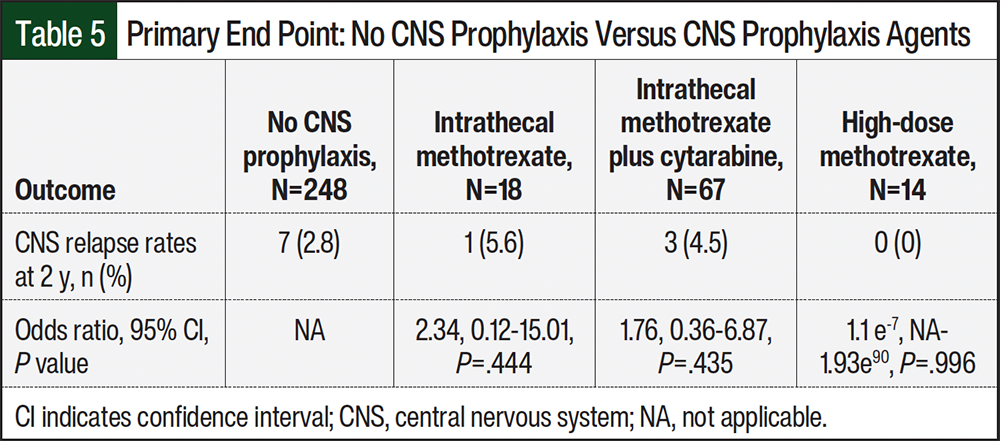
In this study, there was no difference in CNS relapse rates at 2 years or all-cause mortality at 2 years when comparing patients who received CNS prophylaxis with those who did not receive CNS prophylaxis (Table 3). When comparing CNS prophylaxis methods (intrathecal methotrexate, intrathecal methotrexate plus cytarabine, and high-dose methotrexate), there was no significant difference in CNS relapse rates at 2 years (5.6%, 4.5%, and 0%, respectively; Table 5). However, our study shows a significantly higher 2-year all-cause mortality rate in patients who received intrathecal methotrexate with or without cytarabine compared with high-dose methotrexate. It is important to note that our study did not control for confounding variables for the secondary outcomes and conclusions cannot be made from this outcome.
Our study also shows that most of the patients who received high-dose methotrexate had delayed methotrexate clearance and renal impairment. Given the potential AEs associated with high-dose methotrexate that can delay further treatment, this highlights the need for increased monitoring by physicians and pharmacists after patients receive high-dose methotrexate. Although the optimal route of CNS prophylaxis still remains unclear, this study shows a potential benefit with adding CNS prophylaxis to prevent CNS relapse, but definite conclusions cannot be made because of confounding variables. Larger prospective studies are warranted to assess the safety and efficacy of the route of CNS prophylaxis.
After a review of the available literature, there were inconsistent findings on the efficacy of CNS prophylaxis and the optimal CNS prophylaxis route.9-12 A 2021 retrospective study by Bobillo and colleagues evaluated patients with newly diagnosed DLBCL who had a high risk for CNS relapse.9 A total of 295 (50%) patients received at least 1 dose of intrathecal methotrexate and/or cytarabine or high-dose methotrexate. The study showed lower rates of CNS relapse at 1 year in patients who received CNS prophylaxis versus no CNS prophylaxis (2% vs 7.1%, respectively; risk ratio [RR], 0.29; 95% confidence interval [CI], 0.08-0.66). However, the difference was similar at 5 years, with a CNS relapse rate of 5.6% versus 7.5%, respectively (RR, 0.76; 95% CI, 0.35-1.50), indicating that CNS prophylaxis tended to delay CNS relapse rather than prevent it. Furthermore, the study also showed no difference in CNS relapse rates when comparing intrathecal methotrexate with high-dose methotrexate at 5 years (5.2% vs 5.3%, respectively).9 Our study examined the 2-year CNS relapse rates, which suggest the need for future studies to evaluate patients over a 5-year period to determine if CNS prophylaxis prevents or delays progression to CNS lymphoma. In addition, Bobillo and colleagues only evaluated high-risk patients,9 whereas our study included all patients regardless of relapse risk.
In a 2022 large retrospective study, Orellana-Noia and colleagues compared intrathecal methotrexate with high-dose methotrexate in patients with DLBCL regardless of CNS-IPI score.12 The study did not show a difference in CNS relapse rates between intrathecal methotrexate and high-dose methotrexate (5.4% vs 6.8%, respectively; P=.4). In addition, Cheah and colleagues conducted a 2014 multicenter, retrospective analysis of patients with DLBCL and a high risk for CNS relapse.10 Their study defined high risk as ≥2 of the following factors: multiple extranodal sites, elevated serum LDH, and B symptoms or involvement of specific high-risk anatomic sites.10 This definition is not consistent with the current NCCN treatment guidelines.4 The study compared 3 different treatment groups.10 Group 1 received rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone with intrathecal methotrexate; group 2 received rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone with intrathecal methotrexate and 2 cycles of high-dose methotrexate; and group 3 received intrathecal methotrexate and high-dose methotrexate with a dose-intensive systemic antimetabolite-containing chemotherapy backbone.10 The study concluded that the addition of high-dose methotrexate with or without cytarabine was associated with a lower rate of CNS relapse compared with intrathecal methotrexate alone at a median follow-up of 3 years (18.4% vs 6.9%, respectively, vs 2.3% in groups 1-3, respectively; P=.009).10
Last, a 2015 retrospective study by Ferreri and colleagues in 200 patients with DLBCL showed that high-dose methotrexate significantly reduced CNS relapse rates.11 The study stratified patients based on low or high risk for CNS relapse. Their study included 93 low-risk patients and 107 high-risk patients. A total of 40 high-risk patients received prophylaxis (high-dose methotrexate with or without intrathecal methotrexate), of whom 7 received intrathecal methotrexate alone. At a median follow-up of 60 months, 1 (1%) low-risk patient and 9 (8%) high-risk patients progressed to CNS lymphoma (P=.01). Of the high-risk patients, CNS relapse occurred in 8 (12%) of 67 patients who did not receive prophylaxis and in 1 (2.5%) of 40 patients who received prophylaxis, which, in this case, was intrathecal methotrexate alone. There was a significant difference in the rate of CNS relapse in the patients who received inadequate prophylaxis (no prophylaxis or intrathecal methotrexate alone) compared with those who received high-dose methotrexate prophylaxis (12% vs 0%, respectively; P=.03).
Study Limitations
There are several limitations to our study. The study’s retrospective data design could not control for all of the potential confounding variables outside of the baseline characteristics and clinical characteristics collected. In addition, this study did not assess the safety outcomes for the intrathecal prophylaxis group, because the administration of medication via this route is typically in the outpatient setting, and it would not be feasible to follow up on AEs, including CNS-related AEs (eg, headache, back pain, fever).
The CNS prophylaxis arm was small, and there were only 14 patients in the high-dose methotrexate group. In addition, the median number of doses administered in the CNS prophylaxis group was only 3 (range, 1-8 doses). Many patients received intrathecal prophylaxis during their diagnostic lumbar puncture, but if there was no bone marrow involvement, additional CNS prophylaxis was not administered. Those patients who received at least 1 dose of CNS prophylaxis were included in the study, which was a limitation. Last, the baseline characteristics differed significantly between patients in the CNS prophylaxis and no CNS prophylaxis groups, with more high-risk patients in the CNS prophylaxis group.
This study had several strengths, such as having appropriate exclusion criteria based on previous studies and treatment guidelines, being a multicenter study, using a logistic regression model to control for confounding baseline characteristics, and including all patients, irrespective of CNS-IPI score.
Conclusion
This study evaluated CNS relapse rates and mortality in all patients, irrespective of CNS-IPI scores. Patients who received CNS prophylaxis were comprised of more high-risk patients, which suggests that prophylaxis reduces the risk for CNS relapse and mortality similarly to low-risk patients; however, there were many confounders that made it difficult to confirm this conclusion. There was a significantly higher rate of all-cause mortality at 2 years in the patients who received intrathecal prophylaxis, which highlights the need for larger prospective studies to compare the safety and efficacy of the method and route of CNS prophylaxis in the prevention of CNS relapse and mortality. Furthermore, our study demonstrated a high incidence of delayed methotrexate clearance and renal impairment after the administration of high-dose methotrexate. Given the potential AEs with high-dose methotrexate that could delay further treatment, it is important for these patients to be monitored closely.
Acknowledgment
UNC Health Rex provided access to a biostatistician without funds outside of UNC Health Rex.
Author Disclosure Statement
Dr Bunn, Dr Carpenter, Dr Kritzer, Dr Christofferson, Dr Wiser, and Dr Laundon have no conflicts of interest to report.
References
- Padala SA, Kallam A. Diffuse Large B-Cell Lymphoma. StatPearls Publishing; 2023.
- Green K, Munakomi S, Hogg JP. Central Nervous System Lymphoma. StatPearls Publishing; 2024.
- Schmitz N, Zeynalova S, Nickelsen M, et al. CNS International Prognostic Index: a risk model for CNS relapse in patients with diffuse large B-cell lymphoma treated with R-CHOP. J Clin Oncol. 2016;34:3150-3156. doi:10.1200/JCO.2015.65.6520
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: B-cell lymphomas. Version 1.2026. December 22, 2025. Accessed January 20, 2026. www.nccn.org/professionals/physician_gls/pdf/b-cell.pdf
- Villa D, Connors JM, Shenkier TN, et al. Incidence and risk factors for central nervous system relapse in patients with diffuse large B-cell lymphoma: the impact of the addition of rituximab to CHOP chemotherapy. Ann Oncol. 2010;21:1046-1052. doi:10.1093/annonc/mdp432
- Kansara R, Villa D, Gerrie AS, et al. Site of central nervous system (CNS) relapse in patients with diffuse large B-cell lymphoma (DLBCL) by the CNS-IPI risk model. Br J Haematol. 2017;179:508-510. doi:10.1111/bjh.14229
- Howard SC, McCormick J, Pui CH, et al. Preventing and managing toxicities of high-dose methotrexate. Oncologist. 2016;21:1471-1482. doi:10.1634/theoncologist.2015-0164
- Methotrexate. Lexi-Drugs. Lexicomp online. Wolters Kluwer. Accessed September 5, 2025. http://online/lexi.com
- Bobillo S, Joffe E, Sermer D, et al. Prophylaxis with intrathecal or high-dose methotrexate in diffuse large B-cell lymphoma and high risk of CNS relapse. Blood Cancer J. 2021;11:113. doi:10.1038/s41408-021-00506-3
- Cheah CY, Herbert KE, O’Rourke K, et al. A multicentre retrospective comparison of central nervous system prophylaxis strategies among patients with high-risk diffuse large B-cell lymphoma. Br J Cancer. 2014;111:1072-1079. doi:10.1038/bjc.2014.405
- Ferreri AJM, Bruno-Ventre M, Donadoni G, et al. Risk-tailored CNS prophylaxis in a mono-institutional series of 200 patients with diffuse large B-cell lymphoma treated in the rituximab era. Br J Haematol. 2015;168:654-662. doi:10.1111/bjh.13194
- Orellana-Noia VM, Reed DR, McCook AA, et al. Single-route CNS prophylaxis for aggressive non-Hodgkin lymphomas: real-world outcomes from 21 US academic institutions. Blood. 2022;139:413-423. doi:10.1182/blood.2021012888